Abstract
For early-stage oral squamous cell carcinoma (OSCC), the primary treatment modality generally entails surgical resection. However, the role of neck dissection in early-stage OSCC remains controversial. The aim of this study was to evaluate the effect of neck dissection on the prognosis of patients with early-stage OSCC. We identified patients with early-stage OSCC from the Surveillance, Epidemiology, and End Results database spanning from 2004 to 2021. We collected demographic and clinical data, including age, sex, tumor site, marital status, race, residence, income, grade, records of radiotherapy and chemotherapy, neck management modality, survival time, disease-specific survival (DSS), and overall survival (OS). A retrospective cohort study design was employed to differentiate between the neck observation and neck dissection group. Kaplan–Meier survival analysis and Cox regression were utilized to evaluate the association between neck dissection and survival outcome. The study included 12,606 patients, of whom 5135 died (available for OS analysis), and 2477 died from OSCC (available for DSS analysis). Kaplan–Meier survival analysis revealed statistically differences between the observation and neck dissection group in OS (p < 0.0001) and DSS (p = 0.00067). Multivariate analysis confirmed that neck dissection was associated with improved survival, after adjusting for multiple factors such as, age, sex, tumor site, marital status, race, residence, income, grade, records of radiotherapy and chemotherapy, neck dissection was associated with a decreased risk of both DSS (hazard ratio [HR] = 0.73, 95% confidence interval [CI] = 0.66–0.8; p < 0.001) and OS (HR = 0.76, 95% CI = 0.71–0.81; p < 0.001). Patients with early-stage OSCC may derive significant clinical benefit from neck dissection; therefore, neck dissection is recommended for such patients.
Similar content being viewed by others
Introduction
Oral squamous cell carcinoma (OSCC) is the most prevalent malignant neoplasm in the head and neck region, and given its significant impact on patients’ facial structures and physiological functions, OSCC has garnered considerable attention in recent years1,2. According to the latest data, there are over 300,000 new cases of OSCC worldwide each year, and nearly 200,000 patients die from this disease, with a worsening trend3. In accordance with the most recent guidelines from the National Comprehensive Cancer Network (NCCN), patients diagnosed with OSCC should undergo a multidisciplinary treatment, with surgery serving as the cornerstone of therapy, and adjuvant radiotherapy and chemotherapy are recommended to reduce the risk of recurrence in patients with high-risk factors such as lymph node metastasis, lymph vascular invasion, perineural invasion, or close surgical margins4,5.
Early-stage OSCC, classified as stage I-II according to the American Joint Committee on Cancer TNM Staging System (T1-2N0M0), is primarily treated with surgery6. Studies shown that 5-year overall survival (OS) for early-stage OSCC ranged from 60 to 70%, while 5-year disease-specific survival (DSS) ranges from 70 to 80%7,8,9,10, and neck metastasis is a key prognostic factor11. Despite initial clinical examinations and neck imaging studies including MRI and CT scans showing no evidence of positive lymph node metastasis, the incidence of occult neck metastases in early-stage OSCC patients with clinically node-negative (cN0) status ranged from 20 to 30% 12,13. Despite this, there is currently no consensus on the need for neck lymph node dissection in patients with early-stage OSCC, and authoritative guidelines, including those from the National Comprehensive Cancer Network (NCCN) and the Chinese Society of Clinical Oncology (CSCO), do not provide clear guidance14,15. Current research findings have exhibited inconsistencies, some studies demonstrated that patients with early-stage OSCC who underwent selective neck dissection (SND) or sentinel lymph node biopsy (SLNB) in conjunction with primary surgery experience improved OS and DSS14,16,17. For example, Alimujiang Wushou et al. found that cT1N0M0 OSCC patients benefited from elective neck dissection (END) in terms of overall survival and disease-specific survival18. However, other studies indicated that neck dissection did not provide a significant survival benefit for patients with early-stage OSCC, and they pointed that the highly invasive procedure may result in prolonged hospital stays, increased healthcare costs, and complications such as shoulder and neck dysfunction, as well as skin pain and numbness19,20. For example, Qiuyu Wu et al. demonstrated that not all patients with cN0 OSCC benefit from END, and END was only recommended for patients with cN0 tongue squamous cell carcinoma21.As a result, clinical decision about whether to surgically remove the primary site along with neck dissection in early-stage OSCC patients was often confusing22,23.
Given the current conflicting research status, more evidence is needed to guide neck management strategies in patients with early-stage OSCC24. In this study, we conducted a large retrospective cohort analysis involving 12,606 patients with early-stage OSCC, aiming to investigate the effects of neck observation versus neck observation on DSS and OS in early-stage OSCC, using data from the SEER Program.
Results
Patient characteristics
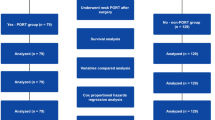
The study cohort included 12,606 eligible patients diagnosed with early-stage OSCC between 2004 and 2021. The detailed patient selection process was summarized in Fig. 1, and patients were divided into observation and neck dissection group. Table 1 provided a comprehensive summary of the baseline demographic characteristics of all participants. Based on the retrospective data obtained from the SEER database, 4264 individuals (33.85%) underwent neck dissection, while 8342 (66.15%) were assigned to the observation group. Specifically, 52.4% of the patients were aged 65 years or older, 61.2% were male, and the majority (86.6%) were white. Tongue cancer was the most prevalent site, accounting for 6803 cases (54.0%). The maximum follow-up period was 10 years, with a median survival duration of 36 months (interquartile range (IQR): 15.0, 75.0). During the follow-up period, 5135 (40.7%) patients died, of which 2477 (19.6%) deaths were attributed to OSCC. There were statistically significant differences in age, sex, tumor site, marital status, race, residence, income, tumor grade, radiotherapy and chemotherapy between the two groups (p < 0.05).
Flow chart of the study.
Cox proportional hazards regression analysis
Univariate analysis revealed significant associations between survival outcome (OS and DSS) and several factors, including age, sex, tumor site, marital status, race, residence, income, grade, radiotherapy, chemotherapy, and neck management modality. And the result indicated that neck dissection was a favorable factor for the survival outcome (Table 2). Multiple regression model was used to evaluate the effects of neck dissection on DSS and OS by adjusting different models (Table 3). Regarding DSS, all models demonstrated a significant reduction in HR, compared to the reference group. Specifically, the risk of death was reduced by 27% after adjusting for age, sex, tumor site, marital status, race, residence, income, tumor grade, radiotherapy, and chemotherapy in Model 3 ([HR] = 0.73, 95%CI = 0.66 ~ 0.8; p < 0.001), indicating high statistical significance. For OS, similar trends were observed, with Model 3 showing a 24% reduction in the risk of death ([HR] = 0.76, 95%CI = 0.71 ~ 0.81); p < 0.001), also highly statistically significant.
Subgroup analysis
Stratification and interaction analyses were employed to evaluate the robustness of neck dissection across various population characteristics. For DSS, significant impacts on survival outcome were observed for age, sex, grade, race and marital status. Interestingly, there were interactions between neck dissection and age, sex, grade and marital status that warrant further investigation (Fig. 2A). Similarly, for OS, age, sex, grade, race and marital status also demonstrated significant effects on survival outcome. Additionally, interactions at age and sex merit further exploration (Fig. 2B). The detailed subgroup analysis data was provided in Supplementary Table 1.
Forest plot illustrating the association between neck dissection and survival outcome across various subgroups: (A) DSS: disease-specific survival; (B) OS: overall survival.
Survival curve analysis
In the entire cohort, 5135 (40.7%) patients died, of whom 2477 (19.6%) succumbed to this cancer. The 5-year and 10-year disease-specific survival (DSS), rates for the neck dissection group were 77.7% and 64.4%, respectively, while the corresponding rates in the control group were 76% and 59% (p = 0.00067). Throughout the entire study period, the neck dissection group exhibited a notably higher probability of survival, particularly during the initial five-year phase (Fig. 3A). The 5-year and 10-year overall survival (OS) rates for patients who underwent neck dissection were 65.4% and 20.9%, respectively, compared to 58.1% and 11.6% in the observation group (p < 0.0001). Throughout the study period, the neck dissection group exhibited a markedly higher survival rate, particularly during the mid-study phase (30–90 months) (Fig. 3B). What’s more, we further explored the effects of SND and SLNB on survival outcome, the results showed that SLNB was significantly superior to SND in DSS and OS throughout the follow-up period (p < 0.05) (Fig. 3C,D).
Survival curve of patients with or without neck dissection. Kaplan–Meier curves of DSS (A) and OS (B) of patients with or without neck dissection; DSS (C) and OS (D) of patients among the observation, SND, and the SLNB group.
Discussion
Oral squamous cell carcinoma (OSCC) is a highly aggressive cancer originating in the mucosal epithelium of the oral cavity and accounts for approximately 90% of all oral cancers25. Not only could the disease cause serious health problems, but it could also affect the patient’s appearance, speech, swallowing ability, and taste, thereby profoundly reducing the patient’s quality of life26. Despite recent advances in treatment strategies, the 5-year survival rate remains around 60%27,28. The situation is further complicated by the fact that many individuals are diagnosed at advanced stages of OSCC. Therefore, for patients with early-stage OSCC, it is necessary to implement more accurate treatment to improve the prognosis of these patients and the overall clinical cure rate of OSCC.
Neck metastasis significantly affects survival outcome in patients with early-stage OSCC; however, it is unclear whether all early-stage OSCC requires neck dissection.
Therefore, the medical community should undertake more extensive and large-scale studies to address this challenge. The aim of this study was to compare the efficacy of neck dissection and neck observation through a retrospective analysis of early-stage OSCC. To date, this study represented the most recent large-scale retrospective cohort analysis, encompassing 12,606 patients with early-stage OSCC, with data current as of 2021. Univariate analysis preliminarily confirmed that neck dissection was a protective factor for DSS and OS, and different model adjustments further confirmed that it was an independent protective factor. Additionally, the stability of our results was validated through subgroup analyses. The results of the KM survival curve analysis demonstrated that neck dissection significantly extended the median survival time for patients with early-stage OSCC, improving both DSS and OS by 5 and 10 years, respectively.
Through multiple analytical methodologies, we determined that neck dissection significantly enhanced survival outcome for patients with early-stage OSCC. Neck metastasis represents the predominant pattern of recurrence in early-stage OSCC, despite the absence of positive lymph nodes in early-stage OSCC, statistical data indicate a potential metastasis rate ranging from 20 to 30% 29,30,31. The study supported early-stage OSCC recommendations for neck dissection, whether SND or SLNB, with improved OS and DSS. The potential reasons may encompass the following aspects: (1)preemptively obstructing the lymph node metastasis pathway to prevent recurrence and metastasis32; (2) For patients at risk of lymph node metastasis, neck dissection may eliminate potential metastases and decrease the likelihood of local recurrence33; (3) Neck lymph node dissection could provide a more accurate staging of the pathology and help to plan subsequent treatment, including adjuvant radiotherapy or chemotherapy34. However, many researchers did not recommend neck dissection, they believed that neck dissection may damage the vague nerve, phrenic nerve, brachial plexus and other important nerves, leading to corresponding complications 35. And vascular damage may also occur, such as the thin wall of the internal jugular vein, unclear anatomical level or rough movement during intraoperative separation, which may lead to massive bleeding of the internal jugular vein rupture, or improper ligation leading to massive bleeding, or even air embolism36. Furthermore, some patients may experience transient dysfunction in the shoulder and neck areas, while a minority of patients might suffer from permanent dysfunction in these regions. And for patients with cN0, neck dissection may result in overtreatment for 70% to 100% of patients, thereby increasing the incidence of treatment-related complications, prolonging hospital stays, and escalating costs37. Despite the potential drawbacks associated with neck dissection, it is imperative to conduct this procedure in early-stage OSCC patients to enhance the prognosis. Of course, to further enhance the management of complications, we suggest conducting multicenter prospective studies to validate best practices and identify strategies that can reduce the incidence of complications. These studies should focus on integrating advanced imaging and molecular techniques to improve preoperative planning and intraoperative decision-making. Additionally, the development of standardized protocols for preoperative assessment, surgical technique, and postoperative care can help minimize variability and improve overall patient outcomes.
Furthermore, our preliminary findings indicated that SLNB may offer advantages over SND.The reason may lie in that SLNB could accurately locate and detect sentinel lymph nodes with minimally invasive technology, which can not only effectively identify latent metastases, avoid unnecessary neck dissection, reduce the incidence of postoperative complications, but also preserve the immune function of neck lymph nodes and help local immune recognition38,39. Considering that SLNB surgery has the characteristics of short time and short hospitalization period, this significantly reduces the occupation of medical resources. In addition, due to the small scope of surgery, fewer postoperative complications, and relatively low overall treatment costs,we recommend that experienced centers conduct further prospective clinical studies to evaluate this potential superiority.
Our study has several limitations. First, the retrospective design of our study was based on data from the SEER database, which may be subject to inaccuracies, particularly concerning detailed treatment protocols. To mitigate this risk, we meticulously excluded patients with incomplete data and conducted subgroup analyses focusing on OS and DSS, thereby validating the robustness of our findings. Second, this study was unable to obtain critical pathological features including vascular invasion, neural invasion, and the depth of tumor infiltration, these factors should be taken into account in subsequent research. Third, the SEER database contains a limited amount of data on SLNB in early-stage OSCC, which impedes comprehensive and in-depth research on SLNB. In the future, our center plans to collaborate with multiple medical institutions to conduct large-sample retrospective analyses and large-scale prospective studies to further validate these findings.
Conclusions
The large-scale cohort study indicated that neck dissection was a significant independent prognostic factor for early-stage OSCC. We recommend that neck dissection be considered as part of early OSCC treatment to potentially improve patient outcomes.
Methods
Data sources
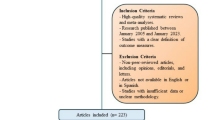
In this large-scale, population-based retrospective study, detailed patient information from the SEER database (17 registries, Nov 2023 submission, 2004–2021) was downloaded using SEER*Stat version 8.4.4 software. Patients with OSCC were identified using ICD-O-3 morphologic codes 8050–8076, 8078, 8083, 8084, 809440,41. Only early-stage primary OSCC cases were included in the analysis. Exclusion criteria were as follows: (1) incomplete survival records (including DSS and OS): To ensure the accuracy and reliability of our findings, patients with incomplete survival records were excluded from the study (N = 113); (2) incomplete neck dissection records: incomplete data regarding neck dissection significantly influenced the analysis outcomes. To ensure the accuracy and reliability of the results, patients with incomplete neck dissection information (N = 448) were excluded; and (3) incomplete AJCC cancer staging records and Late OSCC: Incomplete staging information may cause biased the results, and given that patients with advanced tumors were not included in the study, they were excluded from the analysis (181,366). Since the data involved in our study came from indirect data that was already publicly available, we did not need ethics committee approval.
Surveillance, epidemiology, and end results coding and variable definition
The demographic and clinical data used in the analyses included: age, sex, tumor grade, derived AJCC T, N, M classifications, RX Summary-Surgery Primary Site, RX Summary-Scope Regional LN Surgery, radiation recode, chemotherapy recode, SEER cause-specific death classification, survival months, vital status recode, race recode, marital status at diagnosis, residence, and income. Age groups were categorized as under 65 years and 65 years or older42,43. Race is divided into four categories: white, black, other (including American Indian/Alaska Native and Asian/Pacific Islander) and Unknown. Marital status was recorded as married, others (comprising single, divorced, widowed, unmarried, domestic partner, or separated statuses), and unknown. Tumor grade was categorized as well-differentiated, moderately differentiated, poorly differentiated, and unknown. Residence was classified as urban or rural. Income was were divided into three categories: < $50,000, $50,000–$75,000, and > $75,00040. Tumor sites included lip, tongue, gum, floor of mouth, palate, cheek, and others (overlapping lesions of other/unspecified mouth and mouth, not otherwise specified). Radiotherapy and chemotherapy were categorized as no/unknown or yes. Neck management modality was defined as a binary variable, including observation and neck dissection (1 to 3 regional lymph nodes removed, 4 or more regional lymph nodes removed, SLNB, SLNB with lymph nodes removed at different times, and SLNB with lymph nodes removed at the same/unstated time). The primary study outcome was OS and DSS. OS is the survival time from diagnosis of OSCC to death from any cause44, and DSS is the time from diagnosis of OSCC to death due to OSCC45.
Statistical analyses
Descriptive analysis was conducted on data from all participants. Missing values in the covariates were imputed using appropriate interpolation methods. Categorical variables were presented as frequencies and percentages (%). Comparisons of categorical variables were performed using the chi-square test or Fisher’s exact test, as appropriate. Univariate and multivariable Cox regression analyses were used to assess the correlation between neck dissection and various covariates with OS and DSS. Survival curves were generated using the Kaplan–Meier method. Subgroup analyses were conducted to investigate the relationship between neck dissection and survival outcome, adjusting for potential confounding covariates. Interaction tests within Cox proportional hazards models were utilized to compare hazard ratios (HRs) across subgroups. All statistical analyses were performed using Free Statistics Software version 2.0. Statistical significance was defined as p < 0.05.
Data availability
Data will be made available on request (Should any party require access to the data from this study, they may contact Dr. Yungang He; E-mail:13842693992@163.com).
References
Wang, S. et al. Metal polyphenol nanoparticle-based chemo/ferroptosis synergistic therapy for the treatment of oral squamous cell carcinoma. Bioconjugate Chem. 35(11), 1835–1842. https://doi.org/10.1021/acs.bioconjchem.4c00462 (2024).
He, Y. et al. BANF1 is a novel prognostic biomarker linked to immune infiltration in head and neck squamous cell carcinoma. Front. Immunol. 15, 1465348. https://doi.org/10.3389/fimmu.2024.1465348 (2024).
Li, Q. et al. Disparities in 36 cancers across 185 countries: Secondary analysis of global cancer statistics. Front. Med. 18(5), 911–920. https://doi.org/10.1007/s11684-024-1058-6 (2024).
Shah, H. P. et al. identifying opportunities to deliver high-quality cancer care across a health system: A clinical responsibility. Otolaryngol. Head Neck Surg. 171(2), 445–456. https://doi.org/10.1002/ohn.755 (2024).
Liao, C. T. et al. Identification of a high-risk subgroup of patients with resected pT3 oral cavity cancer in need of postoperative adjuvant therapy. Ann. Surg. Oncol. 18, 2569–2578. https://doi.org/10.1245/s10434-011-1616-4 (2011).
Kim, J. Y. et al. Initial negative resection margin versus revised negative resection margin in patients who underwent surgery without adjuvant therapy for early-stage oral tongue squamous cell carcinoma. Oral Oncol. 159, 107046. https://doi.org/10.1016/j.oraloncology.2024.107046 (2024).
Wushou, A., Yibulayin, F., Sheng, L., Luo, Y. & Yang, Z. C. Elective neck dissection improves the survival of patients with T2N0M0 oral squamous cell carcinoma: A study of the SEER database. BMC Cancer 21, 1–8. https://doi.org/10.1186/s12885-021-09053-3 (2021).
Sinevici, N. & O’sullivan, J. Oral cancer: Deregulated molecular events and their use as biomarkers. Oral Oncol 61, 12–18. https://doi.org/10.1016/j.oraloncology.2016.07.013 (2016).
Chun-Yang, H. et al. Margin to depth of invasion ratio as an indicator for stratifying close margins in early-stage oral squamous cell carcinoma. Oral Oncol. 151, 106726. https://doi.org/10.1016/j.oraloncology.2024.106726 (2024).
Wang, Y., Wang, S., Qu, Y. & Yang, Y. Clinicopathological characteristics, treatment and prognosis of oral adenocarcinoma: A population-based study. Eur. Arch. Oto-Rhino-Laryngol. 280, 3365–3374. https://doi.org/10.1007/s00405-023-07912-2 (2023).
Yasmin, G. et al. A robust and interpretable gene signature for predicting the lymph node status of primary T1/T2 oral cavity squamous cell carcinoma. Int. J. Cancer 150, 450–460. https://doi.org/10.1002/ijc.33828 (2021).
Danilo, D. G. et al. Management of clinically negative neck in early-stage (T1–2N0) oral squamous-cell carcinoma (OSCC): Ten years of a single institution’s experience. J. Clin. Med. 13, 7067. https://doi.org/10.3390/jcm13237067 (2024).
Leandros, V. V. et al. Management of the clinically N(0) neck in early-stage oral squamous cell carcinoma (OSCC): An EACMFS position paper. J. Cranio-maxillofac. Surg. 48, 711–718. https://doi.org/10.1016/j.jcms.2020.06.004 (2020).
Moya-Plana, A. et al. Sentinel node biopsy in early oral squamous cell carcinomas: Long-term follow-up and nodal failure analysis. Oral Oncol. 82, 187–194. https://doi.org/10.1016/j.oraloncology.2018.05.021 (2018).
Koyfman, S. A. et al. Management of the neck in squamous cell carcinoma of the oral cavity and oropharynx: ASCO clinical practice guideline. J. Clin. Oncol. 37, 1753–1774. https://doi.org/10.1200/jco.18.01921 (2019).
Alimujiang, W. et al. Patients with cT1N0M0 oral squamous cell carcinoma benefit from elective neck dissection: A SEER-based study. J. Nat. Compreh. Cancer Netw. 19, 385–392. https://doi.org/10.6004/jnccn.2020.7632 (2020).
Qiuyu, W. et al. Do patients with cN0 oral squamous cell carcinoma benefit from elective neck dissection? A large-scale population-based study. BMC Oral Health 24(1), 32. https://doi.org/10.1186/s12903-023-03632-5 (2024).
Wushou, A. et al. Patients with cT1N0M0 oral squamous cell carcinoma benefit from elective neck dissection: A SEER-based study. J. Nat. Compreh. Cancer Netw. JNCCN 19, 385–392. https://doi.org/10.6004/jnccn.2020.7632 (2021).
Liu, J. Y., Chen, C. F. & Bai, C. H. Elective neck dissection versus observation in early-stage (cT1/T2N0) oral squamous cell carcinoma. Laryngoscope Investig. Otolaryngol. 4(5), 554–561. https://doi.org/10.1002/lio2.301 (2019).
Yubin, C. et al. Elective neck dissection versus wait-and-watch policy for oral cavity squamous cell carcinoma in early stage: A systematic review and meta-analysis based on survival data. J. Oral Maxillofac. Surg. 77, 2154–2167. https://doi.org/10.1016/j.joms.2019.03.015 (2019).
Wu, Q. et al. Do patients with cN0 oral squamous cell carcinoma benefit from elective neck dissection? A large-scale population-based study. BMC Oral Health 24, 32. https://doi.org/10.1186/s12903-023-03632-5 (2024).
Leonardo, F. et al. Controversies in the treatment of early-stage oral squamous cell carcinoma. Curr. Problem Cancer 48, 101056. https://doi.org/10.1016/j.currproblcancer.2023.101056 (2023).
John, T. L. et al. Sentinel lymph node biopsy for management of the N0 neck in oral cavity squamous cell carcinoma. J. Surg. Oncol. 120, 101–108. https://doi.org/10.1002/jso.25494 (2019).
Thoenissen, P. et al. Extent of neck dissection and cervical lymph node involvement in oral squamous cell carcinoma. Front. Oncol. 12, 812864. https://doi.org/10.3389/fonc.2022.812864 (2022).
Yang, B., Wan, Y., Wang, J., Liu, Y. & Wang, S. Construction and validation of a prognostic model based on immune-metabolic-related genes in oral squamous cell carcinoma. Comput. Biol. Chem. 113, 108258. https://doi.org/10.1016/j.compbiolchem.2024.108258 (2024).
Awad, L. et al. Correlation between oral and oropharyngeal resection subsites and impact of reconstruction on speech and swallowing function in head and neck cancer patients: A systematic review. J. Cranio-Maxillofac. Surgery 52(7), 809–820. https://doi.org/10.1016/j.jcms.2024.01.010 (2024).
Yungang, H. et al. Matrisome provides a supportive microenvironment for oral squamous cell carcinoma progression. J. Proteom. 253, 104454. https://doi.org/10.1016/j.jprot.2021.104454 (2021).
Zhen-Hu, R. et al. Prospective observational study of surgery alone for locally advanced oral squamous cell carcinoma: A real-world study. BMC Oral Health 24, 156. https://doi.org/10.1186/s12903-024-03914-6 (2024).
Huang, S. et al. Tumor-stroma ratio is a crucial histological predictor of occult cervical lymph node metastasis and survival in early-stage (cT1/2N0) oral squamous cell carcinoma. Int. J. Oral. Maxillofac. Surg. 51, 450–458. https://doi.org/10.1016/j.ijom.2021.06.011 (2021).
Al-Moraissi, E. A. et al. Management of clinically node-negative early-stage oral cancer: Network meta-analysis of randomized clinical trials. Int. J. Oral. Maxillofac. Surg. 53, 179–190. https://doi.org/10.1016/j.ijom.2023.08.004 (2024).
Seki, M., Sano, T., Yokoo, S. & Oyama, T. Tumour budding evaluated in biopsy specimens is a useful predictor of prognosis in patients with cN0 early stage oral squamous cell carcinoma. Histopathology 70, 869–879. https://doi.org/10.1111/his.13144 (2017).
Tapan, D. P. et al. Efficacy of elective neck dissection in T1/T2N0M0 oral tongue squamous cell carcinoma: A population-based analysis. Otolaryngol. Head Neck Surg. 155, 588–597. https://doi.org/10.1177/0194599816643695 (2016).
Jayaraj, R. et al. Clinical validity and conceptual interpretation of systematic review and meta-analysis on elective neck dissection (END) versus observation for early-stage oral squamous cell carcinoma (OSCC). Oral Oncol. 109, 104764. https://doi.org/10.1016/j.oraloncology.2020.104764 (2020).
Liu, K. Y. et al. Nodal disease burden for early-stage oral cancer. JAMA Otolaryngol. Head Neck Surg. 142, 1111–1119. https://doi.org/10.1001/jamaoto.2016.2241 (2016).
Zanoni, D. K. et al. Survival outcomes after treatment of cancer of the oral cavity (1985–2015). Oral. Oncol. 90, 115–121. https://doi.org/10.1016/j.oraloncology.2019.02.001 (2019).
Al-Moraissi, E. A. et al. Sentinel lymph node biopsy versus elective neck dissection in management of the clinically negative (cN0) neck in patients with oral squamous cell carcinoma: A systematic review and meta-analysis. J. Cranio-Maxillo-Facial. Surgery 52, 141–150. https://doi.org/10.1016/j.jcms.2023.12.012 (2024).
Yang, C., Wang, Z., Shi, L. & Liu, W. Evaluation of neck control strategies for oral squamous cell carcinoma of stage I: Neck dissection or potential immunotherapy. J. Dental Sci. 19(1), 640–644. https://doi.org/10.1016/j.jds.2023.09.006 (2024).
Doll, C. et al. Sentinel lymph node biopsy in early stages of oral squamous cell carcinoma using the receptor-targeted radiotracer (99m)Tc-tilmanocept. Diagnostics 11, 1231. https://doi.org/10.3390/diagnostics11071231 (2021).
Kim, D. H., Kim, Y., Kim, S. W. & Hwang, S. H. Usefulness of sentinel lymph node biopsy for oral cancer: A systematic review and meta-analysis. Laryngoscope 131, E459-e465. https://doi.org/10.1002/lary.28728 (2021).
Harris, J. A., Hunter, W. P., Hanna, G. J., Treister, N. S. & Menon, R. S. Rural patients with oral squamous cell carcinoma experience better prognosis and long-term survival. Oral Oncol. 111, 105037. https://doi.org/10.1016/j.oraloncology.2020.105037 (2020).
Patel, T. D. et al. Efficacy of elective neck dissection in T1/T2N0M0 oral tongue squamous cell carcinoma: A population-based analysis. Otolaryngol. Head Neck Surg. 155, 588–597. https://doi.org/10.1177/0194599816643695 (2016).
Peiwen, W. et al. The role of postoperative radiation therapy in stage I-III male breast cancer: A population-based study from the surveillance, epidemiology, and End Results database. The Breast 65, 41–48. https://doi.org/10.1016/j.breast.2022.06.004 (2022).
Jiazhao, S., Yupeng, D., Xiaoli, K., Gang, R. & Yingjie, W. J. F. P. H. Development and validation of a nomogram to predict cancer-specific survival with unresected cholangiocarcinoma undergoing external radiotherapy. Front. Public Health 11, 1012069. https://doi.org/10.3389/fpubh.2023.1012069 (2023).
Chen, Y. et al. A novel immune-related gene signature to identify the tumor microenvironment and prognose disease among patients with oral squamous cell carcinoma patients using ssGSEA: A bioinformatics and biological validation study. Front. Immunol. 13, 922195. https://doi.org/10.3389/fimmu.2022.922195 (2022).
Ma, X. et al. FERMT2 upregulation in CAFs enhances EMT of OSCC and M2 macrophage polarization. Oral Dis. 30, 991–1003. https://doi.org/10.1111/odi.14610 (2024).
Acknowledgements
We would like to thank all participants, staff and other researchers for their valuable contributions to this study.
Funding
This study was funded by the Technological Innovation Project Assignment of Shapingba District, Chongqing (Grant No. 2024115).
Author information
Authors and Affiliations
Contributions
Yungang He and Haonan Shui contributed to the experiments and data interpretation. Yungang He, Jia Liu, and Yuehang He contributed to writing the original draft and visualization. Qiuzhi Chen and Jian Wu contributed resources, reviewed and edited the manuscript, administered the project, and acquired funding. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
He, Y., Shui, H., Liu, J. et al. Neck dissection improves the prognosis of patients with early-stage oral squamous cell carcinoma. Sci Rep 15, 11640 (2025). https://doi.org/10.1038/s41598-025-96018-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-96018-2