Abstract
The integration of biomechanical and morphological analyses holds tremendous potential for assessing the rupture risk of abdominal aortic aneurysms (AAA). We employed a one-way fluid-structure interaction (FSI) model to distinguish between ruptured AAA (RAAA) and asymptomatic intact AAA (IAAA), focusing on morphological and computational fluid dynamics (CFD) indices. Patient groups with ruptured RAAA and asymptomatic IAAA were matched by diameter, age, and sex. AAA morphology was analyzed via CT segmentation, and biomechanical indices—including wall shear stress (WSS), peak wall stress (PWS), maximum deformation (MD), and other indices—were determined using FSI analysis. Statistical comparisons were performed using paired t-tests or Wilcoxon rank sum tests. Multivariate and LASSO regression analyses identified predictive factors, and a nomogram was developed. Model accuracy was assessed using the area under the curve (AUC). In our study with 66 RAAA and 66 asymptomatic IAAA patients, the tortuosity of the RAAAs was 1.4 times that of the asymptomatic IAAAs (P = 0.0005). The PWS, MD and peak wall rupture index (PWRI) of the RAAAs was 1.18, 1.32 and 1.27 times that of the asymptomatic IAAAs (P = 0.0158, 0.0036, 0.0071). The MD position demonstrated high consistency with RAAA rupture locations (94.12%). Four variables were selected for a nomogram, predicting AAA rupture with an AUC of 0.7604 (95% CI 0.6653–0.8556) and an internal validation AUC of 0.8051 (95% CI 0.6400–0.9703). In this study, we demonstrated that the location of MD is valuable for predicting the rupture location of AAA. We constructed a nomogram incorporating four key predictors—aortic neck length (ANL), intraluminal thrombus volume relative to AAA volume (VILT/VAAA), tortuosity, and MD—that enhances the prediction of AAA rupture risk, offering a more personalized assessment beyond traditional diameter-based methods.
Similar content being viewed by others
Introduction
Abdominal aortic aneurysm (AAA) involves aorta enlargement (> 50% typical diameter)1. The most critical and life-threatening outcome of AAA is its rupture, which results in a staggering mortality rate of approximately 80%2. The significant incidence and mortality rates of ruptured AAA (RAAA) underscore its serious health threat. Current elective repair criteria are based on transverse diameter thresholds (55 mm for men and 50 mm for women)3. Notably, < 10% of AAAs below limits rupture, while some larger ones remain stable4. This highlights the inadequacy of relying solely on aortic diameter for assessing rupture risk and the need for surgical intervention, thus emphasizing the importance of reliable rupture risk assessments for early detection and intervention.
The mechanical failure of AAA involves structural breakdown due to the weakening of the arterial wall, primarily from the degeneration of pathological components such as elastic fibers and collagen. These factors contribute to a decline in the overall strength of the arterial wall, ultimately playing a role in the development of AAA5. This degeneration contributes to a decline in arterial wall strength, which, combined with dynamic factors like high pressure and flow velocity, leads to the abnormal ballooning of the aorta6. The formation and rupture of AAA occur when blood flow stresses exceed the tissue strength of the arterial wall, making the understanding of hemodynamic effects and blood flow profiles crucial for predicting rupture risk.
Numerical simulation is a valuable tool for exploring challenging conditions in human AAA models. Computational fluid dynamics (CFD) enables the investigation of disturbed hemodynamics and frictional shear stresses on vessel walls7. Peak Wall Stress (PWS) serves as an indicator of maximum mechanical tensile stress in AAA walls, notably elevated in symptomatic or ruptured AAAs compared to intact ones8. The Peak wall rupture index (PWRI), which represents the ratio of maximum wall stress to wall strength, may provide a superior metric for assessing rupture risk, offering a more comprehensive evaluation of mechanical stresses relative to wall strength6. Fluid-structure interaction (FSI) studies are essential for modeling the interplay between blood flow and compliant arterial walls, facilitating the exploration of flow characteristics and wall stress distribution within AAAs9. Previous research has shown promise in understanding the interaction between blood flow dynamics and arterial wall mechanics through bidirectional information exchange in FSI models. However, further validation is needed before clinical implementation, as existing studies often rely on geometric and limited biological or hemodynamic parameters.
The research utilized a one-way FSI model integrating laminar-turbulent flow transition to evaluate morphological and hemodynamic index between age, sex, and diameter-matched ruptured and asymptomatic intact AAAs, aiming to pinpoint rupture sites and predict rupture risk.
Method
This retrospective case-control study matched cases of ruptured AAAs 1:1 with controls having asymptomatic intact AAAs. Matching included maximum AAA diameter, age, and sex. In the matching process, the age difference was limited to less than 2 years, and the diameter difference was restricted to less than 5 mm. Ethics and governance approvals were obtained from the Hospital Institute’s Review Board and Ethics Committee. All experiments were performed in accordance with relevant guidelines and regulations.
Patients
Patients with infrarenal AAAs were retrospectively selected from the Vascular Surgery Department database of our hospital (2009–2022). The inclusion criteria were as follows: (1) AAA defined as a diameter of ≥ 30 mm; (2) RAAA group (ruptured AAA and precursors rupture (symptomatic)): Ruptured AAA was defined by the presence of retroperitoneal or peritoneal blood confirmed by CT, evaluated by a vascular specialist, with admission due to abdominal pain and emergency operation within 48 h; Symptomatic AAA was defined as AAA with symptoms requiring immediate repair within 48 h, without CT evidence of rupture. (3) Asymptomatic intact AAA: incidentally discovered AAA, CT angiogram suitable for finite element analysis (FEA). Exclusion criteria included: age < 18 years, thoracoabdominal aneurysm/dissection, unanalyzable CT (due to massive contrast extravasation in the event of rupture), prior vascular repair, rupture of iliac aneurysm, hereditary diseases (e.g., Marfan syndrome), vasculitis, and fungal aneurysms. Patient-specific risk factors such as gender, height, weight, body mass index (BMI), systolic blood pressure (SBP), diastolic blood pressure (DBP), heart rate (HR), hypertension, diabetes, ischemic heart disease, peripheral vascular disease, chronic obstructive pulmonary disease (COPD), stroke, tumor history, and AAA diameter were collected for both groups. Due to the potential for patients in the rupture group to be in a state of shock upon admission, blood pressure measurements after hemodynamic stabilization were utilized.
The studies involving human participants were reviewed and approved by the Xiangya Hospital Institute’s Review Board and Ethics Committee of Xiangya Hospital, Central South University. Written informed consent was obtained from all patients participating in this study (NO.201803481).
CT acquisition
CTA scans of the abdominal aorta were acquired using a multislice helical CT scanner (SIEMENS) with standard radiologic parameters (slice thickness: 0.5–1.0 mm). All CTA images were obtained via an automatic injection system administering a contrast agent intravenously. CT imaging was triggered when the Hounsfield unit at the center of the aorta reached 130 after the injection of contrast. CTA data were exported in the Digital Imaging and Communications in Medicine (DICOM) format.
Assessment of aneurysm morphology
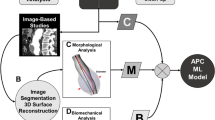
The three-dimensional (3D) model of the AAA was reconstructed from CT data using Mimics Medical 21.0 software (Materialise, Leuven, Belgium). Image segmentation involved defining threshold values to create a segmentation mask in MIMICS software10. The region of interest (ROI) comprised slices from below the lowest renal artery origin to above the aortic bifurcation, excluding accessory arteries. Various thresholds were applied, and optimized thresholds facilitated a region-growing function for personalized aorta mask generation. Manual adjustments, supervised by an expert vascular surgeon, eliminated undesired masks11. The vascular and intraluminal thrombus layers were processed using the same method, with the 3D model derived from the calculation layer (Fig. 1A). MIMICS’ 3D reconstruction function edited the mask to obtain the 3D geometric model for each layer (Fig. 1B). A centerline, based on the patient’s scan, served as a reference for sectioning AAA volumes perpendicular to this line from the renal to the iliac bifurcation levels. Intraluminal thrombus volume (VILT) was calculated by subtracting lumen volume from AAA volume12. Aortic neck length (ANL), aortic neck angle (α), aortic neck angle (β), volume of the abdominal aortic aneurysm (VAAA), and tortuosity were measured (Fig. 1C). Morphological index measurements followed previous studies13,14. The aorta’s 3D surface model was imported into 3-MATIC 13.0 software (Materialise, Leuven, Belgium) for smoothing (Fig. 1B).
The study flowchart and methods for investigating hemodynamics for AAA rupture. Three-dimensional (3D) segmentation produced using finite element analysis on the CT image of an AAA. (A) Geometry Segmentation. The yellow part is the AAA, the blue part is the blood; (B) 3D Segmentation Produced. Using 3-MATIC software smooth aneurysm model; (C) Morphological Measurement. Using MIMICS software measure AAA. Aortic Neck Length (ANL) = The length of the abdominal aortic aneurysm centerline from the plane of the renal artery to the plane of the enlarged segment of the abdominal aortic aneurysm. AAA α = The flow axis of the suprarenal aorta and the infrarenal neck; AAA β = The flow axis of the infrarenal neck and the body of the aneurysm; Tortuosity = One minus (the straight-line distance from the neck to the bifurcation of the iliac artery divided by the distance along the centerline); (D) Mesh Generation. Use ANSYS software to mesh the model and set entrance and exit conditions; (E) Applying Boundary Conditions. Navier Stokes equation. Set dynamic boundary conditions and set the governing equation of the fluid domain to the Navier-Stokes equation; (F) CFD Results of AAA. The left picture shows the fluid part results, and the right picture shows the fluid-structure coupling results. AAA = Abdominal Aortic Aneurysm. Figure created using Microsoft PowerPoint 2019 (Home and Student Edition) (https://www.microsoft.com/) and Microsoft Visio Professional 2019 (https://www.microsoft.com/).
Biomechanical analyses
The AAA 3D model in STEP format was imported into ANSYS 2022R1 and partitioned using the 3D tetrahedral grid method. The artery and intraluminal thrombus (ILT) formed the solid domain, while blood represented the fluid domain. ANSYS Fluent employed the finite volume method for fluid flow modeling, utilizing transient analysis to study pulsating blood flow (Fig. 1E). A time-dependent radial velocity profile at the fluid domain inlet served as a boundary condition (Fig. 1D, E)15. In ANSYS Mechanical, the ‘Transient Structural’ module modeled the AAA wall as a non-linear, hyperelastic, isotropic material15. The mechanical properties of ILT were represented using isotropic material models16. The laminar-turbulent transition was modeled using Menter’s hybrid k−ξ/k−ω SST model and the Gamma-theta transitional model. Solid-fluid coupling was achieved via the integrated multi-field solver (MFX) in ANSYS 2022R1, employing a one-way fluid-structure interaction (FSI) coupling approach. The fluid model solved for dynamics based on initial conditions. More details are provided in the Supplementary material.
Biomechanical indexes
Wall shear stress (WSS), peak wall stress (PWS), and maximum deformation (MD) were directly computed using ANSYS. The calculation methods for oscillatory shear index (OSI), endothelial cell activation potential (ECAP), and relative residence time (RRT) are detailed in prior reports (Fig. 1F) (Supplementary material)17. The peak wall rupture index (PWRI) represents the ratio of maximum wall stress to wall strength. AAA wall strength was evaluated using a statistical model incorporating ILT thickness, AAA diameter, sex, and family history16. These parameters were obtained from tensile testing of human AAA wall specimens, as outlined in previous literature (Supplementary material)16.
Assessment of intraobserver reproducibility
Intraobserver reproducibility for biomechanical indices in asymptomatic intact and ruptured abdominal aortic aneurysms (AAAs) was assessed using randomly selected CT images. The same observer evaluated these images on two occasions, 48 h apart.
Statistical analysis
Variables were compared between the training and validation cohorts using Student’s t-test or the Mann–Whitney U test for continuous variables, the chi-square test or Fisher’s exact test for unordered categorical variables, and the Mann–Whitney U test for ordered categorical variables. Continuous data were presented as median (interquartile range) and standard deviation; categorical data were presented as number (proportion). Age, aortic neck length (ANL), aortic neck angle, heart rate (HR), and diastolic blood pressure (DBP) followed a normal distribution (Shapiro-Wilk test). Paired t-test or Wilcoxon rank sum test was used to compare groups. Least Absolute Shrinkage and Selection Operator (LASSO) regression was employed to reduce data dimensionality and select predictors. Multivariable logistic regression analyzed independent associations between ANL, AAA β, VAAA, VILT, VILT/VAAA, tortuosity, peak wall stress (PWS), wall shear stress (WSS), maximum deformation (MD), relative residence time (RRT), endothelial cell activation potential (ECAP), peak wall rupture index (PWRI), and ruptured AAAs, adjusting for confounders or significant variables between asymptomatic intact and ruptured AAAs18. A predictive model and nomogram for ruptured AAA were developed using multivariable logistic regression. The model’s discriminatory capacity was assessed by the area under the curve (AUC), with internal validation performed via k-fold cross-validation. The code is available at https://github.com/lthcsu/CFDRAAA.git. Model calibration was evaluated using the Hosmer–Lemeshow test. R software (version 4.2.3; R Foundation for Statistical Computing, Vienna, Austria), GraphPad Prism (version 9.5.0, GraphPad Software, La Jolla, CA, USA), and SPSS (version 26.0; IBM SPSS Inc., Chicago, IL, USA) were used for data plotting and statistical analysis. A p-value of < 0.05 was considered statistically significant.
Result
Clinical characteristics of the study sample
A total of 718 patients with AAA with complete medical history and identifiable CTA images can be queried from the hospital electronic case system. 66 patients with a RAAA and 66 with an asymptomatic IAAA were included. Although the diameters of the 8 patients in the RAAA were not matched to similar asymptomatic IAAA, no significant differences were observed in age, sex, and abdominal aortic aneurysm diameter between the two groups. Patients with ruptured AAAs had a higher likelihood of a hypertension diagnosis (Table S1). Due to the historical nature of patients’ baseline data and blood pressure measurements being taken upon admission, hypertension may appear normal post-admission due to medication or medical intervention. Hence, while there is a statistical disparity in hypertension prevalence between groups based on underlying conditions, systolic and diastolic blood pressures show no significant difference.
According to the AAA surgical diameter classification recommended by the guidelines, 41 male patients (87%) in the RAAA group had an AAA diameter greater than or equal to the surgical threshold, with an average diameter of 76.77 ± 17.73 mm. In the asymptomatic IAAA group, 43 patients (91%) had an average diameter of 72.40 ± 14.74 mm. In the RAAA group, 6 male patients (13%) had diameters below the threshold, averaging 44.72 ± 2.37 mm, while 4 patients (9%) in the asymptomatic IAAA group averaged 42.95 ± 2.63 mm. For females, 18 patients (95%) in the RAAA group had an average diameter of 68.65 ± 16.65 mm, and 18 patients (95%) in the asymptomatic IAAA group averaged 64.35 ± 17.18 mm. One female patient (5%) in each group had diameters below the threshold, averaging 38.00 mm and 38.50 mm, respectively. Detailed results are available in Table S2.
More complex morphological structure in the RAAAs
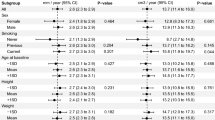
All patients were included in the comparison. As illustrated in Fig. 2 and Table S3, the mean aortic neck length (ANL) for RAAAs was 28.30 mm (95% CI 25.67–30.94), compared to 34.81 mm (95% CI 31.98–37.64) for asymptomatic IAAAs (P = 0.0034). The mean aortic neck angle (AAA β) for RAAAs was 112.90° (95% CI 108.20–117.60), while for asymptomatic IAAAs it was 130.10° (95% CI 114.80–125.20) (P = 0.0464). The mean tortuosity in RAAAs was 0.1661 (95% CI 0.1475–0.1846), compared to 0.1186 (95% CI 0.1018–0.1355) in asymptomatic IAAAs (P = 0.0005). The RAAA group exhibited shorter ANL, a smaller AAA β, and more curved blood vessels. The mean volume of abdominal aortic aneurysm (VAAA) and volume of intraluminal thrombus (VILT) in RAAAs were 328.50 cm2 (95% CI 275.90–381.20) and 136.20 cm2 (95% CI 102.20–170.20), respectively, which were larger than those in asymptomatic IAAAs, which measured 225.20 cm2 (95% CI 183.50–267.00) and 77.60 cm³ (95% CI 58.06–97.14) (P = 0.0034, P = 0.0270).
Compare the morphological results of ruptured abdominal aortic aneurysm (RAAA) and asymptomatic intact abdominal aortic aneurysm (IAAA) for groups. Compared with the diameter-matched asymptomatic IAAA group, the RAAA showed significantly larger volume of abdominal aortic aneurysm (VAAA), volume of intraluminal thrombus (VILT) and tortuosity, smaller aortic neck length (ANL) and aortic neck angle β (AAA β). VILT/VAAA = ratio of volume of intraluminal thrombus to volume of abdominal aortic aneurysm.
Larger PWS, MD and smaller WSS in the RAAAs
Through the forces exerted on the blood flow, intraluminal thrombus, and abdominal aortic aneurysm wall, the results for maximum deformation (MD), peak wall stress (PWS), and wall shear stress (WSS) were obtained (Figure S1). By comparing the blood flow between the RAAAs and the asymptomatic IAAAs, the results for oscillatory shear index (OSI), relative residence time (RRT), and endothelial cell activation potential (ECAP) were also obtained. The mean PWS in RAAAs was 0.5358 MPa (95% CI 0.4837–0.5880), compared to 0.4552 MPa (95% CI 0.4129–0.4976) in asymptomatic IAAAs (P = 0.0158). Additionally, the RAAA group exhibited smaller WSS, with a mean WSS of 0.1859 MPa (95% CI 0.1697–0.2021) compared to 0.2277 MPa (95% CI 0.2021–0.2533) in asymptomatic IAAAs (P = 0.0424). The mean MD in RAAAs was 5.726 mm (95% CI 5.090–6.363) as opposed to 4.348 mm (95% CI 3.772–4.923) in asymptomatic IAAAs (P = 0.0036). Furthermore, the mean peak wall rupture index (PWRI) in RAAAs was 0.6128 (95% CI 0.5370–0.6886) which was higher than the 0.4814 (95% CI 0.4230–0.5398) observed in asymptomatic IAAAs (P = 0.0071). The mean RRT and ECAP in RAAAs were 536.1 (95% CI 407.3–664.9) and 9.849 Pa−1 (95% CI 6.674–13.020), respectively, while in asymptomatic IAAAs they were 376.4 (95% CI 183.50–267.00) and 5.722 Pa−1 (95% CI 4.615–6.830) (P = 0.0422, P = 0.0169). This indicates that RAAAs have larger RRT and ECAP values. These comparisons are illustrated in Fig. 3 and Table S4.
Compare the computational fluid dynamics (CFD) results of ruptured abdominal aortic aneurysm (RAAA) and asymptomatic intact abdominal aortic aneurysm (IAAA) for groups. Compared with the diameter-matched asymptomatic IAAA group, the RAAA showed significantly larger peak wall stress (PWS), maximum deformation (MD), relative residence time (RRT), endothelial cell activation potential (ECAP), and peak wall rupture index (PWRI), smaller wall shear stress (WSS). OSI = Oscillatory shear index.
Significant alignment of MD locations with rupture location in RAAAs
A total of 34 patients with clearly defined rupture locations were included in the analysis. Based on the hydrodynamic model of AAAs and considering the constraining effect of the spine on the AAA wall, we identified the predicted MD locations (Fig. 4). The areas exhibiting maximum deformation, which indicate a high local rupture risk as determined by FEA, were compared with their corresponding rupture CTAs across all 34 patients (Fig. 4, Figure S2). The MD locations in the predicted rupture areas correlated with 32 of the 34 confirmed rupture sites, resulting in an accuracy rate of 94.12%.
The predicted rupture location. (A) Restraining effect of spine on wall deformation of abdominal aortic aneurysm. (B) Comparison between actual rupture position (a) and predicted rupture position (b). The arrow points to the rupture location. Figure created using Microsoft PowerPoint 2019 (Home and Student Edition) (https://www.microsoft.com/).
The Nomogram for predicting AAA rupture risk
From the 14 variables obtained from the morphological and CFD results of patients, four variables were selected based on non-zero coefficients identified through LASSO regression analysis (Fig. 5A, Figure S3A). Multivariable logistic regression further narrowed the selection to the following variables: ANL, VILT/VAAA, and Tortuosity (Table S5).
(A) Predictor selection using the LASSO regression analysis with tenfold cross-validation. Tuning parameter (lambda) selection of deviance in the LASSO regression based on the minimum criteria (left dotted line) and the 1-SE criteria (right dotted line). In the present study, predictor’s selection was according to the 1-SE criteria (right dotted line), where 4 nonzero coefficients were selected. LASSO, least absolute shrinkage and selection operator; SE, standard error. (B) The AUC (representative the discriminatory ability of the model) of the model and the internal validation. It shows the AUC of the predictive model (n = 99). AUC = Area Under the Curve. Threshold = 0.40235. (C) Nomogram for predicting risk of rupture of abdominal aortic aneurysm (RAAA) and its algorithm. First, a point was found for each variable of an abdominal aortic aneurysm (AAA) patient on the uppermost rule, then all scores were added together and the total number of points were collected. Finally, the corresponding predicted probability of RAAA was found on the lowest rule. ANL = Aortic Neck Length; VILT/VAAA = Ratio of Volume of Intraluminal Thrombus to Volume of Abdominal Aortic Aneurysm; MD = Maximum Deformation.
Given the statistical differences in mechanical indices between the two groups in univariate analysis, and the absence of statistical differences in multivariate analysis, significant differences in MD were identified through LASSO regression. Consequently, models including and excluding MD were compared based on their AUC values. It was determined that selecting ANL, VILT/VAAA, Tortuosity, and MD to construct the predictive AAA rupture risk model yielded the highest AUC. The subsequent multivariate logistic regression analysis produced a predictive model with an AUC of 0.7604 (95% CI 0.6653–0.8556). Internal validation utilizing the k-fold cross-validation method demonstrated an AUC of 0.8051 (95% CI 0.6400–0.9703) (Fig. 5B, Figure S3B). A personalized nomogram for predicting AAA rupture probability was developed, incorporating the variables ANL, VILT/VAAA, Tortuosity, and MD (Fig. 5C). Based on the optimal threshold results, patients with a predicted value exceeding 0.40235 were classified as being at higher risk of rupture (Fig. 5B). The proposed model exhibited excellent calibration, as illustrated in Figure S3C. The non-significant P value of 0.487 from the Hosmer–Lemeshow test indicated no statistical deviation, affirming a good fit between the predicted and observed values.
Discussion
Technological advancements have significantly improved the risk assessment of RAAA, particularly through fluid hemodynamics analysis. Despite this progress, aortic diameter remains the primary predictor of AAA rupture. Previous studies using fluid–solid coupling models to assess RAAA risk faced challenges due to limited patient data and insufficient integration of morphological and mechanical indicators, which restricted their predictive capabilities19.
In our study, we identified key morphological differences between RAAA and asymptomatic IAAA patients, including shorter ANL, increased vascular tortuosity, and elevated PWS, MD, and PWRI. We developed a predictive nomogram that incorporates ANL, VAAA, tortuosity, and MD, effectively assessing AAA rupture risk. Notably, around 13% of male patients did not meet the surgical threshold for rupture, aligning with existing literature4, while only one female patient fell below this threshold. These findings highlight the necessity for further research with larger datasets to explore the relationship between aneurysm diameter and rupture risk, particularly in female patients.
Our analysis corroborates previous literature indicating that the rupture group exhibits greater tortuosity and shorter ANL13,19. The impact of neck angle and vascular curvature on wall stress is well-documented, emphasizing the importance of integrating morphological indexes into rupture risk predictions20. Furthermore, our results align with earlier studies showing that diameter-matched ruptured AAAs demonstrate larger VAAA and VILT21.
The established correlation between hemodynamics and AAA rupture is evident in our findings, which show significant differences in PWS, WSS, ECAP, and RRT between RAAA and asymptomatic IAAA groups17. Specifically, elevated PWS in the rupture group suggests its potential role in AAA rupture risk8. Additionally, decreased WSS has been linked to increased rupture risk and poor blood flow, with low WSS associated with pro-inflammatory gene expression and diminished endothelial function22. Hemodynamic instability, indicated by measures such as OSI and ECAP, is crucial for predicting aneurysm progression17,23. Notably, PWRI was significantly higher in the RAAA group, reinforcing its value as a biomechanical predictor of AAA rupture risk24.
CFD models predominantly utilize rigid solid assumptions, which may not accurately reflect blood flow dynamics in AAAs12. Turbulent flow within AAAs can significantly increase wall stress, potentially damaging endothelial cells. Modeling the laminar-turbulent transition is crucial for more effective AAA rupture risk assessment. Despite changes in cardiac cycle conditions, the location of maximum wall stress remains constant in both laminar and SST models15. Previous CFD models overlooked cardiac cycle impact, limiting insights into aortic wall deformation due to pressure changes23. This study introduces a fluid–solid coupling model with a flexible aneurysm wall, integrating actual clinical blood pressure and cardiac cycle data as boundary conditions, while controlling for intergroup blood pressure differences and excluding confounding factors.
The rupture location significantly impacts RAAA prognosis. Studies indicate posterior wall ruptures may reduce bleeding, enabling surgical intervention, while anterior wall ruptures lead to excessive blood loss and increased mortality1. By modeling AAA wall elasticity and dynamic cardiac cycles, we identified deformation patterns that highlight areas prone to rupture. Our findings show a strong correlation (94.12%) between MD and actual rupture sites, suggesting potential for future rupture location prediction. Interestingly, ruptures were more peripheral than expected, likely due to core displacement inducing greater peripheral tensile stress.
Current research suggests that ILT significantly influences the rupture of AAA. The arterial wall morphology of AAA changes with cardiac pulsation, altering the hemodynamic environment and affecting ILT deposition and arterial wall stress. Our findings indicate that the ILT volume in the RAAA group is significantly greater than in the asymptomatic IAAA group, though the VILT/VAAA ratio did not differ significantly, affirming the distinction between RAAA and asymptomatic IAAA, potentially due to differences in AAA volume. ILT is composed of an inner layer (blood-facing), a middle layer, and an outer layer (wall-facing), with tensile strength decreasing from the inner to outer layer25. CFD studies show that ILT can reduce wall stress in AAAs, with ILT volume being a critical factor in rupture risk26,27. Furthermore, ILT presence may lead to degradation of wall matrix proteins, likely due to elevated pro-inflammatory proteins and proteolytic enzymes, promoting AAA growth and rupture28,29. Thus, the predictive role of ILT in AAA rupture remains debated.
In conclusion, biomechanical indicators such as PWS and PWRI are crucial for predicting AAA rupture, with our study revealing significant differences between RAAA and asymptomatic IAAA groups. Despite PWRI lacking significance in multivariate analysis, correlation analysis underscores the impact of morphology on mechanical outcomes24,30. Current predictive models inadequately integrate morphological and biomechanical indices. Our novel model, employing multivariate logistic and LASSO regression, identifies four critical variables for more accurate rupture risk assessments, suggesting that patients scoring above 0.40235 should be considered at high risk for rupture.
Our study has several limitations. Firstly, participants were exclusively recruited from our hospital, highlighting the necessity for validation in diverse cohorts. The challenge of accurately measuring wall strength, which may be more significant than wall stress in the context of AAA rupture, persists due to the lack of reliable non-invasive methods31. While current non-invasive techniques, such as MRI and [18 F] fluorodeoxyglucose positron emission tomography (PET), are valuable, they are not widely adopted in clinical practice32. In our analysis, we emphasized the importance of wall thickness by assuming a uniform aortic wall thickness of 2 mm, which may impact the calculated stress values. Future research should aim to integrate various clinical modalities to improve the accuracy of simulations. This study utilized ex vivo tensile experiments to assess wall strength25, which may not accurately reflect in vivo conditions in AAAs. Artificial intelligence (AI) has been employed to simulate AAA wall strength and predict aneurysm growth33. Future efforts to integrate AI with detection methods could enhance the accuracy of AAA modeling. Additionally, including patients with post-rupture CT scans introduces biomechanical variations that occur before rupture. This factor should be considered when interpreting findings on AAA rupture dynamics34. Furthermore, blood pressures at the entrance and exit boundaries may not accurately reflect pre-rupture conditions, as they could be influenced by prehospital emergency medications. Mansouri et al. have suggested that the distribution of calcification may affect the risk of AAA rupture35. However, in this study, calcification was not considered due to the limitations of the model. Our investigation primarily focused on large AAA diameters, which limits the generalizability of our findings to smaller AAAs6. Exploring risk prediction in smaller AAAs could help identify patients at higher risk, thereby prompting closer monitoring. Nonetheless, large-scale observational studies are necessary to validate this approach.
Conclusion
In this CFD-based investigation comparing 132 patients with ruptured and stable AAAs, the FSI model revealed significant differences in both morphological and biomechanical characteristics. Notably, a strong correlation was identified between the location of maximum deformation and the rupture site, highlighting the potential for precise rupture prediction. Furthermore, a CT imaging-based model that integrates morphological and mechanical indicators—specifically ANL, VILT/VAAA, Tortuosity, and MD—demonstrated robust predictive capabilities. This model enables personalized rupture risk assessment that extends beyond traditional diameter-based indications.
Data availability
The data that support the findings of this study are available on request from the corresponding author, Wei Wang. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
References
McGloughlin, T. M. & Doyle, B. J. New approaches to abdominal aortic aneurysm rupture risk assessment: Engineering insights with clinical gain. Arterioscler. Thromb. Vasc. Biol. 30(9), 1687–1694 (2010).
Sakalihasan, N., Limet, R. & Defawe, O. D. Abdominal aortic aneurysm. Lancet Lond. Engl. 365(9470), 1577–1589 (2005).
Wanhainen, A. et al. Editor’s choice—European Society for Vascular Surgery (ESVS) 2019 clinical practice guidelines on the management of abdominal aorto-iliac artery aneurysms. Eur. J. Vasc. Endovasc. Surg. Off. J. Eur. Soc. Vasc. Surg. 57(1), 8–93 (2019).
Shah, P. K. Inflammation, metalloproteinases, and increased proteolysis: an emerging pathophysiological paradigm in aortic aneurysm. Circulation 96(7), 2115–2117 (1997).
Baxter, B. T. et al. Effect of doxycycline on aneurysm growth among patients with small infrarenal abdominal aortic aneurysms: A randomized clinical trial. JAMA 323(20), 2029–2038 (2020).
Indrakusuma, R. et al. Biomechanical imaging markers as predictors of abdominal aortic aneurysm growth or rupture: A systematic review. Eur. J. Vasc. Endovasc. Surg. 52(4), 475–486 (2016).
Lipp, S. N. et al. Computational hemodynamic modeling of arterial aneurysms: A mini-review. Front. Physiol. 11, 454 (2020).
Khosla, S. et al. Meta-analysis of peak wall stress in ruptured, symptomatic and intact abdominal aortic aneurysms. Br. J. Surg. 101(11), 1350–1357 (2014). discussion 1357.
Lin, S., Han, X., Bi, Y., Ju, S. & Gu, L. Fluid-structure interaction in abdominal aortic aneurysm: Effect of modeling techniques. BioMed. Res. Int. 2017, 7023078 (2017).
Gasser, T. C., Auer, M., Labruto, F., Swedenborg, J. & Roy, J. Biomechanical rupture risk assessment of abdominal aortic aneurysms: Model complexity versus predictability of finite element simulations. Eur. J. Vasc. Endovasc. Surg. 40(2), 176–185 (2010).
Shin, E. et al. Hemodynamics in diabetic human aorta using computational fluid dynamics.
Zambrano, B. A. et al. Association of intraluminal thrombus, hemodynamic forces, and abdominal aortic aneurysm expansion using longitudinal CT images. Ann. Biomed. Eng. 44(5), 1502–1514 (2016).
Georgakarakos, E. et al. The role of geometric parameters in the prediction of abdominal aortic aneurysm wall stress. Eur. J. Vasc. Endovasc. Surg. 39(1), 42–48 (2010).
Chaikof, E. L. et al. Identifying and grading factors that modify the outcome of endovascular aortic aneurysm repair. J. Vasc. Surg. 35(5), 1061–1066 (2002).
Pal, P. Computational modeling of the effects of transient blood flow characteristics and wall thickness on the rupture of abdominal aortic aneurysm. In Mechanics of Solids, Structures and Fluids Vol. 9, p. V009T12A082. (American Society of Mechanical Engineers, 2014). https://asmedigitalcollection.asme.org/IMECE/proceedings/IMECE2014/46583/Montreal,%20Quebec,%20Canada/253921
Vande Geest, J. P., Sacks, M. S. & Vorp, D. A. A planar biaxial constitutive relation for the luminal layer of intra-luminal thrombus in abdominal aortic aneurysms. J. Biomech. 39(13), 2347–2354 (2006).
Mutlu, O. et al. How does hemodynamics affect rupture tissue mechanics in abdominal aortic aneurysm: Focus on wall shear stress derived parameters, time-averaged wall shear stress, oscillatory shear index, endothelial cell activation potential, and relative residence time. Comput. Biol. Med. 154, 106609 (2023).
Venkatasubramaniam, A. K. et al. A comparative study of aortic wall stress using finite element analysis for ruptured and non-ruptured abdominal aortic aneurysms. Eur. J. Vasc. Endovasc. Surg. Off. J. Eur. Soc. Vasc. Surg. 28(2), 168–176 (2004).
Koncar, I. B. et al. Morphological and biomechanical features in abdominal aortic aneurysm with long and short neck-case-control study in 64 abdominal aortic aneurysms. Ann. Vasc. Surg. 45, 223–230 (2017).
Li, Z. & Kleinstreuer, C. Effects of blood flow and vessel geometry on wall stress and rupture risk of abdominal aortic aneurysms. J. Med. Eng. Technol. 30(5), 283–297 (2006).
Spanos, K. et al. Anatomical differences between intact and ruptured large abdominal aortic aneurysms. J. Endovasc. Ther. Off. J. Int. Soc. Endovasc. Spec. 27(1), 117–123 (2020).
Alsiraj, Y. et al. Female Mice With an XY Sex chromosome complement develop severe angiotensin II-induced abdominal aortic aneurysms. Circulation 135(4), 379–391 (2017).
Joly, F., Soulez, G., Lessard, S., Kauffmann, C. & Vignon-Clementel, I. A Cohort longitudinal study identifies morphology and hemodynamics predictors of abdominal aortic aneurysm growth. Ann. Biomed. Eng. 48(2), 606–623 (2020).
Singh, T. P. et al. Comparison of peak wall stress and peak wall rupture index in ruptured and asymptomatic intact abdominal aortic aneurysms. Br. J. Surg. 108(6), 652–658 (2021).
Gasser, T. C., Görgülü, G., Folkesson, M. & Swedenborg, J. Failure properties of intraluminal thrombus in abdominal aortic aneurysm under static and pulsating mechanical loads. J. Vasc. Surg. 48(1), 179–188 (2008).
Ea, F. & Ch, K. K. A. The effect of asymmetry in abdominal aortic aneurysms under physiologically realistic pulsatile flow conditions. J. Biomech. Eng. 125(2) (2003). https://pubmed.ncbi.nlm.nih.gov/12751282/
Arslan, A. C. & Salman, H. E. Effect of intraluminal thrombus burden on the risk of abdominal aortic aneurysm rupture. J. Cardiovasc. Dev. Dis. 10(6), 233 (2023).
Erhart, P. et al. Finite element analysis in asymptomatic, symptomatic, and ruptured abdominal aortic aneurysms: In search of new rupture risk predictors. Eur. J. Vasc. Endovasc. Surg. Off. J. Eur. Soc. Vasc. Surg. 49(3), 239–245 (2015).
Kazi, M. et al. Influence of intraluminal thrombus on structural and cellular composition of abdominal aortic aneurysm wall. J. Vasc. Surg. 38(6), 1283–1292 (2003).
Urrutia, J. et al. Geometric surrogates of abdominal aortic aneurysm wall mechanics. Med. Eng. Phys. 59, 43–49 (2018).
Doyle, B. J., Miller, K., Newby, D. E. & Hoskins, P. R. Commentary: Computational biomechanics–based rupture prediction of abdominal aortic aneurysms. J. Endovasc. Ther. 23(1), 121–124 (2016).
McBride, O. M. B. et al. MRI using ultrasmall superparamagnetic particles of iron oxide in patients under surveillance for abdominal aortic aneurysms to predict rupture or surgical repair: MRI for abdominal aortic aneurysms to predict rupture or surgery-the MA(3)RS study. Open. Heart. 2(1), e000190 (2015).
Forneris, A. et al. AI-powered assessment of biomarkers for growth prediction of abdominal aortic aneurysms. J. Vasc. Surg. 4, 100119 (2023).
Golledge, J. et al. Thrombus volume is similar in patients with ruptured and intact abdominal aortic aneurysms. J. Vasc. Surg. 59(2), 315–320 (2014).
Mansouri, M. et al. CT analysis of aortic calcifications to predict abdominal aortic aneurysm rupture. Eur Radiol. https://link.springer.com/https://doi.org/10.1007/s00330-023-10429-1 (2023).
Acknowledgments
This study is supported by the National Natural Science Foundation of China (No. 81873525&82070491), Clinical Research Project of National Clinical Research Center for Geriatric Disorders; Hunan Provincial Health High-Level Talent Scientific Research Project (No. R2023038).
Author information
Authors and Affiliations
Contributions
TH L: Authors make substantial contributions to conception and design, and acquisition of data, and analysis and interpretation of data; T H: Authors make substantial contributions to conception and design, and acquisition of data, and analysis and interpretation of data; WT L: Authors make substantial contributions to acquisition of data, and analysis and interpretation of data; Y Y: Authors make substantial contributions to analysis and interpretation of data; SR X: Authors make substantial contributions to analysis and interpretation of data; KM W: Authors participate in drafting the article or revising it critically for important intellectual content; Y L: Authors make substantial contributions to conception and design and analysis and interpretation of data; J L: Authors make substantial contributions to conception and design, and analysis and interpretation of data; H B: Authors participate in drafting the article or revising it critically for important intellectual content; Z Y: Authors give final approval of the version to be published; W W: Authors give final approval of the version to be published; All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, T., Hu, T., Lu, W. et al. Morphology and biomechanical index predict the rupture location and rupture risk of abdominal aortic aneurysm. Sci Rep 15, 9604 (2025). https://doi.org/10.1038/s41598-025-94828-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94828-y