Abstract
Traditional gait measurement systems are often limited by factors such as cost, complexity, prolonged setup times, and requirements for specialized training and expertise. Wearable pressure- and motion-sensing insoles have opened new possibilities for accessible gait analysis in real-life conditions. This study evaluated the equivalence of FeetMe insole measurements of gait parameters to those of a laboratory gold standard, optoelectronic motion capture system coupled to force platforms (MoCap/FP). Gait and posture parameters were assessed in 37 healthy adults by FeetMe insoles and by MoCap/FP system simultaneously. Means and variances were compared, and inter-device agreement was assessed for each parameter. Between-device equivalence was demonstrated for all parameters assessed (two one-sided t tests: P < .001). For static parameters, six of 13 variables presented excellent interclass correlation coefficients (ICCs ≥ 0.90) and three had good ICCs (≥ 0.75 to < 0.90). Moreover, 10 of 11 spatiotemporal parameters showed excellent accuracy (ICCs ≥ 0.90), and three of four kinetic parameters showed moderate-to-good accuracy (ICCs between 0.78 and 0.89). In summary, FeetMe can be considered as a valid gait measurement tool compared to the high precision MoCap/FP system and could be used in clinical practice to assess a wide range of gait and posture parameters, overcoming some limitations of traditional systems.
Similar content being viewed by others
Introduction
Gait disturbances are a frequent manifestation of various medical conditions, including neurological diseases (e.g., Parkinson’s disease1,2,3,4,5 and multiple sclerosis6,7,8), cardiovascular diseases (e.g., stroke9), and musculoskeletal diseases (e.g., osteoporosis10). Common gait abnormalities include increased asymmetry, slower walking speed, reduced stride length and foot clearance, decreased arm swing, freezing of gait, ataxia, foot drop, spasticity, and altered coordination11. Assessment of the extent of deviation and variability of the spatiotemporal, kinetic, kinematic and static parameters associated with specific diseases plays an essential role in identifying gait disturbances, assessing the risk of falling, and aiding the monitoring and management of patients with gait disorders1,2,3,6,8,9,12. It can also be useful to predict cardiovascular events and mortality13,14 and to classify pathologies15.
There are several kinds of devices available to measure gait parameters but instrumented quantitative gait analysis is usually carried out using non-wearable devices such as walkways, treadmills, and optoelectronic motion capture systems, sometimes coupled to force platforms (MoCap/FPs). Walkway systems with embedded pressure-activated sensors, are commonly used for the measurement of spatiotemporal parameters of gait in clinical practice and research settings for different patient populations, with GAITRite walkway being the most widely used system16. Although GAITRite has been shown to be a reliable, accurate, and valid tool and was considered as the gold standard of walkways to assess spatiotemporal parameters16,17,18,19, it has some limitations. Primarily, its measuring capabilities are limited to spatiotemporal parameters exclusively and it lacks the capability to evaluate gait under real-world conditions. Furthermore, its use is restricted to specialized centers due to many factors, including cost, limited availability, and the need for trained personnel to operate the system. Finally, even in clinical settings, the restricted length of the walkway drastically limits the distance over which gait parameters are assessed.
MoCap/FP systems are widely regarded as “high precision” tools used in laboratory settings or expert centers for measuring gait for research purposes20,21,22,23. The MoCap component of the system employs passive reflective markers placed on the patient’s body, along with high-frequency cameras, to capture three-dimensional motion data. On the other hand, FPs provide measurements of three-dimensional forces and torques applied to the ground. The combined use of these devices enables an extensive analysis of various aspects of walking, including kinematic, kinetic, and spatiotemporal gait parameters. Although MoCap/FP systems offer numerous advantages and provide a broader depiction of gait than walkway systems, they still have a few disadvantages. Just like the walkways, these systems are restricted to indoor laboratory environments, which may not fully reflect the true complexity of gait in real-life settings. As they typically measure a limited number of strides in controlled conditions, they may struggle to capture the natural, cycle-by-cycle variations in gait. Moreover, these systems are expensive, lack portability, and are cumbersome to set up. They also require dedicated space, specialized training, and expertise, as well as time-consuming data processing, restricting their availability and use to a select number of well-equipped centers.
Given the above-mentioned constraints of the gold standard GAITRite and high precision MoCap/FP systems, wearable devices (e.g., insole-based systems, inertial measurement units), and digital tools (e.g., depth cameras and applications for phones and smartwatches using signals from device-embedded accelerometers) have been developed and assessed as possible alternatives for gait monitoring in clinical practice11. Commercially available insole-based systems can generally be classified into two main categories. The first category comprises insoles equipped with pressure or load sensors, (pedar24, XSENSOR25, LoadSol26, and Medilogic27). These systems enable the measurement of plantar pressure distribution and ground reaction force. The second category involves inertial measurement unit-based insoles (PODOSmart28), able to capture spatiotemporal gait parameters. Notably, certain insoles, such as Insole329, integrate both sensor types, facilitating the measurement of both plantar pressure distribution and spatiotemporal gait parameters. Among these systems, the FeetMe connected insoles were specifically developed with the aim of allowing gait assessments in a wider range of clinical settings and in real-life conditions30. The validity and reliability of FeetMe connected insoles to measure spatiotemporal gait parameters has already been evaluated in comparison to the GAITRite walkway system in a variety of populations and conditions31,32,33,34. Thus, the aim of the current work was to assess the validity of FeetMe insoles for measuring gait metrics compared with a system that provides more accurate data than GAITRite and for measuring kinetic and postural parameters not provided by GAITRite. In other words, determine if the FeetMe insoles could provide gait and posture parameter measurements comparable to those obtained with the high precision, marker-based MoCap/FP instruments, while being simpler and more accessible for use in clinical practice and research settings.
Methods
Study objectives
The main objective of this study was to demonstrate the equivalence of the measurements obtained by the FeetMe insoles to those acquired by the MoCap/FP system during the static stabilometry and walking tests. The following gait parameters were studied:
-
static parameters: unipodal and bipodal center of pressure (CoP) in the anterior-posterior (AP) and medial-lateral (ML) directions, total body weight, body weight distribution, and asymmetry;
-
kinetic parameters: the vertical ground reaction force (vGRF) through its passive (weight acceptance or heelmax load) and active (push-off or toemax load) peaks, and the dynamic CoP trajectory dimensions in the AP and ML directions during stance phase;
-
spatiotemporal: heel strike (HS) and toe off (TO) events, stride length, velocity, cadence, and stance, swing, step, stride, single-support and double-support duration.
Study design
This prospective, comparative validation, single-center study was conducted at the Functional Unit for Movement Analysis at Saint-Maurice Hospital (Saint-Maurice, France) between February 2022 and November 2022. Healthy volunteers were included in the study if they met the following inclusion criteria: (i) were aged 18 to 80 years, (ii) had a shoe size between 35 and 46 (with the aim of having at least one volunteer for each shoe size), (iii) had a body weight between 40 kg and 120 kg (with the aim of having at least two volunteers allocated to one of nine body weight groups: 40–45, 46–55, 56–65, 66–75, 76–85, 86–95, 96–105, 106–115, and 116–120 kg), (iv) were able to walk unaided for 30 min, and (v) were comfortable with using a smartphone. Participants were not included if they (i) had cognitive or behavioral problems limiting communication or participation in the study, (ii) were admitted to a health or social institution for purposes other than research, (iii) had undergone major surgery or orthopedic surgery, or other interventions during the previous 3 months that had a possible effect on walking, or (iv) had a chronic disease affecting walking, (v) were not covered by any social security scheme.
Potential participants were contacted over the phone or via e-mail to confirm their interest in participating in the study and were provided with an explanation of the study content. Individuals who met the inclusion criteria were invited to participate. Participants could be withdrawn from the study if they failed to meet the inclusion criteria, met any of the non-inclusion criteria, withdrew consent, or were not covered by any social security scheme. The study consisted of one 60-min visit, during which each participant performed one static test in a controlled posture, referred to as the static stabilometry test, and four walk tests with different cadences conducted using a metronome, referred to as dynamic walking tests. Detailed descriptions of the tests can be found in the intervention section.
Ethical considerations
The study was approved by the Personal Protection Committee (CPP Ile de France IV, approval ID: 21.03042.000046-MS03). It was performed in accordance with the Helsinki Declaration and all subsequent amendments, Good Clinical Practice (GCP) guidelines, and local regulatory requirements (registration number: ID-RCB 2021-A02409-32, ClinicalTrials.gov Identifier: NCT06027762). Prior to proceeding with the study, each volunteer provided written informed consent for data collection and analysis according to the stated research goals. The study data were de-identified with an individual alphanumeric code for each participant and were handled according to French L. 1121-3 public health legislation. No compensation was provided.
Instrumentation
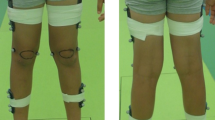
An optoelectronic MoCap system (Vicon NEXUS and MX-T40, Vicon Motion Systems Ltd, Unit 6, Oxford Industrial Park, Oxford, UK) combined with FPs (AMTI BP400600-1000, 176 Waltham Street, Watertown, USA) was used as the reference system for capturing real-time body movement. The MoCap system comprised a set of reflective markers fixed to the participants shoes (Fig. 1(a)) and 10 cameras arranged around an area containing four FPs (60 cm x 40 cm each) that were positioned to create a straight, obstacle-free, flat walkway measuring 6 m long and 2 m wide (Fig. 1(c)).
(a) Participant during non-assisted walking on force platforms with FeetMe insoles inside the shoes and reflective markers for the Vicon optoelectronic motion capture (MoCap) system attached to the outside of the shoes. (b) FeetMe insoles. (c) Schematic representation of the walk trajectory on the force plates and the position of MoCap cameras. (d) Aquashoes from Decathlon with reflective markers and FeetMe insoles inside the shoes.
The test system, FeetMe insoles (a Class Im CE [93/42/EC] medical device available in European sizes 35–46 corresponding to 224–296 mm; Fig. 1(c)) can calculate gait parameters in real time based on the signals from 18 capacitive pressure sensors and a 6-axis inertial measurement unit (IMU). The parameters can be sent to a smartphone via a Bluetooth Low Energy (BLE) link when the insoles are used in the connected mode, or be stored in the internal memory when no phone is connected (standalone mode). Given that the data collected as part of this study could provide valuable insights for the future development of the device, the raw, unprocessed signals from the pressure sensors and IMU were transmitted to the smartphone via BLE and then by Wi-Fi to a medical service-certified secure server. Measures of static, kinetic, and spatiotemporal gait parameters were then computed on a PC using an algorithm identical to that embedded in the insoles, since the embedded algorithms are not running when raw signals are being transferred to the smartphone. Results of this computation was used for the comparison of the two tools.
FeetMe insoles were inserted into participant’s footwear (i.e., the shoes with reflective markers; Fig. 1(d)) for simultaneous measurements by the two systems. The gait parameters evaluated with both the reference (MoCap/FP) and test (FeetMe) systems are listed in Table 1. The sampling rates were 100 Hz for the MoCap system, 1000 Hz for the FPs and 110 Hz for the FeetMe insoles. To reduce the impact of the shoe sole on the pressure distribution received by the FPs and ensure the CoP trajectory between the FeetMe insoles and the MoCap /FP system was comparable, soft sole shoes (aqua shoes from Decathlon) were used during the trial.
The locations and use of reflective markers (shown in Fig. 1(d)) are described here. Markers containing the letters FF are placed on the forefoot and markers containing the letters HL are placed on the heel. FF_L2/R2 and HL_L/R are located on the length extremities and FF_L/R_Lat and FF_L/R_Med are located on the width extremities to detect the position of the foot in relation to the force plate zone during walking; HL_L/R_Lat, HL_L/R_Med, FF_L1/R1 and HL_L/R are used to determine the orientation and origin of the insole-based reference frame located at the centre of the white circle on the FeetMe insoles. The position of the origin is visible on the insole surface and is consistent across insoles relative to other insole components, making it an appropriate choice for the reference frame origin. These markers represent the insole signals in the MoCap-based reference frame, allowing comparison of parameters such as the center of pressure (CoP).
Intervention
Each participant underwent a 1-minute static stabilometry test, followed by four consecutive sets of dynamic walking tests. All tests were conducted while the participants were wearing shoes fitted with the FeetMe insoles on the inside and reflective markers attached to the outside. Thus, throughout all the tests, both systems simultaneously captured signals to allow a direct between-system comparison of the estimated gait parameters. To achieve synchronization between the two systems, before each test the participants were asked to stand with one foot positioned on FP No.3 and the other on FP No.4 and perform a jump, which created an impulse signal on both the FeetMe insoles and the MoCap/FP system. This sharp signal was later used to synchronize the data between the two systems using a purpose-built algorithm (as illustrated in Figure S2(a)). The parameters evaluated simultaneously by the FeetMe test and MoCap/FP reference systems are listed in Table 1.
Static stabilometry test: the static stabilometry test consisted of participants standing still on the FPs. After the synchronization step, the participants were instructed to stand on the FPs, and an investigator ensured that their feet were positioned with a 30° angle between them and a 2-cm distance between their heels35 (as shown in Figure S2(b)). The participants were asked to remain motionless in this position for one minute while parameters were recorded.
Dynamic walking test: the dynamic walking test consisted of participants walking over the FPs at different cadences and involved at least 20 passages (10 round trips). A passage was defined as a walk path in sequence over FP No.1, 2, 3, and 4, or in the inverse order No.4, 3, 2, and 1 (as illustrated in Fig. 1(b)). To reach their natural walking pace, each participant began walking 2 m before the active area covered by the FPs and walked 2 m beyond the active area. After the synchronization step, the participants were asked to perform the 10 round trips (20 passages) at four different cadences: 40 beats per minute (bpm), 75 bpm, and 110 bpm by following the rhythm of a metronome, and a free spontaneous walk without a controlled or imposed cadence. They were also asked to ensure that they placed one foot on only one FP at a time and did not walk on the border of the FPs. Recordings were done with both systems for each cadence.
Data preparation and processing
A quality check on the frequency of FeetMe insole signals was done to ensure there was no significant data loss and that the sampling rate was regular enough, so that the signals were of sufficient quality to apply FeetMe algorithms. Data loss can be detected under two conditions: when the time difference between the two subsequent samples is greater than 30ms or when the percentage of time difference between 15 ms and 30 ms is greater than 10% over the whole test record. If data loss was detected for a test, checks were conducted to determine if this loss occurred during one stride. This stride was then not analyzed due to poor signal quality.
Static stabilometry test: for the static stabilometry test recordings, an algorithm was used to eliminate data corresponding to the synchronization step and the transitory phase between the jump and stable static state. Data corresponding to the stable static phase in a controlled position was kept for analysis, which corresponded to 30 to 40 s out of the full one-minute duration of each recording.
Dynamic walking test: for the dynamic walking test recordings, the data collected from the two gait recording systems were temporally aligned and segmented into single strides for each participant. Only gait events and gait cycles fully detected by the MoCap/FP system were included in the comparative analysis. This selection was made because many gait parameters require a full gait cycle to calculate their values. A gait cycle was deemed detected by the MoCap/FPs only if both the initial and terminal HS events of the gait cycle were correctly detected. This means that any gait cycle that started on a FP, but ended outside of any FP was disregarded, as the terminal HS of this cycle would not be detected by the system (Figure S2(d)). To the contrary, the initial HS and TO events from this cycle were included in the analyses, as these events would be detected by the FP on which the gait cycle began. As a result, data collected when participants walked outside the FPs, for example to make a turn, were not considered. In addition, it was ensured that for all analyzed data, only one foot touched one FP at a time and did not exceed the FP boundary. Having only one foot touching one FP at a time also ensured that one FP reflected vGRF for one single foot during a stance period. Accordingly, the number of data points for the parameters requiring a gait cycle of a single foot for their estimation (e.g., stride duration, stride length, stance duration, swing duration, velocity, and cadence) corresponded to the number of valid gait cycles detected by the MoCap/FP system. On the other hand, the number of data points for parameters requiring information from both feet (e.g., step duration, single-support duration, and double-support duration) would be lower because each data point required at least one valid gait cycle for one foot and one valid stance for the opposite foot to be detected simultaneously. For the analysis of vGRF accuracy, only the precision of the max peaks (“heelmax load” and “toemax load”, see Figure S2(c)) was quantified, thus vGRF data for which it was not possible to clearly identify the two peaks with either the MoCap/FP system or insoles were excluded.
Data processing: the pressure and motion sensors of the FeetMe insoles generated 24 time series during a test: 18 from each pressure sensor and six from each axis of the IMU (see Figure S3). A Savitzky-Golay filter36 (3rd degree polynomial) was applied to each pressure sensor signal, and a special algorithm automatically eliminated offsets for each signal. Once the signals were pre-processed, the main FeetMe algorithms to calculate static, kinetic, and spatiotemporal gait parameters were applied. Figure S4 shows the corresponding data flowchart. In particular, CoP and vGRF were calculated from the interpolated pressure maps as a weighted spatial average and a spatial integral respectively. Gait events were detected based on the pressure and motion data and stride length was estimated using Kalman filter32.
On the MoCap/FP side, the gait was segmented in cycles using gait events detected thanks to the force plates. In particular, HSs were deemed detected when force signal exceeded a threshold of 10 N, while an opposite principle was applied to detect TOs. Spatiotemporal parameters were calculated from the positions of retroreflective markers and using gait events. While kinetic parameters were calculated in a standard way directly from the force and moments signals.
Statistical analysis
Quantile-quantile (Q-Q) plots and the Shapiro–Wilk test (with a 95% confidence level; CI) were used to assess the distribution and normality of the body weights of the participants. After identifying appropriate gait cycles, values of the parameters for all cycles were calculated. The equivalence of averages of measurements produced by each system was assessed using paired two one-sided t tests (TOSTs). The testing hypothesis was as follows: H0: |µF - µM| ≥ δ (delta) vs. H1: |µF - µM| < δ, where µF and µM are the averages of measurements by the FeetMe and MoCap/FP systems, respectively. Delta is the margin of equivalence which corresponds to the maximum acceptable deviation between the averages of the studied device (FeetMe) and the reference device (MoCap/FP). The acceptable deviation was determined independently for each parameter based on the requirements of its field of application. The values of δ for all parameters are presented in Table 1. A Levene test was used to assess significant differences between the standard deviations (SD) of MoCap/FP and FeetMe device data. The testing hypothesis was as follows: H0: σF = σM vs. H1:σF ≠ σM, where σF and σM are the variances of FeetMe and MoCap/FP measured parameters, respectively. The significance level alpha (α) for both the TOST and Levene tests was set at 0.05. If the P-value was greater than α, H0 was accepted; if the P-value was less than or equal to α, H0 was rejected.
The bias (i.e., systematic error), 95% limits of agreement, Pearson correlation coefficient, mean absolute error (MAE), mean relative absolute error (MRAE), intraclass correlation coefficient (2,1) (ICC) and its 95% CIs, and coefficient of determination (r2) were also determined for each parameter measured by the FeetMe insoles versus the MoCap/FP system. In addition, agreement between the reference and test systems was analyzed using Bland-Altman and linear regression plots.
The following criteria were used for the Pearson correlation coefficient and r2 to assess the degree of correlation 37: <0.30 negligible, ≥ 0.30–<0.50 low, ≥ 0.50–<0.70 moderate, ≥ 0.70–<0.90 good, and ≥ 0.90–<1.00 excellent correlation. For the inter-device ICCs, values below 0.50 were deemed to indicate poor validity, values ≥ 0.50 to < 0.75 to indicate moderate validity, values ≥ 0.75 and < 0.90 to indicate good validity and values ≥ 0.90 to indicate excellent validity, as previously described37.
All data and statistical analyses were performed using Python software (version 3.8).
Results
Study population
A total of 37 healthy volunteers, including 14 females and 23 males, were included in the study (Table 2). The age of the participants ranged from 23 to 71 years, with a mean age of 44 years. The average height and body weight (± SD) were 173 ± 11 cm and 78 ± 20 kg, respectively. The body weight of the participants displayed a normal distribution (Shapiro–Wilk test, P = .568; Figure S5(e)).
Data for one of the participants were excluded from the analysis of static parameters due to incomplete data capture caused by a technical fault with the mobile application. Data from two of the participants were also excluded from the analysis of kinetic and spatiotemporal parameters due to material failure. The analysis of kinetic parameters was conducted using a total of 11,515 data points. For parameters that relied on data from only one foot, there were 5250 available data points; whereas for those that required data from both feet, 4782 data points were available for analysis.
Accuracy of static parameters
Results of the comparative analysis of the static parameters estimated by FeetMe and the MoCap/FP system during the static stabilometry tests are presented in Table 3. All static parameter measurements made by the FeetMe device were within the acceptable margin of equivalence (TOST P-values < 0.001), and there were no significant differences in the variance of the measured parameters between the two devices (Levene P-values > 0.5; Table 3).
Bland-Altman analyses of FeetMe and MoCap/FP measurements of static stabilometry parameters: (a) bCoP AP dim, (b) bCoP ML dim, (c) total weight, and (d) weight ratio. The solid line represents the mean difference between the systems, with the upper and lower lines (dotted lines) representing the limits of agreement. bCoP AP = anterior-posterior bipodal center of pressure, bCoP ML = medial-lateral bipodal center of pressure, dim = dimensions.
The Pearson correlation coefficient values ranged from 0.67 to 1.0, with 46% (6/13) of the values being ≥ 0.90, indicating an excellent level of correlation between the two.
systems. Very good-to excellent coefficient of determination values (r² between 0.74 and 0.99) were obtained for most of parameters, with very high levels of correlation for bipodal CoP ML center (0.90) and unipodal CoP ML center (0.99). Moderate-to-excellent inter-device agreement was also obtained for the static parameters, with ICCs ranging between 0.65 for weight ratio and asymmetry, to 1.0 for unipodal CoP ML center. The limits of agreement, bias, and measures of MAE and MRAE for all parameters are also provided in Table 3 and examples of Bland-Altman analyses of bias are shown in Fig. 2.
The most accurate measures were those obtained for bipodal CoP AP dim and bipodal CoP ML dim, with bias reaching 0 cm and moderately narrow limits of agreement ([-0.5, 0.4] and [-0.6, 0.5]; Table 3; Fig. 2(a) and 2(b)) and good-to-excellent reliability (ICCs of 0.89 and 0.91, respectively; Table 3), indicating good-to-excellent agreement between the devices.
Regarding total weight and weight distribution between sides, the FeetMe insoles tended to overestimate these parameters in the static test, with bias values of 8.7 kg and 4.4 kg, respectively (Table 3; Fig. 2(c) for total weight). However, the FeetMe insoles still showed good levels of correlation with the reference system (ICC values of 0.81 and 0.79, respectively). On the other hand, the weight ratio bias was very low (-1%; Fig. 2(d)), but the levels of correlation were only moderate (ICC: 0.65; Table 3).
Accuracy of kinetic parameters
Results of the comparative analysis of the kinetic parameters estimated by the two systems during the walking test are shown in Table 4. No significant between-system differences were observed for any of the kinetic parameter means (all TOST P-values were < 0.001), but there was significantly greater variability of the unipodal CoP ML and AP, and “toemax load” measurements made by FeetMe than of those made by the reference method (Levene P-values < 0.05). The Pearson correlation coefficient values for the kinetic parameters ranged between 0.50 and 0.89 and indicated a low level of correlation between the measurements obtained from the two systems for unipodal CoP ML (0.50), but good-to-excellent correlations for unipodal CoP AP (0.81), heelmax load (0.88), and toemax load (0.89). The r2 and ICC values also showed that the agreement between the two devices was poor for unipodal CoP ML (r2: 0.23 and ICC: 0.4). These values were moderate-to-good for unipodal CoP AP, heelmax load, and toemax load (r2 values of 0.66, 0.77, and 0.80; and ICCs of 0.78, 0.87, and 0.89, respectively). The limits of agreement, bias, and the measures of MAE and MRAE for all kinetic parameters are also provided in Table 4.
Analysis of the bias of individual measurements on Bland-Altman plots (Fig. 3), revealed no clear trends in the differences for dynamic unipodal CoP AP dim (Fig. 3(a)), but indicated a potential trend toward a more significant underestimation of dynamic unipodal CoP ML dim for higher values (Fig. 3(b)). These observations were confirmed by linear regression analysis (Figure S6(a) and (b)). Linear regression analysis of the heelmax load and toemax estimates indicated that FeetMe tended to slightly overestimate values for these parameters for lighter participants and underestimate them for heavier ones (Figure S6(c) and (d)).
Bland-Altman analyses of kinetic parameters recorded by the FeetMe and MoCap/FP systems during walking. (a) Dynamic uCoP AP dim during a stance, (b) Dynamic uCoP ML dim during a stance, (c) Heelmax load and (d) Toemax load (n = 11,515 for all measurements). The solid line represents the mean differences between the systems, with the upper and lower lines (dotted lines) representing the limits of agreement. Each point represents the value for a given stride. uCoP AP = anterior-posterior of unipodal center of pressure, uCoP ML = medial-lateral of unipodal center of pressure, dim = dimensions, N = number.
Accuracy of Spatiotemporal parameters
Comparative analysis of the measurements of the spatiotemporal parameters estimated by the two systems are shown in Table 5. No statistically significant differences were observed between the two devices for any of the parameter means (all TOST P-values were < 0.001). The results of the Levene tests indicated that there were no significant differences in the variance for HS, TO, stride duration, stance duration, cadence, and step duration (P-values > 0.5). However, there was significantly greater variability of the stride length, velocity, swing duration, and single- and double-support duration parameters measured by FeetMe compared to the reference method (Levene P-values < 0.05; Table 5). The results indicated an excellent level of correlation for the spatiotemporal parameters between the two systems, as evidenced by the high Pearson correlation coefficients, r2, and ICCs, with values for all parameters except stride length of 0.97 or higher (Table 5). The Pearson correlation coefficient, ICC and r2 values (0.82, 0.73, and 0.67, respectively) for stride length indicated a good-to-moderate level of correlation between systems for this parameter. The limits of agreement, bias, and the measures of MAE and MRAE for all parameters are provided in Table 5.
Analysis of the Bland-Altman and linear regression plots revealed no strong trends for most parameters (examples of plots for stride length; stance duration, velocity, and double-support duration are shown in Fig. 4 and S3). However, for double-support duration, Bland-Altman analysis indicated a slight trend toward underestimation with longer durations (Fig. 4(d)). The linear regression analysis for double-support duration confirmed this conclusion (Figure S3(d)).
Bland-Altman per stride analyses of the FeetMe and MoCap/FP measurements of spatiotemporal parameters (a) stride length (StrideLen; stride n = 5250), (b) stride duration (StanceDur; stride n = 5250), (c) velocity (stride n = 5250) and (d) double-support duration (DoubleSupDur; stride n = 4782). The solid line represents the mean difference between the systems, with the upper and lower lines (dotted lines) representing the limits of agreement. Each point represents the value for a given stride.
Discussion
Principal results
This study investigating the FeetMe insoles for measuring the various static, kinetic, and spatiotemporal parameters used in gait assessments showed that most of the the FeetMe measurements were valid and accurate when compared to the high precision, laboratory-grade, gold standard MoCap/FP (Vicon/AMTI) reference system. Equivalence of the measurements made by FeetMe to the measurements made by the reference system was demonstrated for all parameters assessed. In addition, strong inter-device correlations were observed for most parameters. Nine of the 13 static parameters presented good (≤ 0.75-<0.90) or excellent (≥ 0.90) ICC values and almost half (46%) presented excellent Pearson coefficient values (≥ 0.90). Good-to-excellent levels of correlation were also observed for three of the four kinetic parameters. Finally, excellent levels of inter-device correlation were observed for all the spatiotemporal parameters assessed, except for stride length, which showed moderate-to-good agreement (ICC: 0.73 and Pearson coefficient:0.82). Our study therefore provided strong evidence of the validity and accuracy of the FeetMe insoles for measuring a wide range of gait parameters (with only a few exceptions), even when compared to the high precision MoCap/FP system.
The good-to-excellent levels of inter-device agreement for the majority spatiotemporal parameters measured in the current study were in the same range or higher than those reported in previous studies assessing the validity and reliability of FeetMe insoles compared to the gold standard GAITRite walkway system31,32,33. Indeed, similar to the results of the current study, excellent levels of agreement (coefficient values ≥ 0.95) were observed between FeetMe and the GAITRite reference system for spatiotemporal parameters in healthy volunteers in the study conducted by Jacobs et al.32. Good-to-excellent levels of correlation between the FeetMe and GAITRite measures for stance duration, velocity and cadence have also been observed in patients recovering from stroke (ICCs: ≥ 0.77 for stance duration and ≥ 0.95 for velocity and cadence)31, compared to excellent correlations (ICCs: ≥ 0.99) for these parameters in the current study. FeetMe insoles have also been shown to be a valid and reliable tool for measuring spatiotemporal gait parameters in both single and cognitive dual-task conditions when compared to the GAITRite system in patients with Parkinson’s disease33. In both task conditions, good-to-excellent correlation coefficients were obtained for stride-to-stride comparative analyses of velocity, cadence, step duration, stride duration, and stance duration (r2 values from 0.79 to 0.97), compared to excellent r2 values (0.97 to 1.0) for all these parameters in the current study.
It is important to note that a few parameters demonstrated a lesser degree of agreement. Namely, even though static bCoP AP center showed good level of correlation (Pearson coefficient: 0.76), the agreement was only moderate (ICC 0.64) and, above all, the lower boundary of ICC was as low as 0.2. Similar observation holds for Total weight and Weight by side, for which noticeable bias of nearly 9 kg and 4.5 kg, respectively, is evident. The impact of this bias depends on the measured weight, which may vary considerably between the patients. Nevertheless, the ICC and Pearson correlation coefficients for these 2 parameters remained high (Pearson coefficients: 0.88 and 0.86, ICCs: 0.81 and 0.79 respectively), still indicating good agreement despite the presence of a systematic error. While this systematic error represents a clear limitation of the studied device, it is important to note that, unlike the other parameters assessed, precise Total Weight measurement is widely available, easily accessible, and does not require specialized context. In this context, it becomes evident that this limitation does not decrease the device’s relevance for gait and posture analysis. Furthermore, two other static parameters, Weight ratio and Asymmetry showed only moderate level of correlation and moderate agreement (Pearson coefficients: 0.65 and ICCs: 0.67 for both parameters). As for dynamic parameters, uCoP ML stands out as the one that demonstrated poor agreement (ICC: 0.4) and correlation at the limit between moderate and low (Pearson coefficient: 0.5).
The only spatiotemporal parameter for which inter-device agreement between FeetMe and the MoCap/FPs was consistently lower than that reported in the previous studies comparing FeetMe and GAITRite was stride length. Indeed, excellent inter-device agreements for stride length (≥ 0.96) were observed in the FeetMe and GAITRite comparison studies31,32,33, compared to good (Pearson correlation: 0.82) to moderate agreement (ICC: 0.73 and r2: 0.67) in the current study. In addition, we observed a bias of 0.05 m for the FeetMe measurement of stride length compared to that measured by the MoCap/FP system, whereas a smaller bias of -0.013 m was reported in the prior study comparing FeetMe to the GAITRite system in healthy volunteers32. Even though the discrepancy between the findings of the two studies is noteworthy, the value of the bias itself accounts for only around 4% of a typical stride length38. The observed differences between the two results can be attributed to the fact that the algorithms of the FeetMe system were fine-tuned using the GAITRite system as a reference, aiming to closely align the measurements obtained with the two systems. Consequently, inconsistencies between the GAITRite and MoCap/FP systems likely provide a plausible explanation for the divergent conclusions between our work and the study conducted by Jacobs et al., particularly concerning the noticeable bias observed in our study. Investigating the origins of the bias in more detail could be valuable, as it may contribute to improving the device’s performance. This could also serve as a topic for a future study.
Comparison with prior work
The performance of the FeetMe insoles can also be compared to that of other recently developed wearable gait analysis systems. Typically, inertial sensors integrated into insoles (e.g., PODOSmart28) or worn on various body parts such as the feet, ankles, lower legs, or torso (e.g., Mobility Lab39, Physilog40, and Shimmer3 42) are used to estimate spatiotemporal parameters. Conversely, pressure sensors integrated into insoles (e.g., Pedar24, Insole3 29, LoadSol26, and Medilogic27) are used for estimating kinetic and static parameters. However, some wearable solutions, such as the FeetMe insoles or Insole3 devices41, incorporate both inertial and pressure sensor technologies. As a result, these integrated systems have the capability to provide a comprehensive set of parameters encompassing all the forementioned aspects of gait analysis.
Pressure-sensing insoles, such as Pedar and Medilogic, have been shown to provide reliable estimates of CoP when compared to FPs, although both systems were found to provide less accurate measurements for CoP ML than for CoP AP during walking or foot-rocking tests24,42. In our current study, FeetMe provided good-to-excellent estimates of all CoP parameters in the static test (all Pearson coefficients and ICC values between 0.73 and 1), but the level of correlation for CoP ML during walking was lower than that for CoP AP. As with other systems, this was likely due to the small magnitude of the dynamic CoP ML measurements (< 1 cm in some cases), as well as the limited spatial resolution available in the ML direction. Similar to FeetMe, both the LoadSol and Insole3 pressure-sensing insoles have been found to provide accurate estimates of vGRF parameters when compared to FP reference systems, with good-to-excellent correlations for the LoadSole insoles (ICCs: 0.80–0.99; 200 Hz model), and excellent correlations (ICCs: 0.94–0.99) for the Insole3 system26,41. Except for stride length, the FeetMe insoles also demonstrated similar or better performance than other inertial sensor-based systems for spatiotemporal parameters. Indeed, the excellent correlations between FeetMe and the MoCap/FP system (ICCs ≥ 0.93–1.0) for widely assessed parameters (e.g., cadence, velocity, stride duration) were similar to those observed previously for Physilog (r2: 0.93 for cadence and velocity), PODOSmart (ICCs: 0.92 for velocity to 0.99 cadence), and Shimmer3 (r2: 0.99 for velocity and 1.0 stride duration) compared to MoCap systems28,40,43, and for Mobility Lab (ICCs: 0.90 for velocity and 0.99 for cadence) compared to the GAITRite walkway39. Moreover, the findings from the current study indicate that FeetMe may outperform other inertial sensor-based devices for the measurement of some temporal parameters, particularly double-support duration, with moderate (r2: 0.76) and poor (r2: 0.1) correlations for this parameter being observed previously for Physilog and Mobility Lab39,40, compared to the very high levels of correlation observed for FeetMe (all correlations ≥ 0.97). In comparison to the Insole3, a system integrating both inertial and pressure sensors, the FeetMe insoles exhibited closely aligned performance in terms of measurement bias across various spatiotemporal parameters44. For instance, considering different walking paces, the Insole3 displayed biases ranging from − 0.16 to 0.203 strides per minute for cadence, -5 ms to 4 ms for stride duration, 0.42 m/s to 0.46 m/s for velocity, and 0.053 m to 0.057 m for stride length. Conversely, FeetMe demonstrated respective biases of 0.1 strides per minute, -3 ms, 0.03 m/s, and 0.05 m for the same parameters. It is thus noteworthy that, unlike the numerous other gait analysis tools discussed above, which predominantly incorporate a single type of sensor, the inclusion of both built-in inertial measurement units and pressure sensors in the FeetMe insoles enables an elevated level of performance. The demonstrated accuracy and reliability of this dual-sensor approach may make FeetMe a more suitable and simpler tool for simultaneously collecting data on a wider set of parameters during gait analysis.
Clinical relevance
The analysis of hallmark deviations and variability of these static, kinetic, and spatiotemporal gait parameters is a valuable tool for supporting the monitoring and diagnosis of numerous conditions. Spatiotemporal characteristics are known to be highly relevant in multiple disease fields. For instance, in Parkinson’s disease they can indicate disease progression, treatment responsiveness2, and early-stage disease45. They can also serve as functional biomarkers in amyotrophic lateral sclerosis (ALS)46, and can predict the risk of falling12 or cognitive decline47 in older adults. Assessment of spatiotemporal gait parameters can also be used to classify pathologies, as has been demonstrated in patients with adolescent idiopathic scoliosis15. Slow gait speeds and faster rates of gait speed decline have also been shown to be predictors of an increased risk of cardiovascular events in patients with myocardial infarction13 and of mortality in well-functioning older adults14. Kinetic gait parameters like GRF and CoP are also widely used in conditions such as ataxia, Parkinson’s disease, and osteoarthritis5,10,48,49. In particular, Alam et al. were able to successfully distinguish controls from patients with Parkinson’s disease with machine learning classifiers that use vGRF and CoP signals5. Likewise, Wiik et al. demonstrated a substantial vGRF asymmetry between affected and non-affected limbs in patients with knee osteoarthritis particularly evident at the weight acceptance peak10. These findings underscore the potential utility of vGRF measurements for the screening and rehabilitating of individuals at risk of developing, or already presenting, early-stage knee arthrosis. Assessing static balance is useful for quantifying stability and predicting the risk of falling in both the elderly population and patients with Parkinson’s disease50,51. The development of tools that can provide reliable and accurate measures of static, kinetic, and spatiotemporal gait parameters is therefore of great clinical relevance.
The Vicon MoCap system used as the reference in our study is widely regarded as a highly accurate and precise tool for measuring movement21,22,23. The demonstrated equivalence of the measurements estimated by FeetMe and the MoCap/FP system therefore provides strong evidence of a sufficient level of accuracy of the FeetMe data. The accuracy of quantitative measurements of gait parameters is important in clinical practice, as there is a need to ensure that the systems used are appropriate for detecting minimal clinically important differences (MCIDs). MCID values represent the threshold below which a change is considered insignificant from the patient’s or clinician’s perspective, and therefore indicate the smallest change that a device needs to be able to detect for practical use in clinical settings. MCID values for various gait parameters and diseases can be found in the literature. As an example, for comfortable gait velocity, MCID values in the literature range from 0.08 m/s to 0.26 m/s for patients with conditions such as hip fracture, stroke, and multiple sclerosis52. Considering the equivalence margin of delta of 0.04 m/s for gait velocity used in the current study, our findings clearly demonstrate that the FeetMe device would be able to accurately detect even subtle alterations in patients with these conditions.
In addition to monitoring gait in controlled clinical and laboratory settings, it is important to evaluate a patient’s gait in real-world settings to obtain results more representative of their day-to-day state and draw accurate conclusions. The European Medicines Agency (EMA) has even accepted the 95th percentile of gait velocity measured in real-world settings as a secondary endpoint53. It is therefore crucial to make gait analysis accessible to a wider range of clinicians, without requiring extensive training, and to enable these analyses to be performed in real-life conditions without sacrificing accuracy. The FeetMe insoles have several advantages over the reference MoCap/FP system in this context, most notably the FeetMe system is user-friendly and does not require any specialized skills or training for operation. Its integrated-sensor design allows the insoles to be placed in any type of footwear and be used in diverse conditions. Therefore, unlike the reference system, which typically requires space, specialized skills and significant setup times, and is typically restricted to laboratory or expert clinical settings, the FeetMe system can be easily and routinely used by clinicians working with pathologies that require regular gait analysis to provide systematic and long-term follow-up of patients. The system may as well enable monitoring of gait patterns during daily activities in real-world environment.
Strengths and limitations
The current study has several strengths. The inter-device validity approach used in the current study clearly demonstrated that aside for a limited number of parameters FeetMe system provides high level of accuracy. Compared to previous validation studies, the performance indicators assessed in the current study were calculated by comparing the values of parameters from FeetMe and from the reference device for each individual stride, rather than being based on per test averages. This approach allowed us to assess more accurately the variability of the measurements and any potential discrepancies between the two devices. In addition, the fact that we did not perform any prior calibration of the gait measurement system evaluated and the inclusion of a diverse range of participant ages and weights in the study, enhances the generalizability and representativeness of our study results. However, the present study has several limitations that need to be considered when interpreting our results and that could serve as a guide for future research. Our study only included healthy volunteers and did not consider age-matched individuals with pathological conditions, thus limiting the generalizability of our findings to healthy populations. Furthermore, the study did not evaluate the repeatability of the measurements or the influence of specialized footwear, such as orthopedic shoes. These aspects should be investigated in further study. Nevertheless, the promising results of our study suggest that the use of FeetMe in clinical applications is warranted. The system could become an integral part of standard care for conditions requiring quantified gait analysis for patient evaluation as well as for remote patient monitoring following hospital discharge or between on-site visits. It would therefore be worthwhile exploring the use of these wearable sensors in real-life settings outside of the laboratory, as this could provide valuable “real-world” insights for both healthy individuals and those with medical conditions.
Conclusions
This study established the validity of FeetMe device in measuring of static stability parameters, spatiotemporal and kinetic gait parameters by comparing it to the gold standard MoCap/FP system in a laboratory setting. A concurrent data collection with both systems was carried out in 37 healthy adults aged from 23 to 71 years, with acquired parameters compared stride by stride. In total, 28 gait and stability parameters were considered in this work. Equivalence between the two systems was demonstrated and good to excellent inter-device agreement and correlation together with low systematic errors were found across most of the parameters compared, with only a few exceptions.
These findings position FeetMe solution as a viable alternative to high-precision force platforms and optoelectronic motion capture systems for assessing balance and gait parameters. Combining this level of accuracy with user-friendly simplicity, FeetMe can help overcome typical barriers associated with traditional systems and help to make long-term monitoring of real-world gait during daily activities more commonplace.
The study outcomes therefore serve as a strong motivation for further research to offer additional evidence of the validity and repeatability of the device’s measurements especially among participants with impaired walking, balance, or cognition or those wearing orthotics or orthopedic shoes. Moreover, our results identify measurements that require further performance improvements. Finally, particular emphasis should be placed on investigating the performance of FeetMe insoles in real-world conditions across diverse settings and scenarios, including hospital environment as well as patients’ homes. In summary, FeetMe has the potential to emerge as an essential tool for clinicians working with pathologies that require regular gait analysis, as well as a highly relevant system for research purposes.
Data availability
The data sets generated during and analyzed during this study are available from the corresponding author on reasonable request.
Abbreviations
- ALS:
-
Amyotrophic lateral sclerosis
- AP:
-
Anterior-posterior
- BLE:
-
Bluetooth low energy
- bpm:
-
beats per minute
- CI:
-
Confidence interval
- CoP:
-
Center of pressure
- EMA:
-
European Medicines Agency
- FF:
-
Forefoot
- FP:
-
Force platform
- GCP:
-
Good clinical practice
- HL:
-
Heel
- ICC:
-
Intraclass correlation coefficient
- IMU:
-
Inertial Measurement Unit
- MAE:
-
Mean absolute error
- MCID:
-
Minimal Clinically Important Differences
- ML:
-
Medial-lateral
- MoCap:
-
Motion capture system
- MRAE:
-
Mean relative absolute error
- PC:
-
Personal computer
- Q-Q:
-
Quantile-quantile
- SD:
-
Standard deviation
- TOST:
-
Two one-sided t-tests
- vGRF:
-
vertical ground reaction force
- WL:
-
Weight on the left foot
- WR:
-
Weight on the right foot
References
Abrami, A. et al. Using an unbiased symbolic movement representation to characterize Parkinson’s disease States. Sci. Rep. 10, 7377. https://doi.org/10.1038/s41598-020-64181-3 (2020).
Agurto, C., Heisig, S., Abrami, A., Ho, B. K. & Caggiano, V. Parkinson’s disease medication state and severity assessment based on coordination during walking. PLoS One. 16, e0244842. https://doi.org/10.1371/journal.pone.0244842 (2021).
Goetz, C. G. et al. Movement disorder society-sponsored revision of the unified Parkinson’s disease rating scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov. Disord. 23, 2129–2170. https://doi.org/10.1002/mds.22340 (2008).
Hausdorff, J. M., Cudkowicz, M. E., Firtion, R., Wei, J. Y. & Goldberger, A. L. Gait variability and basal ganglia disorders: Stride-to-stride variations of gait cycle timing in Parkinson’s disease and Huntington’s disease. Mov. Disord. 13, 428–437. https://doi.org/10.1002/mds.870130310 (1998).
Alam, M. N., Garg, A., Munia, T. T. K., Fazel-Rezai, R. & Tavakolian, K. Vertical ground reaction force marker for Parkinson’s disease. PLoS One. 12, e0175951. https://doi.org/10.1371/journal.pone.0175951 (2017).
Filli, L. et al. Monitoring long-term efficacy of Fampridine in gait-impaired patients with multiple sclerosis. Neurology 88, 832–841. https://doi.org/10.1212/WNL.0000000000003656 (2017).
Wurdeman, S. R., Huisinga, J. M., Filipi, M. & Stergiou, N. Multiple sclerosis affects the frequency content in the vertical ground reaction forces during walking. Clin. Biomech. (Bristol Avon). 26, 207–212. https://doi.org/10.1016/j.clinbiomech.2010.09.021 (2011).
Kurtzke, J. F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 33, 1444–1452. https://doi.org/10.1212/wnl.33.11.1444 (1983).
Balaban, B. & Tok, F. Gait disturbances in patients with stroke. PM&R 6, 635–642. https://doi.org/10.1016/j.pmrj.2013.12.017 (2014).
Wiik, A. V., Aqil, A., Brevadt, M., Jones, G. & Cobb, J. Abnormal ground reaction forces lead to a general decline in gait speed in knee osteoarthritis patients. World J. Orthop. 8, 322–328. https://doi.org/10.5312/wjo.v8.i4.322 (2017).
Hulleck, A. A., Mohan, M., Abdallah, D., Rich, N. E., Khalaf, K. & M. & Present and future of gait assessment in clinical practice: Towards the application of novel trends and technologies. Front. Med. Technol. 4, 901331. https://doi.org/10.3389/fmedt.2022.901331 (2022).
Verghese, J., Holtzer, R., Lipton, R. B. & Wang, C. Quantitative gait markers and incident fall risk in older adults. J. Gerontol. Biol. Sci. Med. Sci. 64, 896–901. https://doi.org/10.1093/gerona/glp033 (2009).
Matsuzawa, Y. et al. Association between gait speed as a measure of frailty and risk of cardiovascular events after myocardial infarction. J. Am. Coll. Cardiol. 61, 1964–1972. https://doi.org/10.1016/j.jacc.2013.02.020 (2013).
White, D. K. et al. Trajectories of gait speed predict mortality in well-functioning older adults: The health, aging and body composition study. J. Gerontol. Biol. Sci. Med. Sci. 68, 456–464. https://doi.org/10.1093/gerona/gls197 (2013).
Boulcourt, S. et al. A gait functional classification of adolescent idiopathic scoliosis (AIS) based on spatio-temporal parameters (STP). Gait Posture. 102, 50–55. https://doi.org/10.1016/j.gaitpost.2023.03.002 (2023).
Parati, M. et al. The reliability of gait parameters captured via instrumented walkways: A systematic review and meta-analysis. Eur. J. Phys. Rehabil Med. 58, 363–377. https://doi.org/10.23736/s1973-9087.22.07037-x (2022).
Bilney, B., Morris, M. & Webster, K. Concurrent related validity of the gaitrite walkway system for quantification of the spatial and temporal parameters of gait. Gait Posture. 17, 68–74. https://doi.org/10.1016/s0966-6362(02)00053-x (2003).
Menz, H. B., Latt, M. D., Tiedemann, A., Mun San Kwan, M. & Lord, S. R. Reliability of the gaitrite walkway system for the quantification of temporo-spatial parameters of gait in young and older people. Gait Posture. 20, 20–25. https://doi.org/10.1016/S0966-6362(03)00068-7 (2004).
Webster, K. E., Wittwer, J. E. & Feller, J. A. Validity of the gaitrite walkway system for the measurement of averaged and individual step parameters of gait. Gait Posture. 22, 317–321. https://doi.org/10.1016/j.gaitpost.2004.10.005 (2005).
Mirek, E., Rudzinska, M. & Szczudlik, A. The assessment of gait disorders in patients with Parkinson’s disease using the three-dimensional motion analysis system vicon. Neurol. Neurochir. Pol. 41, 128–133 (2007).
Tsushima, H., Morris, M. E. & McGinley, J. Test-retest reliability and inter-tester reliability of kinematic data from a three-dimensional gait analysis system. J. Jpn Phys. Ther. Assoc. 6, 9–17. https://doi.org/10.1298/jjpta.6.9 (2003).
Windolf, M., Gotzen, N. & Morlock, M. Systematic accuracy and precision analysis of video motion capturing systems–exemplified on the Vicon-460 system. J. Biomech. 41, 2776–2780. https://doi.org/10.1016/j.jbiomech.2008.06.024 (2008).
McGinley, J. L., Baker, R., Wolfe, R. & Morris, M. E. The reliability of three-dimensional kinematic gait measurements: a systematic review. Gait Posture. 29, 360–369. https://doi.org/10.1016/j.gaitpost.2008.09.003 (2009).
Weizman, Y., Tan, A. M. & Fuss, F. K. Accuracy of centre of pressure gait measurements from two pressure-sensitive insoles. Proceedings 2, 277 (2018).
Parker, D., Andrews, J. & Price, C. Validity and reliability of the XSENSOR in-shoe pressure measurement system. PLoS One. 18, e0277971 (2023).
Renner, K. E., Williams, D. S. B. & Queen, R. M. The reliability and validity of the Loadsol((R)) under various walking and running conditions. Sens. (Basel). 19. https://doi.org/10.3390/s19020265 (2019).
DeBerardinis, J., Dufek, J. S., Trabia, M. B. & Lidstone, D. E. Assessing the validity of pressure-measuring insoles in quantifying gait variables. J. Rehabil Assist. Technol. Eng. 5, 2055668317752088. https://doi.org/10.1177/2055668317752088 (2018).
Ziagkas, E. et al. A novel tool for gait analysis: validation study of the smart insole PODOSmart((R)). Sens. (Basel). 21 https://doi.org/10.3390/s21175972 (2021).
Cramer, L. A. et al. Validity and Reliability of the Insole3 Instrumented Shoe Insole for Ground Reaction Force Measurement during Walking and Running. Sensors (Basel) 22, 2203. https://doi.org/10.3390/s22062203 (2022).
FeetMe® Insoles. Available online: https://feetmehealth.com/insoles/. Accessed on 02 April 2023.
Farid, L. et al. FeetMe(R) monitor-connected insoles are a valid and reliable alternative for the evaluation of gait speed after stroke. Top. Stroke Rehabil. 28, 127–134. https://doi.org/10.1080/10749357.2020.1792717 (2021).
Jacobs, D. et al. Evaluation of the validity and reliability of connected insoles to measure gait parameters in healthy adults. Sens. (Basel). 21 https://doi.org/10.3390/s21196543 (2021).
Parati, M. et al. Validation of pressure-sensing insoles in patients with Parkinson’s disease during overground walking in single and cognitive dual-task conditions. Sens. (Basel) 22. https://doi.org/10.3390/s22176392 (2022).
Granja Domínguez, A. et al. Study for the validation of the FeetMe® integrated sensor insole system compared to GAITRite® system to assess gait characteristics in patients with multiple sclerosis. PLoS One. 18, e0272596. https://doi.org/10.1371/journal.pone.0272596 (2023).
Kapteyn, T. S. et al. Standardization in platform stabilometry being a part of posturography. Agressologie 24, 321–326 (1983).
Savitzky, A. & Golay, M. J. E. Smoothing and differentiation of data by simplified least squares procedures. Anal. Chem. 36, 1627–1639. https://doi.org/10.1021/ac60214a047 (1964).
Koo, T. K. & Li, M. Y. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J. Chiropr. Med. 15, 155–163. https://doi.org/10.1016/j.jcm.2016.02.012 (2016).
Barker, S., Craik, R., Freedman, W., Herrmann, N. & Hillstrom, H. Accuracy, reliability, and validity of a Spatiotemporal gait analysis system. Med. Eng. Phys. 28, 460–467. https://doi.org/10.1016/j.medengphy.2005.07.017 (2006).
Morris, R. et al. Validity of mobility lab (version 2) for gait assessment in young adults, older adults and Parkinson’s disease. Physiol. Meas. 40, 095003. https://doi.org/10.1088/1361-6579/ab4023 (2019).
Homan, K., Yamamoto, K., Kadoya, K., Ishida, N. & Iwasaki, N. Comprehensive validation of a wearable foot sensor system for estimating Spatiotemporal gait parameters by simultaneous three-dimensional optical motion analysis. BMC Sports Sci. Med. Rehabil. 14, 71. https://doi.org/10.1186/s13102-022-00461-x (2022).
Cramer, L. A. et al. Validity and reliability of the insole3 instrumented shoe insole for ground reaction force measurement during walking and running. Sens. (Basel) 22. https://doi.org/10.3390/s22062203 (2022).
DeBerardinis, J., Neilsen, C., Lidstone, D. E., Dufek, J. S. & Trabia, M. B. A comparison of two techniques for center of pressure measurements. J. Rehabil Assist. Technol. Eng. 7, 2055668320921063. https://doi.org/10.1177/2055668320921063 (2020).
Kluge, F. et al. Towards mobile gait analysis: Concurrent validity and Test-Retest reliability of an inertial measurement system for the assessment of Spatio-Temporal gait parameters. Sens. (Basel). 17 https://doi.org/10.3390/s17071522 (2017).
Ganguly, A., Olmanson, B. A., Knowlton, C. B., Wimmer, M. A. & Ferrigno, C. Accuracy of the fully integrated Insole3’s estimates of Spatiotemporal parameters during walking. Med. Eng. Phys. 111, 103925. https://doi.org/10.1016/j.medengphy.2022.103925 (2023).
Del Din, S. et al. Gait analysis with wearables predicts conversion to Parkinson disease. Ann. Neurol. 86, 357–367. https://doi.org/10.1002/ana.25548 (2019).
Inam, S., Vucic, S., Brodaty, N. E., Zoing, M. C. & Kiernan, M. C. The 10-metre gait speed as a functional biomarker in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 11, 558–561. https://doi.org/10.3109/17482961003792958 (2010).
Byun, S. et al. Gait variability can predict the risk of cognitive decline in cognitively normal older people. Dement. Geriatr. Cogn. Disord. 45, 251–261. https://doi.org/10.1159/000489927 (2018).
Ienaga, Y., Mitoma, H., Kubota, K., Morita, S. & Mizusawa, H. Dynamic imbalance in gait ataxia. Characteristics of plantar pressure measurements. J. Neurol. Sci. 246, 53–57. https://doi.org/10.1016/j.jns.2006.02.002 (2006).
Martino, G. et al. Locomotor patterns in cerebellar ataxia. J. Neurophysiol. 112, 2810–2821. https://doi.org/10.1152/jn.00275.2014 (2014).
Patel, M., Pavic, A. & Goodwin, V. A. Wearable inertial sensors to measure gait and posture characteristic differences in older adult fallers and non-fallers: A scoping review. Gait Posture. 76, 110–121. https://doi.org/10.1016/j.gaitpost.2019.10.039 (2020).
Barbieri, F. A. et al. Postural control, falls and Parkinson’s disease: Are fallers more asymmetric than non-fallers? Hum. Mov. Sci. 63, 129–137. https://doi.org/10.1016/j.humov.2018.10.008 (2019).
Bohannon, R. W. & Glenney, S. S. Minimal clinically important difference for change in comfortable gait speed of adults with pathology: A systematic review. J. Eval Clin. Pract. 20, 295–300. https://doi.org/10.1111/jep.12158 (2014).
EMA. European Medicines Agency. Qualification opinion on stride velocity 95th centile as a secondary endpoint in Duchenne muscular dystrophy measured by a valid and suitable wearable device (2019). Available at: https://www.ema.europa.eu/en/documents/scientific-guideline/qualification-opinion-stride-velocity-95th-centile-secondary-endpoint-duchenne-muscular-dystrophy_en.pdf (Date accessed: 08 June 2023).
Acknowledgements
The authors express their gratitude to all volunteers who agreed to participate in this study. Additionally, the authors would like to thank Gilles Monneret for advising on statistical analysis, Gabriel Suazo for his support with data analysis and Drs Emma Pilling and Marielle Romet (Santé Active Edition – Synergy Pharm) for medical writing assistance and language editing.
Author information
Authors and Affiliations
Contributions
Conceptualization P.H., A.M., C.C. and R.P.; methodology P.H., A.M. and R.P.; formal analysis, P.H.; investigation P.H., R.C. and R.P; writing—original draft preparation, figures P.H. and A.M.; writing—review and editing A.M and R.P.; supervision C.C.; project administration P.H. and R.P. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The FeetMe company was a partner in this study: P.H. R.C. and A.M. were employees of the FeetMe company at the time of the study. The FeetMe company provided partial financial support for this study through access to their insoles. C.C. and R.P. have no conflicts of interest to disclose and have no link of interest to the FeetMe company.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Huang, P., Mostovov, A., Cohen, R. et al. Comparison of feetme insoles with a motion capture system coupled to force plates for assessing gait and posture. Sci Rep 15, 13476 (2025). https://doi.org/10.1038/s41598-025-96878-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-96878-8