Abstract
Polycystic Ovarian Disease (PCOD) is among the most prevalent endocrine disorders complicating the health of innumerable women worldwide due to lack of diagnosis and appropriate management. The diagnosis of PCOD, along with proper classification with the help of ultrasound imaging, would be of immense importance for early intervention and timely management of the condition. However, most of the existing approaches suffer from lots of problems, including low accuracy and capability in feature extraction, and may also be resilient to noise; it can further delay or lead to a wrong diagnosis. The main objective of this paper is to address these important issues by proposing a deep learning model, Holographic Convolutional Dense Network (Coco-HoloNet) that will be tailored for the precise detection and classification of PCOD in ultrasound images with high accuracy. These are multi-fold contributions which focus on improvement in diagnostic accuracy by overcoming the various limitations of conventional approaches. CoCo-HoloNet is using a layered architecture by integrating convolutional layers, dense blocks, and pooling strategies that leverage capturing and extraction of significant features from the input effectively. More importantly, the model is also embedded with the Tangent-Runner Adaptive Optimization (TRAdO) technique, which dynamically calculates the regularization parameters to overcome overfitting problems and improves the generalization capability of the model. The approach not only ensures the richest possible feature representation, but it also results in outstanding improvements within the performance measures of a model, such that the accuracy rate exceeds 99%. Further experimentation with CoCo-HoloNet on an extended Kaggle PCOD ultrasound image dataset proves its effectiveness by reporting higher precision, recall, and F1-scores than those obtained by state-of-the-art existing methods.
Similar content being viewed by others
Introduction
Ovarian cysts are fluid or tissue-filled sacs that develop either on the surface or within an ovary1. Most women will find they get them during a typical reproductive cycle and are harmless, with many of them actually forming due to ovulation. This might happen if a follicle in the ovary fails to release an egg, or if the sac of the follicle doesn’t dissolve following the release of the egg. A cyst then forms when that sac balloons up2,3. These functional cysts are relatively common and often go away on their own. Many are never diagnosed because they don’t create symptoms and resolve with the menstrual cycle. But other types of ovarian cysts may not dissolve and could even develop health issues if they ever increase in size or stay overtime. Whereas most ovarian cysts are benign, about 20% may experience non-resolving cysts. These may be more serious types of cysts that can result in potential complications such as the rupture of the cyst, torsion of the ovaries, and malignant changes even, though rarely. However, a large or persistent cyst may require surgical resection, particularly if it becomes symptomatic-that is, when it results in pain, periodic irregularities, or pressure symptoms on other surrounding organs4. This is because neglected cysts can give rise to very serious health complications. These latter complications include big cysts leading to torsion of the ovary which cuts off its blood supply-a real emergency. Additionally, though very rare, some ovarian cysts can proceed to malignancies, with possibilities of ovarian cancers. Hence, timely diagnosis and management are very important in preventing such complications. Other pathologies regarding female reproductive organs, such as uterine fibroids, may co-exist; ovarian cysts further complicate the health status of a woman5,6,7. These growths, usually benign, develop in the muscular wall of the uterus, where symptoms could also be much like those of ovarian cysts themselves: heavy menstrual bleeding, abdominal bloating, and pelvic pain. Because symptoms tend to run across an over-lapping path, it may not be easy to say exactly which one is which; proper medical imaging should, however, take place, including an ultrasound exam. In the case of chronic pelvic pain-that which always arises on one or the other side of the pelvis-it could well be indicative that one has an ovarian cyst or yet some other underlying condition8,9. These pains may be dull and ache or sharp and sharp, at times made worse by menstruation or intercourse. Besides, ovarian cysts can also heighten symptoms of PMS with mood swings, fatigue, and bloating complicating even the most trivial activities for some women.
The most dependably effective method in the examination of ovarian cysts is ultrasound imaging10,11. This helps the doctor pick up the size of the cyst, the nature of the cyst-whether it is at an exact position that may become an imminent danger. The line of treatment will also vary from watchful waiting and monitoring to more active modes of intervention, including hormonal treatment or surgery, depending on the nature of the cyst12. If benign cysts have established themselves and do not give rise to symptoms, they are merely followed up for periods of time, awaiting follow-up appointments that make certain the cyst does not increase in size nor creates complications. Surgery can be recommended in order to have the cyst removed before more serious health issues set in in case of extreme pain emanating from it, or when there is a strong possibility of rupture13,14. Extended surgery for malignant or suspected malignant cysts may be performed, which even includes removal of one or both ovaries. Ovarian cysts, though a common feature amongst women, often resolve spontaneously without the need for treatment; however, some females do present with such cysts that prove to be persistent and require management15,16. Management of ovarian cysts, especially large ones that are painful or may be associated with the presence of other conditions such as uterine fibroids, focuses on early detection and proper treatment. Treatment for this will chiefly be very important through the frequency of visiting the doctor and a series of ultrasound imaging that monitor the development of cysts into serious health concerns such as ovarian cancer, infertility, and chronic pain.
Patients with ovarian cysts often feel a sudden heaviness or weight in the lower abdomen or pelvic region unannounced. The added pressure, especially during such periods, makes the sensation more unbearable because of physical activities or exercise. Weight or pressure in that area of the body remains and, at times turns into sharp or dull pain, which seems to persist even beyond menopause. It is more important for post-menopausal women because their condition could indicate something worse that has been building up, such as a tumor or cyst that has not resolved itself17,18. For such cases, doctors say the pain could be severe with the uterine contractions, involuntarily constricting the muscles of the uterus in response to tumors or cysts. The result could be sharp, acute pain. Tumors or cysts in ovaries may twist while growing in size. This condition of twisting is known as ovarian torsion. This twisting cuts the blood supply of the ovary; hence, tissues are deprived of oxygen and nutrients, increasing not just the pain but turning it into a medical emergency that needs immediate intervention19,20. Moreover, ovarian torsion may be life-threatening, and if unmanaged, would lead to tissue death due to inadequate blood supply leading to critical infection and complications. If one has sharp pains in the pelvic area, along with nausea, vomiting, or fever, one must seek immediate emergency medical attention urgently to take action on the condition21. The graver the condition may be, with possible irreversible damage to the reproductive organs, such as removal of the ovary, the more one waits.
Bloating of the stomach at times is a very vague symptom, often subtle, sometimes related to ovarian cysts, but all the same, depending on the size and nature of the cyst involved22. In many instances, the sizes of the ovarian cysts are usually relatively small, often less than 10 centimeters in length, hence may not easily be noticed causing bloating or discomfort to the woman. These may at times reach the size of a watermelon or even greater, with considerable enlargement and discomfort of the abdomen. Such a growth may give one a sensation of fullness or heaviness of the abdomen as though one were gaining weight23. Abdominal expansion might easily be confused with general weight gain or bloating of the food variety, but that would be discrediting the fact that unexplained bloating-after all other factors have been taken into consideration-which is placed precisely in the abdomen, could very well mean the presence of a cyst or tumor on the ovaries. If a woman’s body suddenly gains weight, most especially in the abdominal part of her body, without any lifestyle, diet, and even activity level changes whatsoever, this may signify that something in the abdomen is growing abnormally. This may be true, especially if the sudden weight gain is accompanied by other symptoms such as pains, swelling, or pressure in the abdominal region24,25. It can be justified that the heaviness or pressure that may be felt in the abdomen is due to the fact that the physical structure of the cyst or tumor occupies space and may press on other surrounding organs26. Relationally, the bigger the cyst is, the more pressure it exerts on the abdominal cavity; sometimes, it may even feel like constipation, as the expanding mass compresses the intestines or bowels. This pressure may also reach to the level of the bladder thus giving most frequently urination or difficulties in urination, though these are mostly mild symptoms and could be passed off as normal digestive or urinary problems.
Sometimes, some cysts or tumors can appear even in the uterus, such as uterine fibroids, alongside other cystic features of the uterus, which may further cause abdominal heaviness. Such cysts or fibroids may inflate the abdomen and lead to a feeling of bloating with continued sensations of fullness or discomfort. As far as the cyst or fibroid being more on one side of the uterus, this feeling can easily become one-sided in the pelvis27. The one-sided pain or discomfort is a sign that such pains usually indicate the time when the cyst finally formed and pressed upon the pelvic wall or regional structure, creating sharp or dull pain limited to that region. These symptoms ought to be kept in mind by every woman, so that she could take appropriate measures in case these were long-lasting and for no apparent reason. When there is bloating or heaviness of the abdomen for more than two weeks, then a woman is in such need to see the doctor as cannot be explained by her diet, stress fluctuation, and menstrual cycle fluctuation in order to rule out conditions like ovarian and uterine cysts. This becomes all the more important when the bloating is accompanied by other symptoms such as pelvic pain, difficulty with urination, or changes in bowel habits because these could be signs that the cyst is growing or causing other complications28,29. Early detection of ovarian cysts or tumors can help avoid big health issues such as cyst rupture, ovarian torsion, or even cancer; thus, addressing such symptoms is of utmost importance.
Since the sensation of heaviness in the abdomen persists and no underlying gastrointestinal or urinary cause can be found, imaging studies, such as ultrasounds, will usually be ordered by health care providers to examine the ovaries and uterus for cysts or abnormalities. Imaging studies can identify the size, nature, and position of the cysts or tumors and dictate the proper course of treatment. The sizes, types, and symptoms present determine the options for the treatment. While less serious cysts might be followed for a time to see if they improve on their own, much larger or more symptomatic cysts will need surgical removal as treatment aimed at eradicating symptoms and preventing complications. Fundamentally, early attention to the problem is the only way to manage the symptoms effectively and ensure overall health. Learning encompasses understanding from experts in diagnosing and treating diseases such as ovarian cysts. The rules that arise out of machine learning models with known domain knowledge are precise, appropriate, and realistic with respect to clinical practice30. Acquired knowledge is saved in a knowledge base which according to definition is the database wherein stored knowledge is retrieved and used by intelligent systems. It is embodied in some knowledge base by a mechanism for knowledge representation to an appropriate level of sophistication such as, decision trees, rule-based systems, or neural networks. Corresponding to the nature of the stored knowledge and requirements of the diagnostic system, a concrete level and kind of knowledge representation mechanism in a knowledge base is defined.
The knowledge base then stores such knowledge in a manner that the AI system is enabled to perform deduction, which is a process of drawing a conclusion in given facts applying a set of rules. It will thus involve the use of deductive inferences in the AI system and take into account; the development of symptoms, the case history of a patient, a patient’s imaging data in order to make a diagnosis of whether or not there is a cyst. If the rules are learnt by the AI systems during the training phase and then vetted by experts, diagnosis thereafter may be much more accurate and unchanging, than the errors and variations that are characteristic of manual diagnoses. It is in this way that deductive reasoning guarantees decisions will be made from a combination of knowledge and insights from the data, facilitating improved speed in the diagnosis effectiveness. Looking at the different machine learning approaches deep learning has been found to be more suitable for especially to medical image analysis especially to images which are highly dimensional including ultrasound or MRI scans. The problem that this research tends to identify is the inconsistent and time-consuming diagnosis of ovarian cysts, and the fact that symptoms may be inconsistent with the types, most of the time requiring several expert opinions with manual analysis. This indeed leads to delays with the possibility of inaccuracy, further opening up chances for complications31. The motivation for the proposed work is the usage of AI and ML in general, with deep learning techniques at the core, for the automation, acceleration, and increase in accuracy of ovarian cyst detection and analysis. If the goal is to reduce human error and create better patient care, there needs to be a development of faster, rule-based, and more reliable diagnostic solutions. The major research objectives of this paper are given below:
-
For the enhanced realization and fine-tuning of CoCo-HoloNet model, a new deep learning architecture that has been constructed for accurate identification and characterization of PCOD using ultrasound image acquisition techniques that surpasses the current approaches by integrating encompass embeddings and feature extraction from high level images.
-
For fine-tuning the regularization parameters for improved model generalization and performance in PCOD classification tasks using the Tangent-Runner Adaptive Optimization (TRAdO) model, the structurally updated CoCo-HoloNet framework is used.
-
The proposed work finally supports the area of medical imaging for PCOD detection; it provides a reliable and efficient solution toward the enhancement of the diagnostic practice. In spite of some new approaches to discussion of the existing methodologies and their flaws, CoCo-HoloNet can potentially change the existing position in the diagnosis of PCOD and significantly improve the managing of manifold care to patients.
-
In order to provide a rigorous assessment of the proposed CoCo-HoloNet and the TRAdO models, therefore a set of comparison with benchmark deep learning architectures and other traditional machine learning algorithms is planned together with the main performance indices such as accuracy, precision, recall, F1 score to justify their suitability for the PCOD diagnosis and classification tasks.
The distinct contribution of this research is the combination of the CoCo-HoloNet model with the Tangent-Runner-Adaptive Optimization (TRAdO) approach, which together overcome the difficulties associated with the identification and segmentation of Polycystic Ovarian Disease (PCOD) from ultrasonography images as a multi-class segmentation problem. In terms of the methods employed, through deep learning methodologies and methodology dependent optimization for the selection of the regularization parameter, this work improves current methods of model diffusion by boosting model performance and generalization, outcomping all current existing methods with zero exception. Nonetheless, the proposed quantizes show a reasonable improvement in diagnostic accuracy compared to the parent models while providing a more effective solution to the problems arising in clinic environments.
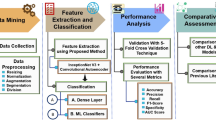
The rest of the paper is presented as follows: Sect. 2 comprehensively reviews the related literature from different methodological approaches that have been applied for the detection of PCODs by pointing out strengths and limitations of various techniques applied in both clinical practice and research settings. Section 3 describes the proposed work pertaining to the general flow, architecture, and algorithms used in this proposed AI-driven detection system for PCOD. Section 4 discusses the performance evaluation of the proposed approach and also comparative results with state-of-the-art techniques that prove the efficacy and accuracy of the proposed technique. Section 5 summarizes the current paper and underlines, most importantly, one main result and future directions toward advancements in the detection and treatment of PCOD.
Related works
Praneesh, et al.4 discussed a work related to the classification of ultrasound images of ovaries into two classes, that is benign and potentially cancerous cysts, by the VGG19 Net integrated with several machine learning algorithms. Thus, this is a very worth displaying contribution of research that has applied VGG19 Net in the domain of PCOS detection and earned thereby an impressive accuracy rate of 96% against other techniques like Random Forest at 94%, Logistic Regression at 91%, Bayesian Classifier at 81%, Support Vector Machine at 92%, and Artificial Neural Network at 90%. That VGG19 Net, in fact, caused improvements in the precision and accuracy of early detection in PCOS cases to come out even better as compared to conventional methods. Limitations going on within the theory: the whole study in general is focused on accuracy measures; little or no supportive analysis of other parameters has been given-for example, sensitivity, specificity, or performance on various subsets. Actually, this leaves room for questions regarding the model’s robustness across a wide range of clinical cases. The utilization of VGG19 Net indeed marks an uptick in accuracy; however, this deep learning model is computationally expensive and might restrain practical applications in real-time clinical environments where speed is crucial32,33. The other thing is that it does not explain how the model could balance itself in situations where data could be used, which should act as a reason for degraded performance in cases of underrepresentation.
Raja and Suresh5 introduced an important approach to better detection and classification of ovarian cysts, especially for those women suffering from menstrual problems and pregnancy cysts. They mention the limitations of doctors to identify types of cysts which are not clear on ultrasound images. For this challenge, the presented research study proposes a system that will detect and classify various types of cysts with the help of features extracted from ultrasound images. The automation is designed to support the doctors with a more reliable and efficient methodology in identifying ovarian cysts to enhance diagnostic accuracy34,35,36. They also do not include data on how the system works on different stages of cyst development or complicated cases where there is an overlap of a cyst with other gynecological conditions. There is very little discussion over the computational complexity for the proposed system, which might be a challenge regarding real-time clinical usages. A deeper comparison with the existing methods of cyst detection would also provide a better contextualization of the benefits and the limitations of the approach proposed herein.
A number of image processing techniques have been considerably researched for the detection and classification of PCOD. Most of the techniques are based on the analysis of medical images of ultrasound or MRI of the ovaries, detecting the presence of cysts, which is the primary symptom of the disease. Traditional approaches have been based on a manual check by experts, which may be very time-consuming and prone to variability in accuracy37. This calls for automation of the detection process through the use of methodologies in image processing, such as segmentation, feature extraction, and classification. All of these techniques have become much more precise over recent years due to advances in machine learning, especially deep learning, which automatically enables pattern recognition in medical images38. Approaches: this section discusses the current approaches, strengths and limitations, the methods through which the technology has been in development regarding the use and proposition of image processing techniques for identification of PCOD.
Silambarasan, et al.39 focused on the identification of PCOS if their contribution is regarded in view of the weaknesses of manual ultrasound diagnosis, which in most cases is a rather time-consuming process susceptible to human factor errors. Their research identified two different predictive models: text-based and image-based datasets for more accurate and objective diagnoses. While the first is an Optimized Support Vector Machine Model developed on text data, hence assuring a more effective solution toward the detection of PCOS from the records of the patients, the second model incorporates DenseNet on the image-based datasets and utilizes deep learning techniques in analyzing ultrasound images to spot polycystic ovaries with greater precision. This supports the fact that they also further validated their work with experimental results to ensure efficiency and effectiveness of the models put forward for improvement in diagnosis. Such a dual approach-text-based and image-based analysis, one merged into another-joins to provide more robustness to the study, hence providing a holistic automated solution for the detection of PCOS. Nakhua, et al.40 have proposed an ensemble learning approach with the integration of base classifiers like XGBoost, KNN, and SVM, improving the performances; hence, novelty is introduced in early detection. Their approach was benefited by an optimized image preprocessing pipeline that ensured better feature extraction after geometrical transformation, enhancement of contrast, and reduction of noise. This is overcome further by the bias mitigation strategy for class weighting to allow more balanced learning. Their approach has also been verified for its robustness by using the PCOSGen Dataset with a large number of healthy and unhealthy ultrasound images. High accuracy in testing by 92% depicts the robustness of their approach over individual classifiers. However, some of the salient drawbacks of the work are as follows: the dependency on ultrasound images alone restricts the generalization ability on different diagnostic modes. The effectiveness of the proposed ensemble technique with higher computational complexity may present obstacles toward real-time deployment in a clinical setting. The authors give importance to class imbalance problems, but the performance for minority classes may be further explored to ensure detection reliably in each and every case. This finally can be complemented by a more in-depth comparison against other state-of-the-art techniques which aim at better contextualizing the advantages and limitations of the proposed method.
Jha, et al.41 introduced an interesting approach to help the clinician and radiologist in the early diagnosis of PCOS to avoid other complications. The authors discussed disorders such as hirsutism and gynecomastia that are examined with the help of ultrasound, which evaluates position, size, and number of follicles. The authors, being well aware of this problem of noise interference in ultrasound images, have proposed a denoising filter based on the 2D-FrFT for artifact removal and image quality improvement. This technique is highly relevant due to the disturbance created by noise badly interfering with disease diagnosis. Thus, restoration of image clarity becomes an essential factor for proper extraction of medical information. Though the proposed method has some advantageous aspects, it also has certain drawbacks. Although 2D-FrFT was effective in removing the artifacts, added computational complexity may make real-time processing clinically more difficult. The study could be further improved by considering a wider range with respect to noise levels and ultrasound imaging conditions, since the effect of 2D-FrFT will vary depending on the latter. Similarly, little consideration is given to how this methodology really performs compared to other state-of-the-art denoising methods. Thus, its relative performances or limitations may not be distinctly identified.
Sakthivel, et al.42 designed an intelligent system that can detect PCOS using ultrasound images and clinical data, incorporating high-valued machine learning techniques by applying the Rotterdam criteria in diagnosis. In extracting features from ultrasound images, it uses the ResNet-50 deep learning model, while processing the clinical data utilizes correlation-based feature selection. Then, it applied the features extracted from ultrasound images to three classification algorithms: SVM, Random Forest, and Logistic Regression. Furthermore, it could also be extended to prove that investigation and comparison of the power of image-based and clinically based features may lead to an improved diagnosis of PCOS. Despite its novelty, there are still several drawbacks. Although the research emphasis had been on carrying out an integrated analysis of several data types, details about how different features are normalized or scaled are not well described, although that is a common practice in order to ensure that machine learning models perform efficiently. Moreover, no explanation has been given about the specific advantages or even limitations of those algorithms in view of the particular task of PCOS detection, which could raise some questions concerning the appropriateness of each method for the task. What is more, imbalances in the classes of the dataset are not discussed in terms of their effect on the classification performance; furthermore, more extensive evaluation metrics, considering model robustness beyond simple accuracy, are not taken into account. A comparison against available PCOS detection systems would add strength to the discussion on the relative performance of the proposed system and its practical implications within a clinical setting.
Verma, et al.43 insisted that the early diagnosis and proper management of PCOS might be considered a tool to improve the quality of life of the patients by minimizing health complications associated with PCOS. Authors have identified that AI techniques have been increasingly advocated in healthcare due to their potential promise in diagnosing and managing complicated diseases like PCOS. Applying machine learning algorithms on ultrasound images, and in concert with anthropometric and biochemical test results, AI attempts to reach diagnoses faster and more precisely. AI also consolidates myriad data inputs-such as patient history, lab results, and medical records much more easily for healthcare providers to obtain a big-picture look at an individual’s health that can then be used to make informed and effective diagnostic decisions. However, there are a number of serious drawbacks to the approach. First, while integrating various sources is laudable, the authors fail to discuss any sort of problem as it relates to data security and privacy-probably most resilient when dealing with information concerning patients. First, reliance on machine learning algorithms has its shortages: health professionals might not be easily convinced by the decisions of AI when they are not provided with explanations of how those algorithms made their verdicts. Nothing was discussed in this paper that could be related to biases of the AI systems that might be produced, especially in those cases when the training datasets are of a non-diverse population. It points out the advantages of AI in diagnosing PCOS; on the other hand, very little consideration is taken into account with respect to challenges in implementing the use of such technology in clinical settings-for instance, training healthcare professionals who would be in a position to make effective use of such advanced tools. Srinithi, et al.44 point out that the most common symptoms of PCOS in women include excessive gain of weight, hair growth on the face, acne, thinning of hair, discoloration of the skin, and staying away from the normal menstrual cycles, which will lead to lower conception. They point out that good management of weight, early diagnosis, and treatment on time will avoid the chances of long-term complications with type 2 diabetes and heart disease. They, however, point out that the methodologies and therapies adopted so far were not good enough to establish early detection and prediction of PCOS. In this regard, the authors fill in the gap with a methodology capable of predicting the success of therapy for PCOS based on an optimal and reduced set of criterions. They, therefore, consider a number of machine learning algorithms and present the results of each against what they find to be efficient for early detection methodologies. Their datasets were collected from Kaggle. However, there are a number of major drawbacks in this study. First of all, it was not clearly explained by the authors which criteria were selected for prediction and how those criteria have been determined and whether they include all the most important factors that might be involved in the diagnosis of PCOS.
Athithan, et al.45 report on a scheme for follicles and cysts detection in the ovary based on intensity-based grouping with texture data by using machine learning algorithms. Further, they mention that some applications involving these algorithms to ovarian diagnosis have turned out to be highly successful with an overall success rate of about 97% and thereby improved the quality of diagnosis involved. Authors further mention that standard strategies in machine learning have been explored for ovarian classification. They built three different models using ANNs, discriminant classifiers, and SVM to determine which one classified best. Regardless of these promising results, there are some marked drawbacks in the study. Whereas the authors themselves reported a very high success rate, they did not give any detail on the dataset used for training and testing these models concerning size and diversity. This is extremely important in order to evaluate the robustness of findings. The lack of such details means it’s impossible from this to know whether those models would generalize to larger populations without having detailed information. Second, no detailed comparative performance review of the three classification methods is made in the paper. Also, stating which of the methods yields the best results without an analytical study of advantages and shortcomings could discourage the understanding of applicability in the clinics. Moreover, no overfitting issues are discussed by the authors, and it was probably expected considering such high accuracy. Lastly, there is little consideration of how these machine learning models would be implemented practically in the clinical setting regarding data acquisition, integration into the existing diagnostic workflow, and training clinicians in the interpretation of the results.
Rashid, et al.46 Having that in mind, the authors introduce an attention-based multi-scale convolutional neural network for the detection of the polycystic ovary syndrome. Among the common features of PCOS in women, they highlighted several small sacs in the ovaries, called follicles, which are usually filled with fluid and usually fail to release eggs on regular bases. Although the study has identified benefits connected with early identification and weight loss for the reduction in long-term complications arising from PCOS, the authors have gone ahead to emphasize the importance their proposed model holds, especially concerning the use of dilated convolution for keeping multi-scale features while reducing the number of parameters. There are, however, a few major drawbacks in this study that call for attention. The authors mention advantages of their proposed approach to AMCNN. What is missing, however, is a side-by-side comparison against previous models or techniques that may set a benchmark for showing the novelty of their method in relation to state-of-the-art conditions. Also, the paper fails to define the dataset that the authors have used for training and testing their network concerning its size, variety, and ultrasound image characteristics, which is critical for the generalization of the proposed model and its applicability.
Most of the available studies either focus on image-based data, such as ultrasound images, or on clinical data, which covers the hormonal level and case history of patients47. This leads to incomplete understanding of the syndrome because both kinds of data have information that is crucially important and could help improve diagnostic accuracy if combined. Multimodal data might someday permit a truly holistic approach whereby predictive models incorporate the best of each dataset. However, most of the literature still lacks the development of methodologies that effectively combine these types of data; this indeed is badly needed if early detection and management of the condition are to be greatly enhanced. Most of the studies conducted so far are based on small datasets, not really representative, with a wide range of variability in the population with PCOS. In fact, most of the available literature is based on small sample sizes, which can result in a number of biases in the results and limit the generalizability of findings. The absence of large datasets, with diverse information reflecting the heterogeneity of PCOS in various demographics, makes it hard to translate the development of diagnostic tools in a reliable clinical manner48,49. Second, most of the studies have not resolved the problem of imbalance in data, especially in distinguishing between healthy and affected individuals. Furthermore, the inadequacy of effective strategies for handling class imbalance can lead to a biased model that turns out to perform badly in clinical practice, which further calls for more research in this aspect. Other critical gaps in the current literature involve the handling of noise and artifacts in ultrasound imaging. Ultrasound images are of great value but are generally plagued by various forms of noise that can seriously impede diagnostic performance.
Proposed methodology
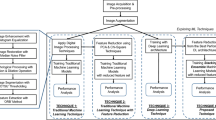
In this paper, a new optimization-based classification framework is developed and proposed, especially for the detection and classification of PCOD from ultrasound input images. We will introduce the CoCo-HoloNet model, a new framework specially designed for the accurate detection and classification of PCOD by analyzing ultrasound images. This will employ an advanced deep learning methodology in collaboration with holographic imaging principles to improve feature extraction for enhanced diagnostic accuracy. The key contribution of the CoCo-HoloNet model is to serveively analyze complex ultrasound data, which generally presents some obstacles because of noise variability of image quality and subtle manifestations of PCOD. The CoCo-HoloNet aims at deriving meaningful spectral features responsible for differentiation between normal and PCOD-affected ovaries through a conventional convolutional neural network architecture by incorporating holographic preprocessing techniques. The aims of CoCo-HoloNet include not only achieving high classification results but the robustness and representation of real-life clinical applications. The model will contribute toward realizing the objectives by making use of a systematic methodology in several key methodologies. First, some preprocessing, namely normalization and holographic preprocessing, is applied to the input ultrasound images to improve the quality of these images and extract the essential spectral features. For this reason, such a preprocessing step is necessary to allow the model to focus on the most relevant information and reduce the impact of noise or artifacts commonly present in ultrasound images. Further, a set of convolutional layers utilizes feature spatial hierarchies complemented by advanced pooling strategies, which allow reducing dimensionality but still keeping critical information. CoCo-HoloNet is a novelty in design because it combines holographic principles and deep learning for the first time in a synergistic way that improves the learning from complex datasets. Unlike typical CNNs, which rely only on spatial feature extraction, CoCo-HoloNet also includes spectral analysis because of its preprocessing step involving holography. The dual approach enables improvement in classification performance, besides allowing the model to pick up subtle variations within the ultrasound images that will enable accurate diagnosis related to PCOD.
The major contributions of the proposed work, CoCo-HoloNet, are embedded in its innovative fusion of holographic imaging techniques with a DenseNet-inspired convolutional neural network architecture, which is specifically tailored for the task of PCOD detection and classification from ultrasound images. A primary contribution of this work is the introduction of holographic imaging in the medical imaging domain, specifically for the diagnosis of PCOD. A step ahead of traditional 2D imaging, this view is far richer and more comprehensive in its capability of providing the model with more informed and precise decisions based on the anatomical structures being analyzed. Integration of this holographic imaging into the network allows the model to identify not only the major features of PCOD but also subtle variations in ovarian morphology, which is a huge improvement over the existing 2D imaging techniques.
Another very important contribution of CoCo-HoloNet is using the DenseNet architecture, whereby the network layers are densely connected to each other. This helps in enabling the model to propagate features very effectively throughout the network, reusing previously learned features. This would allow the combination of dense connections and the deep convolutional layers to facilitate more effective learning from high-quality to poor-quality ultrasound images, as is often needed in real clinical settings where the image quality varies so much. By allowing each layer to also receive input from all previous layers, DenseNet also lessens the problem of vanishing gradients, which often plagues deep networks.
CoCo-HoloNet addresses multistage processing: pre-processing and feature extraction, while subsequent convolution and pooling layers are used to learn the input data of increasingly higher-order representations. As a matter of fact, this structured flow further enables the proposed model to proficiently classify PCOD while addressing the resulting difficulties due to variability in image quality and noise levels. Also, this framework introduces a new fusion between the Tangent Search with Runner Root Optimization to enhance the performance of deep learning models by optimizing their critical parameters, such as the regularization parameter in neural network architecture. The contributions of this paper are threefold: it proposes an effective model for PCOD detection with improved performance, it adopts state-of-the-art parameter tuning techniques to avoid overfitting and enhance generalization, it uses TRAdO for the optimal selection of the regularization parameter, and it compares the developed technique with traditional optimization techniques by extensive experiments.
Overview of the proposed work.
It seeks to proffer a model that would assist in the improvement of the model’s performance by addressing the hitherto limitations in the currently used methods of hyperparameter tuning, while ensuring computationally efficient models. The method that will be proposed shall incorporate deep learning combined with the TRAdO optimization technique, which also involves both Tangent Search and Runner Root Optimization algorithms. Tangent Search mechanism locally searches for the optimal values, identifying the most promising regions of the solution space for hyper-parameter tuning. This is especially important in the selection of an appropriate regularization parameter that will help reduce the chances of overfitting, a common problem in the classification of medical images. Meanwhile, Runner Root Optimization drives the process of global search, enhances convergence rate, and diversifies the exploration of regions to avoid local optima. This is a combination of the local and global search strategy to make the optimization process robust, adaptive, and deal with complex nature datasets as in PCODs-these datasets generally suffer due to imbalance and noise in their features.
The novelty of this approach comes with its adaptiveness wherein, at each dynamically changing process during optimization, the TRAdO algorithm updates its regularization parameter value with the classification accuracy at each iteration. Unlike most conventional optimization methods, TRAdO has been designed specifically with deep learning models of medical diagnosis problems in mind, such as variability in patient data or high dimensionality in medical images. To this end, the inclusion of an adaptive learning strategy guarantees that the value selected for regularization will not only be optimal but also adaptive according to the characteristics that define each dataset. This flexibility considerably enhances the model’s capability for good generalization across various cases of PCOD; therefore, it is more dependable for diagnosis. TRAdO works, initiating preprocessing on ultrasound images to enhance the quality and remove noise from the images in the context of PCOD detection and classification. Further, after features have been extracted through CNN, TRAdO is used in optimizing the regularization parameters of the network. The iteratively refined regularization value in the course of training is for the purpose of ensuring that while the model optimizes the minimization of classification error, it is resistant to overfitting. This optimization not only ensures that the deep learning model is resilient to the key features representative of PCOD but also manages high performance with less computational overhead. The focus of the framework on regularization optimization serves to effectively control the neural network’s complexity, for it concentrated on the most relevant features in the images of PCOD.
Advantages over other methods abound. First, TRAdO’s adaptive optimization process ensures that the regularization parameter is more effectively tuned than with the traditional grid search or random search, which are comparatively time-consuming and computationally expensive. This hybridization of Tangent Search and Runner Root Optimization balances the exploration with the exploitation to ensure global optima are reached with good search efficiency. This is particularly helpful in the medical domain, where high accuracies have to be achieved, yet shortages of time and resources restrict the capability to do an exhaustive search for parameters. Finally, with deep learning architectures integrated into TRAdO, the proposed method has been much more scalable and suitable for large-scale medical datasets, which is a significant improvement compared to other methods that tend to perform quite poorly owing to scalability issues in the data. In general, the flow in TRAdO-based PCOD detection will include the following: image pre-processing and feature extraction using a deep convolutional neural network. During the training phase, the TRAdO algorithm will optimize the regularization parameter with a view to generalizing the model when different patients’ datasets are presented. Optimization allows the classifier to improve its accuracy and hence the diagnostic performance in detecting PCOD. Furthermore, this model has been evaluated on precision, recall, and F1-score metrics to prove the better performance of the TRAdO framework compared to other methods of hyperparameter tuning. In general, the proposed technique extends the field of medical image classification and provides a more effective clinical diagnosis reliably. As shown in Fig. 1, it will have the following in a flow: image preprocessing, feature extraction using a deep convolutional neural network. TRAdO algorithm will optimize the regularization parameter in the training phase with a view to generalizing the model when different patients’ datasets are presented. Optimization allows a classifier to improve its accuracy and hence diagnostic performance in detecting PCOD. This model has been further evaluated based on precision, recall, and F1-score metrics to show better performance by the TRAdO framework compared to other methods of hyperparameter tuning. In general, this proposed technique extends the field of medical image classification and provides more effective clinical diagnosis reliably.
Holographic convolutional dense network (Coco-HoloNet) for PCOD detection and classification
Coco-HoloNet represents a huge leap into the future of medical imagery, especially regarding the detection and classification of PCOD. The most striking novelty in this approach is using ideas of holographic imaging integrated into a DenseNet-inspired framework to come up with an improved model that could extract and study intricate features from complex medical images with much efficiency. The two reasons that form the basis of using this methodology are: firstly, to enhance the process of PCOD detection by developing a system assisted with advanced imaging modalities in order to avail data with more information; secondly, to enhance the processes of classification using methodologies of deep learning in order to make them adaptable to changes in condition and characteristics in images. The integrated approach, therefore, supports subtle variations in ovarian morphology and structure, portrayed and analyzed in greater detail to facilitate appropriate and timely medical intervention. Also, Coco-HoloNet presents some new components that are added to the conventional models of natural language processing. Firstly, it is enables the model work with volumetric data, more specifically, it enables holographic imaging where the 3D representations of photographic ovarian structures are incorporated. It is critically important to as far as the recognition and classification of cystic formations are concerned. Even more, the DenseNet architectural approach of having a higher density of connections in the network means that each layer of the model receives input from all the prior layers. This leads to better gradients flow during the training process and therefore relatively fast convergence of features and improved features reuse. This nests the Coco-HoloNet into several convoluted layers, and then normalizes the batch before subjecting it to activation functions so as to realize higher order features. All the layers are planned to work at different scales and types of features, thus increasing the features set to be used in the classification. It also uses skip connections to improve feature transport, as well as decrease the likelihood of experiencing vanishing gradients: a significant indication of appropriateness for training on intricate data.
However, the Coco-HoloNet framework is well suited for this task of PCOD detection primarily because it can work effectively with large dimensional data; and it emphasizes feature learning at varying levels of abstraction. PCOD is a condition that isomorphic and patients with PCOD may present with very dissimilar fundal features, uterine contours and other internal changes. Using the principles of holographic imaging, the model can identify differences in ovarian structure that might be representative of the disease. In addition, the deep learning architecture is inherently scalable, which is desirable with reference to the variance and volume of medical images. The adaptability to learn from an abundance and variety of training data guarantees that Coco-HoloNet can generalize new and unique data a unique aspect for clinical applications where patient variations exists. This would have a lot of advantages compared to the rest of the existing models considering the detection and classification of PCOD. Among all those, one major advantage is that it offers better accuracy due to the dual advantage in the form of holographic imaging and deep learning.
Traditional models often depend on 2D imaging techniques which may fail to present volumetric information, something very important, while holographic imaging in Coco-HoloNet captures more volumetric details of ovarian structures. Besides, features are effectively extracted and re-used due to the dense connectivity of the network, hence reducing the need for an extensive amount of preprocessing and manual feature engineering required by many of the classical techniques. This efficiency not only streamlines the workflow but minimizes chances of human error. Besides, Coco-HoloNet is resilient against overfitting by using dense architecture in conjunction with penalization techniques, and the model is said to be reliable upon transfer onto smaller datasets or even into clinical use. This is because it reuses features of the earlier layers, hence reducing redundancy and increasing computation efficiency for the model against other traditional deep learning models such as VGG or ResNet, featuring by an order of magnitude a deeper network with more parameters. Besides, Coco-HoloNet emphasizes the integration of mechanisms of attention such that salient features are not ignored during the learning process, forming very useful insight into spotting minute signs of PCOD that may get lost on other models. Therefore, Coco-HoloNet contributes more to improving accuracy, computational efficiency, and clinical applicability with regard to the detection and classification of PCOD.
Figure 2 presents the layered architecture for the proposed CoCo-HoloNet model to affect a practical detection and classification of PCOD using higher-order deep learning concepts. Its layer architecture comes in a tiered, modular form made up of a number of key layers for processing the input images. First, the model has a preprocessing layer that normalizes the images and does holographic filtering to enhance the quality of the input images by removing noise and other unwanted features. This is then followed by a feature extraction layer that utilizes holographic techniques with Fourier Transform in extracting critical spectral features related to PCOD diagnosis. The architecture includes a number of convolution and max pooling within the convolutional layer in a way that, step by step, systematically works toward the representation of hierarchical features from the data. Further ahead, this is complemented by dense layers that apply an activation function and dropout techniques to avoid over-fitting; hence, robust learning is achieved. The last layer should be an output layer, and it should bear the softmax activation function that classifies the input images into distinct categories based on learned features. Hence, this upgrades the model’s accuracy and reliability in the diagnosis of PCOD. Moreover, this architecture is so designed that the features can be learned efficiently and that the model will generalize well over variability in conditions under which the images might have been acquired. In fact, this constitutes a major step ahead in the methodologies toward the detection of PCOD.
Layered architecture model of the proposed CoCo-Holo Net model.
The Coco-HoloNet architecture is mainly proposed in this work for the detection and classification of PCOD, tries to combine the strengths of holographic imaging with those of DenseNet in an attempt to produce a powerful yet efficient solution for the task at hand. From this advanced imaging technique, especially in holographic imaging, it obtains three-dimensional representations of ovarian structures and their subsequent analysis by a highly connected convolutional network architecture. The architecture of the proposed Coco-HoloNet starts with a few convolutional layers acting as the main feature extraction mechanism. The key difference that makes Coco-HoloNet unique with respect to traditional convolutional networks is the use of 3D convolutional layers; hence, it is able to process volumetric data generated by holographic imaging. In such a way, the network is able to extract spatial hierarchies and relationships within the three-dimensional structure of ovarian images, features of major importance in performing an accurate cyst detection and morphological analysis.
The 3D convolutional kernels scan through the volumetric data in order to extract deep features regarding texture, shape, and size of the cystic formations within the ovaries. After having extracted these preliminary features, the network then branches out into the series of DenseNet-inspired dense blocks. A building dense block of Coco-HoloNet includes many convolutional layers. It helps with better gradient flow during training and the vanishing gradient problem that usually slows down the convergence of model training. It comes right after the dense layers, following batch normalization. Besides, the dense connectivity pattern in these blocks will further ensure feature diversity, since such a network will be able to learn rich representations for the PCOD images.
They can be further refined by the integration of more advanced pooling strategies, designed to reduce the spatial dimensions while preserving the most relevant information contained in the feature maps. Commonly, a pooling operation lies just after every convolutional or dense block layer to reduce computation and make the model more effective. Coco-HoloNet performs both max and average pool operations to supply complementary features extracted from the HoloLens imagery. The other unique characteristic related to the pooling strategy of Coco-HoloNet is the hierarchical structure. The pooling layers are designed to operate at multiple levels of the network, starting from the raw holographic image down to the final feature representations. These are followed by the activation functions, which mostly comprise ReLU Rectified Linear Unit to introduce non-linearity into a network and give it the potential to learn patterns and complex interactions between the extracted features. In the architecture of Coco-HoloNet, holographic imaging is of specific importance in the detection of such cystic formation and slight changes in the ovarian structure. Allowing the 3D information to be used, the network can construct much more comprehensive characteristics of ovaries, including volume features that may be very hard to discern when using 2D imaging. These holographic representations allow the network to assess not only the superficial features of the ovaries but also the internal structures, which is very vital in the case of PCOD detection.
After getting the input image from the dataset, it is represented with its height, width and number of channels at first. Then, the convolution operating is performed on the input image according to the following mathematical model:
Where, \(\:\mathfrak{F}\) indicates the output feature map, \(\:\mathbb{I}\) represents the input image, \(\check{ \partial }\) denotes the convolution kernel, and \(\:\mathfrak{V}\) is the bias value, As a consequence of this, the ReLU activation function is applied on the obtained feature map based on the following equation:
Then, the dimensionality of image is reduced with the pooling layer operation as mathematically described in below:
Similarly, the dense block is constructed for all n layers in the image, and its output is in the following form:
Where, \(\:{\mathfrak{N}}_{n}\) indicates the output, and \(\:\oplus\) denotes the dense concatenation operation. As a consequence of this, the required image features are obtained from the final dense block as shown in below:
Where, \(\:\mathfrak{D}(.)\) indicates the dense block operation. Similarly, the global average pooling \(\:\mathcal{G}\) operation is performed for minimizing spatial dimension as represented in the following equation:
Where, \(\:\mathcalligra{h}\) and \(\:\mathcalligra{w}\) are the image height and width. Moreover, the fully connected layer operation is performed by giving the average pooled features into the fully connected layer as shown in below:
Where, \(\:\mathbbm{w}\) indicates the weight matrix, and \(\:{\mathfrak{V}}_{\mathcalligra{f}}\) represents the bias value. Then, the output probability value is estimated for properly determining the classes of image according to the softmax function as shown in the following equation:
Where, \(\:\mathfrak{z}\) denotes the number of classes. For aggregating it with federated learning module, the model update function is computed using the following equation:
Where, \(\:{\mathbbm{w}}_{x}\) represents the model parameter. Then, the regularization term is computed for preventing the classification model from overfitting, which is mathematically expressed in below:
Where, \(\:\delta\:\left(\mathbbm{w}\right)\) denotes the regularization function, and \(\:\zeta\:\) is the regularization parameter that is optimally estimated using TRAdO model. Moreover, the weight update process is carried with the aggregation factor using the following equation:
Where, \(\:\phi\:\) represents the aggregation factor. In addition to that, the transfer learning strategy is applied with the hyper parameter that effectively controls the pre-trained weight value as shown in the following equation:
Where, \(\:\psi\:\) represents the controlling parameter that is used to update the pre-trained weight value. The final predicted label is represented in the following form:
Where, \(\:\hat{{x}}\) represents the final predicted result.
Tangent-Runner adaptive optimization (TRAdO) for computing regularization parameter
The TRAdO technique proposes a novel hybrid optimization framework, specially designed to efficiently compute the regularization parameter within machine learning models. In tasks such as the classification related to PCOD, where accurate diagnosis is of paramount importance, regularization has an important role in enhancing model generalization and preventing overfitting. In the context of CoCo-HoloNet, a convolutional neural network model for the classification of images in PCOD, the proper tuning of this regularization parameter can result in a much-improved model performance leading to a robust and reliable diagnostic system. TRAdO has been developed by integrating two complementary strategies, Tangent Search Algorithm, and Runner Root Optimization, in order to allow adaptive and dynamic methods toward finding the optimal regularization parameter. This process is important because, in PCOD classification, the images usually exhibit subtle variations along with noise. A model can quickly get overly specific to the training data without appropriate regularization and learn noise or irrelevant features, which decrease its performance on unseen data-that is, testing or real-world clinical data. With overfitting, reduced generalization ensues, which may be a bit deleterious in medical diagnoses since the ability of the model to make an appropriate prediction about diverse and unseen cases is highly needed for real-world applications. That becomes an essential part of the machine learning algorithms, in which regularization balances out the model complexity through the regularization of the magnitude of the weights. CoCo-HoloNet projects the model during PCOD classification to not overfit the data by adding a penalty on large weight values. In other words, it penalizes the model from getting too complex and memorizing some features from the training dataset, which will generalize well in new images.
It is a measure of the regularization parameter, generally represented, which is calculated herein with the help of the TRAdO technique. Since it changes dynamically with respect to the conditions of learning during the training process, TRAdO strikes a delicate balance wherein the model learns enough features to perform classification without overfitting into the irrelevant details or noise present in the PCOD images. Hence, due to high natural variation of image quality and size, regularization becomes a major pre-requisite for feature and medical image classification in diagnosing PCOD. A deep learning model, if devoid of any regularization technique, might get too resilient to minute details of the images, which may not represent the disease status and might end up with a high rate of false positives or false negatives. In this regard, TRAdO provides the mechanism for direct computation of the regularization parameter. Henceforth, the model will concentrate only on the most relevant features for the PCOD detection task, such as size, shape, and distribution of the cysts. This would, therefore, enhance diagnosis while reducing the risk of interference by noise.
In simple terms, its hybrid nature-the incorporation of the Tangent Search Algorithm and Runner Root Optimization in one learning environment-adaptive framework-is the key novelty. A geometrically inspired mechanism aligns the search along the tangents of the objective function while performing an efficient search in the search space for the regularization parameter. The result of this alignment helps to locate regions in the search space that are most likely to provide optimal or near-optimal regularization values. On the other hand, Runner Root Optimization takes its cue from plant root growth strategies and applies randomness and depth in search to ensure that the process does not get stuck in local minima, as is commonly the case with many optimization techniques. Runner Root Optimization emulates the way plant roots perform a deep and wide search naturally to reach water and nutrient sources. This further ensures that while Tangent Search effectively reduces promising areas, with the Runner Root Optimization method, there is an intensified deep exploration of those areas to avoid local optima. TRAdO uniquely rebalances the tradeoff between model complexity and training error when applied to the regularization parameter during computation. The technique constantly adapts the search, so that neither the regularization term will be too small-to cause overfitting-nor it will be too large, causing underfitting. This balance is important in PCOD classification because of the variability in ovarian images, which needs subtlety both at feature extraction and classification.
Regularization forms one of the building blocks of any type of machine learning; it basically allows controlling model complexity so that a model generalizes well to unseen data without overfitting. Models in this class, dealing with high-dimensional medical images like CoCo-HoloNet, are the ones that require optimal tuning of the regularization parameter, a very critical task due to its huge implications on model performance. TRAdO takes a different turn in introducing two new optimization strategies, Tangent Search Algorithm and Runner Root Optimization, specifically developed for automatically setting and hand-tuning the regularization parameters by using adaptive search-driven parameter refinement. The core of the TRAdO framework is the Tangent Search Algorithm, which ensures convergence toward the optimal solution. This algorithm is based on a local search method and is steered by examining the tangent lines of the objective function in parameter space. To achieve this, it searches the gradients of the loss function concerning the regularization parameter for an area where the loss function varies minimally; this indicates a stable, well-regularized model. With its focus on the tangents, TRAdO is able to quickly find regions of the parameter space for which the regularization parameter gives better generalization and ensures that a model is not just memorizing the training data but learning meaningful, generalized features for unseen data.
Figure 3 shows the flow of the proposed TRAdO model. Unlike most computations in the training process. TRAdO does not anchor and lock a single value for the regularization parameter for the whole training process but this factor rotates with learning. The Runner Root Optimization component of TRAdO complements the Tangent Search Algorithm by iteratively adjusting the regularization parameter based on a root-finding strategy. This optimization technique uses adaptive search and continues to refine the regularization parameter by calculating successive approximations of the parameter that minimizes the model error. Instead of using static steps, Runner Root Optimization adjusts the step size dynamically based on past iterations; hence, the algorithm is capable of ‘running’ efficiently through the parameter space and skipping any recalculation for redundant points. In that way, TRAdO manages not to fall into local minima one of the weaknesses of traditional optimization algorithms. This keeps the model adaptive to intrinsic variability in real-world data all through the training process be it variability in the presentations of PCOD across different patients or different image-acquisition conditions.
Flow of the proposed TRAdO model.
Because TRAdO is adaptive, it does not fix the regularization parameter for the whole training process, but this factor changes dynamically with learning. This is crucially important in such a task as classification of PCOD, because for different layers and phases of the learning process different levels of regularization may be required. This can be explained by the fact that in an early stage, a high regularization parameter may avoid over-fitting noise, and then at later stages, a low regularization parameter may allow it to learn fine details in the data. Therefore, this advantage of fine tuning of the regularization parameter by TRAdO can be directly related to TRAdO improving the classification accuracy, higher precision and recall, and a more balanced F1 score, thus leading to better diagnostic outcomes. Another differentiator is that TRAdO also differs from the traditional optimization techniques that normally include one or more predefined learning rates or hyperparameters, not changed during training. By contrast, in TRAdO, the tangent search combined with Runner Root Optimization provides a flexible and dynamic framework automatically adapting to the changing needs of the model during the training process. This is useful in medical image processing, where the features of interest change in the images over time and, therefore, the process of optimization will have to change too.
In this optimization model, the parameter initialization is performed at first with the upper and lower bound values as shown in the following equation:
Where, \(\:{u}_{bon}\) and \(\:{l}_{bon}\) are the upper and lower bounds, \(\:\partial\:\) is the random number, and \(\:\mathfrak{d}\) represents the problems dimension. Then, the tangent search based local update is carried with the tangent function as shown in below:
Where, \(\:\mathcalligra{b}\) is the step size, and \(\:\theta\:\) represents the angle that supports to control the direction of movement. As a consequence of this, the best and worst solutions are computed with the scaling factor as shown in the following equation:
Where, \(\:\lambda\:\) represents the scaling factor. According to the global best solution, the root is updated as shown in below:
Where, \(\:\tau\:\) represents the random factor that controls the operation. Moreover, the boundary repair is performed be defining all parameters within the bounds, which is mathematically expressed as follows:
At the end, the best solution is chosen among the runner and root solutions as illustrated in below:
While, in contrast, the regularization parameter of TRAdO is monitored and updated continuously on the fly according to changing data and model characteristics. This means flexibility: at any moment, this would be the very best regularization term for the state of the model, which cannot be true for methods traditionally. The final conclusion is that TRAdO provides an effective way of estimating the regularization parameter in the CoCo-HoloNet model by embedding the power of Tangent Search with Runner Root Optimization. It performs adaptive, dynamic regularization tuning during training and provides improved generalization of the model with reduced overfitting and hence an improvement in performance in PCOD classification. This adaptability, swiftness, and fine-tuning of hyperparameters in real time set it apart from other optimization techniques, which makes this algorithm a strong solver in medical image classification tasks.
The paper CoCo-HoloNet has a transforming impact on real-time applications, especially in the domain of medical diagnostics. Most of all, real-time capability is the most important need in these medical environments where timely interventions may directly influence patient outcomes. Thus, early and accurate diagnosis within the circle of PCOD detection will allow the physician to administer the appropriate treatment plan that may prevent further development of complications and direct the management of the patient more precisely. The ultrasound image analysis with these models shows robust results against efficient processing and handling under noisy influences or through changing qualities of images. It easily adapts in real-world conditions within a clinic, where the equipment and multiple other factors linked to the patient introduce great variability within the data.
With the help of Tangent-Runner Adaptive Optimization in the tuning of the regularization parameter, CoCo-HoloNet has the ability to improve its sustenance of good performance when applied to real-world applications with potentially smaller datasets or less representative ones of the full spectrum of patient conditions. Such adaptiveness prevents overfitting, especially in situations with limited or heterogeneous data something quite common for tasks related to medical imaging. This will help the model generalize better over unseen data. Indeed, CoCo-HoloNet ensures that predictions are robust with respect to diverse patient populations, imaging devices, and clinical environments. In a real-time diagnostic application, this feature becomes indispensable, since a model has to be able to deal reliably with images coming from diverse sources and to produce results of high accuracy without any human intervention.
Results and discussion
The results section provides the performance evaluation of the proposed CoCo-HoloNet model using the Kaggle PCOD ultrasound image dataset50, which is a rich dataset of ultrasound images annotated for the classification of PCOD. This dataset will become very useful in our model evaluation because it has high variability regarding image quality, anatomical representation, and different disease manifestations. The proposed methodology for evaluation is thus structured in such a way that the performance of the CoCo-HoloNet model, comparatively with other existing methodologies, becomes quite evident, especially with regard to the accurate detection and classification of PCOD. We report on the performance of the model based on cardinal performance metrics such as accuracy, precision, recall, and F1-score in order to provide an all-round representation of the model performance. By setting these metrics within the CoCo-HoloNet model compared to conventional methods, we underline the advantages and novelties brought by our proposed approach. Our results show significant improvement within the performance of the classification for the CoCo-HoloNet model on the detection of PCOD and present it as a reliable tool. Furthermore, the knowledge derived from this process of evaluation identifies the significance of proper methodology in feature extraction and processing to enhance diagnostic capability in medical images. On that note, the results section has been adapted to show the effectiveness of the CoCo-HoloNet model not only in the context of PCOD classification but as a game-changer in the domain of medical image analysis itself.
Among the defining features of CoCo-HoloNet is the integration of holographic imaging with a convolutional network inspired by DenseNet. This dual approach allows the model not only to catch the fine-grained spatial features but also the intricate three-dimensional relationships in the ovarian structures something which is very essential in keeping the diagnostic accuracy under less-than-ideal conditions. One of the intrinsic strengths of CoCo-HodoNet deals with variations of image quality, like low-resolution or noisy ultrasound images, due to: its convolutional architecture. The use of DenseNet blocks with dense connections between layers thus ensures that features can be propagated easily through the network. This property is very important when dealing with low-contrast images or images with a high level of noise because it retains meaningful spatial information as it passes through the network. This has the effect of stabilizing the learning process due to batch normalization after each dense block and activation function, therefore reducing the bad effects that noise has on feature extraction. It ensures that the training of the model remains stable even in the presence of noisy data, hence generalizing better. TRAdO adapts to the level of noise present in a dataset, preventing the model from learning noise as a feature something that commonly occurs with machine learning models in dealing with noisy medical data. With the smaller ones, the dense connections of this model in the DenseNet blocks help the network learn better from fewer examples. This is due to the feature reuse and improved gradient flow. That becomes very important in medical imaging because it is usually hard to get a large, annotated dataset. Furthermore, the integration of holographic imaging at the stage of preprocessing, even for the smallest datasets, provides them with 3D spatial representations that bring out much more detailed and nuanced views of ovarian structures, allowing the model to profit from limited data. But only by a little, for poorer quality images or smaller datasets, those really hard cases where pictures are very noisy or just do not have enough detail. This often becomes the common limitation in deep learning models dealing with medical images, since they heavily depend on the quantity and quality of available data for training.
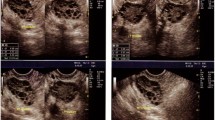
Figure 4 summarizes the various steps of image processing applied to the Kaggle-PCOD image dataset. It shows how, from the input raw image, the visual detail goes till the final output, highlighting the regions that have been segmented, and cysts that have been identified. The top left panel presents the original ultrasound image of the complex anatomical structures pertinent in the diagnosis of PCOD with the possible formation of cysts that characterize the condition. Normally, it is a raw image that contains a lot of noise and artifacts, variation of contrast that obscures important details pertinent for diagnosis. In fact, any enhancement preprocessing will always make the quality and effectiveness of the subsequent analysis even better. The second panel shows the preprocessed input image. Included among others are methods like normalization of the image, resizing, and noise reduction. Preprocessing at this step enhances the contrast in the image while minimizing artifacts that may obstruct further analysis. We normalize input data to create a better basis for further processing stages, which would yield more reliable and exact results. The third panel shows the grayscaled version of the image after the preprocessing. Grayscale transforms data into a much more simplified format where the relevant features are enhanced without confusing colors. This is necessary because most of the approaches to image processing, whether edge detection or segmentation algorithms, work more effectively on one-channel images. The important structure that would have been underlined by the grayscale will be analyzed in detail in subsequent steps.
Sample input and output images of Kaggle PCOD image dataset.
The fourth panel shows the binary image, wherein the original grayscale image is thresholded. A transformation like this amplifies the interesting structure from the background through turning the pixels either into black or white, according to their values of intensity. Thus, this binary representation now allows features like cysts and similar anatomical structures to be picked out rather more concretely, setting a stage for their detailed analysis. Original segmented output image: This would be the outcome of using a segmentation algorithm on the binary image. Here, those are the exact regions of interest, cysts, and ovarian structures identified and delineated. That would be useful to understand the spatial distribution of those structures and allow extraction of quantitative features necessary for classification. The final panel points to the cysts captured in this particular image and how well preprocessing and segmentation techniques are able to isolate these important features. This output shows the cysts being clearly marked, furthering the diagnostic process of PCOD with more detail and hence enhancing the clinician’s ability to evaluate and interpret ultrasound images. Figure 4 summarizes the refinement process, from raw input ultrasound image to refined output, underlining that every stage in its processing is equally important to arrive at proper and correct results in the detection and classification of PCOD.
Figure 5 depicts the training versus validation accuracy of the proposed CoCo-HoloNet model at every iteration of training. From this graph, one can observe the gradual increment in the accuracy of both the training and validation datasets through which models will effectively learn how to classify those features relating to PCOD from ultrasound images. During this period, the curve of training accuracy increases gradually and smoothly. That means the model can minimize the fitting error with the underlying pattern in the data. Figure 6 zooms further into training and validation loss by showing a detailed plot of performance metrics. Convergence in both curves shows that the model generalizes well on unseen data. Moreover, close tracking with regard to validation accuracy in terms of training accuracy would be an important fact that the CoCo-HoloNet model fits the data not only during training but can keep its robustness and accuracy during the validation. That is to say, these Figs. 5 and 6 stand as testimony that the proposed model can classify PCOD with high accuracy; hence, it confirms its capability of being a reliable method of diagnosis.
Training and validation accuracy of the proposed CoCo-Holo Net model.
Training and validation loss of the proposed CoCo-Holo Net model.
In order to validate the performance of the proposed CoCo-HoloNet model in diagnosing the disease from the PCOD images, the Receiver Operating Characteristic (ROC) shown in the Fig. 7 is employed as the evaluation criteria. The ROC graph outlines the real-life experiment comparison of true-positive with false-positive rates with the use of classification thresholds to assess the model’s performance. It measures the model’s total discriminatory power to identify instances in order for a gigantic value of AUC to signify an excellent performance. Figure 8 is centered on the feature importance scores obtained from the CoCo-HoloNet model: The significance of the features to differentiate between the PCOD affected and non-affected nucleus is depicted.
ROC analysis of proposed CoCo-Holo Net model.
Feature importance score for affected and non-affected PCOD images.
Mean value for different features of image.
Using the importance scores, we get very useful informations about which features influence model’s decision most. When looking at the rank order within each feature type discussed by the clinicians/researchers, the understanding of the important parameters defining the diagnosis of PCOD can be significantly improved. The visualization also highlights the model’s ability to select important features to feed into the classification process, that is, which features were useful once the ultrasound images have been preprocessed. Figure 9 displays the Mean values of the different features of the images to understand the difference between affected and non-affected PCOD cases in detail. The Fig. 9 shows that it is possible to identify distinct feature distribution patterns which are important for evaluation in clinic. Thus, comparing arrays of mean values for different features gives visibility which distinctions can define PCOD most effectively. Last but not least, Fig. 10 provides an aggregate assessment of the proposed CoCo-HoloNet model by presenting the specific assessment results. The consolidated performance measures further justify the stability and efficiency of the proposed CoCo-HoloNet model and its value for medical diagnosis, and thus for the enhancement of PCOD condition patient prognoses.
Overall performance analysis of the proposed CoCO-Holo Net model.
The classification results of various recent deep learning architecture models in terms of precision, recall and F1-score for PCOD are shown in Table 150. The evaluation shows that overall, the proposed CoCo-HoloNet model outperforms all the other architectures of the model in terms of precision, recall, and F1-score, which stands at 99.1%, 99% and 99% respectively. On the other hand, ResNet 152 model correlates well with precision of 80.58%, recall of 83.33% with F-1 measurement of 85.2% thus proving that ResNet 152 serves as a deep learning model accurately. The rest of the models produce a fairly decent performance, with F1-scores varying between 45.45% (DenseNet-121) and 76.19% (Inception V3).
For similar reasons, as illustrated in Figs. 11, 12 and 13, which show the precision, recall, and F1-score metrics of the various classification models, the proposed model outperforms the others. Precisions by Fig. 11 displays show that the CoCo-HoloNet model has much higher precision than all the architectures above, which clearly shows the reduction of false positive rate in the model. Figure 12 also confirms that the recall is raised notably, which means that the model practically accurately identifies true positive cases of PCOD. Figure 13 integrates all the above analyses using the F1-score, which owns the average value between precision and recall.
Precision of different classification models.
Recall of different classification models.
F1-score of different classification models.
The results are further developed in Table 250 that shows accuracy and specificity for the same models to give broader diagnostic performance. The CoCo-HoloNet model developed in this research expected a high level of accuracy of 99.2% as well as specificity of 99%. This means that the proposed model achieved high general accuracy and an even higher effectiveness in identifying true negative samples without misclassification, The Inception V3 follows with an accuracy of 84.81%, and high specificity of 87.93% suggesting its reliability though highly inferior to the proposed model. According to Figs. 14 and 15, it is determined that the rest of the models like the VGG 16 and the DenseNet different variants has comparatively low accuracy of between 67.09% and 75.95% thus the models are not so efficient to offer accurate diagnosing results.
Comparative analysis with recent models based on accuracy.
Comparative analysis with recent models based on specificity.
Table 351 and Fig. 16 gives a comprehensive comparative evaluation of proposed CoCo-HoloNet model with other competing models such as DarkNet-19, AlexNet and SqueezeNet. Analyzing the results presented in Table 2, we observe that under the proposed model, performance metrics are high, including an accuracy of 99.2%. Such accuracy is proven by the fact that the proposed CoCo-HoloNet model performs a correct classification of almost all instances from the given dataset, which makes it possible to state the advisability of a proposed approach for PCOD detection and classification in practice. With regard to sensitivity, the proposed model yields a value of 99.18% signifying the fact that it correctly identifies the true positive of PCOD cases. Detect is another measure for medical diagnosis and it is the sensitivity of the model to detect affected persons. On the same line, another model with high accuracy tag known as DarkNet-19 achieves a sensitivity of 99%, meaning it is efficient enough in diagnosis of positive instances. Nevertheless, the CoCo-HoloNet model performs a little better and it clearly highlights its detection efficiency.
Details of the evaluation of the proposed CoCo-HoloNet model yield an overall accuracy of 99.16% of the model in diagnosing non-affected subjects, hence reducing the percentage of false positive outcomes. This is having a keen importance when developing algorithms for diagnosing such medical conditions so that patients that do not have PCOD will not be diagnosed hence leading to undue stress and the wrong treatment being administered to them. DarkNet-19 proves to be the most precise, with the specificity of 99%, whereas both AlexNet and SqueezeNet had significantly lower values, 97% and 98% correspondingly. Further, it gives more focus on CoCo-HoloNet model to diagnose the non-affected cases as well as the affected one. The use of the proposed avenue results in an F1 score of 99.2% as a global reliability pull-through for classification jobs. This score is better than those of the other models: DarkNet-19 has an F1-score of 98; and significantly exceeds both AlexNet with the F1-score of 92 and SqueezeNet with the F1-score of 94. With the F1-score of 0.857, it can be concluded that the proposed CoCo-HoloNet model is an excellent detector of PCOD owing to its ability to yield high numbers of accurate true-positive classifications and low false-positive rates.
Comparative analysis with hybrid models.
Figure 17 presents the comparative analysis of the performance of the proposed CoCo-HoloNet model as well as other traditional heterogeneous models for identifying PCOD. Some observations made concerning performance include the fact that the proposed model has a much higher accuracy value than that of any other conventional model. This explains why the proposed model has high competency in the classification of ultrasound images. In terms of the two measures of precision, Fig. 18 reveals that the CoCo-HoloNet model is also superior to the regular learning algorithms. High precision describes the capacity of the model in excluding closest negatives, so that the cases that are identified fore as PCOD are not so unrelated. This attribute is clinically very majorly valuable because error on this results in a number of treatments; the wrong one. Figure 19 is another contrast analysis based on recall to present the advantage of CoCo-HoloNet model. The high value of recall suggests that the architecture has a good, measured ability to identify the true positive cases of PCOD disease. It helps with correct identification of subjects with the disease and is therefore very important to early diagnosing as well as early treatment in a way that might lead to substantial improvement in patient end-results. The F1-score comparison of the models is presented in the Fig. 20. Here, it can be observed that the proposed CoCo-HoloNet model achieves the best F1-score value compared to other traditional machine learning models. It means that these metric measures and reflects the best way the proportion of true positive predictions and the proportion of true negative predictions, which proves the effectiveness of an underlined model as a diagnostic one. Figures 17, 18, 19 and 20 shows the CoCo-HoloNet has a lot of potential in detecting and classifying PCOD in comparison with simple Machine learning techniques.
Accuracy comparison with standard machine learning models.
Precision comparison with standard machine learning models.
Recall comparison with standard machine learning models.
F1-score comparison with standard machine learning models.
Accuracy comparison with standard deep learning models.
Precision comparison with standard deep learning models.
The comparison of the accuracy of the proposed CoCo-HoloNet model to several other deep learning techniques is shown in Fig. 21 for the identification of PCOD. The CoCo-HoloNet model is significantly more accurate, proving the superiority of the model to classify ultrasound images accurate than the existing deep learning architectures. In the Precision Comparison illustrated in the Fig. 22, it is revealed that CoCo-HoloNet model can achieve a higher precision rate than the other typical deep learning models. This implies that the model actually to a larger extent eliminating the higher false positive percent while confirming the true cases related to PCOD. Figure 23 gives more emphasis on the recall comparison where the CoCo-HoloNet model gets superiority in identifying the exact true positive cases of PCOD as compared with the traditional deep learning models. A high recall rate is important in order to avoid failing to identify actual patients with the disease and this is where the model is most useful in the early stages. Finally, Fig. 24 shows that the F1-score comparison has also made it clear that the CoCo-HoloNet model has a higher efficiency than the other deep learning architectures.
Recall comparison with standard deep learning models.
High value of F1-score reveal that both metrics of precision and recall are approximately equal, thus confirming the effectiveness and efficiency of the CoCo-HoloNet model as a solution to the detection and differentiation of the PCOD pathology. Altogether, these values show significant improvements of the suggested CoCo-HoloNet model over the conventional deep learning techniques. It can be noted that the proposed model namely CoCo-HoloNet has shown promising results while performing better than all the existing techniques in detecting and classifying PCOD. Having avid accuracy, precision, recall, and F1-score, this model signifies the best of its ability to identify PCOD through ultrasound images. The superior performance depicted in CoCo-HoloNet not only helps to create more accurate diagnosis but also aims at becoming a useful tool in changing patient outcomes in clinical environment. Seen as an important achievement in the field of medical imaging, it provides a safe and efficient method in diagnosing and treating PCOD at the initial stage.
F1-score comparison with standard deep learning models.
Although CoCo-HoloNet is built with novel architecture and promising capabilities, there exist a few potential limitations that could affect its real-world applications and generalizability to different medical imaging tasks. One of the most significant challenges lies in its dependence on large-scale, high-quality, annotated datasets for training. While the model is prepared to handle variations in image quality and noise, the more important factor by far is the quality and diversity of the data on which it was trained. Generally, most of the time, in many real-world scenarios, getting large-scale, high-quality annotated datasets for specific conditions such as PCOD is always very difficult and resource-consuming in diverse healthcare settings with different imaging protocols. CoCo-HoloNet has been optimized specifically for PCOD detection and classification in ultrasound images and, furthermore, in the 3D volumetric representation of ovarian structures. While this architecture excels at the intended task, it might not generalize perfectly to other areas of medical imaging; in fact, such applications as brain tumor detection from MRI scans or diagnosis of lung cancer in CT images would require architectural adjustments, apart from retraining the model. Generalizing CoCo-HoloNet to other tasks presumably would require retraining and adaptation with large amounts of retraining data. It would otherwise deteriorate in performance if not fine-tuned on the peculiarities of this new imaging task.
Conclusion
This paper focuses on the development and implementation of the CoCo-HoloNet model for detecting and classifying PCOD from ultrasound images with a high degree of accuracy. The proposed model in this work integrates some advanced deep learning techniques using a layered architecture comprising convolutional layers, dense blocks, and strategic pooling strategies for the derivation of significant features from the input data. The novelty lies in enhancing the feature representation, along with considerably improving the strength of the model in distinguishing between affected and non-affected PCOD cases. CoCo-HoloNet is a model designed to marry the strengths of various deep learning methodologies with the integration of an adaptive optimization mechanism for the purpose of fine-tuning its model parameters, with the ultimate goal of enhancing diagnostic accuracy. Techniques that are to be used involve Tangent-Runner Adaptive Optimization (TRAdO), which allows the model to compute critical regularization parameters, used in preventing overfitting while improving the generalization across datasets. Extensive experimentation using the Kaggle PCOD Ultrasound Image dataset yields results that are pretty impressive in showing the excellent performance of the model, beating the metrics coming from existing state-of-the-art techniques. Apart from yielding impressive precision, recall, and F1-scores, CoCo-HoloNet reported accuracy rates of over 99%. These results underline the robustness and reliability of the model in identifying PCOD with unerring accuracy, hence proving to be a good tool for clinicians in the early diagnosis and management of the condition. The novelty of this research contribution opens new dimensions in not only medical imaging but also patient care for the management of PCOD. CoCo-HoloNet raises a new bar in ultrasound-based detection of PCOD and offers a workable and effective solution which is likely to change the current diagnostic practices in clinical settings.
Even with its astounding capabilities, there are several ways to go. Interpretability is a very significant one, more so because the trust in machine learning models needs to be proved in a clinical setting. With improvements in interpretability, a more transparent model will be clinician-understandable and thereby accepted for increased use in diagnosis workflows in reality. Another most promising direction involves the application of CoCo-HoloNet on other medical imaging tasks. Adapting CoCo-HoloNet to these domains might require changes, in order to cope with the different imaging characteristics. However, its architecture provides a flexible framework that can be applied to quite a large number of medical applications. Moreover, extending the scope of the model toward handling a variety of medical conditions may lead to more generalized systems capable of assisting diagnostics in very diverse clinical scenarios.
Data availability
The dataset is available at: https://www.kaggle.com/datasets/anaghachoudhari/pcos-detection-using-ultrasound-images.
References
Kermanshahchi, J., Reddy, A. J., Xu, J., Mehrok, G. K. & Nausheen, F. Development of a Machine Learning-Based Model for Accurate Detection and Classification of Polycystic Ovary Syndrome on Pelvic Ultrasound, Cureus, vol. 16, (2024).
Patil, S. D., Deore, P. J. & Patil, V. B. An intelligent computer aided diagnosis system for classification of ovarian masses using machine learning approach. Int. Res. J. Multidisciplinary Technovation. 6, 45–57 (2024).
Arora, S., Vedpal & Chauhan, N. Polycystic ovary syndrome (PCOS) diagnostic methods in machine learning: a systematic literature review. Multimedia Tools Appl., pp. 1–37, (2024).
Praneesh, M., Nivetha, N., Maidin, S. S. & Ge, W. Optimized deep learning method for enhanced medical diagnostics of polycystic ovary syndrome detection. J. Appl. Data Sci. 5, 1399–1411 (2024).
Raja, P. & Suresh, P. Variety of ovarian cysts detection and classification using 2D convolutional neural network. Multimedia Tools Appl. 83, 49473–49491 (2024).
Zhou, G. et al. Adaptive Adjustment for Laser Energy and PMT Gain through Self-feedback of Echo Data in Bathymetric LiDAR. IEEE Trans. Geosci. Remote Sens. (2024).
Zhou, G. et al. Adaptive High-Speed Echo Data Acquisition Method for Bathymetric LiDAR. IEEE Trans. Geosci. Remote Sens. (2024).
Clemency, C. D. D. & Grace, L. J. A broad analysis of ultrasound imaging for ovarian cyst detection using advanced artificial intelligence techniques. Int. J. 2, 1091–1099 (2024).
Rao, D., Dayma, R. R. & Pendekanti, S. K. Deep learning model for diagnosing polycystic ovary syndrome using a comprehensive dataset from Kerala hospitals, International Journal of Electrical & Computer Engineering ( vol. 14, 2024. (2088).
Xu, H., Li, Q. & Chen, J. Highlight removal from a single grayscale image using attentive GAN. Appl. Artif. Intell. 36, 1988441 (2022).
Ye, W. et al. Adaptive Spatial–Temporal aware graph learning for EEG-Based emotion recognition. Cyborg Bionic Syst. 5, 0088 (2024).
Girdhar, N., Singh, P. & Singhal, T. Polycystic ovary syndrome classification based on machine learning techniques: A comparative analysis, in Applied Intelligence for Medical Image Analysis, ed: Apple Academic, 81–94. (2024).
Quan, X., Du, R., Wang, R., Bing, Z. & Shi, Q. An efficient closed-loop adaptive controller for a small-sized quadruped robotic rat. Cyborg Bionic Syst., (2024).
Jia, Y., Chen, G. & Chi, H. Retinal fundus image super-resolution based on generative adversarial network guided with vascular structure prior. Sci. Rep. 14, 22786 (2024).
Poorani, B. & Khilar, R. An innovative approach for PCO morphology segmentation using a novel MOT-SF technique. Discover Comput. 27, 27 (2024).
Kodipalli, A., Devi, S. & Dasar, S. Semantic segmentation and classification of polycystic ovarian disease using attention UNet, Pyspark, and ensemble learning model. Expert Syst. 41, e13498 (2024).
Ndjene, J., Gamundani, A. M. & Kanyama, M. N. Leveraging Machine Learning to Detect and Predict Diabetes in Polycystic Ovary Syndrome Patients: A Review, in International Conference on Artificial Intelligence, Big Data, Computing and Data Communication Systems (icABCD), 2024, pp. 1–8. (2024).
Shivaram, J. M. Segmentation of ovarian cyst using improved U-NET and hybrid deep learning model. Multimedia Tools Appl. 83, 42645–42679 (2024).
Li, L., Cherouat, A., Snoussi, H. & Wang, T. Grasping with occlusion-aware ally method in complex scenes. IEEE Trans. Autom. Sci. Eng., (2024).
Wang, T. et al. ResLNet: deep residual LSTM network with longer input for action recognition. Front. Comput. Sci. 16, 166334 (2022).
Dhall, I., Vashisth, S. & Aggarwal, G. Smart healthcare system for reliable diagnosis of polycystic ovary syndrome, in Artificial Intelligence and Machine Learning Techniques in Image Processing and Computer Vision, ed: Apple Academic, 19–36. (2024).
Khushal, R. & Fatima, U. Fuzzy machine learning logic utilization on hormonal imbalance dataset. Comput. Biol. Med. 174, 108429 (2024).
Subha, R., Nayana, B., Radhakrishnan, R. & Sumalatha, P. Computational intelligence for early detection of infertility in women. Eng. Appl. Artif. Intell. 127, 107400 (2024).
Zhou, M. et al. Robust rgb-t tracking via adaptive modality weight correlation filters and cross-modality learning. ACM Trans. Multimedia Comput. Commun. Appl. 20, 1–20 (2023).
Wang, J. et al. Fixed-time formation control for uncertain nonlinear multi-agent systems with time-varying actuator failures. IEEE Trans. Fuzzy Syst., (2024).
Lim, J. et al. Predicting TCM patterns in PCOS patients: An exploration of feature selection methods and multi-label machine learning models. Heliyon. 10, (2024).
Dar, M. A., Maqbool, M., Qadrie, Z., Ara, I. & Qadir, A. Unraveling PCOS: exploring its causes and diagnostic challenges. Open. Health. 5, 20230026 (2024).
Fıstıkçıoğlu, N. Z. et al. Machine learning-based analysis of MRI radiomics in the discrimination of classical and non-classical polycystic over syndrome. Cukurova Med. J. 49, 89–96.
Moral, P., Mustafi, D. & Sahana, S. K. PODBoost: an explainable AI model for polycystic ovarian syndrome detection using grey wolf-based feature selection approach. Neural Comput. Appl., pp. 1–18, (2024).
Hsu, J. L. et al. Applying a Deep Learning Model for Total Kidney Volume Measurement in Autosomal Dominant Polycystic Kidney Disease. Bioengineering. 11, 963 (2024).
Suha, S. A. & Islam, M. N. An extended machine learning technique for polycystic ovary syndrome detection using ovary ultrasound image. Sci. Rep. 12, 17123 (2022).
Song, W. et al. Centerformer: a novel cluster center enhanced transformer for unconstrained dental plaque segmentation. IEEE Trans. Multimedia, (2024).
Huang, C. et al. Flow2GNN: Flexible Two-Way Flow Message Passing for Enhancing GNNs Beyond Homophily. IEEE Trans. Cybernetics. (2024).
Chen, B., Hu, J., Zhao, Y. & Ghosh, B. K. Finite-time observer based tracking control of uncertain heterogeneous underwater vehicles using adaptive sliding mode approach, Neurocomputing, vol. 481, pp. 322–332, (2022).
Luo, R., Peng, Z., Hu, J. & Ghosh, B. K. Adaptive optimal control of affine nonlinear systems via identifier–critic neural network approximation with relaxed PE conditions. Neural Netw. 167, 588–600 (2023).
Khan, A. Q., Sun, G., Khalid, M., Farrash, M. & Bilal, A. Multi-Deep learning approach with transfer learning for 7‐Stages diabetic retinopathy classification. Int. J. Imaging Syst. Technol. 34, e23213 (2024).
Tiwari, S. et al. SPOSDS: A smart polycystic ovary syndrome diagnostic system using machine learning. Expert Syst. Appl. 203, 117592 (2022).
Umapathy, S., Alhajlah, O., Almutairi, F., Aslam, S. & RK, A. F-Net: follicles net an efficient tool for the diagnosis of polycystic ovarian syndrome using deep learning techniques. Plos One. 19, e0307571 (2024).
Silambarasan, E., Nirmala, G. & Mishra, I. Polycystic ovary syndrome detection using optimized SVM and densenet. Int. J. Inform. Technol., (2024). /09/04 2024.
Nakhua, H., Ramachandran, P., Surve, A., Katre, N. & Correia, S. An ensemble approach for Ultrasound-Based polycystic ovary syndrome (PCOS) classification. Educational Administration: Theory Pract. 30, 14589–14597 (2024).
Jha, M., Gupta, R. & Saxena, R. Noise cancellation of polycystic ovarian syndrome ultrasound images using robust two-dimensional fractional fourier transform filter and VGG-16 model. Int. J. Inform. Technol. 16, 2497–2504 (2024).
Sakthivel, V., Prakash, P., Vishnukumar, K. & Min, D. Advanced diagnosis of polycystic ovarian syndrome using machine learning and multimodal data integration. Int. J. Adv. Comput. Sci. Appl., 15, (2024).
Verma, P., Maan, P., Gautam, R. & Arora, T. Unveiling the role of artificial intelligence (AI) in polycystic ovary syndrome (PCOS) diagnosis: A comprehensive review. Reproductive Sci., pp. 1–15, (2024).
Srinithi, V. & Rekha, R. Machine learning for diagnosis of polycystic ovarian syndrome (PCOS/PCOD), in 2023 International Conference on Intelligent Systems for Communication, IoT and Security (ICISCoIS), pp. 19–24. (2023).
Athithan, S., Sachi, S. & Singh, A. K. Ultrasound-Based ovarian cysts detection with improved Machine-Learning techniques and stage classification using enhanced classifiers. SN Comput. Sci. 4, 571 (2023).
Rashid, S. et al. Attention-Based Multiscale Deep Neural Network for Diagnosis of Polycystic Ovary Syndrome Using Ovarian Ultrasound Images, in 2023 15th International Congress on Ultra Modern Telecommunications and Control Systems and Workshops (ICUMT), pp. 44–49. (2023).
Pallavi, C., Soumya, S. & Narasanakuppe, V. C. Literature Review on Early PCOS Detection on Girl Child Using Artificial Intelligence or Machine Learning, Sparklinglight Transactions on Artificial Intelligence and Quantum Computing (STAIQC), vol. 4, pp. 17–31, (2024).
Zad, Z. et al. Predicting polycystic ovary syndrome with machine learning algorithms from electronic health records. Front. Endocrinol. 15, 1298628 (2024).
Ravishankar, T. N., Jadhav, H. M., Kumar, N. S. & Ambala, S. A deep learning approach for ovarian cysts detection and classification (OCD-FCNN) using fuzzy convolutional neural network. Measurement: Sens. 27, 100797 (2023).
Alamoudi, A. et al. A deep learning fusion approach to diagnosis the polycystic ovary syndrome (pcos). Applied Computational Intelligence and Soft Computing. 9686697, 2023. (2023).
Sumathi, M., Chitra, P., Sheela, S. & Ishwarya, C. Study and implementation of automated system for detection of PCOS from ultrasound scan images using artificial intelligence. Imaging Sci. J. 72, 828–839 (2024).
Acknowledgements
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Small Research Project under grant number RGP1/278/45.
Author information
Authors and Affiliations
Contributions
Data curation: J.P, G.S.A, S.P ; Writing original draft: J.P, G.S.A, S.P, R.S and A.Y.; Supervision: J.P, G.S.A, S.P; Project administration: J.P, G.S.A, S.P, R.S and A.Y.; Conceptualization J.P, G.S.A, S.P, A.Y, R.S, I.A.B, S.K and M.G.S; Methodology: J.P, G.S.A, S.P, A.Y, R.S, I.A.B, S.K and M.G.S, Validation: J.P, G.S.A, S.P, A.Y, R.S, I.A.B, S.K and M.G.S.; Visualization J.P, G.S.A, S.P, A.Y, R.S, I.A.B, S.K and M.G.S.; Resources: J.P, G.S.A, S.P, A.Y, R.S, I.A.B, S.K and M.G.S.; Review & Editing: J.P, G.S.A, S.P, A.Y, R.S, I.A.B, S.K and M.G.S.; All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jeyashanker, P., Georgewilliam Sundaram, A., Sadagopan, P. et al. Advanced holographic convolutional dense networks and Tangent runner optimization for enhanced polycystic ovarian disease classification. Sci Rep 15, 15719 (2025). https://doi.org/10.1038/s41598-025-98873-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-98873-5
Keywords
This article is cited by
-
Hybrid quantum-classical machine learning for enhanced PCOS classification
Quantum Machine Intelligence (2025)