Abstract
This study aims to compare the effectiveness and safety of surgical interventions for secondary glaucoma following penetrating keratoplasty (PKP) through a network meta-analysis. PubMed, Cochrane Central Register of Controlled Trials, Embase, Web of Science, Scopus, and Google Scholar databases were searched for articles published from inception to July 16, 2024. We included studies that compared the effects of trabeculectomy, glaucoma drainage devices (GDDs), or cyclophotocoagulation (CPC) on graft failure, intraocular pressure (IOP), or the number of glaucoma medications required for patients with post-PKP glaucoma. Based on data synthesized from 13 cohort studies, we observed that valved GDDs are not associated with a higher risk of graft failure (risk ratio [RR], 1.49; 95% confidence interval [CI], 0.89 to 2.48) compared with trabeculectomy with antifibrotic agents. Valved GDDs demonstrated greater benefits in achieving successful IOP control (RR, 1.33; 95% CI, 1.08 to 1.64), reducing IOP from baseline (mean difference [MD], − 3.08; 95% CI, − 5.36 to − 0.81), and reducing the number of glaucoma medication usage from baseline (MD, − 0.53; 95% CI, − 0.94 to − 0.12) compared with trabeculectomy with antifibrotic agents. In conclusion, valved GDDs may be the preferred surgical option for managing post-PKP glaucoma.
Similar content being viewed by others
Introduction
Secondary glaucoma is a critical complication of corneal transplantation and the second most common cause of graft failure1. The incidence of post-keratoplasty glaucoma ranges from 10 to 35%2, indicating that aggressive control of intraocular pressure (IOP) after corneal transplantation is essential. However, it is often challenging due to postoperative inflammation, iridocorneal angle decrease, peripheral anterior synechiae formation, severance of elastic fibers, and steroid use3,4,5,6. Among various keratoplasty techniques, penetrating keratoplasty (PKP) demonstrates a higher incidence of secondary glaucoma compared with Descemet’s membrane endothelial keratoplasty (DMEK), Descemet stripping automated endothelial keratoplasty (DSAEK), and deep anterior lamellar keratoplasty (DALK)7. Given the potentially high incidence and severe complications associated with elevated IOP after PKP, active IOP control is imperative.
In patients with an elevated IOP refractory to medical treatment, early surgical intervention is necessary. Recent review articles2,8 provided suggestions for selecting the optimal surgery. However, these conclusions were predominantly drawn from studies with small sample sizes, single-arm designs, and variable outcomes. Each study utilized different criteria to evaluate the postoperative outcomes, such as graft failure rates, extent of IOP reduction, and decrease in the amount of medication used for managing glaucoma. Therefore, determining the most suitable treatment from options such as trabeculectomy with or without antifibrotic agents, valved or non-valved glaucoma drainage devices (GDDs), and cyclophotocoagulation (CPC) remains challenging. Therefore, a comprehensive assessment that incorporates all available evidence and compares existing surgical options is urgently needed.
We conducted a comprehensive network meta-analysis of cohort studies to examine the effects of trabeculectomy, GDDs, and CPC on patients with secondary glaucoma after PKP. Our outcomes include graft failure, successful IOP control, IOP change, and change in number of glaucoma medications required. By analyzing the findings of existing studies, we aimed to guide ophthalmologists in selecting the preferred intervention for patients with secondary glaucoma following PKP.
Materials and methods
Study design
This systematic review and network meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) 2020 guidelines, PRISMA Extension Statement9, and Meta-analyses of Observational Studies in Epidemiology (MOOSE) guidelines10. The checklists are provided in Supplementary Table S1–S4. The predefined study protocol was officially published in the International Prospective Register of Systematic Reviews (PROSPERO) under the registration number CRD4202452742411.
Data sources and search strategy
We searched PubMed, the Cochrane Central Register of Controlled Trials, Embase, Web of Science, Scopus, and Google Scholar databases from inception to July 16, 2024. Our search strategy is outlined in Supplementary Table S5, with keywords verified by an expert librarian.
Study selection
The inclusion criteria were as follows: (i) studies comprising individuals diagnosed with glaucoma after PKP; (ii) studies including trabeculectomy with or without antifibrotic agents (such as mitomycin C or 5-fluorouracil), valved or non-valved GDDs, CPC, Ex-PRESS mini shunt, XEN gel stent, or iStent as the intervention group; (iii) studies incorporating another surgical option or medical treatment as the comparison group; (iv) studies evaluating graft failure, IOP, and the number of glaucoma medications used as outcomes.
The exclusion criteria were as follows: (i) studies that investigated the combined effects of multiple treatments; (ii) studies that did not describe the surgical intervention employed, including the use of antifibrotic agents in trabeculectomy or the type of GDD implanted; and (iii) case-control studies, cross-sectional trials, crossover studies, single-arm trials, case series, or case reports. No language or publication time frame restrictions were applied. For studies not published in English or Chinese, we used Google Translate to interpret the content.
Two reviewers (SYP and CCC) independently screened each study for eligibility. Discrepancies were resolved through discussions or by consulting another reviewer (YYC) for suggestion.
Outcomes
The outcomes of this study were (i) occurrence of graft failure, generally defined as the presence of corneal edema; (ii) successful control of IOP level at the end of follow-up, defined as an IOP of ≤ 21 mmHg regardless of concurrent medication usage; (iii) IOP reduction from baseline, calculated by subtracting the baseline IOP value from the follow-up IOP value; and (iv) reduction in the number of glaucoma medications used from baseline, calculated by subtracting the baseline from the follow-up number of glaucoma medications used.
Quality assessment
We assessed the quality of each included cohort study using the Risk Of Bias In Non-randomized Studies - of Interventions (ROBINS-I) tool12. Reporting bias, indirectness, imprecision, heterogeneity, and incoherence were evaluated using the web-based application, Confidence in Network Meta-Analysis (CINeMA)13. Two reviewers (SYP and CCC) independently evaluated the quality of each study, with disagreements resolved through discussion or guidance from another reviewer (YYC).
Data extraction, synthesis, and analysis
Two reviewers (SYP and CCC) recorded and verified the data from the included studies. The number of eyes with an event was extracted as binary outcomes, while the mean and standard deviation (SD) were recorded as continuous outcomes. If a specific outcome was observed across all individuals in a particular treatment group, adjustments were made by increasing the total sample size by 1 and adding 0.5 to the number of events within each group. This adjustment was performed to prevent the occurrence of a negative variance in the treatment arm, which could impede the analysis. In studies that did not present the results of changes in SD from baseline, we computed this value using paired t-tests in SAS 9.4 if raw data were available. If raw data were unavailable, we calculated this value using the standard error, 95% confidence interval (CI), or baseline and final SD values based on previous recommendations14,15. When studies only provided their analytical results in figures, the online application “WebPlotDigitizer” (https://automeris.io/WebPlotDigitizer) was utilized to extract the numerical data.
A frequentist network meta-analysis was performed to evaluate the comparative effectiveness of different surgical options. The combination treatment of trabeculectomy and antifibrotic agents was assigned as the reference group as it was the recommended first-line surgical treatment in previous review articles2,8,16. Effect estimates were presented as risk ratios (RRs) for binary outcomes and mean differences (MDs) for continuous outcomes, each with a corresponding 95% CI. We also performed a subgroup analysis by stratifying follow-up durations into < 4 years and ≥ 4 years. The follow-up duration for each study was determined based on the longest follow-up period reported.
Data were synthesized using the random-effects model. The “netmeta” package17 in R (version 4.3.1) was used to perform the frequentist network meta-analysis. Ranking probabilities for treatment options were reported using p-scores, where a higher p-score indicates a more favorable treatment option. Publication bias was assessed by visual inspection of funnel plot asymmetry, using the functions incorporated in the “netmeta” package17 in R (version 4.3.1).
Meta-regression analysis
To evaluate the potential impact of confounders on our outcomes, we conducted a meta-regression analysis. The covariates in the meta-regression included age, sex, follow-up duration, time from PKP to surgery, preoperative IOP, and the number of preoperative glaucoma medications used. A p-value < 0.05 suggests that the covariates do not significantly influence the outcomes.
Sensitivity analysis
Sensitivity analysis was performed to evaluate the effect of cohort studies that resembles case series designs. Among the studies we included, two were classified as case series by the authors18,19. However, we still considered them as cohort studies and included them in the analysis, as they are both comparative instead of non-comparative studies. We conducted a sensitivity analysis by excluding these two studies. We also conducted another sensitivity analysis including only studies that reported outcome events based on the number of patients rather than the number of eyes.
Additionally, we performed a Bayesian network meta-analysis using the same studies and outcomes due to small sample sizes and high heterogeneity across studies. Models were fitted using the Markov chain Monte Carlo (MCMC) algorithm. The analysis was conducted using four chains, 5000 burn-in iterations, 100,000 actual simulation iterations, and a thin of 10. A random effects model was applied. Empirical informative prior distributions for heterogeneity were used, with semi-objective chosen for outcome type and non-pharmacological comparison chosen for comparison type. Results were presented as RRs for binary outcomes and MDs for continuous outcomes, along with 95% credible intervals (CrIs). The Surface Under the Cumulative RAnking (SUCRA) score, indicating the ranking probability of different surgical interventions, was also computed.
Convergence was assessed using trace plots, density plots, and Gelman-Rubin-Brooks plots, with an overall Potential Scale Reduction Factor (PSRF) < 1.05 indicating good convergence. Inconsistency was evaluated using the nodesplit method, where a p-value < 0.05 for the difference between direct and indirect effect estimates suggesting the presence of inconsistency.
Results
Literature search
We retrieved 3682 studies from the databases. After sequential screening of title, abstract, and full texts, 13 cohort studies were included in the final analysis18,19,20,21,22,23,24,25,26,27,28,29,30. The eligible studies consisted of a combined sample size of 562 eyes, which compared the effects of trabeculectomy with or without antifibrotic agents, valved or non-valved GDDs, CPC, Ex-PRESS mini shunt, and medical treatment. Among the eligible studies, mitomycin C was the most commonly used antifibrotic agent, the Ahmed glaucoma valve implant was the most used valved GDD, and the Baerveldt glaucoma implant was the most used non-valved GDD. The study selection process is provided in Supplementary Figure S1, which was constructed based on the PRISMA 2020 Flow Diagram9. The baseline characteristics of eligible studies (Supplementary Table S6), definition of outcomes (Supplementary Table S7), and outcome data (Supplementary Table S8) are also included in the supplementary material. Given the lack of studies exploring the effects of XEN gel stent or iStent in managing post-keratoplasty glaucoma, we could not include them in our analysis.
Quality assessment results
In the risk of bias assessment for each cohort study using ROBINS-I, all studies were classified as having a “low risk of bias” or “moderate risk of bias” (Supplementary Figure S2). Studies rated as having a “moderate risk of bias” primarily lacked adequate reporting of baseline characteristics for each intervention group or failed to present the handling of missing data. However, instances of bias due to deviations from the intended interventions or improper measurement of outcomes rarely occurred. The evaluation of within-study bias, reporting bias, indirectness, imprecision, heterogeneity, and incoherence using CINeMA are presented in Supplementary Table S9.
Network meta-analysis results
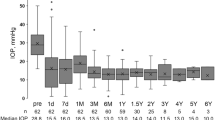
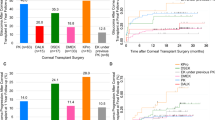
Nine studies reported the findings of graft failures (Fig. 1A). The results suggest that the graft failure risk of valved GDDs (RR, 1.49; 95% CI, 0.89 to 2.48), non-valved GDDs (RR, 1.06; 05% CI, 0.54 to 2.06), and CPC (RR, 1.64; 95% CI, 0.95 to 2.83) is comparable to that of the trabeculectomy surgery with antifibrotic agents. Medication (RR, 2.29; 95% CI, 1.30 to 4.01) and trabeculectomy without antifibrotic agents (RR, 1.73; 95% CI, 1.08 to 2.77) significantly increase the risk of graft failure compared with trabeculectomy with antifibrotic agents (Fig. 2A). Considering the risk of graft failure, values for ranking probabilities indicate that trabeculectomy with antifibrotic agents is the best treatment option (Supplementary Figure S3A).
Network plots. Network plots visualize the available treatment comparisons within the included studies for each outcome. The diameter of each node reflects the corresponding sample size, whereas the thickness of each connecting line is proportional to the number of participants. GDD glaucoma drainage device, CPC cyclophotocoagulation.
Forest plots. Forest plots depict the results of the network meta-analysis for each outcome. We determined TRBC with antifibrotics as the reference treatment. The blocks in the figures represent either risk ratios or mean differences, with error bars denoting the 95% confidence intervals. The treatments in each plot were arranged based on their p-scores, which indicate treatment rankings. CI confidence interval, IOP intraocular pressure, TRBC trabeculectomy, GDD glaucoma drainage device, CPC cyclophotocoagulation.
Eight studies reported the number of patients with successful control of IOP (Fig. 1B). Compared with trabeculectomy with antifibrotic agents, valved GDDs demonstrate a significant favorable effect (RR, 1.33; 95% CI, 1.08 to 1.64), non-valved GDDs exhibit a non-significant effect (RR, 1.24; 95% CI, 0.92 to 1.66), while trabeculectomy without antifibrotic agents are significantly ineffective (RR, 0.34; 95% CI, 0.12 to 0.97) compared to trabeculectomy with antifibrotics (Fig. 2B). According to ranking probabilities, valved GDDs are the most effective treatment for successful IOP control, followed by non-valved GDDs (Supplementary Figure S3B).
The findings from nine studies investigating the reduction of IOP from baseline (Fig. 1C) report that valved GDDs are significantly effective (MD, − 3.08; 95% CI, − 5.36 to − 0.81), while trabeculectomy without antifibrotic agents are significantly ineffective (MD, 3.86; 95% CI, 0.41 to 7.32) compared with trabeculectomy with antifibrotic agents (Fig. 2C). Ranking probability values demonstrate that valved GDDs are the optimal treatment for reducing the change in IOP from baseline (Supplementary Figure S3C).
The change in the number of glaucoma medications used was assessed in six studies (Fig. 1D). The synthesized results reveal that valved GDDs (MD, − 0.53; 95% CI, − 0.94 to − 0.12), non-valved GDDs (MD, − 0.83; 95% CI, − 1.51 to − 0.15), and CPC (MD, − 0.90; 95% CI, − 1.64 to − 0.16) are significantly superior to trabeculectomy with antifibrotic agents in reducing the number of glaucoma medications required. Trabeculectomy without antifibrotic agents (MD, 0.85; 95% CI, 0.26 to 1.44) is significantly less effective in reducing the number of glaucoma medications required compared to trabeculectomy with antifibrotic agents (Fig. 2D). According to ranking probabilities, CPC is the best treatment option for reducing the number of glaucoma medications used from baseline, followed by non-valved GDDs in second place and valved GDDs in third (Supplementary Figure S3D). The results of each pairwise treatment comparison are presented in Table 1. Funnel plots are shown in Supplementary Figure S4.
The subgroup analysis, which stratified studies by follow-up duration, shows that studies with follow-up < 4 years yield results that are mostly consistent with those of the main analysis. However, there are no statistically significant differences between trabeculectomy without antifibrotics and trabeculectomy with antifibrotics in terms of change in IOP (MD, 2.49; 95% CI, −0.80 to 5.78) or change in number of glaucoma medications required (MD, 1.00; 95% CI, −0.17 to 2.17). Additionally, no statistically significant differences were found between trabeculectomy with antifibrotics and any other treatment options for follow-up durations ≥ 4 years (Supplementary Figure S5).
Meta-regression results
The meta-regression analysis shows no significant association of age, sex, follow-up duration, time from PKP to surgery, preoperative IOP, or the use of preoperative glaucoma medications with the risk of graft failure or successful IOP control (all p-value ≥ 0.05). Supplementary Table S10 outlines the detailed results of the meta-regression analysis.
Sensitivity analysis results
Sensitivity analysis indicates that after excluding studies resembling case series designs18,19, the results are generally unaffected (Supplementary Figure S6a). Another sensitivity analysis demonstrates that after excluding studies evaluating outcomes in eyes instead of patients, valved GDDs remain the optimal choice (Supplementary Figure S6b).
The comparative effectiveness of different treatment options on graft failure, successful IOP control, change in IOP from baseline, and change in number of glaucoma medications were re-assessed using Bayesian network meta-analysis. Supplementary Table S11 outlines the results of each pairwise comparison for each treatment arm. No significant difference in graft failure risk is observed between trabeculectomy with antifibrotic agents and other surgical interventions. Compared to trabeculectomy with antifibrotics, valved GDDs show a trend toward improved successful IOP control (HR, 1.37; 95% CrI, 0.99 to 1.96), while trabeculectomy without antifibrotics is associated with a significantly lower likelihood of successful IOP control (HR, 0.31; 95% CrI, 0.07 to 0.78). The findings for IOP change from baseline align with those from the main analysis, showing that valved GDDs achieve a significantly greater reduction in IOP compared to trabeculectomy with antifibrotics (MD, −3.26; 95% CrI, −5.14 to −1.28). Likewise, the evaluation of changes in the number of glaucoma medications from baseline follows a similar trend to the main analysis; however, no statistically significant differences are observed between trabeculectomy with antifibrotics and valved GDDs, non-valved GDDs, or CPC (Supplementary Figure S6c). Ranking probabilities assessed through the Bayesian approach are generally consistent with the findings of the frequentist network meta-analysis (Supplementary Figure S6d).
Convergence of our MCMC simulation was confirmed through trace plots exhibiting stable estimates across the four chains, density plots of posterior effect size estimates following a normal distribution, and the overall PSRF in the Gelman-Rubin-Brooks plots gradually decreasing to below 1.05 (Supplementary Figure S6e). No evidence of inconsistency was observed between direct and indirect estimates, as no p-value was below 0.05 for any outcome (Supplementary Figure S6f).
Discussion
This network meta-analysis using the frequentist approach contributes to our understanding of the preferred surgical approach for post-PKP glaucoma. Our study has three major findings: (i) the risk of graft failure is similar across valved GDDs, non-valved GDDs, and trabeculectomy with antifibrotic agents; (ii) valved GDDs demonstrate superior effectiveness in controlling IOP compared with trabeculectomy with antifibrotic agents; and (iii) valved and non-valved GDDs exhibit greater reduction in the number of glaucoma medications used compared to trabeculectomy with antifibrotic agents. Although the effectiveness of valved GDDs in reducing the number of glaucoma medications is not evident when analyzed using a Bayesian model, a trend still suggests that valved GDDs may be superior to trabeculectomy with antifibrotics for this outcome. In summary, we observed that valved GDDs exhibit a favorable effect on most key outcomes compared with trabeculectomy with antifibrotic agents.
Association of valved GDDs with the risk of graft failure
Our analysis suggests that the risk of graft failure is similar across valved GDDs, non-valved GDDs, CPC, and trabeculectomy with antifibrotic agents. These findings are consistent across studies with follow-up durations of either < 4 years or ≥ 4 years. By contrast, previous studies have reported that GDDs are associated with a high risk of graft failure2,8. These discrepancies may suggest that, although GDDs show a slightly higher risk of graft failure, the difference is not statistically significant. The absence of graft failure risk associated with valved GDDs may be attributed to their effectiveness in controlling IOP and minimizing the risk of hypotony. Maintaining a well-controlled IOP could potentially preserve the endothelial cells in the corneal graft31,32, thereby reducing the risk of corneal graft failure. Therefore, our findings suggest that valved GDDs may emerge as a preferred surgical approach for the management of post-PKP glaucoma.
Effectiveness of valved GDDs in IOP reduction
In our study, valved GDDs are significantly superior to trabeculectomy with antifibrotic agents in controlling IOP and reducing the number of required glaucoma drugs. The effectiveness of GDDs in reducing IOP has also been observed in a previous meta-analysis33. Compared with patients undergoing trabeculectomy, those receiving GDD implantations exhibit less postoperative inflammation and scarring of the sclera and conjunctiva, which are factors that may induce inadequate aqueous humor drainage34. Moreover, both the tube and plate of GDDs can facilitate efficient aqueous humor passage, ensuring a sustained outflow tract for fluid drainage even in the presence of fibrosis around the tube35.
Additionally, while previous studies have suggested that non-valved GDDs demonstrated a greater reduction in IOP levels compared with valved GDDs in patients with primary glaucoma36,37, this pattern was not observed in our study. We hypothesize that this discrepancy may be resulted from inflammatory cells produced after corneal transplantation surgery, which may easily migrate to the subconjunctival area through the non-valved GDD tubes38. Compared to valved GDDs, non-valved GDDs might lead to increased relocation of inflammatory cells to the subconjunctival space due to the absence of a valve, particularly shortly after surgery when inflammation is most active. This may result in increased scar formation and subsequent obstruction of the tubes in non-valved GDDs. However, further studies are needed to validate this hypothesis.
Our subgroup analysis reveals no significant differences between valved GDDs and trabeculectomy with antifibrotics in achieving successful IOP control in studies with follow-up durations ≥ 4 years. This finding may indicate that the long-term effectiveness of valved GDDs gradually diminishes due to the fibrous reaction around the bleb and end-plate39. Studies examining the IOP-lowering effect of valved GDDs have also observed that as follow-up durations increased, success rates gradually declined within the same population40,41.
Valved GDDs as the preferred surgical approach
Given the excellent effectiveness of valved GDDs in controlling IOP, reducing the need for glaucoma medications, and minimizing the risk of graft failure, valved GDDs may be considered the preferred surgical approach over trabeculectomy with antifibrotic agents for patients with post-PKP glaucoma.
Performing trabeculectomy before implanting GDDs may be unfavorable owing to its potential to cause conjunctival fibrosis and scar formation38. This pathological change can negatively affect GDD implantation by complicating the insertion process, causing inadequate tube coverage, and inducing plate encapsulation42. Prioritizing valved GDDs over trabeculectomy may improve surgical success and postoperative prognosis in patients with post-PKP glaucoma.
Use of antifibrotic agents in performing trabeculectomy
In addition to highlighting the remarkable effectiveness and safety of valved GDDs, our findings suggest that trabeculectomy without antifibrotic agents is ineffective and poses safety concerns. Unlike trabeculectomy with antifibrotic agents, trabeculectomy without antifibrotic agents is linked to a significantly higher risk of graft failure, lesser reduction in IOP, and a larger number of glaucoma medications required. This finding aligns with the longstanding practice of applying antifibrotic agents such as mitomycin C during glaucoma filtration surgery43. Antifibrotic agents can reduce fibrotic reactions, prevent subconjunctival scarring, and enhance functional bleb formation44,45. Therefore, conducting trabeculectomy without the use of antifibrotic agents should be avoided.
Limitations and strengths
Our study has several limitations. First, we predominantly included cohort studies due to the absence of identified randomized controlled trials (RCTs). This may arise from the inherent challenges in executing double-blinded RCTs in surgical settings, which is a typical limitation in this domain46. Although these cohort studies provide the most reliable evidence under these constraints, they lack the controlled conditions for RCTs. Therefore, RCTs should be conducted in the future. Second, the eligible cohort studies are all retrospectively designed, with some resembling a case series design. This may arise from the smaller number of individuals with post-PKP glaucoma, which complicates the planning of cohort studies. Third, the number of studies included and the sample size are relatively low, especially for less investigated comparisons such as Ex-PRESS mini shunt. These conditions may arise from the lack of investigations into this particular topic, highlighting the need for more future studies to address this issue. Fourth, there are inconsistent results between the frequentist and Bayesian network meta-analysis regarding the effectiveness of valved GDDs in achieving successful IOP control and reducing the number of glaucoma medications required. Further studies are needed to clarify whether valved GDDs provide benefits across these outcomes. Fifth, although we attempted to assess other outcomes such as visual acuity improvement, endothelial cell density, hypotony, or other postoperative complications, the available data were too limited to enable analysis. We thereby propose that future studies could include these crucial prognostic parameters into evaluation.
Despite these limitations, our study has several strengths. First, our findings reveal the potential benefits of valved GDDs, which are of great importance to ophthalmologists in selecting the preferred surgical techniques for post-PKP glaucoma. Second, by synthesizing the available studies with standard statistical methods, we offer evidence on this topic based on a larger sample size than individual studies. Third, this network meta-analysis is the first of our knowledge to compare different surgical approaches for post-PKP glaucoma, thereby enhancing the current understanding in this field and paving the way for future research.
Future research
Recent studies have introduced the potential benefits of mini shunts, such as the Ex-PRESS shunt, or devices for microinvasive glaucoma surgeries (MIGS), such as XEN gel stent or iStent47 in glaucoma management. The Ex-PRESS mini shunt has been proposed as a viable option for post-keratoplasty glaucoma owing to its smaller and thinner design compared with GDDs, which may provide greater stability and reduce postoperative inflammation2,42. Several studies had investigated the outcomes in patients with post-keratoplasty glaucoma who underwent Ex-PRESS mini shunt implantation. Ledesma-Gil et al. conducted a prospective study with 17 participants and observed that mean IOP changed from 35.94 mmHg to 12.76 mmHg after Ex-PRESS mini shunt insertion48. Ates et al. collected 15 cases and found that after Ex-PRESS mini shunt implantation, the mean IOP decreased from 41.46 mmHg to 12.06 mmHg, 93% of patients successfully achieved IOP below 21 mmHg, the average number of glaucoma medications required reduced from 3.20 to 0.26, and all grafts that were clear preoperatively remained clear postoperatively49. Other studies indicated that XEN stent is also a promising option for post-keratoplasty glaucoma management. In a case series of 14 eyes analyzed by Oliver et al., IOP decreased from 32.7 mmHg to 12.5 mmHg and number of glaucoma medications required reduced from 4.0 to 0.4 after XEN stent implantation50. Although the Ex-PRESS mini shunt and XEN stent appear to be viable alternative treatments for secondary glaucoma following keratoplasty, we could not include studies with a non-comparative design in our analysis, which hinders effective statistical comparison. Therefore, future studies should investigate this area further.
Among the eligible studies, CPC was used less frequently compared with trabeculectomy and GDDs for post-PKP glaucoma, possibly due to the relatively serious complication from CPC. In recent years, micropulse transscleral CPC (MP-TSCPC) has been introduced as a novel option in treating refractory glaucoma51,52. Several single-arm studies had demonstrated that MP-TSCPC was effective in controlling IOP and caused minimal complications when applied to patients with post-keratoplasty glaucoma53,54,55,56. In studies comparing the effects of MP-TSCPC and traditional continuous wave transscleral CPC (CW-TSCPC), the post-keratoplasty glaucoma group was seldom investigated. However, research conducted on patients with open-angle glaucoma and refractory glaucoma demonstrated favorable results. Compared with CW-TSCPC, MP-TSCPC was more effective in reducing IOP and carried a lower complication rate57,58. This may be explained by the ON/OFF cycles of MP-TSCPC. The OFF cycle represents the thermal relaxation time, which prevents temperature rise and result in less tissue destruction compared to the continuous mode59.
Additionally, the Paul Glaucoma Implant (PGI), a novel non-valved GDD, has demonstrated comparable effectiveness in reducing IOP levels and the risk of postoperative hypotony in patients with glaucoma compared with other GDDs60. However, studies on the PGI are limited, particularly regarding its application in patients with post-keratoplasty glaucoma. Therefore, we suggest that future studies explore the potential efficacy of PGI in patients with post-keratoplasty glaucoma.
Conclusions
Compared with trabeculectomy with antifibrotic agents, valved GDDs may be more effective in controlling IOP and preserving the corneal grafts. Therefore, we propose that valved GDDs may be the preferred surgical approach for the treatment of post-PKP glaucoma.
Data availability
The data supporting the findings of this study are available from the corresponding author under reasonable request.
Abbreviations
- CI:
-
Confidence interval
- CPC:
-
Cyclophotocoagulation
- DALK:
-
Deep anterior lamellar keratoplasty
- DMEK:
-
Descemets membrane endothelial keratoplasty
- DSAEK:
-
Descemets stripping automated endothelial keratoplasty
- GDD:
-
Glaucoma drainage devices
- IOP:
-
Intraocular pressure
- MD:
-
Mean difference
- MIGS:
-
Microinvasive glaucoma surgeries
- MOOSE:
-
Meta-analyses of observational studies in epidemiology
- MP-TSCPC:
-
Micropulse transscleral cyclophotocoagulation
- PGI:
-
Paul glaucoma implant
- PKP:
-
Penetrating keratoplasty
- PRISMA:
-
Preferred Reporting Items for Systematic Reviews and Meta-analyses
- ROBINS-I:
-
Risk of bias in non-randomized studies-of interventions
- RR:
-
Risk ratio
- SD:
-
Standard deviation
- TSCPC:
-
Transscleral cyclophotocoagulation
References
Williams, K. A. et al. Risk factors for human corneal graft failure within the Australian corneal graft registry. Transplantation 86, 1720–1724 (2008).
Abdelghany, A. A., D’Oria, F. & Alio, J. L. Surgery for glaucoma in modern corneal graft procedures. Surv. Ophthalmol. 66, 276–289 (2021).
Zimmerman, T., Olson, R., Waltman, S. & Kaufman, H. Transplant size and elevated intraocular pressure. Postkeratoplasty Arch. Ophthalmol. 96, 2231–2233 (1978).
Kersey, J. P. & Broadway, D. C. Corticosteroid-induced glaucoma: A review of the literature. Eye 20, 407–416 (2006).
Dada, T. et al. Post-penetrating keratoplasty glaucoma. Indian J. Ophthalmol. 56, 269–277 (2008).
Lewis, P. N. et al. Three-dimensional arrangement of elastic fibers in the human corneal stroma. Exp. Eye Res. 146, 43–53 (2016).
Saini, C. et al. Incidence and risk factors for glaucoma development and progression after corneal transplantation. Eye 37, 2117–2125 (2023).
Baltaziak, M., Chew, H. F. & Podbielski, D. W. Ahmed, I. I. K. Glaucoma after corneal replacement. Surv. Ophthalmol. 63, 135–148 (2018).
Page, M. J. et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Int. J. Surg. 88, 105906 (2021).
Stroup, D. F. et al. Meta-analysis of observational studies in epidemiology—A proposal for reporting. JAMA 283, 2008–2012 (2000).
Pan, S. Y. & Lu, Y. A. Comparison of surgical management in glaucoma following corneal transplantation. PROSPERO 2024 CRD42024527424 Available from: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42024527424.).
Sterne, J. A. et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355, i4919 (2016).
Nikolakopoulou, A. et al. CINeMA: An approach for assessing confidence in the results of a network meta-analysis. PLoS Med. 17, e1003082 (2020).
Higgins, J. et al. Chapter 6: Choosing effect measures and computing estimates of effect. In Cochrane Handbook for Systematic Reviews of Interventions version 6.4. J Higgins, Editors. Cochrane. Accessed 11 Sept 2023. (2023). Available from www.training.cochrane.org/handbook
Taylor, K. What can I do if I have a missing mean, standard deviation or sample size? Accessed September 11, (2023). Available from https://www.cebm.ox.ac.uk/resources/data-extraction-tips-meta-analysis/missing-mean-standard-deviation-or-sample-size
Ayyala, R. S. Penetrating keratoplasty and glaucoma. Surv. Ophthalmol. 45, 91–105 (2000).
Balduzzi, S. et al. Netmeta: An R package for network meta-analysis using frequentist methods. J. Stat. Softw. 106, 1–40 (2023).
Ayyala, R. S. et al. Comparison of mitomycin C trabeculectomy, glaucoma drainage device implantation, and laser neodymium: YAG cyclophotocoagulation in the management of intractable glaucoma after penetrating keratoplasty. Ophthalmology 105, 1550–1556 (1998).
Arroyave, C. P., Scott, I. U., Fantes, F. E., Feuer, W. J. & Murray, T. G. Corneal graft survival and intraocular pressure control after penetrating keratoplasty and glaucoma drainage device implantation. Ophthalmology 108, 1978–1985 (2001).
Akdemir, M. O., Acar, B. T., Kokturk, F. & Acar, S. Clinical outcomes of trabeculectomy vs. Ahmed glaucoma valve implantation in patients with penetrating keratoplasty. Int. Ophthalmol. 36, 541–546 (2016).
Elhofi, A. & Helaly, H. A. Graft survival after penetrating keratoplasty in cases of trabeculectomy versus ahmed valve implant. J. Ophthalmol. 2018, 9034964 (2018).
Ishioka, M. et al. Trabeculectomy with mitomycin C for post-keratoplasty glaucoma. Br. J. Ophthalmol. 84, 714–717 (2000).
Li, H., Ren, J., Dong, L., Yang, X. & Zhu, L. Comparison in clinical outcomes of trabeculectomy and Ahmed glaucoma valve implantation in patients with penetrating keratoplasty. Recent. Adv. Ophthalmol. 41, 149–153 (2021).
Oztutuncu, O. et al. Surgical management of glaucoma following different keratoplasty techniques. Int. Ophthalmol. 42, 2829–2840 (2022).
Purtskhvanidze, K. et al. Ten-Year outcome of glaucoma drainage device surgery after penetrating keratoplasty. J. Glaucoma. 30, e108–e113 (2021).
Raj, S. et al. Comparison of Short-term outcomes of aurolab aqueous drainage implant with Ahmed glaucoma valve in Post–Penetrating keratoplasty glaucoma: A retrospective Follow-up study at a tertiary care center. Ophthalmol. Glaucoma. 2, 172–177 (2019).
Sihota, R., Sharma, N., Panda, A., Aggarwal, H. C. & Singh, R. Post-penetrating keratoplasty glaucoma: Risk factors, management and visual outcome. Aust N Z. J. Ophthalmol. 26, 305–309 (1998).
Wy, S., Kim, Y. K., Jeoung, J. W. & Kim, M. K. Clinical outcomes in maximum tolerated medical therapy in penetrating keratoplasty for bullous keratopathy. Front. Med. 9, 810848 (2022).
Yakin, M. et al. Outcomes of trabeculectomy and glaucoma drainage devices for elevated intraocular pressure after penetrating keratoplasty. Cornea 37, 705–711 (2018).
Yildirim, N., Gursoy, H., Sahin, A., Ozer, A. & Colak, E. Glaucoma after penetrating keratoplasty: Incidence, risk factors, and management. J. Ophthalmol. 2011, 951294 (2011).
Liu, M. & Hong, J. Risk factors for endothelial decompensation after penetrating keratoplasty and its novel therapeutic strategies. J. Ophthalmol. 2018, 1389486 (2018).
Hollander, D. A. et al. Graft failure after penetrating keratoplasty in eyes with Ahmed valves. Am. J. Ophthalmol. 150, 169–178 (2010).
Tandon, A. et al. Surgical management for postkeratoplasty glaucoma: A meta-analysis. J. Glaucoma. 23, 424–429 (2014).
Grehn, F. et al. Factors affecting the outcome of trabeculectomy: An analysis based on combined data from two phase III studies of an antibody to transforming growth factor beta2, CAT-152. Ophthalmology 114, 1831–1838 (2007).
Dueker, D. & Robinson, J. Procedural treatments: Glaucoma drainage devices. In Pearls of Glaucoma Management. JA Giaconi, Editors. Springer. (2010).
Budenz, D. L. et al. Five-year treatment outcomes in the Ahmed Baerveldt comparison study. Ophthalmology 122, 308–316 (2015).
Christakis, P. G. et al. The Ahmed versus Baerveldt study: Five-Year treatment outcomes. Ophthalmology 123, 2093–2102 (2016).
Yamanaka, O., Kitano-Izutani, A., Tomoyose, K. & Reinach, P. S. Pathobiology of wound healing after glaucoma filtration surgery. BMC Ophthalmol. 15 (Suppl 1), 157 (2015).
Hong, C. H., Arosemena, A., Zurakowski, D. & Ayyala, R. S. Glaucoma drainage devices: A systematic literature review and current controversies. Surv. Ophthalmol. 50, 48–60 (2005).
Purtskhvanidze, K., Saeger, M., Treumer, F., Roider, J. & Nölle, B. Long-term results of glaucoma drainage device surgery. BMC Ophthalmol. 19, 14 (2019).
Gupta, S. et al. Long-term outcomes of glaucoma drainage devices for glaucoma post-vitreoretinal surgery with silicone oil insertion: A prospective evaluation. Graefes Arch. Clin. Exp. Ophthalmol. 254, 2449–2454 (2016).
Barton, K. et al. Aqueous Shunts: Choice of Implant. In Glaucoma, 2nd Edition. TM Shaarawy, Editors. Elsevier. (2015).
Vinod, K. et al. Practice preferences for glaucoma surgery: A survey of the American glaucoma society. J. Glaucoma. 26, 687–693 (2017).
Shao, C. G., Sinha, N. R., Mohan, R. R. & Webel, A. D. Novel therapies for the prevention of fibrosis in glaucoma filtration surgery. Biomedicines 11, 657 (2023).
Zhang, X., Song, Y., Liebmann, J. & Weinreb, R. N. A modified technique in applying sponge soaked with mitomycin C in trabeculectomy. Asia Pac. J. Ophthalmol. (Phila). 10, 548–552 (2021).
McCulloch, P., Taylor, I., Sasako, M., Lovett, B. & Griffin, D. Randomised trials in surgery: Problems and possible solutions. BMJ 324, 1448–1451 (2002).
Zhang, D. et al. Trends in operating room-based glaucoma procedures at a single eye center from 2016–2020. Int. J. Ophthalmol. 16, 208–214 (2023).
Ledesma-Gil, J., García-Rodríguez, M. L., Gurria, L. U., Graue-Hernández, E. O. & Navas, A. Glaucoma Mini-Shunt implantation after keratoplasty. J. Glaucoma. 26, 315–319 (2017).
Ates, H., Palamar, M., Yagci, A. & Egrilmez, S. Evaluation of Ex-PRESS mini glaucoma shunt implantation in refractory postpenetrating keratoplasty glaucoma. J. Glaucoma. 19, 556–560 (2010).
Oliver, C., Gupta, P., Yeung, S. N. & Iovieno, A. Outcomes of XEN stent in patients with glaucoma and previous corneal transplantation. J. Glaucoma. 32, 750–755 (2023).
Tan, A. M. et al. Micropulse transscleral diode laser cyclophotocoagulation in the treatment of refractory glaucoma. Clin. Exp. Ophthalmol. 38, 266–272 (2010).
Williams, A. L. et al. Clinical efficacy and safety profile of micropulse transscleral cyclophotocoagulation in refractory glaucoma. J. Glaucoma. 27, 445–449 (2018).
Subramaniam, K., Price, M. O., Feng, M. T. & Price, F. W. Micropulse transscleral cyclophotocoagulation in keratoplasty eyes. Cornea 38, 542–545 (2019).
Lee, J. H. et al. Clinical outcomes of micropulse transscleral cyclophotocoagulation in patients with a history of keratoplasty. J. Ophthalmol. 2020, 6147248 (2020).
Zemba, M. et al. Micropulse transscleral cyclophotocoagulation for glaucoma after penetrating keratoplasty. Diagnostics 12, 1143 (2022).
Kang, J. M., Rose-Nussbaumer, J., Schallhorn, J. M., Hwang, D. G. & Han, Y. Clinical outcomes of micropulse transscleral cyclophotocoagulation in post-keratoplasty patients. Invest. Ophthalmol. Vis. Sci. 60, 2826–2826 (2019).
Fili, S., Vastardis, I., Perdikakis, G. & Kohlhaas, M. Transscleral cyclophotocoagulation with MicroPulse® laser versus cyclophotocoagulation with continuous diode laser in patients with open-angle glaucoma. Int. Ophthalmol. 42, 525–539 (2022).
Aquino, M. C. D. et al. Micropulse versus continuous wave transscleral diode cyclophotocoagulation in refractory glaucoma: A randomized exploratory study. Clin. Exp. Ophthalmol. 43, 40–46 (2015).
Souissi, S. et al. An update on continuous-wave cyclophotocoagulation (CW-CPC) and micropulse transscleral laser treatment (MP-TLT) for adult and paediatric refractory glaucoma. Acta Ophthalmol. (Copenh). 99, e621–e653 (2021).
Weber, C. et al. Clinical outcomes of the PAUL® glaucoma implant: One-year results. Clin. Exp. Ophthalmol. 51, 566–576 (2023).
Acknowledgements
All authors express their gratitude to the Center of Evidence-Based Medicine, the Biostatistics Group, Department of Medical Research in Taichung Veterans General Hospital, and the chief librarian of Taichung Veterans General Hospital Medical Library, Yu-Hui Su, who provided invaluable assistance in refining our search strategy.
Funding
This study was supported by Taichung Veterans General Hospital (grant number: TCVGH-1136902B). The funding organizations played no role in this study.
Author information
Authors and Affiliations
Contributions
SYP and CCC developed the fundamental concepts for this study. SYP and CCC performed the article screening, risk of bias assessment, and data extraction. SYP conducted data analysis, created visual representations in graphs, and wrote the initial draft. YYC offered substantial technical support for data analysis and performed meta-regression analysis. CCC edited the manuscript and supervised the entire study. YAL, HEH, PTT, HJL, CYW, CJC, and IJW provided valuable expertise and experience. All authors discussed the results, provided feedback on the findings, and endorsed the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Pan, SY., Chen, YY., Lu, YA. et al. A systematic review and network meta-analysis of surgical interventions for glaucoma following penetrating keratoplasty. Sci Rep 15, 13783 (2025). https://doi.org/10.1038/s41598-025-95997-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-95997-6