Abstract
This study evaluated the factors predisposing women to a more substantial functional decline than men in the chronic phase of stroke. Sex differences in functional worsening and improvement, defined as an increase and decrease in one or more modified Rankin Scale scores between the 3-month and each assessment point throughout 5 years after ischemic stroke were examined using data from a multicenter prospective stroke registry in Japan. Logistic regression analysis was performed to estimate the risk of unfavorable outcomes in women after adjusting for potential confounders. The interactions between sex and confounders were also assessed. Among 6848 patients who survived for 5 years poststroke, 39.3% were female. Female survivors were more likely to experience unfavorable functional outcomes throughout 5 years post-stroke than male survivors. The higher risk of functional worsening in women than men was more apparent among patients aged > 75 years and those without limb weakness (P for interaction for age 0.04 and for limb weakness 0.03). Older female patients, frequently experiencing frailty and multimorbidity, should be targeted in poststroke interventions to reduce the burden of long-term disability after stroke. Female patients without apparent motor impairment might also benefit from physical activity programs to maintain muscle strength.
Similar content being viewed by others
Introduction
Although age-standardized stroke mortality has declined over the past decades, stroke-related disability remains a major public health concern worldwide1,2. The burden of disability among female stroke survivors has drawn more attention3,4,5. Women experience their initial stroke at an older age and have a higher lifetime risk of stroke than men since they live longer than men and stroke incidence increases with age3,6. Moreover, women have worse functional outcomes after stroke than men4,5,7,8,9,10.
Many studies on sex differences in functional outcomes assessed the functional status at one time-point rather than on functional status change after stroke8,9,10. We recently reported that the female sex was associated with a higher risk of functional decline in the chronic phase of ischemic stroke after adjusting for age, prestroke disability, stroke subtype, and stroke severity11. In previous studies, including ours, no sex difference was observed in stroke recurrence8,10,11. Therefore, other biological, clinical, and social factors might be involved in the sex difference in poststroke functional decline. Female patients tend to be older, harbor multiple comorbid diseases, and experience more severe neurological impairment due to stroke7,12. However, the extent to which these factors contribute to poststroke functional decline in women remains unclear. One possible explanation is that female patients with stroke are generally frailer, which may limit their capacity for recovery compared to male patients9. Given the estimated rapid increase in female patients with stroke in aging populations7, elucidating factors related to female patients’ long-term functional decline is urgently needed to identify potential targets for postacute interventions to reduce sex-related disparities.
This study aimed to explore clinical factors associated with a higher risk of long-term functional decline poststroke in women. We analyzed the functional status change in 3-month ischemic stroke survivors up to 5 years poststroke and examined patient characteristics’ influence on sex differences in functional decline in the chronic phase of stroke using a multicenter hospital-based prospective registry of acute stroke in Japan.
Results
Sex differences in baseline characteristics
Baseline characteristics were assessed in 6848 patients who were followed up for the 5 years after ischemic stroke onset and survived for the 5 years poststroke. The mean (standard deviation) age was 69.0 (12.0) years, and 2691 patients (39.3%) were females. Significant sex differences were observed in the unadjusted comparisons (Table 1).
We first compared the clinical factors that might contribute to sex differences in the long-term functional status. Women were older and had lower BMI than men. Women had dyslipidemia and chronic kidney disease more frequently than men, whereas men had diabetes mellitus, smoking and drinking habits, ischemic heart disease, and a history of stroke more frequently than women. Women experience a severe stroke more frequently than men. Regarding neurological impairment, arm, leg, and limb weakness and aphasia were more prevalent in women than in men. Women received antidyslipidemic drugs more frequently but less frequently received antidiabetics and antiplatelets at discharge than men. Functional dependency at 3 months poststroke was more frequent in women than in men.
Sex differences in functional status during the 5 years poststroke
The proportion of dependent patients was consistently higher in women than in men throughout the 5 years poststroke (Fig. 1). A higher risk of disability in women was still observed after adjusting for multiple confounders (Table S1). Using the definition of functional disability as an modified Rankin Scale (mRS) score of 2‒5 instead of 3‒5 did not alter the results (OR of functional dependency at 5 years, 1.62; 95% CI 1.36–1.94; P < 0.001).
Functional status change during the 5 years poststroke by sex. The functional status was evaluated using the modified Rankin Scale (mRS). The proportion of patients with each mRS score among surviving patients at each follow-up time point is shown according to sex.
We further investigated whether the poor functional status in women was because of accelerated deterioration or hindered improvement in functional status after stroke. Consequently, functional worsening was more frequent, and functional improvement was less frequent in women than in men at all follow-up time points. These sex differences in functional worsening and improvement remained significant, even after adjusting for possible confounders (Table 2). Similar results were observed when applying different definitions of poor and good functional outcomes (Table S2).
Factors related to worse functional status in women
Subgroup analyses were conducted to explore factors related to worse poststroke functional status in women. The higher risk of functional deterioration in women than in men was more pronounced among patients ≥ 75 years. Additionally, a significant interaction was present between sex and motor impairment; the increased risk of functional worsening in women was only found in patients without limb weakness and not found in those with limb weakness. A female disadvantage was observed among normal-weight and obese patients rather than among underweight patients, although the P for interaction did not reach statistical significance. Similarly, a higher risk of functional worsening in women was found in patients who were independent at 3 months poststroke rather than in those who had already been dependent at 3 months poststroke, although no significant interaction was detected (Table 3). Regarding functional improvement, effect modification by age was found; a lower OR for functional improvement was observed only in older patients. No significant interactions were observed between sex and the other factors (Table 4). Regarding the 5-year functional dependency, the effect modification by age was similar to that for functional worsening. (Table S3). The association between sex and long-term functional status change was further evaluated using propensity score-matched analysis to exclude the effect of age and three-month functional status. A higher risk of functional worsening and a lower chance of functional improvement in female patients were consistently observed in propensity-score matched cohorts (Table S4).
No sex difference was observed in stroke recurrence during the 5-year poststroke period with 13.7% in female patients and 12.7% in male patients. Notably, the association between female sex and a higher risk of functional worsening persisted even in a cohort of patient who did not experience stroke recurrence during the 5-year poststroke period (Table S5).
Discussion
This study examined data from a multicenter, hospital-based, prospective registry of patients with acute stroke and found that female survivors of ischemic stroke experienced more frequent functional worsening and less frequent functional improvement between 3 months and 5 years poststroke than males. These sex differences remained significant, even after adjusting for confounding factors. An effect modification by age was found; the higher risk of functional worsening in women than men was more marked in patients ≥ 75 years than in younger ones. Heterogeneity according to neurological impairment was also found; the increased risk of functional worsening in women was significant in patients without limb weakness but not significant in those with limb weakness.
Here, long-term functional outcomes after stroke were worse in women than in men, which agrees with previous studies4,5,7,8,9. Some studies reported older age, prestroke disability, and higher stroke severity as possible causes of worse functional outcomes in women5,8,9. However, in our study, female sex remained associated with a higher risk of poor functional outcomes even after considering age and functional status at 3 months poststroke, which reflected prestroke functional status and stroke severity. The existing literature suggests that no sex difference was found in stroke recurrence in the long-term8,10,11, and the same finding was confirmed in our analysis. Therefore, recurrence does not appear to cause worse outcomes in women. Additionally, our study demonstrated that women experienced more frequent functional decline than men after surviving for 3 months following an ischemic stroke, and this sex difference was significant even among patients who were independent at 3 months poststroke. Our findings imply that a good functional status after the acute phase of stroke does not necessarily guarantee good long-term outcomes in female patients. Previous studies have reported lower mortality among female patients at three months poststroke and throughout the subsequent 5 years8,9. Taken together, female patients are more likely to survive but with functional disability rather than succumb to the disease, compared with their male counterparts.
We also found that the association between the female sex and functional worsening was more pronounced in patients without limb weakness at discharge than in those with limb weakness. Probably, the sex difference in functional outcome is apparent when neurological impairment caused by the index stroke event is not devastating, and functional recovery is possible, whereas the difference between the sexes becomes less clear when the stroke is significantly severe to leave little possibility of recovery. Given the finding that even female patients who survive stroke events without lasting motor impairment experience functional decline in the following years more frequently than male patients, reducing stroke severity by acute stroke treatment may be insufficient to diminish sex differences in long-term functional outcomes. Therefore, enhancing the functional recovery of female patients with stroke in postacute care by promoting focused interventions is crucial. Rehabilitation programs aiming at maintaining muscle strength should be provided even for female patients without apparent motor impairment. Managing comorbidities that impair physical function, such as musculoskeletal and cardiovascular diseases, is also important.
The older age of female patients with stroke can be an obstacle to realizing functional recovery13. We found that sex-related differences in functional worsening were more pronounced in older patients than in younger ones. A study also reported that higher age was associated with poor functional recovery after ischemic stroke compared to younger age in female patients, but not in male patients14. The reduced recovery capacity in women might be related to frailty. Prior studies reported higher prevalence and greater degree of frailty in women compared to men for any age group15,16. The loss of physiological reserve due to frailty may impair the ability of female patients to cope with a stressors, such as stroke16,17. Evidence suggests that frailty negatively impacts stroke recovery18, and one study found that frailty status was associated with the functional decline in female patients rather than in male patients19. This finding confirms that frail women with reduced resilience to stressors, such as a stroke event, are more prone to poststroke functional deterioration than men. Therefore, more attention should be given to older frail female patients in organizing poststroke health-care services as targeting this vulnerable population could help prevent long-term disability, thereby reducing the burden on both patients and healthcare systems.
This study had some limitations. First, some patients were lost to follow-up during the 5-year poststroke period, which may have caused a selection bias (Table S6). However, the percentage of lost patients (13%) was relatively low compared to other studies on long-term poststroke outcomes. Additionally, excluding patients who died during the 5-year poststroke period may have resulted in a study cohort with milder stroke and less neurological impairment (Table S7); therefore, our findings may not be generalizable to all patients with stroke. Second, mRS was used to assess long-term functional status changes poststroke. Although some concerns over interrater variability have been presented on mRS20, trained staff assigned the mRS scores using a standardized form to improve the reliability of mRS in our study21. Third, the possibility of residual confounding could not be precluded. In particular, we could not examine the role of socioeconomic factors, including educational level, living status, and access to healthcare services, due to insufficient information. One study suggested that living alone may negatively impact acute stroke care outcomes; however, its long-term effect remains unclear22. Future research is warranted to properly addressing socioeconomic factors. Lastly, because of the FSR design, our study patients may not represent all patients with stroke; further studies in other settings should confirm our findings’ generalizability.
In conclusion, female patients with stroke were more likely to experience functional decline between 3 months and 5 years poststroke than male patients. Functional worsening in women was more pronounced in older patients than in younger ones and patients with little motor impairment than in those with severe impairment. These findings suggest that older female patients have a lower chance of recovery poststroke, and this poorer recovery in women is not due to stroke-induced neurological deficits, but rather due to frailty, which compromises resilience to debilitating diseases, including stroke. Targeted postacute interventions for female stroke survivors, such as exercises to maintain muscle strength even in those without apparent motor impairment and management of comorbidities that limit physical activities, are urgently needed to address the growing burden of stroke in a rapidly aging society.
Methods
Study design and setting
This study used data from the Fukuoka Stroke Registry (FSR), a multicenter, hospital-based, longitudinal acute stroke registry in Fukuoka, Japan (UMIN Clinical Trials Registry 000,000,800), as detailed elsewhere22,23. The FSR prospectively enrolled patients with acute stroke admitted to seven participating hospitals (see Appendix) within 7 days of stroke onset. Each participating hospital obtained ethics committee approval for the FSR (Kyushu University Institutional Review Board for Clinical Research, 22,086–00; Kyushu Medical Center Institutional Review Board, R06-03; Clinical Research Review Board of Fukuokahigashi Medical Center, 29-C-38; Fukuoka Red Cross Hospital Institutional Review Board, 629; St. Mary’s Hospital Research Ethics Review Committee, S13-0110; Steel Memorial Yawata Hospital Ethics Committee, 06–04-13; Kyushu Rosai Hospital Institutional Review Board, 21–8). Stroke was defined as the sudden onset of nonconvulsive and focal neurological deficits persisting for ˃24 h. Ischemic stroke diagnosis was confirmed using computed tomography and/or magnetic resonance imaging. Written informed consent was obtained from all participants during their hospital stay. Permission was obtained from the next of kin or legal guardian as a proxy for patients who could not consent. The present study was performed according to the Ethics of Clinical Research (Declaration of Helsinki).
Study population
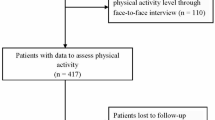
Overall, 11,487 patients with ischemic stroke were enrolled in the FSR between June 2007 and September 2016. Here, we evaluated sex differences in poststroke outcomes among patients who survived for 3 months after an ischemic stroke to focus on functional status changes in the chronic phase of stroke. Accordingly, 10,881 patients who survived for 3 months and for whom data at 3 months poststroke were available were included in this study. During the follow-up period between 3 months and 5 years after the index stroke, 1411 patients were lost to follow-up, and 2622 died. The remaining 6848 patients were included in the functional status analyses. Figure S1 shows a flow diagram of the patient selection process.
Clinical assessment
Information on baseline characteristics, such as demographics, risk factors, comorbidities, body mass index (BMI), and history of stroke before the index stroke onset, was collected on admission or during hospitalization for the index stroke. The definitions of risk factors and comorbidities have been described in detail previously23,24. The mechanism of ischemic stroke was classified according to the criteria of the Trial of Org 10,172 in Acute Stroke Treatment.25 Information on antihypertensive, antidiabetic, and antidyslipidemic drugs, antiplatelets, and anticoagulants usage at discharge was recorded.
Several factors affecting long-term functional recovery poststroke were dichotomized and used as variables of interest, including age, body weight, neurological impairment, and functional status at 3 months poststroke. Old age was defined as age ≥ 75 years at the onset of stroke. Underweight and obesity were defined as having BMI < 18.5 and ≥ 25 kg m2, respectively, according to the criteria for Asians proposed by the regional office for the Western Pacific Region of the World Health Organization26. Neurological impairment, such as arm and/or leg weakness and aphasia, was assessed using the National Institutes of Health Stroke Scale (NIHSS) at discharge by trained neurologists. Arm weakness, leg weakness, limb weakness, and aphasia were defined as scores of ≥ 1 for the NIHSS item of right arm or left arm motor function or both, the NIHSS item of right leg or left leg motor function or both, at least one item among the NIHSS items of the four limbs’ motor function, and the NIHSS item of language, respectively. Functional status at 3 months poststroke was measured using the mRS27. Patients with an mRS score of 3‒5 were considered dependent5,9.
Outcome measurement
The main study outcomes were functional worsening and improvement during the 5 years poststroke. Functional worsening and improvement were defined as an increase and decrease in one or more mRS scores between the 3-month and each assessment point poststroke, respectively. Patients with an mRS score of 5 at 3 months poststroke, in whom worsening would result in death, were excluded from the analysis of functional worsening. Patients with an mRS score of 0 at 3 months, for whom further improvement could not be expected, were excluded from the functional improvement analysis. Additionally, functional dependency, defined as an mRS score of 3–5 at each assessment point over 5 years, was evaluated.
Study participants were interviewed by telephone at 3, 6, and 12 months from stroke onset and yearly subsequently for up to 5 years in person or, if not possible, through their caregivers. Information on survival status and stroke recurrence, including ischemic and hemorrhagic stroke, was obtained. The functional status of surviving patients at each follow-up time point was assessed using a structured interview form for the mRS. The interviews were performed by trained research nurses blinded to patients’ clinical backgrounds.
Statistical analysis
Regarding baseline variables, comparisons between men and women were performed using the χ2 test for categorical variables and the t-test or the Wilcoxon rank-sum test for continuous variables, as appropriate. Regarding poststroke outcomes, the results were first stratified by sex, and logistic regression analyses were subsequently used to examine the effect of sex on outcomes. Odds ratios (ORs) and 95% confidence intervals (CIs) for each outcome in women were estimated using men as a reference. Potential confounding factors were selected based on clinical relevance and the univariate analysis results. Multivariate analyses included age, BMI, hypertension, diabetes mellitus, dyslipidemia, atrial fibrillation, smoking, drinking, ischemic heart disease, chronic kidney disease, history of stroke, ischemic stroke subtype (cardioembolism vs. non-cardioembolism), medications at discharge, and the mRS score at 3 months poststroke. The functional status at 3 months poststroke reflects prestroke disability and stroke severity. No data were missing on the items used in the analyses.
Effect modifications according to age, body weight, 3-month functional status, and neurological impairment were also assessed. Interaction was evaluated by adding the interaction term sex × the variable of interest.
Propensity score-matched analysis was performed to evaluate the association between sex and long-term functional status change without bias caused by sex differences in age and three-month functional status. Logistic regression modeling was used to calculate propensity scores incorporating age and functional status at three months poststroke. Female and male patients underwent one-to-one nearest neighbor (greedy type) matching of the standard deviation of the propensity score logit with a caliper width of 0.25. Matching was performed without replacement and unpaired cases and controls that did not meet the matching criteria were excluded. Each propensity score-derived matched pair was assigned a unique pair identification number, and 1043 matched-pair identification numbers were selected.
Sensitivity analysis was performed using an mRS score of 2–5 instead of 3–5 as the definition of functional dependency. In addition, sensitivity analyses were performed to evaluate the long-term functional status change using different outcomes (e.g., poor and good). Poor functional outcome was defined as an mRS score of 3–5 at 5 years poststroke among patients with an mRS score of 0–2 at three months poststroke and good functional outcome was defined as an mRS score of 0–3 at 5 years poststroke among patients with an mRS score of 4–5 at three months poststroke. Furthermore, sex differences in long-term functional change were examined in a cohort of patients without stroke recurrence during the 5-year poststroke period.
Patient characteristics were compared between the lost and studied patients and between patients who died and those studied to assess for potential selection biases.
All statistical analyses were conducted using Stata 15 software (StataCorp LLC, College Station, TX, USA). All tests were two-sided, and statistical significance was considered at P < 0.05.
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to the sensitive nature of the data collected for this study but are available from the corresponding author on reasonable request.
References
Barker-Collo, S. et al. Sex differences in stroke incidence, prevalence, mortality and disability-adjusted life years: Results from the Global Burden of Disease Study 2013. Neuroepidemiology 45, 203–214 (2015).
Feigin, V. L., Norrving, B. & Mensah, G. A. Global burden of stroke. Circ Res. 120, 439–448 (2017).
Bushnell, C. D. Stroke and the female brain. Nat. Clin. Pract. Neurol. 4, 22–33 (2008).
Gall, S. L., Tran, P. L., Martin, K., Blizzard, L. & Srikanth, V. Sex differences in long-term outcomes after stroke: Functional outcomes, handicap, and quality of life. Stroke 43, 1982–1987 (2012).
Gall, S. et al. Focused update of sex differences in patient reported outcome measures after stroke. Stroke 49, 531–535 (2018).
Kelly-Hayes, M. et al. The influence of gender and age on disability following ischemic stroke: The Framingham study. J. Stroke Cerebrovasc. Dis. 12, 119–126 (2003).
Reeves, M. J. et al. Sex differences in stroke: Epidemiology, clinical presentation, medical care, and outcomes. Lancet Neurol. 7, 915–926 (2008).
Renoux, C. et al. Confounding by pre-morbid functional status in studies of apparent sex differences in severity and outcome of stroke. Stroke 48, 2731–2738 (2017).
Phan, H. T. et al. Factors contributing to sex differences in functional outcomes and participation after stroke. Neurology 90, e1945–e1953 (2018).
Medlin, F. et al. Sex differences in acute ischaemic stroke patients: clinical presentation, causes and outcomes. Eur. J. Neurol. 27, 1680–1688 (2020).
Irie, F. et al. Sex differences in long-term functional decline after ischemic stroke: a longitudinal observational study from the Fukuoka Stroke Registry. Cerebrovasc. Dis. 52, 1–8 (2023).
Bonkhoff, A. et al. Female Stroke: sex differences in acute treatment and early outcomes of acute ischemic stroke. Stroke 52, 406–415 (2021).
Paolucci, S. et al. Is sex a prognostic factor in stroke rehabilitation? A matched comparison. Stroke 37, 2989–2994d (2006).
Hankey, G. J. et al. Rate, degree, and predictors of recovery from disability following ischemic stroke. Neurology 68, 1583–1587 (2007).
Gordon, E. H. et al. Sex differences in frailty: a systematic review and meta-analysis. Exp. Gerontol. 89, 30–40 (2017).
Park, C. et al. The Science of frailty: sex differences. Clin. Geriatr. Med. 37(4), 625–638 (2021).
Duncan, P. W. et al. Health status of individuals with mild stroke. Stroke 28, 740–745 (1997).
Evans, N. R. et al. The importance of frailty in stroke and how to measure it. Stroke 56(1), e8–e11 (2025).
Winovich, D. T. et al. Factors Associated with ischemic stroke survival and recovery in older adults. Stroke 48, 1818–1826 (2017).
Quinn, T. J. et al. Reliability of the modified Rankin Scale: a systematic review. Stroke 40, 3393–3395 (2009).
Shinohara, Y. et al. Modified Rankin scale with expanded guidance scheme and interview questionnaire: Interrater agreement and reproducibility of assessment. Cerebrovasc. Dis. 21, 271–278 (2006).
Reeves, M. J. et al. Impact of living alone on the care and outcomes of patients with acute stroke. Stroke 45, 3083–3085 (2014).
Kamouchi, M. et al. Prestroke glycemic control is associated with the functional outcome in acute ischemic stroke: the Fukuoka Stroke Registry. Stroke 42, 2788–2794 (2011).
Kumai, Y. et al. Proteinuria and clinical outcomes after ischemic stroke. Neurology 78, 1909–1915 (2012).
Adams, H. P. Jr. et al. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in acute stroke treatment. Stroke 24, 35–41 (1993).
Anuurad, E. et al. The new BMI criteria for Asians by the Regional Office for the Western Pacific Region of WHO are suitable for screening of overweight to prevent metabolic syndrome in elder Japanese workers. J Occup Health 45, 335–343 (2003).
Banks, J. L. & Marotta, C. A. Outcomes validity and reliability of the modified Rankin scale: Implications for stroke clinical trials: a literature review and synthesis. Stroke 38, 1091–1096 (2007).
Acknowledgements
We thank all the FSR investigators and their hospitals for participating in this study and all the clinical research coordinators (Hisayama Research Institute for Lifestyle Diseases) for their help in obtaining informed consent and collecting clinical data.
Funding
This study was supported by the Japan Society for the Promotion of Science (JSPS), KAKENHI (Grant Numbers: JP21K19648, JP21H03165, JP21K10330, JP22K10386, and JP24K02669) from the Japanese Ministry of Education, Culture, Sports, Science and Technology..
Author information
Authors and Affiliations
Consortia
Contributions
F.I. contributed to the design and conception of the study, literature review, analysis and interpretation of the data, and drafting of the manuscript. K.N. contributed to the design of the study, and analysis and interpretation of the data. R.M. contributed to the design and conception of the study, acquisition, analysis and interpretation of the data, and critical revision of the manuscript for intellectual content. Y.W., and T.A. participated in the design and conception of the study, interpreted the data, and revised the manuscript for intellectual content. T.K. participated in the design and conception of the study, supervised the running of the F.S.R., and revised the manuscript for intellectual content. M.K. supervised the study, interpreted the data, and critically revised the manuscript for intellectual content. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Irie, F., Nakamura, K., Matsuo, R. et al. Factors related to sex differences in long-term functional decline after acute ischemic stroke. Sci Rep 15, 13400 (2025). https://doi.org/10.1038/s41598-025-97668-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-97668-y