Abstract
This study explores new screening strategies to enhance liver cancer screening effectiveness. In a prospective study, 2605 participants underwent baseline, 6-months self-reported, and 1-year follow-up screenings using abdominal ultrasonography, AFP, AFP-L3%, and DCP. The results demonstrated the GALADUS protocol exhibited superior performance with higher AUC (0.935 vs. 0.836; DeLong P < 0.001), sensitivity (91.0% vs. 70.8%; P < 0.001), detection (3.1% vs. 2.4%; P < 0.001), and early diagnosis rates (64.2% vs. 58.7%) compared to the AFP/US protocol. Notably, among individuals with an aMAP score ≥ 60, GALADUS had significantly outperformed AFP/US in AUC (0.923 vs. 0.826; DeLong P < 0.001), sensitivity (94.2% vs. 69.6%; P < 0.001), detection (9.7% vs. 7.2%; P < 0.001), and early diagnosis rates (63.1% vs. 54.2%). However, for those with an aMAP score < 60, GALADUS offered no significant advantages. Introducing the “aMAP triage” protocol, combining GALADUS for aMAP ≥ 60 and AFP/US for aMAP < 60, further enhanced AUC to 0.925 (DeLong P < 0.001), improved sensitivity by 19.1% (89.9% vs. 70.8%; P < 0.001), and increased detection (3.1% vs. 2.4%; P < 0.001) and early diagnosis rates (65.0% vs. 58.7%), being cost-effective compared to GALADUS. In conclusion, this study highlights the potential of a stratified precision screening strategy in identifying high-risk individuals, applying tailored early detection protocols to improve liver cancer screening efficacy.
Similar content being viewed by others
Introduction
Liver cancer ranks among the most prevalent malignancies globally, with an estimated 905,700 new cases and 830,200 deaths reported worldwide in 20201. Regrettably, recent projections indicate a projected increase of over 55% in both new cases and deaths by 20402. What is even more discouraging is the dismal 5-year survival rate for liver cancer, which stands at a mere 12.1%. This poor prognosis is largely attributed to delayed clinical consultations by patients, leading to missed opportunities for timely treatment3,4. Conversely, radical resection in early-detected liver cancer has demonstrated a cure rate of up to 70%5. Consequently, screening and monitoring for liver cancer have been acknowledged as pivotal public health strategies for facilitating early diagnosis and treatment, ultimately aiming to enhance prognosis. In clinical practice, the implementation process of liver cancer screening entails identifying a suitable at-risk population (i.e., risk stratification) and subsequently administering early detection tests repeatedly to this population at regular intervals (i.e., early detection)6,7.
Initially, international guidelines and consensus recommended the implementation of risk stratification for the general population based on high-risk factors for liver cancer (such as hepatitis B, hepatitis C, and cirrhosis, among others) and the conduct of screening for high-risk populations. However, the current “one-size-fits-all” screening approach inevitably poses a significant healthcare burden, as it necessitates screening or monitoring of all patients with the same intensity, amidst an overall shortage of medical resources and unreasonable regional distribution. To shift from the current “one-size-fits-all” screening paradigm to an “individualized precision” screening approach, several objective liver cancer risk prediction models have emerged, ready for application in population risk stratification. In 2020, Fan et al. developed a novel liver cancer risk prediction model, abbreviated as aMAP (age, Male, Albumin–bilirubin, and Platelet), which was derived from inexpensive routine blood and liver function indices and displayed excellent performance in stratifying the 5-year risk of liver cancer development8. This study is the first to apply the aMAP score for risk stratification in a community population at high risk for liver cancer. Furthermore, the lack of efficient and affordable early detection protocols for liver cancer has resulted in unsatisfactory screening outcomes. In 2002, Japan began supplementing their traditional screening protocol, which comprised alpha-fetoprotein (AFP) and ultrasonography (US), with the serological detection of lens culinaris agglutinin-reactive fraction of alpha-fetoprotein (AFP-L3%) and des-gamma-carboxyprothrombin (DCP)9. While this protocol achieved superior screening effectiveness compared to AFP and US alone, it has yet to demonstrate ideal detection capabilities10. Previous research proposed the incorporation of two demographic characteristics, namely age and gender, to construct the GALADUS score after combining AFP and US with AFP-L3% and DCP, anticipating further improvement in liver cancer detection11. However, the GALADUS score was initially established and utilized solely in a hospital-based population, and community-based studies aimed at identifying indicators such as detection rate and misdiagnosis rate of liver cancer in the screening population have been limited12. The present study is the first to apply the GALADUS score, which is more intensive than the traditional screening protocol, for the early detection of liver cancer, with the objective of enhancing liver cancer detection, particularly at earlier stages.
In the current study, we attempted to transform the “one-size-fits-all” screening strategy into an “individualized precision” screening strategy. After implementing risk stratification utilizing the aMAP score, we explored precise early detection protocols tailored to various risk stratification outcomes, aiming to effectively control screening costs while enhancing liver cancer detection.
Results
During the baseline screening phase, among the 2605 participants, 172 tested positive, and 72 were subsequently diagnosed with liver cancer. At the six-month self-reported screening stage, of the 2533 participants who were free of a liver cancer diagnosis at baseline, 1595 voluntarily reported their screening outcomes (self-reported screening compliance rate: 62.9%). Of these, 45 tested positive, and 7 were diagnosed with liver cancer. Subsequently, a follow-up screening was conducted on patients who remained undiagnosed for liver cancer one year post-enrollment. Among the 2526 patients eligible for the follow-up screening, 2469 participated (follow-up screening compliance rate: 97.7%). During this follow-up, 52 tested positive, and 9 were diagnosed with liver cancer. For the remaining 57 individuals who did not participate in the follow-up screening, both active and passive follow-up measures were implemented. Consequently, one additional case of liver cancer was detected, and no deaths from any cause were observed (Fig. 1).
Flow chart of study enrollment and screening.
Comparison of liver cancer detection rates based on various characteristics
After confirming the diagnosis in 223 individuals who tested positive at least once during screening, 89 of the 2,605 enrolled participants were ultimately identified as having liver cancer (referral rate: 8.5%; liver cancer detection rate: 3.4%). Among these individuals, detection rates were notably higher in males, those with HBV and HCV co-infection, those not undergoing anti-HBV therapy, and those with cirrhosis (P < 0.05). Furthermore, detection rates demonstrated a steady increase with age (35–50, 50–60, 60–70, and ≥ 70 years; P-trend < 0.001) and smoking degree (no, mild, and severe; P-trend = 0.018). In terms of liver cancer staging, 56 cases were classified as BCLC stage 0 + A (early liver cancer) among all patients with liver cancer (early diagnosis rate: 62.9%). However, no significant difference in liver cancer staging (early stage vs. middle and advanced stages) was observed among patients grouped based on various characteristics (Table 1).
Regarding each screening indicator, the majority of participants exhibited low levels of AFP, AFP-L3%, and DCP, with 91.8%, 97.3%, and 95.1% of participants respectively possessing AFP concentrations below 7 ng/mL, AFP-L3% percentages below 5%, and DCP levels below 40 ng/mL. Furthermore, liver ultrasonography (US) was negative in 94.8% of the individuals. Upon comparing groups with and without liver cancer, we observed that AFP, AFP-L3%, and DCP levels, as well as GALADUS scores, were significantly elevated in liver cancer patients compared to non-liver cancer patients (P < 0.001). The rank-based elevation of AFP, AFP-L3%, and DCP demonstrated a rising trend in the detection rate of liver cancer (P-trend < 0.001). Additionally, the detection rates of liver cancer were notably higher among participants with a positive US compared to those with a negative US (P < 0.001). Regarding liver cancer stage, patients with early-stage disease exhibited lower levels of AFP and AFP-L3% and lower GALADUS scores compared to those with middle and advanced disease stages. However, no significant differences were observed in DCP levels or US findings. Furthermore, early diagnosis rates decreased progressively with the increasing rank of AFP levels (P-trend = 0.006; Table 2).
Enhancement of screening efficacy following the adoption of the GALADUS protocol, which integrates DCP and AFP-L3% detection into the traditional screening protocol
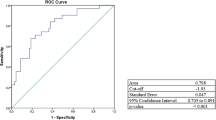
The traditional AFP/US protocol is typically employed when conducting liver cancer screening in actual clinical practice. After incorporating DCP and AFP-L3% detection, GALADUS scores were computed and, for the first time, attempted to be integrated into liver cancer screening. In consideration of the practicality of liver cancer screening, the GALADUS score was categorized into two groups using a threshold of − 0.7, determined as the optimal cutoff value via ROC curve analysis (GALADUS protocol).
As a novel liver cancer screening protocol, the GALADUS protocol demonstrated a significant enhancement in screening effectiveness compared to the traditional AFP/US protocol across the entire population [Categorical NRI (95% CI): 0.197 (0.113–0.281)]. Furthermore, the AUC of the GALADUS protocol was notably superior to that of the AFP/US protocol (0.935 vs. 0.836, DeLong P < 0.001), exhibiting a 20.2% increase in sensitivity (91.0% vs. 70.8%; P < 0.001) while maintaining nearly identical specificity (95.9% vs. 96.4%; P = 0.091) (Table 3). Additionally, the GALADUS protocol exhibited significantly higher detection rates (3.1% vs. 2.4%; P < 0.001) and early diagnosis rate (64.2% vs. 58.7%) compared to the AFP/US protocol, along with a modestly elevated referral rate (7.1% vs. 5.9%; P < 0.001). There was no significant difference in NNR between the two protocols (2.3 vs. 2.4; Fig. 2).
In total population: (a) Flowchart of AFP/US protocol, (b) Flowchart of GALADUS protocol, (c) Comparison of screening effectiveness between AFP/US protocol and GALADUS protocol, (d) ROC curves for AFP/US protocol and GALADUS protocol.
Differences in screening performance between the AFP/US and GALADUS protocols stratified according to an aMAP Score of 60
After stratified exploration based on the aMAP score, the current study found that the optimal threshold for the aMAP score in liver cancer risk stratification was 60.1, with an AUC of 0.824, a sensitivity of 77.53%, and a specificity of 77.03%. This threshold aligns with the recommendations for intensive screening or surveillance established when the aMAP score was introduced (Figure S1).
For participants with an aMAP score below 60, the GALADUS protocol demonstrated no significant difference in liver cancer screening efficacy compared to the AFP/US protocol [Categorical NRI (95% CI): 0.059 (− 0.036–0.155)], exhibiting a comparable AUC (0.888 vs. 0.858, DeLong P = 0.236) and sensitivity (80.0% vs. 75.0%; P = 1.000) along with a marginally higher specificity (97.6% vs. 96.7%; P = 0.004) (Table 3). Additionally, the GALADUS protocol showed no difference in detection rate (0.8% vs. 0.8%; P = 1.000) but a slightly lower early diagnosis rate (68.8% vs. 73.3%) compared to the traditional AFP/US protocol. Notably, it had a decreased referral rate (3.2% vs. 4.1%; P < 0.001) and a lower NNR (3.9 vs. 5.3; Fig. 3).
In populations with an aMAP score < 60: (a) Flowchart of AFP/US protocol, (b) Flowchart of GALADUS protocol, (c) Comparison of screening effectiveness between AFP/US protocol and GALADUS protocol, (d) ROC curves for AFP/US protocol and GALADUS protocol.
Among those with an aMAP score of ≥ 60, our findings clearly demonstrated that the GALADUS protocol exhibited significantly superior screening efficacy compared to the AFP/US protocol [Categorical NRI (95% CI): 0.195 (0.091–0.298)]. The AUC of the GALADUS protocol was significantly higher than that of the AFP/US protocol (0.923 vs. 0.826; DeLong P < 0.001), showcasing a 24.6% increase in sensitivity (94.2% vs. 69.6%; P < 0.001), with only a marginal decrease in specificity (90.5% vs. 95.7%; P < 0.001) (Table 3). Furthermore, the GALADUS protocol demonstrated a significantly improved detection rate (9.7% vs. 7.2%; P < 0.001) and early diagnosis rate (63.1% vs. 54.2%) compared to the AFP/US protocol. Additionally, it had a slightly increased referral rate (18.3% vs. 11.1%; P < 0.001), with a negligible difference in NNR (1.9 vs. 1.5; Fig. 4).
In populations with an aMAP score ≥ 60: (a) Flowchart of AFP/US protocol, (b) Flowchart of GALADUS protocol, (c) Comparison of screening effectiveness between AFP/US protocol and GALADUS protocol, (d) ROC curves for AFP/US protocol and GALADUS protocol.
Implementing a stratified screening protocol based on the aMAP score: a novel and promising screening strategy
The personalized “aMAP triage” protocol, in which the AFP/US protocol was implemented for individuals with an aMAP score of < 60 and the GALADUS protocol for those with a score of ≥ 60, exhibited significantly superior screening effectiveness compared to the AFP/US protocol. Specifically, it demonstrated a Categorical NRI (95% CI) of 0.179 (0.097–0.261), indicating a significant increase in the AUC from 0.836 to 0.925 (DeLong P < 0.001). Additionally, there was a 19.1% improvement in sensitivity, from 70.8–89.9%(P < 0.001), with only a minor 1.2% decrease in specificity, from 96.4 to 95.2% (P < 0.001) (Table 3). Concurrently, the “aMAP triage” protocol significantly outperformed the AFP/US protocol in terms of both the detection rate (3.1% vs. 2.4%; P < 0.001) and the early diagnosis rate (65.0% vs. 58.7%), while maintaining a nearly comparable NNR (2.5 vs. 2.4). Although a slight increase in the referral rate was observed (7.7% vs. 5.9%; P < 0.001), the enhancements in the detection rate, particularly the early diagnosis rate, are of much greater significance for liver cancer management.
Compared to the GALADUS protocol, the “aMAP triage” protocol exhibited lower screening costs while achieving similarly high sensitivity (89.9% vs. 91.0%; P = 1.000), detection rate (3.1% vs. 3.1%; P = 1.000), and early diagnosis rate (65.0% vs. 64.2%). The specificity of the “aMAP triage” protocol was only marginally reduced by 0.7% (95.2% vs. 95.9%; P < 0.001), and there was a slight increase in the referral rate (7.7% vs. 7.1%; P < 0.001). Notably, the “aMAP triage” protocol remained a relatively efficient screening tool that warrants promotion, as it demonstrated no statistically significant difference in screening effectiveness compared to the GALADUS protocol, as indicated by the Categorical NRI (95% CI) of − 0.018 (− 0.041 to 0.004) (Table 3; Fig. 5).
Furthermore, the accuracy parameters of these three screening protocols were further evaluated in various subgroups (e.g., female/male, over/under 60 years of age, HBV/HCV), and the results of these analyses were presented in Table S1 and S2.
In total population: (a) Flowchart of “aMAP triage” protocol, (b) Screening effectiveness of “aMAP triage” protocol, (c) ROC curve for “aMAP triage” protocol.
Discussion
As a public health measure, liver cancer screening should consider not only the screening benefits but also economic costs. Various cost-effective screening strategies for different types of cancer, such as colorectal cancer13, breast cancer14, prostate cancer15, employ a screening protocol of appropriate intensity after assessing individual risks and implementing risk stratification. In the process of exploring the optimal strategy for liver cancer screening, we adopted the aforementioned concept of risk stratification. This study is the first to utilize aMAP scores, which incur no or very low costs, for risk stratification in a population at high risk for liver cancer. Surprisingly, the aMAP scores demonstrated exceptional performance in risk stratification, and the cutoff values explored in this study were entirely consistent with those already established. After risk stratification, we sought appropriate early detection protocols for different risk-stratified populations. Among these protocols, a more expensive protocol that incorporates AFP-L3% and DCP into the traditional screening protocol can provide additional benefits for liver cancer screening in individuals with higher risk stratification. Accordingly, we established a personalized stratified screening strategy based on individual risk, taking into account both the cost and the effectiveness of screening. New efforts are currently underway to integrate other cancer biomarkers with traditional screening protocols, owing to the unsatisfactory screening effectiveness of AFP in conjunction with US. Among these biomarkers, AFP-L3% and DCP demonstrate the most promise for screening purposes. Previous studies have revealed that the incorporation of AFP-L3% detection can decrease the incidence of misdiagnosis in AFP-negative liver cancer cases and can effectively compensate for the limitations of AFP by offering high specificity in the early diagnosis of liver cancer (> 95%)16,17. Furthermore, some studies have demonstrated that DCP can enhance the diagnosis of AFP-negative liver cancer and significantly improve the sensitivity for liver cancer diagnosis, irrespective of the presence of HBV or HCV infection and whether the population consists entirely of liver cancer patients or early liver cancer patients16,18,19,20. To improve the screening of liver cancer, particularly early liver cancer, while maintaining appropriate referral protocols, the current study attempted to simultaneously detect AFP-L3% and DCP in addition to the traditional protocol, in order to calculate the GALADUS score. Overall, the GALADUS score, which incorporates the most comprehensive set of indicators, exhibited exceptional screening performance, with a sensitivity (91% vs. 95%) and specificity (96% vs. 91%) comparable to those reported in previous diagnostic studies11. The GALADUS protocol demonstrated significantly better detection and early diagnosis rates of liver cancer compared to the traditional protocol.
In the context of our research, the implementation of the GALADUS protocol would inevitably incur higher screening costs, despite the improved screening effectiveness achieved by incorporating the additional detection of AFP-L3% and DCP. Through in-depth exploration based on the concept of precise stratified screening, we determined that individuals with an aMAP score of ≥ 60 are at a greater risk of developing liver cancer. In such cases, the GALADUS protocol was able to detect more patients with liver cancer, particularly early liver cancer, compared to the AFP/US protocol. Therefore, we recommend an individualized stratified screening strategy termed “aMAP triage”. For participants with an aMAP score of < 60, the traditional AFP screening protocol combined with US is utilized. Conversely, for participants with an aMAP score of ≥ 60, the GALADUS protocol, which demonstrates superior screening effectiveness, is implemented.
The “aMAP triage” protocol, which allocates limited medical resources efficiently and is readily adoptable, exhibited significantly better detection and early diagnosis rates than the AFP/US protocol, while incurring costs comparable to those of the GALADUS protocol. The “aMAP triage” protocol, which allocates limited medical resources efficiently and is readily adoptable, exhibited significantly better detection and early diagnosis rates than the AFP/US protocol, while incurring lower costs than the GALADUS protocol.
The present study boasts several strengths. As is well-known, determining the effectiveness of a liver cancer screening protocol necessitates reference to the gold standard, which encompasses pathological findings or imaging diagnoses. However, conducting diagnostic examinations for liver cancer in individuals within the community who have a high likelihood of not having liver cancer would undoubtedly result in substantial harm, including psychological burden, radiation exposure, and other adverse effects. In this study, diagnostic examinations were only conducted on participants with positive screening results. During the 1-year follow-up, we identified that some early-stage liver cancer patients were missed due to their early-stage disease at baseline. Ultimately, the cumulative discovery of liver cancer across three screening sessions was employed as the gold standard to assess the effectiveness of the screening protocol, aiding in the identification of a more precise protocol for detecting early-stage liver cancer. Concurrently, non-liver cancer patients were determined based on three harmless and negative screening tests, which not only reduced costs but also avoided harmful diagnostic tests for a significant number of patients with chronic liver disease. To the best of our knowledge, this is the first cancer biomarker study to implement active screening using AFP, AFP-L3%, and DCP in a community population to date. In addition to demonstrating sensitivity and specificity according to the screening method, the current study also provides further indicators for the screened population, such as referral rate, detection rate, and early diagnosis rate, offering valuable insights for the future promotion of innovative liver cancer screening protocols in high-risk populations.
However, this study has limitations that merit discussion. Firstly, the sample size was small and comprised individuals from a limited geographical area, necessitating the conduct of a validation study with larger samples across multiple regions. Secondly, despite the application of the four indicators—AFP, AFP-L3%, DCP, and US—through three screening tests, unavoidable missed diagnoses of liver cancer may still exist. Thirdly, the participants in this study were predominantly patients infected with hepatitis B and C, necessitating further research on the applicability of the screening protocol to other high-risk populations for liver cancer, such as those with metabolic dysfunction-associated fatty liver disease or alcoholic liver disease. Fourthly, the cost comparison between screening protocols in this study was merely a rough and direct cost estimate, lacking precise cost calculation. Future health economics studies are required to further elucidate the conclusion. In conclusion, the GALADUS protocol, which incorporates the detection of AFP-L3% and DCP, demonstrates enhanced effectiveness in liver cancer screening when compared to traditional screening protocols. However, it also entails an augmentation in screening costs. A novel and promising stratified screening strategy, termed “aMAP triage,” has been proposed. This methodology advocates for the adoption of varying intensities of screening protocols, tailored according to risk stratification based on the aMAP score. It is anticipated to markedly elevate the detection rate and early diagnosis rate of liver cancer, while concurrently achieving cost savings in screening. This approach further facilitates the concentration of resources on pivotal prevention and control measures, rendering it a more efficient alternative.
Methods
Study design and participants
This prospective screening study was conducted among a community-based population at high risk for liver cancer in Jilin Province, China, spanning from 2019 to 2020. Enrolled subjects underwent identical follow-up screening one year after the baseline screening conducted by our team. Between the baseline screening and the one-year follow-up, a self-reported screening was conducted at a local hospital six months post-enrollment to obtain pertinent screening results. Participants with a positive baseline screening, a positive self-reported screening at six months, or a positive follow-up screening at one year underwent confirmatory tests for a diagnosis of liver cancer. Conversely, individuals with three negative screenings or a positive screening followed by a negative confirmatory test were classified as non-liver cancer cases. In brief, individuals meeting the following inclusion criteria were invited to participate in the liver cancer screening: (1) aged 35–75 for men or 40–75 for women; (2) HBV-infected patients with hepatitis B surface antigen (HBsAg) positivity, HCV-infected patients with hepatitis C antibody (HCV-Ab) positivity and detectable HCV RNA, or liver cirrhosis patients with negative HBsAg and HCV-Ab. Of the 2653 individuals who met the inclusion criteria, 48 were excluded due to: (1) a pre-existing diagnosis of malignant tumors, including liver cancer, at the time of enrollment; (2) the presence of other severe illnesses with an anticipated survival of less than one year; (3) inability to self-care (Fig. 1). Ultimately, 2605 participants were enrolled in the liver cancer screening study. The present study adhered to the standards and principles outlined in the Declaration of Helsinki and received approval from the Ethics Committee of the First Hospital of Jilin University (approval number: 19K043-001). Written informed consent was obtained from each participant prior to enrollment.
Screening and diagnosis procedures
Uniformly trained investigators conducted face-to-face interviews utilizing a standardized questionnaire after obtaining informed consent from the participants. In addition to gathering demographic data (i.e., sex, age, and education level), the questionnaire assessed health-related behaviors (i.e., smoking, alcohol consumption, and physical activity) alongside the participants’ history of diagnosis and treatment, as well as their family history of hepatic diseases or malignancies. Medical professionals with appropriate training conducted measurements of height, weight, and waist circumference. All participants underwent abdominal ultrasonography (Mindray M9; Shenzhen, China) and testing to determine serum levels of alpha-fetoprotein (AFP), AFP-L3%, and des-gamma-carboxy prothrombin (DCP) using the Hotgen MQ60plus system (Beijing, China). Furthermore, standard diagnostic tests were performed, including routine blood tests, liver function tests, and assessments of HBV DNA or HCV RNA levels. The ultrasonography was conducted by an attending physician with over five years of experience in ultrasound imaging to detect the presence of space-occupying liver lesions and liver cirrhosis. Follow-up screening was conducted on all participants with negative baseline screening results and/or diagnoses after one year, adhering to the same protocol as the baseline screening. Participants who tested positive for AFP, AFP-L3%, DCP, or had abnormal findings on ultrasonography at the baseline or follow-up screening were referred to tertiary hospitals for confirmatory diagnosis using dynamic computed tomography, dynamic magnetic resonance imaging, or liver biopsy. Based on clinical practice guidelines, these individuals received appropriate therapeutic interventions. Between the baseline screening and the one-year follow-up screening, participants were provided with written instructions and reminded via telephone to undergo serum AFP, AFP-L3%, and DCP level testing, as well as liver ultrasonography, six months after enrollment. They were also urged to upload their screening results to us via telephone follow-up. Subsequently, specialist physicians from our team reviewed the screening results reported by the participants and provided recommendations for further diagnosis and treatment.
Definition of positive screening protocols
To enhance the detection of liver cancer, a screening protocol incorporating AFP, AFP-L3%, DCP, and US was implemented during the baseline screening, self-reported screening at 6 months, and follow-up screening at 1 year in the present study. Individuals exhibiting any of the following characteristics were classified as AFP/AFP-L3%/DCP/US-positive: (1) serum AFP ≥ 400 ng/mL, AFP-L3% ≥ 15%, or DCP ≥ 200 ng/mL regardless of the US-detected nodule; (2) US-detected nodule of ≥ 2 cm in size, aside from typical liver cysts and hepatic hemangiomas, regardless of serum AFP, AFP-L3%, and DCP levels; and (3) US-detected nodule of ≥ 1 cm in size, aside from typical liver cysts and hepatic hemangiomas, with serum AFP ≥ 200 ng/mL, AFP-L3% ≥ 10%, or DCP ≥ 100 ng/mL. Furthermore, individuals who did not meet the criteria for AFP/AFP-L3%/DCP/US-positive but exhibited any of the following were designated as suspected AFP/AFP-L3%/DCP/US-positive and were mandated to undergo repeated liver cancer screening within 3 months until they were definitively classified as positive or negative for clinical screening: (1) serum AFP ≥ 20 ng/mL, AFP-L3% ≥5%, or DCP ≥ 40 ng/mL regardless of the US-detected nodule; (2) US-detected nodule of ≥ 1 cm in size, aside from typical liver cysts and hepatic hemangiomas, regardless of serum AFP, AFP-L3%, and DCP levels. AFP and US, the conventional screening protocol for liver cancer, have been widely employed for liver cancer screening. Consequently, individuals are categorized as AFP/US-positive or suspected AFP/US-positive based on AFP and US results according to the aforementioned criteria. If an individual is identified as suspected AFP/US-positive, repeated screening using AFP in conjunction with US within a 3-month period is necessary. Additionally, to ascertain whether the novel screening protocol GALADUS enhances liver cancer detection, this study utilized the traditional screening protocol AFP/US as a comparator. Simultaneously, GALADUS-positive individuals were defined based on the optimal cutoff value of the GALADUS score for liver cancer screening.
Definition and calculation of key indicators
A smoker was defined as someone who has smoked at least one cigarette per day for six months or more. They were further divided into mild smokers (< 20 and < 30 pack-years for women and men, respectively) and severe smokers (≥ 20 and ≥ 30 pack-years for women and men, respectively)21. Alcohol abusers were defined individuals who regularly consumed one or more drink per day for more than six months22. Physically active individuals were defined as those who exercised at least 30 min for at least 3 times a week. In addition, the aMAP and GALADUS scores were calculated as follows: (1) aMAP score = {[0.06 × Age (years) + 0.89 × Sex (Male:1, Female:0) + 0.48 × ALBI − 0.01 × platelets (103/mm3)] + 7.4} / 14.77 × 100, ALBI = [log10 bilirubin (µmol/L) × 0.66] + [albumin (g/L) × −0.085]8; (2) GALADUS score= -12.79 + 0.09 × Age (years) + 1.74 × Sex (Male:1, Female:0) + 2.44 × log10 (AFP) + 0.04 × AFP-L3% + 1.39 × log10 (DCP) + 3.56 × US (positive:1, negative:0)11 ; (3) referral rate = (number of patients with positive screening who need to be referred for diagnosis / number of patients participated in liver screening) ×100%; (4) detection rate = (number of patients diagnosed with liver cancer / number of patients participated in liver screening) × 100%; (5) early diagnosis rate = (number of patients diagnosed with very early or early stage liver cancer / number of patients diagnosed with live cancer) × 100%; and (6) NNR = number of patients with positive screening who need to be referred for diagnosis / number of patients diagnosed with liver cancer.
Statistical analysis
Categorical variables were presented as frequencies with percentages, after which differences among the groups were analyzed using the Chi-square or Fisher’s exact test for unordered categorical variables (i.e., the majority of demographic characteristics and US) and the Chi-square trend test for ordered categorical variables (i.e., the levels of age, AFP, AFP-L3% and DCP). Quantitative variables, such as AFP, AFP-L3%, DCP, and GALADUS, were presented as median and interquartile range (IQR) and analyzed using the Mann–Whiney U test. The screening protocol was evaluated by creating a receiver-operating characteristic (ROC) curve and calculating the area under the curve (AUC), sensitivity (Se), specificity (Sp), positive predictive value (PPV), and negative predictive value (NPV). The DeLong test was used to compare differences among ROC curves, and improvements in the screening model after adding indicators were presented using the net reclassification improvement index (NRI). After determining the optimal aMAP score cutoff threshold for liver cancer risk stratification based on the ROC curve, the paired Chi-square test was used to compare sensitivity, specificity, referral rates, and detection rates of different screening protocols. Statistical analysis and plotting were performed using SPSS version 24 (IBM, Chicago, IL, USA) and R version 4.0.5 software (R-project, Vienna, Austria). For all tests, a two-tailed P < 0.05 indicated statistical significance unless there were multiple comparisons requiring correction via the Bonferroni method.
Data availability
The data used in this study are not publicly available, but can contact Professor Jiang jing (jiangjing19702000@jlu.edu.cn) for more information.
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249. https://doi.org/10.3322/caac.21660 (2021).
Rumgay, H. et al. Global burden of primary liver cancer in 2020 and predictions to 2040. J. Hepatol. 77, 1598–1606. https://doi.org/10.1016/j.jhep.2022.08.021 (2022).
Zheng, R. et al. Cancer incidence and mortality in China, 2016. J. Natl. Cancer Cent. 2 1–9. https://doi.org/10.1016/j.jncc.2022.02.002 (2022).
Zeng, H. et al. Changing cancer survival in China during 2003-15: a pooled analysis of 17 population-based cancer registries. Lancet Glob Health. 6, e555–e567. https://doi.org/10.1016/S2214-109X(18)30127-X (2018).
Allaire, M. & Nault, J. C. Advances in management of hepatocellular carcinoma. Curr. Opin. Oncol. 29, 288–295. https://doi.org/10.1097/CCO.0000000000000378 (2017).
Lee, Y. T., Fujiwara, N., Yang, J. D. & Hoshida, Y. Risk stratification and early detection biomarkers for precision HCC screening. Hepatology https://doi.org/10.1002/hep.32779 (2022).
Singal, A. G. et al. International liver cancer association (ILCA) white paper on hepatocellular carcinoma risk stratification and surveillance. J. Hepatol. https://doi.org/10.1016/j.jhep.2023.02.022 (2023).
Fan, R. et al. aMAP risk score predicts hepatocellular carcinoma development in patients with chronic hepatitis. J. Hepatol. 73, 1368–1378. https://doi.org/10.1016/j.jhep.2020.07.025 (2020).
Kudo, M. Japan’s successful model of nationwide hepatocellular carcinoma surveillance highlighting the urgent need for global surveillance. Liver Cancer. 1, 141–143. https://doi.org/10.1159/000342749 (2012).
Oeda, S. et al. Optimal Follow-up of patients with viral hepatitis improves the detection of Early-stage hepatocellular carcinoma and the prognosis of survival. Intern. Med. 55, 2749–2758. https://doi.org/10.2169/internalmedicine.55.6730 (2016).
Yang, J. D. et al. GALAD score for hepatocellular carcinoma detection in comparison with liver ultrasound and proposal of GALADUS score. Cancer Epidemiol. Biomarkers Prev. 28, 531–538. https://doi.org/10.1158/1055-9965.EPI-18-0281 (2019).
Pepe, M. S. et al. Phases of biomarker development for early detection of cancer. J. Natl. Cancer Inst. 93, 1054–1061. https://doi.org/10.1093/jnci/93.14.1054 (2001).
Lansdorp-Vogelaar, I. et al. Risk-stratified strategies in population screening for colorectal cancer. Int. J. Cancer. 150, 397–405. https://doi.org/10.1002/ijc.33784 (2022).
Clift, A. K. et al. The current status of risk-stratified breast screening. Br. J. Cancer. 126, 533–550. https://doi.org/10.1038/s41416-021-01550-3 (2022).
Hendrix, N. et al. Clarifying the trade-offs of risk-stratified screening for prostate cancer: A cost-effectiveness study. Am. J. Epidemiol. 190, 2064–2074. https://doi.org/10.1093/aje/kwab155 (2021).
Sterling, R. K. et al. Utility of lens culinaris agglutinin-reactive fraction of alpha-fetoprotein and des-gamma-carboxy prothrombin, alone or in combination, as biomarkers for hepatocellular carcinoma. Clin. Gastroenterol. Hepatol. 7, 104–113. https://doi.org/10.1016/j.cgh.2008.08.041 (2009).
Zhou, J. M., Wang, T. & Zhang, K. H. AFP-L3 for the diagnosis of early hepatocellular carcinoma: A meta-analysis. Med. (Baltim). 100, e27673. https://doi.org/10.1097/MD.0000000000027673 (2021).
Feng, H., Li, B., Li, Z., Wei, Q. & Ren, L. PIVKA-II serves as a potential biomarker that complements AFP for the diagnosis of hepatocellular carcinoma. BMC Cancer. 21, 401. https://doi.org/10.1186/s12885-021-08138-3 (2021).
Song, P. et al. Clinical utility of simultaneous measurement of alpha-fetoprotein and des-gamma-carboxy prothrombin for diagnosis of patients with hepatocellular carcinoma in China: A multi-center case-controlled study of 1,153 subjects. Biosci. Trends. 8, 266–273. https://doi.org/10.5582/bst.2014.01116 (2014).
Liu, S. et al. Diagnostic performance of AFP, AFP-L3, or PIVKA-II for hepatitis C virus-associated hepatocellular carcinoma: A multicenter analysis. J. Clin. Med. 11 https://doi.org/10.3390/jcm11175075 (2022).
Li, X. et al. Association between tea consumption and risk of cancer: a prospective cohort study of 0.5 million Chinese adults. Eur. J. Epidemiol. 34, 753–763. https://doi.org/10.1007/s10654-019-00530-5 (2019).
Otani, T. et al. Alcohol consumption, smoking, and subsequent risk of colorectal cancer in middle-aged and elderly Japanese men and women: Japan public health Center-based prospective study. Cancer Epidemiol. Biomarkers Prev. 12, 1492–1500 (2003).
Acknowledgements
The authors would like to thank the participants of the liver cancer screening for providing their individualized information and a blood sample.
Funding
This study was supported by the National Natural Science Foundation of China (No.82204116) and the Scientific and Technological Development Program of Jilin Province (No.20200403098SF).
Author information
Authors and Affiliations
Contributions
Study concept: HH, JJ. Study design: HH, YW, ZJ, YZ, YP, JJ. Acquisition of data: HH, YW, ZJ, KS, YC, YS, DL, HL, JY, YW. Resources: all authors. Statistical analysis: HH, JJ. Drafting manuscript: HH, JJ. Critical revision of manuscript: all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
He, H., Wu, Y., Jia, Z. et al. A stratified precision screening strategy for enhancing hepatitis B- and C-associated liver cancer detection: a prospective study. Sci Rep 15, 11396 (2025). https://doi.org/10.1038/s41598-025-95795-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-95795-0