Abstract
Ultramafic rocks are an abundant source of cations for CO2 mineralization (e.g., Mg) and elements for sustainability technologies (e.g., Ni, Cr, Mn, Co, Al). However, there is no industrially useful process for dissolving ultramafic materials to release cations for CO2 sequestration or mining them for energy-critical elements. Weathering of ultramafic rocks by rainwater, release of metal cations, and subsequent CO2 mineralization already naturally sequesters CO2 from the atmosphere, but this natural process will take thousands to hundreds of thousands of years to remove excess anthropogenic CO2, far too late to deal with global warming that will happen over the next century. Mechanical acceleration of weathering by grinding can accelerate cation release but is prohibitively expensive. In this article we show that gluconic acid-based lixiviants produced by the mineral-dissolving microbe Gluconobacter oxydans accelerate leaching of Mg2+ by 20× over deionized water, and that leaching of Mg, Mn, Fe, Co, and Ni further improves by 73% from 24 to 96 h. At low pulp density (1%) the G. oxydans biolixiviant is only 6% more effective than gluconic acid. But, at 60% pulp density the G. oxydans biolixiviant is 3.2× more effective than just gluconic acid. We demonstrate that biolixiviants made with cellulosic hydrolysate are not significantly worse than biolixiviants made with glucose, dramatically improving the feedstock available for bioleaching. Finally, we demonstrate that we can reduce the number of carbon atoms in the biolixiviant feedstock (e.g., glucose or cellulosic hydrolysate) needed to release one Mg2+ ion and mineralize one atom of carbon from CO2 from 525 to 1.
Similar content being viewed by others
Introduction
The accumulation of over a trillion tonnes of anthropogenic CO2 in the Earth’s atmosphere1,2,3,4 driving global climate change is one of the most pressing challenges of our time. To address this, the IPCC’s Special Report in 2018 stresses the necessity of deploying carbon dioxide removal (CDR) technology capable of removing tens of gigatonnes of CO2 from the atmosphere annually, paired with carbon-neutral energy technologies to prevent global warming from exceeding 1.5˚C above pre-industrial levels5.
Mineralization of CO2 is a highly attractive approach for permanent sequestration of CO2, as it converts atmospheric carbon into solid minerals as a permanent sink6,7. Carbon mineralization proceeds through a two-step process. First, ultramafic rocks are dissolved by weathering to release magnesium cations (Note S1). In the second step, magnesium ions are reacted with dissolved carbonate to form magnesite (MgCO3), a stable mineral that permanently stores CO28 (Note S1). A schematic of a proposed indirect carbonation method is shown in Fig. 1.
Proposed system for bio-accelerated weathering of ultramafic materials for carbon mineralization. This work focuses on the rate-limiting step, the metal leaching stage (enclosed in red box), where G. oxydans biolixiviant leaches magnesium ions and other metals from dunite. Downstream carbonation results in magnesite formation for CO2 storage, and metal recovery done chemically or electrochemically can extract energy-critical metals for commercial use.
Naturally occurring weathering of ultramafic rocks and subsequent carbon mineralization is thought to be responsible for removal of up to hundreds of millions of tonnes of CO2 each year8,9,10,11. In fact, because ultramafic rocks are so abundant12,13, reaction of all of the metal cations in all surface-accessible ultramafic material could capture 100 trillion tonnes of CO2 as carbonate minerals14 (at least 58× the excess CO2 in the atmosphere), making them a strong candidate for gigatonne-scale CDR15,8,16. However, natural carbon mineralization is partially offset by oxidation of rock organic carbon11,17. As a result, the time to naturally regulate atmospheric CO2 levels is estimated to be between tens and hundreds of thousands of years8,11,18,19,20.
The large-scale deployment of carbon-neutral energy technologies will also be necessary to control climate change5, but it will require vast quantities of metals21. In addition to metal cations needed for CO2 sequestration, ultramafic minerals are a potential low-grade ore source for energy-critical metals such as nickel (batteries and stainless steel), chromium (concentrated solar and geothermal energy), manganese (steel and batteries), cobalt (batteries), and copper (wiring and wind turbines)8,21,22,23,24,25 (Figure S2, Tables S1 and S2). This presents an opportunity to extract energy-critical metals from ultramafic rock while sequestering CO2.
However, extracting metals from ultramafic rocks poses a significant challenge. In aqueous mineral carbonation, the rate of carbon mineralization is limited by the slow kinetics of the leaching of metal ions from ultramafic sources14,26,27. While mechanical acceleration of ultramafic weathering through crushing, grinding, heating, chemically treating, and spreading over land is promising for its high sequestration potential and improved kinetics8,16,28,29,30, it requires large area footprints and high energy inputs involved in crushing, grinding, heating, and pressurizing, making it prohibitively expensive31. Likewise, technologies to extract the energy-critical elements present in low concentration in ultramafic rocks present formidable economic and environmental challenges24,32,33.
We hypothesize that microbially-accelerated mineral dissolution could overcome the slow kinetics and high costs14 of the dissolution of ultramafic material, releasing magnesium ions at a rate several orders of magnitude greater than in nature34,35. Over the past two decades, microbial mineral dissolution has revolutionized mining copper and gold from low-grade ores—a process known as biomining. Bacteria such as Acidithiobacillus ferrooxidans and related species are responsible for producing about 15% of the world’s copper supply and about 5% of its gold supply36 through iron-redox-mediated bioleaching, a process in which microbes alter the redox state of metals to extract them from mineral substrates37,38. Significant research has been performed to improve industrial-scale Cu bioleaching with consortia including A. ferrooxidans and non-domesticated microbes, resulting in processes that are fairly mature. While copper biomining is the only industrialized biomining process, organisms and processes are in development for rare earth elements (REE)39,40,41,42,43,44 and lithium45,46.
Biolixiviants produced by Gluconobacter oxydans are a strong candidate for a sustainable and strong catalyst for CO2 mineralization using ultramafic rocks11,47. A recent study from our team comparing the dunite dissolution performance of three mineral-dissolving microbes, Gluconobacter oxydans, Sphingomonas desiccabilis, and Penicillium simplicissimum, concludes that G. oxydans outperformed the other two in the extraction of all metals from dunite11.
G. oxydans efficiently produces a gluconic acid-based mineral-dissolving solution, called a biolixiviant, when glucose is present47,48. Previous studies have shown that biolixiviants from G. oxydans can be more effective than just gluconic acid at leaching REE42,49. In a recent article, our team identified genes that control the effectiveness of the G. oxydans biolixiviant at dissolving neodymium phosphate (synthetic monazite) but do not notably change its pH50. These results suggest that biologically-produced lixiviants could contain additional components, such as metal chelators, redox molecules, and flavins that improve overall metal leaching at fixed pH, allowing for more efficient and low-resource metal leaching. This could avoid the use of strong acids that can have harmful environmental effects such as acid mine drainage51.
Second, it is suspected that microbes interact with ultramafic materials in the wild, accelerating weathering processes52,53,54,55. Furthermore, laboratory mixed cultures have been shown to dissolve ultramafic material56. Meanwhile, in two companion articles, we demonstrate that three easily culturable isolated species (G. oxydans, Penicillium simplicissimum and Sphingomonas desiccabilis) can all accelerate the dissolution of ultramafic material11 and characterize mineral phase transformations due to bioleaching by G. oxydans57.
However, there has been little systematic study of microbial dissolution of ultramafic minerals. Furthermore, it remains unclear under what conditions a biolixiviant could be more effective than just an organic acid and why; could genetic engineering improve ultramafic bioleaching; what impact process parameters have on biological dissolution of ultramafic materials; the extraction efficiencies for energy critical elements; utilization of low-cost feedstock; and the carbon source demand of large-scale ultramafic bioleaching. To address these knowledge gaps, we provide some of the first systematic observations of dissolution of ultramafic minerals by the mineral-dissolving microbe, G. oxydans B58.
Results and discussion
In this work, we perform a study of G. oxydans bioleaching capability on dunite, an ultramafic rock composed of greater than 90% olivine (see composition in Figure S2, and Tables S1 and S2). For accelerated weathering technologies, olivine-rich dunite is the preferred feedstock due to olivine having the highest CO2 sequestration potential by mass, excellent carbonation kinetics, and high abundance58,59.
This study benchmarks the bioleaching efficacy of glucose-based and lignocellulosic sugar-based biolixiviants produced by G. oxydans on dunite, comparing conditions of whole-cell and filtered (cell-free) biolixiviants, varying pulp densities, aerobic and anaerobic conditions, and short-term and long-term leaching. This study also investigates how a high-performing mutant strain of G. oxydans could improve leaching in ultramafic minerals, serving as a preliminary study of how genetic engineering could further improve bioleaching for carbon mineralization. We demonstrate that the effectiveness advantage of the G. oxydans biolixiviant over gluconic acid alone grows with increasing pulp density. Finally, we demonstrate that the number of carbon atoms that need to be supplied by sugar feedstocks can be reduced to match the number of carbon atoms potentially sequestered as a mineral.
G. oxydans biolixiviants accelerate magnesium and energy-critical metal leaching by 20× over deionized water after 24 h
Biolixiviants from wild-type Gluconobacter oxydans effectively leached magnesium from ultramafic rocks (Fig. 2A). The efficiency of extraction measures how much of the available metal is leached from the mineral, and is calculated from the ratio of metal concentrations in the leachate and the original concentration in the source multiplied by the pulp density (ratio of solid mass to liquid volume [g/L]),
Deionized water is used as a control for these experiments since it estimates the improvements made by bio-accelerated leaching to leaching from environmental water sources, such as rainwater. In traditional accelerated carbon mineralization with ultramafic rocks, crushed rocks are dispersed over land, where water from the environment is the solvent for magnesium8,29,30. Comparing the concentrations of magnesium between leaching with biolixiviants and water provides a rough estimate for the order of magnitude in which carbon mineralization is accelerated by G. oxydans.
After 24 h of leaching with dunite at 1% pulp density, magnesium was leached with an extraction efficiency of 11% (300 ppm), corresponding to an increase of 20× when compared to the deionized water control (Fig. 2A). The pH of wild-type biolixiviants was 2.39 ± 0.02 across all experiments using wild-type biolixiviants. At the start of each leaching experiment, the initial concentrations of all metals in biolixiviants and controls are below detection limits. Figure S3 shows how the metal concentrations in controls and G. oxydans leachates diverge within the first few hours of leaching, starting with initial metal concentrations of zero.
Leaching dunite at 1% pulp density with wild-type (WT) G. oxydans biolixiviant improves leaching of magnesium by 20-fold when compared to leaching with deionized water (control). However, the G. oxydans biolixiviant is only 6% better than just gluconic acid at the same pH. Bioleaching of dunite at 1% pulp density shaking at 22 ˚C for 24 h was performed with wild-type G. oxydans biolixiviant, gluconic acid diluted to the same pH with deionized water, and a deionized water control. Panels (A) to (E) show leaching concentrations and extraction efficiencies of Mg, Mn, Fe, Co, and Ni. Wild-type G. oxydans biolixiviants slightly improved metal leaching compared to gluconic acid across all metals. Stars denote significant difference between gluconic acid leaching and wild-type biolixiviant leaching by a Welch’s two-sample t-test, where, p < 0.05 (*), p < 0.01 (**), p < 0.001 (***).
Bioleaching of metals by G. oxydans increases by 73% from 24 to 96 h
The most efficiently extracted metal at 24 h was Mn (Fig. 2B), extracting over 15% of Mn from dunite (corresponding to a leachate concentration of 1.6 ppm). Fe, Co, and Ni were also leached highly, with efficiencies between 11% and 15% (Figs. 2C, D, and E, respectively). Al and Cr were also leached, but much less efficiently (Figure S4). Cr was the least efficiently extracted metal, with extraction of no more than 0.3% under any condition (Figure S4 right column). We speculate that chromium is significantly less leached since the chromite inclusions within dunite are relatively more stable than olivine and resistant to acid dissolution60.
When leaching for longer than 24 h, bioleaching efficiency continues to increase (Fig. 3). In a 96-hour bioleaching analysis of dunite at 1% pulp density, bioleaching increased between 24 and 96 h by an average of 73% for Mg, Mn, Fe, Co, and Ni (Fig. 3). After 96 h, the leaching of magnesium in biolixiviant is 42 times that of water (Fig. 3). This shows that biolixiviants from G. oxydans improve the overall leaching of metals at a rate faster than water.
Increasing leaching time to 96 h increases extraction efficiency by an average of 73% between 24 and 96 h for Mg, Mn, Fe, Co, and Ni. Biolixiviants from wild-type G. oxydans leached dunite at 1% pulp density for 96 h, shaking at 22 °C. Mg, Mn, Fe, Co, and Ni concentrations were measured from leachates every 24 h for 96 h. Error bars indicate standard deviation of measurements from duplicate measurements.
The G. oxydans biolixiviant is only 6% better than gluconic acid at 1% pulp density
A secondary control was performed using only gluconic acid diluted with deionized water to the same pH as the acid biolixiviants produced by G. oxydans. Reed et al.42 and Antonick et al.49 demonstrated that biolixiviants produced by G. oxydans were noticeably more effective at bioleaching than gluconic acid alone. However, recent anecdotal evidence suggests this is not the case under all conditions. In our hands, when bioleaching ultramafic materials at low pulp density (1%), G. oxydans-produced biolixiviants only slightly outperform gluconic acid in leaching most metals (Fig. 2). Wild-type G. oxydans biolixiviants leached only 6% more magnesium compared to pure gluconic acid that was diluted with ultrapure water to the same pH after 24 h of leaching. Furthermore, at least with low pulp density leaching, filtering cells from the biolixiviant did not significantly affect bioleaching, at least under the conditions of these tests (Fig. 4). This was done to assess if there was a significant residual effect of microbial activity on leaching after biolixiviant production. Furthermore, stoppering flasks during leaching to prevent gas exchange did not significantly affect bioleaching (Figure S5). Later in the article, we ask if there are conditions under which the G. oxydans biolixiviant has a significant performance advantage for bioleaching ultramafic materials.
Filtering cells from the G. oxydans biolixiviant does not notably reduce the bioleaching of metals from dunite after 24-hour leaching. We compared Bioleaching of 1% dunite by sugar water (col. 1), with whole cell lixiviant (col. 2), and with a biolixiviant that was clarified by filtration with a 0.22 μm syringe filter (col. 3). Filtered and unfiltered biolixiviants were at identical pH, produced from duplicate colonies, and were added to ultramafic minerals in identical conditions. Filtering biolixiviants before bioleaching resulted in similar metal concentrations when compared to whole-cell biolixiviants when leaching dunite. A Welch’s two-sample t-test shows that no significant difference was detected between the unfiltered and filtered groups, with p-values ranging from 0.2 to 0.9. This indicates that based on a 24-hour leaching study, the presence of G. oxydans does not significantly improve or diminish the effect of the biolixiviant on leaching in short term leaching.
Cellulosic hydrolysate can be used as an alternative sugar feedstock for bioleaching
Techno-economic analysis shows that glucose feedstock for biolixiviant production is the biggest cost driver in REE biomining with G. oxydans, accounting for almost half of the total cost61. Furthermore, the sheer scale that carbon sequestration needs to operate at, means that enormous amounts of feedstock will be required, potentially placing a significant demand on the world’s biomass supply62.
Lignocellulose, found in agricultural waste, can be a renewable carbon source for microbial growth and production once monomeric sugars are released through acid pretreatment and enzymatic hydrolysis, making a glucose-rich cellulosic hydrolysate63,64. Lignocellulosic sugars are being increasingly investigated as a potential alternative sugar source for several applications, including biofuels/bioethanol production and substrates for cellular production. Utilizing lignocellulose-derived sugars could serve as a high glucose replacement feedstock that would greatly improve the cost-efficiency of bioleaching for carbon capture technologies65,66. G. oxydans has been shown to be able to convert the glucose and xylose sugars in cellulosic hydrolysate into gluconic acid and xylonic acid at extremely high efficiencies67,68,69. Replacing pure glucose with cellulosic hydrolysate could further improve the economic and environmental viability of bio-accelerated weathering.
G. oxydans has been shown to efficiently produce gluconic acid and xylonic acid from lignocellulosic hydrolysate feedstocks67,68,69, making industrial-scale, bio-accelerated carbon mineralization far more economically feasible. We investigated if biolixiviants produced from cellulosic hydrolysate, rather than pure glucose, could be effective at leaching dunite. The cellulosic hydrolysate (CH) used in this study is a glucose-rich solution produced from the enzymatic hydrolysis of concentrated corn stover (Table S3).
Biolixiviants produced from cellulosic hydrolysate were on average 28% less effective in leaching magnesium compared to glucose-based biolixiviants (Fig. 5). Leaching of Mn, Fe, Co, and Ni was also reduced by an average of 52% compared to glucose-based biolixiviants (Fig. 5). Dunite was added at 1% pulp density, and a leaching comparison between CH-based and glucose-based lixiviants was conducted in parallel and in identical conditions. The cellulosic hydrolysate added was diluted with deionized water so that the total sugar concentrations were equal to the amount of glucose added for glucose-based biolixiviants.
Use of cellulosic hydrolysate as a biolixiviant feedstock does not considerably reduce bioleaching efficiency. Panels (A) to (E) show comparison of bioleaching of Mg, Mn, Fe, Co, and Ni by Gluconobacter oxydans when using glucose and cellulosic hydrolysate (CH) as as biolixiviant feedstock. CH-based biolixiviant leaches magnesium on average at 72% the efficacy of glucose-based biolixiviant. Leaching was performed with 1% dunite shaking at 22 °C for 24 h. Stars denote significant difference compared with glucose-based biolixiviants by a Welch’s two-sample t-test, where, p < 0.05 (*), p < 0.01 (**), p < 0.001 (***).
Despite a 28% average reduction in Mg leaching, we were unable to detect a statistically meaningful difference between glucose- and CH-based leaching of Mg using a Welch’s two-sample t-test. This was due to the small sample size and variability in CH-based biolixiviant leachate measurements. Data collected for CH-based biolixiviants resulted in greater variability than data with glucose-based biolixiviants. ICP-MS analysis of cellulosic hydrolysate samples showed that the CH used in this study contains trace concentrations of metals, possibly as a result of the manufacturing process (Table S4). When calculating the amounts of metal leached from CH-based biolixiviants, the concentrations of metal in a cellulosic hydrolysate control study were subtracted from the total detected to eliminate metal concentrations that were not extracted from dunite. In low pulp density studies, the concentrations of metals in the cellulosic hydrolysate feedstock interfered with metals leached, resulting in higher overall error.
Cellulosic hydrolysate produced from corn stover contains several toxins such as organic acids, furan derivatives, and phenolic compounds that could inhibit the growth of G. oxydans and its production of biolixiviant70. Cellulosic hydrolysate also significantly increases viscosity, resulting in lower oxygen transfer rates that limit the fermentation of glucose to gluconic acid68. Despite this, leaching results show that G. oxydans can produce a biolixiviant from cellulosic hydrolysate capable of leaching metals at concentrations significantly greater than controls; however, further analysis and experimentation are needed to show if CH can be a viable replacement to glucose without a significant reduction in performance.
Although cellulosic hydrolysate contains mild antibiotic properties due to its acidity and certain toxic compounds, this could be advantageous for the industrial scale use of it as a feedstock for biolixiviant production. G. oxydans has high acid resistance and is capable of growing and producing biolixiviant with cellulosic hydrolysate, while many other microbes have low tolerance mechanisms for it. This could allow for cellulosic hydrolysate to be used as an ultra-low-cost feedstock while also preventing the growth of competing microbes. The toxic components of cellulosic hydrolysate could also be a genetic engineering target for G. oxydans, where mutants that have better resistance mechanisms can be selected for to improve biolixiviant production.
G. oxydans mutant strain ΔpstS, P112:mgdh improved magnesium leaching by 12% over wild-type
A preliminary study using the mutant strain G. oxydans ΔpstS, P112:mgdh43 shows that under identical growth and leaching conditions, mutant strains of G. oxydans can be more effective than wild-type in leaching magnesium and energy-critical metals from dunite.
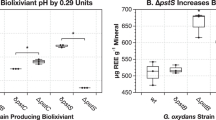
A bioleaching analysis over 96 h using dunite at 1% pulp density was conducted with biolixiviant from wild-type and mutant G. oxydans. Bioleachate samples were taken and analyzed every 24 h up to 96 h using ICP-MS. The strain used is G. oxydans ΔpstS, P112:mgdh, which was engineered to improve acid production for metal leaching43. It contains a clean deletion of the pstS gene which is involved in phosphate signaling and transport44, and an up-regulation with promoter P112 of the mgdh gene (Membrane-bound Glucose DeHydrogenase) which is critical for converting glucose into gluconic acid71. The ΔpstS, P112:mgdh mutant was previously shown to decrease the pH of biolixiviants compared to wild-type, and it increases the overall leaching of REE43. The biolixiviant produced from the ΔpstS, P112:mgdh mutant had a pH of 2.01 ± 0.02 across all replicates, while the pH of wild-type biolixiviants were 2.39 ± 0.02.
The analysis shows that ΔpstS, P112:mgdh mutant biolixiviant increases magnesium leaching by an average of 12% over all samples taken between 24 and 96 h of leaching. Mn, Fe, Co, and Ni had an average improvement in leaching of 12% using mutant biolixiviant compared to wild-type (Fig. 6).
Bioleaching efficiency of magnesium increased by an average of 12% when leaching with a high performing G. oxydans mutant. The G. oxydans mutant contains a deletion of pstS phosphate signaling and transport gene, alongside up-regulation of the mgdh gene that codes for the Membrane-bound Glucose DeHydrogenase (MGDH) with the P112 promoter (G. oxydans ΔpstS, P112:mdgh). Leaching was done with dunite at 1% pulp density shaking at 22 °C over 96 h. Stars and dots denote a statistically significant difference between wild-type G. oxydans and G. oxydans ΔpstS, P112:mdgh mutant by a Welch’s two-sample t-test, where, p < 0.1 (.), p < 0.05 (*), p < 0.01 (**), p < 0.001 (***).
The entire genome of G. oxydans is yet to be completely characterized, and several other genes may exist that are critical in controlling metal leaching from ultramafic rocks. Future work to characterize the genome of G. oxydans in terms of its functions related to ultramafic leaching is vital to engineer better mutant strains that can improve the performance of bio-accelerated weathering. The ΔpstS, P112:mgdh mutant is engineered to maximize gluconic acid production; however, studies have shown that for rare earths leaching, G. oxydans mutants can be screened to identify genes that specifically affect rare earth extraction50. Our research group has created a quality-controlled (QC) whole genome knockout collection for G. oxydans B58, containing a collection of 2,733 transposon disruption mutants, each with a different nonessential gene disrupted. This QC collection enables a high-throughput screen of the G. oxydans genome, selecting for significant changes to dunite leaching efficacy. This will provide us with a roadmap for genetically engineering G. oxydans for greater ultramafic bio-accelerated weathering capability.
G. oxydans biolixiviants are 3.2× more effective than gluconic acid at 60% pulp density
At higher pulp densities and longer leaching times, Gluconobacter oxydans biolixiviants significantly outperformed gluconic acid at the same pH. We compared Gluconobacter oxydans biolixiviants and gluconic acid metal leaching with 5%, 10%, and 60% dunite pulp density, where leachate metal concentrations were measured every 24 h for 72 h. The gluconic acid was diluted with ultrapure deionized water to match the pH of biolixiviants, and leaching was performed in identical conditions with the same mineral substrates (DUN-2). After 72 h of leaching, wild-type biolixiviants leached 1.74 times more magnesium than gluconic acid from 5% dunite, 2.87 times more from 10% dunite, and 3.25 times more from 60% dunite (Fig. 7). At higher pulp densities, Gluconobacter oxydans biolixiviants exhibit significantly higher leaching efficiency than gluconic acid. This analysis suggests that metal leaching from dunite is not only controlled by acidolysis, but might contain other biologically-synthesized components that amplify extraction50.
At 60% pulp density, the wild-type G. oxydans biolixiviant was 3.2× more effective at bioleaching dunite than gluconic acid diluted to the same pH with pure water. A comparative bioleaching study was conducted comparing the leaching efficacy of wild-type G. oxydans biolixiviants and just gluconic acid at the same pH. Magnesium concentrations in leachate were measured after 24, 48, and 72 h of leaching with 5% (A), 10% (B), and 60% (C) dunite (DUN-2). At higher dunite pulp densities, the improvement of Gluconobacter oxydans biolixiviants compared to gluconic acid was amplified. After 72 h, biolixiviants were 74% better than gluconic acid at leaching 5% dunite, 1.87 times better at leaching 10% dunite, and 2.25 times better at leaching 60% dunite. Stars denote significant difference between gluconic acid leaching and wild-type biolixiviant leaching by a Welch’s two-sample t-test, where, p < 0.05 (*), p < 0.01 (**), p < 0.001 (***).
Magnesium leaching relative to glucose input can be greatly improved by process and genetic engineering
Glucose is the cost-limiting factor in utilizing G. oxydans biolixiviant as a weathering accelerant61. Furthermore, the sheer scale at which carbon sequestration must operate requires enormous amounts of microbial feedstock, possibly enough to make a significant demand on the world’s biomass supply62. Therefore, it is essential to prioritize maximizing carbon dioxide sequestration while minimizing feedstock consumption in order to mitigate the strain on worldwide biomass resources.
We define the carbon investment to carbon sequestration ratio, kseq, as the number of carbon atoms in the feedstock (e.g., glucose) needed to liberate one magnesium ion (and hence sequester the single atom of carbon in a CO2 molecule11,62,
Lower kseq values indicate that more magnesium was leached given a certain glucose input, while high kseq values indicate relatively lower magnesium leaching for the same amount of feedstock consumed. A kseq value of 6 for a glucose feedstock means that one glucose molecule is necessary to liberate one magnesium ion (which has the potential to sequester 1 CO2). A kseq value of 1 means that a minimum of one feedstock carbon atom is needed to eventually sequester one CO2 molecule. The statement that each liberated magnesium ion results in one CO2 molecule sequestered assumes a 100% carbonation rate in a future downstream process that converts magnesium in leachate into magnesite. kseq only illustrates the lower limit of sugar input necessary to sequester CO2.
We can use kseq to estimate the lower limit price of carbon sequestration and the total amount of feedstock needed to sequester a useful amount of CO2 (e.g., 20 gigatonnes of CO2 per year). The mass of feedstock, Mfeedstock, needed to sequester a given mass of CO2, \(\:{M}_{{CO}_{2}}\),
where, MWfeedstock is the molecular weight of the feedstock; \(\:{\text{M}\text{W}}_{{\text{C}\text{O}}_{2}}\) is the molecular weight of CO2, and nC, feedstock is the average number of carbon atoms in each molecule of the feedstock. Likewise, the cost to sequester a given mass of CO2,
Where pfeedstock is the price per unit mass of the feedstock.
In this work, we succeeded in reducing kseq from 525 to 1.0 (almost 3 orders of magnitude) (Fig. 8). With 1% dunite and 24 h of leaching, the approximate kseq is 525 carbon invested/magnesium leached. One method to reduce kseq is to increase the time of leaching. After 96 h of leaching, the magnesium concentration increased by 81% when compared to 24 h leaching at the same pulp density (Fig. 3). When increasing the time of leaching to 10 days, the magnesium concentration improved by an average of 147% (Fig. 8). Increasing the pulp density of dunite also reduces kseq. When compared to 1% pulp density of dunite, increasing to 10% pulp density resulted in a 5-fold increase in magnesium leaching after 24 h (Fig. 8). When combining 10% pulp density with 10 days of leaching, magnesium leaching increased by 9.2 times that of 1% pulp density leached for 1 day (Fig. 8), resulting in a kseq of 57, since glucose input remained unchanged. For the lowest kseq values achieved, fine-particle dunite at a pulp density of 60% was leached for 19 days with three different methods (Fig. 8 Leaching Attempts #12–14). The first method (Fig. 8 Leaching Attempt #12) was direct addition of 60% dunite, the second method (Leaching Attempt #13) was a 2-step dunite addition, where 30% dunite was added on day 0 and another 30% added on day 10, and the third method (Leaching Attempt #14) was a 2-step dunite addition where the first 30% dunite feed was removed from the leachate before the second 30% addition on day 10. All three leaching methods resulted in a kseq equal to or less than 1.0, with the lowest kseq being 0.96 on Leaching Attempt #13 in Fig. 8.
Process and genetic improvements reduce the amount of feedstock carbon needed to potentially sequester one CO2 molecule from 525 to 1. Increasing leaching time, pulp density, and reducing average particle sizes were able to reduce kseq (glucose carbon investment vs. carbon sequestration ratio) by several orders of magnitude. Increasing dunite pulp density from 1–10% had a larger impact in reducing kseq than increasing leaching time from 1 to 10 days. Combining the ΔpstS, P112:mdgh double mutant with high pulp densities and long leaching times resulted in a kseq value of 1 in three experiments (Col. 10–12). Leaching attempts #1–9 used DUN-1 and attempts #10–12 used DUN-3 with a particle size of less than 74 microns as the leaching substrate.
What implications do these results have for CO2 sequestration? For kseq = 525, when using glucose as a feedstock, the cost of sequestering 1 tonne of CO2 is $358,000, a long way above the US Department of Energy’s (DOE) target of $100 per tonne72, and well above existing technologies31. On the other hand, if we could reduce kseq to 1 (as in Fig. 8) at industrial scale, the cost of carbon sequestration (at least the microbial feedstock part of the cost) through bio-accelerated weathering starts to become comparable to (still high) current technologies, at $682 per tonne31. If we could reduce kseq to 0.15 (about a 6-fold reduction over our best attempt, much smaller than the reduction we achieved in the course of writing this article) then we could reduce the feedstock cost for sequestering 1 tonne of CO2 to $100. This would not account for the costs of mining ultramafic material, but nor does it account for the offset cost of energy-critical metal recovery. However, using this to sequester 20 gigatonnes of CO2 a year5 would require 2 billion tonnes of glucose, over 10 times current global annual sugar production73.
Sugars derived from cellulosic biomass are potentially much more abundant than glucose that is derived from cereal crops like corn, sugar cane, and sugar beet74. While there is no widespread production of cellulosic sugars today that is not immediately diverted to cellulosic ethanol production, this could happen in the future. Furthermore, it is estimated that cellulosic sugar production could be as low as $100 per tonne75 (in contrast to around $1,000 per tonne for glucose). Although cellulosic sugars are abundant, they are not limitless. Slade et al.76, made estimates of global bioenergy supply, and concluded that several tipping points would occur as the amount of biomass withdrawn from the biosphere for bioenergy (or some other chemical production, such as carbon sequestration) increased, forcing increasingly unpalatable choices on global society including mass adoption of vegetarian diets, mass deforestation, population control, or combinations thereof. Their analysis suggests that the first global agricultural tipping point (choosing one of the three unpalatable options) would occur when biomass was withdrawn at a rate of around 7 gigatonnes per year62. Assuming that cellulosic sugars are on average a 50:50 mix of C5 and C6 sugars, then to reduce the cellulosic biomass demand to sequester 20 gigatonnes of CO2 to 3.5 gigatonnes, kseq needs only to be reduced to 0.25.
Although kseq serves as a useful metric for displaying sugar investment in terms of CO2 sequestration potential, it is only a theoretical value whose real value will depend on the conversion rates of downstream CO2 mineralization. Since glucose inputs are a major cost driver in bio-accelerated weathering61, kseq can help estimate lower-limit costs of scaled operations. Future scale-up work will be necessary to formulate more accurate estimates of cost before they can be compared with other direct CO2 mineralization technologies.
Possibilities for downstream carbon mineralization
On its own, G. oxydans bioleachate saturated with magnesium ions will be difficult to convert into stable carbonate minerals. Due to the nature of acid leaching, the final pH of biolixiviants post-leaching tended to be low (4–5) in our experiments, making carbon mineralization chemically unfavorable. Since pH has a significant influence on the precipitation of carbonates due to the lower availability of carbonate ions in acidic conditions77,78, pH adjustments are required to increase carbonate mineral yields downstream. Increasing the concentration of gaseous CO2 injected or sparged through the leachate increases the total dissolved inorganic carbon (DIC); however, this reduces the pH of the leachate, reducing carbonate ion availability and decreasing the saturation index of carbonate minerals2,77.
A possible downstream pathway to adjust the pH of leachates to make carbon dioxide complexation chemically favorable is to add ammonia from biological or natural sources to synthesize magnesite, MgCO3, or nesquehonite, MgCO3·3H2O. Adding ammonia to CO2-sparged magnesium chloride solutions at ambient temperatures was shown to completely convert free magnesium ions into nesquehonite in about ten minutes79. Furthermore, ammonia can be recycled in this process through recovery of ammonia using activated carbon, which has been shown to recover ammonium chloride salts and separate them into ammonia and hydrochloric acid at near ambient conditions80. Another possibility is to simply add sodium hydroxide that has been electrically synthesized from carbon-neutral pathways.
Microbes that accelerate carbon mineralization, such as cyanobacteria, have been observed to accelerate mineralization in legacy alkaline mine tailings34,35. Coupling bio-accelerated weathering from Gluconobacter oxydans with biotic downstream processes could maximize carbonate precipitation in a low-resource and energy-efficient process that could be genetically engineered further.
Scale-Up and future steps
In order to show that bio-accelerated weathering is a feasible option for gigatonne-scale CO2 sequestration, future work will be required to show that a scaled-up bioprocess is possible without a significant reduction in efficiency compared to the bench-scale experiments shown here.
Bench-scale lab results rarely have equal yields and efficiencies when scaled up to pilot- or industrial-scale bioprocesses. Being an obligate aerobe, G. oxydans requires air exchange and consistent mixing for optimal growth. Furthermore, optimized mixing of the mineral substrate with biolixiviants, rate of glucose supplementation, rate of mineral addition, and optimized particle sizes for leaching are yet to be thoroughly investigated. To address these scale-up challenges, all process parameters will need to be investigated in bioreactor experiments.
Another consideration is how cell viability changes throughout the leaching process. G. oxydans has been shown to be resilient in high metal concentrations11, unlike several other microbes. However, having high pulp densities of dunite would be a growth challenge for G. oxydans. Despite this, cell viability during leaching may not be a critical parameter for the effectiveness of leaching by G. oxydans biolixiviants. For short term leaching, it is shown that having cells present in the biolixiviant has an insignificant effect on leaching (Fig. 4). Since biolixiviants are produced in an independent step before leaching begins (Figure S1), it is not essential to have live cells present during leaching. However, possible direct microbe-mineral interactions and continued acid production may have an effect on long-term leaching and scaled processes.
Overcoming the deceleration of leaching over time is also a future scale-up challenge to address. The rates of leaching were shown to slow down over the 96 h they were monitored (Fig. 3). As ultramafic minerals are dissolved by biolixiviants, passivation layers can form from amorphous silica (Note S1) or other mineral byproducts. Furthermore, as the solution nears saturation of metal cations, dissolution rates are expected to stagnate. Future investigations into overcoming the passivation layers and developing integrated mineralization technologies to allow for continuous leaching will be necessary for scale-up.
Utilizing lignocellulosic-based sugars as an alternative sugar feedstock also poses a challenge in scale-up. Although G. oxydans has been shown to effectively produce biolixiviants from cellulosic hydrolysate, the state of lignocellulosic sugar production is underdeveloped. Large-scale production of sugars derived from second-generation biomass (lignocellulose) is still limited, and developments in improving efficiency and yield are ongoing. Advances in the supply chain of lignocellulosic biomass and optimization in pretreatment and enzymatic hydrolysis will be necessary in the near future for the large-scale implementation of lignocellulosic-based sugars for bio-accelerated weathering66.
Conclusion
This article shows that G. oxydans possesses the capability to bioleach metals for energy technologies and carbon mineralization from dunite. At low pulp densities (1%), the G. oxydans organic acid-based biolixiviant is 20× more effective at Mg leaching than deionized water. At high pulp densities (60%), the biolixiviant is 3.2× better than even gluconic acid alone. This suggests that microbially-accelerated weathering of ultramafic materials could be an environmentally-friendly method for supplying Mg for mineralization of CO2, and metals like Ni and Co for sustainable energy technologies at rates that can far exceed equivalent abiotic methods.
Several pH swing methods have been shown to be highly effective for greatly accelerating olivine carbonation7,81. However, scaling up these technologies to meet the world’s CO2 removal requirements may involve high energy costs82 and environmental drawbacks like those found in strong acid leaching involved with large scale energy-critical metal mining. Other methods of accelerated weathering involving energy-intensive preprocessing of mineral feedstocks by crushing, grinding, and chemically treating ultramafic rocks also come with their own environmental and scaling drawbacks. In situ accelerated ultramafic weathering can result in environmental leaching and environmental drainage of harmful doses of nickel, chromium, and cobalt59,83,84,85. These methods also involve slow kinetics that depend on high energy inputs to be accelerated and enormous land use7,28,82,86.
Bio-accelerated weathering may be an alternative approach to mineral weathering for carbon mineralization. When engineered further, this strategy may prove an economically-viable and scalable option that avoids the environmental concerns with in situ mechanically-accelerated weathering and strong acid leaching51,59,84,85,87, as well as the energy costs involved in high temperature and pressure reactions14,82. Energy requirements for heating in common abiotic carbonation processes, such as ammonium salts-driven pH swings and HCl pH swings, often result in CO2 emissions greater than the amount sequestered82. G. oxydans-based bioleaching was shown to be effective at ambient temperature and pressure, removing a major energy burden for this process.
In this article, we were able to reduce the number of carbon atoms in glucose needed to sequester one molecule of CO2 from 525 to 1. However, further work needs to be done to optimize G. oxydans for enhanced weathering for carbon mineralization. Maximizing the leaching of magnesium while minimizing carbon inputs and lowering the economic costs are key steps to making this technology viable for eliminating the several gigatonnes of carbon dioxide in excess on our planet.
Materials and methods
Ultramafic samples were collected from the Day Brook Dunite (35˚ 58’ 0.45” N, 82˚ 16’ 58.60” W) in western North Carolina88. The chemically weathered exterior of the dunite was removed using a tile saw (151991, MK Diamond Products, Inc., Torrance, CA). The trimmed interior was crushed into powder using a SPEX SamplePrep 8000M mill and a tungsten carbide vial. Before bioleaching experiments, the mineralogy of powder from two samples was analyzed via x-ray diffraction (XRD) with a Bruker D8 Advance ECO powder diffractometer at Cornell Center for Materials Research (CCMR). The instrument is equipped with a Copper K-α X-ray source. Analyses were made at 5 to 60 degrees, with an increment of 0.019 degrees and a dwell time of 0.3 seconds per step. To mitigate Fe fluorescence the detector’s lower discriminator was raised from 0.110 V to 0.182 V. XRD shows that sample DUN-1 consists of 64 wt. % olivine, 27 wt. % lizardite, 5 wt. % antigorite, and 4 wt. % talc. Sample DUN-2 contains 37 wt. % olivine, 59 wt. % lizardite, 2 wt. % talc, and 2 wt. % meixnerite. Sample DUN-3 contains 92 wt. % olivine, 5 wt. % antigorite, 2 wt. % lizardite, and 1 wt. % talc. Major and trace element compositions of the bulk rock were determined at the Hamilton Analytical Laboratory in Clinton, NY via XRF and LA-ICPMS and are shown in Tables S1 and S2.
Three separate dunite samples were used in bioleaching studies. DUN-1 was used in the leaching studies shown in Figures 2, 3, 4, 5, 6, and Supplementary Figures S4 and S5. Due to the depletion of DUN-1 in previous experiments, DUN-2 was used to produce the data used in Figure 7. DUN-3 was used in Leaching Attempts #10 and #12-14 in Figure 8 and Supplementary Figure S3.
Microorganisms and media
Gluconobacter oxydans strain NRRL B-58 (GoB58) was obtained from the American Type Culture Collection (ATTC), Manassas, VA. In all experiments, GoB58 was cultured in Yeast-Peptone-Mannitol media (YPM; 5 g L-1 yeast extract (C7341, Hardy Diagnostics, Santa Maria, CA), 3 g L-1 peptone (211677, BD, Franklin Lakes, NJ), 25 g L-1 mannitol (BDH9248, VWR Chemicals, Radnor, PA)).
Gluconobacter oxydans mutant strain ΔpstS, P112:mdgh contains a clean deletion of the gene encoding the periplasmic phosphate-binding protein, PstS, and an up-regulation of the membrane-bound glucose dehydrogenase gene with promoter P112. This mutant strain was previously constructed by Schmitz et al.43.
Growth and organic acid production
GoB58 was inoculated into YPM liquid culture then incubated for 24 h at 30 °C and 200 rpm in a shaking incubator (Infors HT Multitron). Cultures were then back-diluted to OD590 of 0.05 in a total volume of 25 mL YPM in 50 mL flasks. The flasks were then incubated at 30 °C and 200 rpm until the OD590 reaches approximately 1.5 after about 48 h. Cultures were then split into 7.5 mL triplicates with 7.5 mL of 40% glucose added to each triplicate, for a total volume of 15 mL. The glucose and bacterial mixture was then incubated for 48 h at 30 °C and 200 rpm, starting biolixiviant production. After 48 h, the pH of each flask is measured (VWR Symphony B10P.).
Cellulosic hydrolysate (National Renewable Energy Laboratory, Golden, CO) was used in some leaching studies as an alternative glucose feedstock. In experiments using cellulosic hydrolysate, it was diluted to 80% concentration so that the total sugar concentrations (Table S2) would be equal to 40%. Then 7.5 mL of 80% cellulosic hydrolysate was added in replacement of 7.5 mL of 40% glucose.
Bioleaching studies
Bioleaching experiments were conducted with dunite at a pulp density of 1–10% wt/vol, as specified. Minerals were added to 15 mL of biolixiviant, then agitated at room temperature in a shaking incubator for 24 h. After 24 h, the ultramafic rock and biolixiviant slurry was centrifuged and the bioleachate was extracted for ICP-MS analysis.
Analytical methods
Leachates from bioleaching experiments were filtered through a 0.45 μm AcroPrep Advance 96-well filter plate (8029, Pall Corporation, Show Low, AZ, USA) by centrifugation at 1,500 × g for 5 min, then diluted to 1% with 2% trace metal grade nitric acid (JT9368, J.T. Baker, Radnor, Pennsylvania, USA). Samples were analyzed with an Agilent 7800 ICP-MS. The concentrations of metals were measured against a custom ICP-MS standard solution containing 10 mg/L of Al, As, B, Ca, Cd, Co, Cr, Cu, Fe, K, Mg, Mn, Mo, Na, Ni, P, Pb, S, Sb, Se, Si, Sn, and Zn (High Purity Standards, North Charleston, SC) and an ICP-MS internal standard (5188–6525, Agilent Technologies, Santa Clara, CA). Quality checks were done with intermittent standards and blanks as specified in the ICP-MS guidelines and protocols89.
Limitations of the study
This study was designed to establish a baseline understanding of ultramafic rock leaching by G. oxydans. The starting ultramafic rocks were chosen due to their high olivine/forsterite content and low complexity (lower content of other minerals). More industrially-applicable and abundant starting materials, such as lower olivine-containing forms of dunite, ultramafic mine tailings, and highly serpentinized ultramafic rocks will be investigated in future studies.
This study only observes G. oxydans leaching over short periods at relatively low pulp densities. While some experiments did include high pulp densities for long periods (Fig. 8), a rigorous examination of how leaching evolves over long periods was not conducted. Future experimentation and cost analysis is necessary to determine optimal mineral pulp densities and leaching times.
This study focuses on the extracted metal content found in solution during leaching. A complete mass balance that monitors the starting and ending mineral substrates will be addressed in future studies. Future studies will also include surface area measurements of starting and post-leaching mineral substrates.
Statistics
A Welch’s two-sample t-test was performed to determine statistical significance in comparative bioleaching studies. A Welch’s two-sample t-test was done since some studies, such as a comparison of sugar feedstocks (Fig. 5), resulted in differences in variances between the two groups tested. The Welch’s t-test is more appropriate in instances when the variances between two groups cannot be considered as equal. Significance was displayed in all figures as dots and stars, where p < 0.1 (.), p < 0.05 (*), p < 0.01 (**), p < 0.001 (***).
Data availability
Datasets generated and analyzed during this study are included as Dataset S1. Data for plots are deposited on GitHub at https://github.com/barstowlab/article-037-ultramafic-bioleaching and are archived on Zenodo90.
References
Keller, D. P. et al. The effects of carbon dioxide removal on the carbon cycle. Curr. Clim. Change Rep. 4, 250–265. https://doi.org/10.1007/s40641-018-0104-3 (2018).
Lackner, K. S. & Azarabadi, H. Buying down the cost of direct air capture. Ind. Eng. Chem. Res. 60, 8196–8208. https://doi.org/10.1021/acs.iecr.0c04839 (2021).
Andrew, R. M. & Peters G. P. (2024).
Measuring & analyzing greenhouse gases: Behind the scenes, (2022). https://gml.noaa.gov/outreach/behind_the_scenes/gases.html
IPCC et al. Global warming of 1.5°C. An IPCC Special Report on the impacts of global warming of 1.5°C above pre-industrial levels and related global greenhouse gas emission pathways, in the context of strengthening the global response to the threat of climate change, sustainable development, and efforts to eradicate poverty et al. (2018).
Pan, S. Y. et al. An innovative approach to integrated carbon mineralization and waste utilization: A review. Aerosol Air Qual. Res. 15, 1072–1091. https://doi.org/10.4209/aaqr.2014.10.0240 (2015).
Park, A. H. A. & Fan, L. S. CO2 mineral sequestration: physically activated dissolution of serpentine and pH swing process. Chem. Eng. Sci. 59, 5241–5247. https://doi.org/10.1016/j.ces.2004.09.008 (2004).
Ciais, P. et al. In Climate Change 2013: the Physical Science Basis. Contribution of Working Group I To the Fifth Assessment Report of the Intergovernmental Panel on Climate Change Ch6 (Cambridge University Press, 2013).
Gaillardet, J., Dupré, B., Louvat, P. & Allègre, C. J. Global silicate weathering and CO2 consumption rates deduced from the chemistry of large rivers. Chem. Geol. 159, 3–30. https://doi.org/10.1016/S0009-2541(99)00031-5 (1999).
Hartmann, J. & Bicarbonate-fluxes CO2-consumption by chemical weathering on the Japanese Archipelago — Application of a multi-lithological model framework. Chem. Geol. 265, 237–271. https://doi.org/10.1016/j.chemgeo.2009.03.024 (2009).
Plante, L. et al. Cross-species comparison of ultramafic rock bio-accelerated weathering performance. bioRxiv, 2024.2011.2019.624384 (2024). https://doi.org/10.1101/2024.11.19.624384
Kelemen, P. & Matter, J. In situ carbonation of peridotite for CO2 storage. Proc. Natl. Acad. Sci. U.S.A. 105, 17295–17300. https://doi.org/10.1073/pnas.0805794105 (2008).
Nicolas, A., Boudier, F., Ildefonse, B. & Ball, E. Accretion of Oman and united Arab Emirates ophiolite – Discussion of a new structural map. Mar. Geophys. Res. 21, 147–180. https://doi.org/10.1023/A:1026769727917 (2000).
Kelemen, P., Benson, S. M., Pilorgé, H., Psarras, P. & Wilcox, J. An overview of the status and challenges of CO2 storage in minerals and geological formations. Front. Clim. 1 https://doi.org/10.3389/fclim.2019.00009 (2019).
Power, I. M. et al. Carbon mineralization: from natural analogues to engineered systems. Rev. Mineral. Geochem. 77, 305–360. https://doi.org/10.2138/rmg.2013.77.9 (2013).
Renforth, P. The negative emission potential of alkaline materials. Nat. Commun. 10, 1401. https://doi.org/10.1038/s41467-019-09475-5 (2019).
Zondervan, J. R. et al. Rock organic carbon oxidation CO2 release offsets silicate weathering sink. Nature 623, 329–333. https://doi.org/10.1038/s41586-023-06581-9 (2023).
Archer, D. et al. Atmospheric lifetime of fossil fuel carbon dioxide. Annu. Rev. Earth Planet. Sci. 37, 117–134. https://doi.org/10.1146/annurev.earth.031208.100206 (2009).
Colbourn, G., Ridgwell, A. & Lenton, T. M. The time scale of the silicate weathering negative feedback on atmospheric CO2. Glob. Biogeochem. Cycles. 29, 583–596. https://doi.org/10.1002/2014gb005054 (2015).
Röhl, U., Westerhold, T., Bralower, T. J. & Zachos, J. C. On the duration of the Paleocene-Eocene thermal maximum (PETM). Geochem. Geophys. Geosyst. 8 https://doi.org/10.1029/2007GC001784 (2007).
IEA. The Role of Critical Minerals in Clean Energy Transitions. (2021).
Dilshara, P. et al. The role of nickel (Ni) as a critical metal in clean energy transition: applications, global distribution and occurrences, production-demand and phytomining. J. Asian Earth Sci. 259 https://doi.org/10.1016/j.jseaes.2023.105912 (2024).
Hamilton, J. L. et al. Accelerating mineral carbonation in ultramafic mine tailings via direct CO2 reaction and heap leaching with potential for base metal enrichment and recovery. Econ. Geol. 115, 303–323. https://doi.org/10.5382/econgeo.4710 (2020).
Li, J., Hitch, M., Power, I. & Pan, Y. Integrated mineral carbonation of ultramafic mine Deposits—A review. Minerals 8 https://doi.org/10.3390/min8040147 (2018).
Wilson, S. & Hamilton, J. L. Fizzy ore processing sequesters CO(2) while supplying critical metals. Proc. Natl. Acad. Sci. U S A. 119, e2212424119. https://doi.org/10.1073/pnas.2212424119 (2022).
Sanna, A., Uibu, M., Caramanna, G., Kuusik, R. & Maroto-Valer, M. M. A review of mineral carbonation technologies to sequester CO2. Chem. Soc. Rev. 43, 8049–8080. https://doi.org/10.1039/c4cs00035h (2014).
Stokreef, S., Sadri, F., Stokreef, A. & Ghahreman, A. Mineral carbonation of ultramafic tailings: A review of reaction mechanisms and kinetics, industry case studies, and modelling. Clean. Eng. Technol. 8 https://doi.org/10.1016/j.clet.2022.100491 (2022).
Knapp, W. J. et al. Quantifying CO(2) removal at enhanced weathering sites: a multiproxy approach. Environ. Sci. Technol. 57, 9854–9864. https://doi.org/10.1021/acs.est.3c03757 (2023).
Rashid, M. I. et al. Aqueous carbonation of peridotites for carbon utilisation: a critical review. Environ. Sci. Pollut Res. Int. 29, 75161–75183. https://doi.org/10.1007/s11356-022-23116-3 (2022).
Wogelius, R. A. & Walther, J. V. Olivine dissolution at 25°C: effects of pH, CO2, and organic acids. Geochim. Cosmochim. Acta. 55, 943–954. https://doi.org/10.1016/0016-7037(91)90153-V (1991).
National Academies of Sciences, E., and Medicine & Division on Earth and Life Studies. Ocean Studies Board; Board on Chemical Sciences and Technology; Board on Earth Sciences and Resources; Board on Agriculture and Natural Resources; Board on Energy and Environmental Systems; Board on Atmospheric Sciences and Climate; Committee on Developing a Research Agenda for Carbon Dioxide Removal and Reliable Sequestration. (National Academies Press (US) Copyright 2019 by the National Academy of Sciences. All rights reserved., (2018).
Teir, S., Kuusik, R., Fogelholm, C. J. & Zevenhoven, R. Production of magnesium carbonates from serpentinite for long-term storage of CO2. Int. J. Miner. Process. 85, 1–15. https://doi.org/10.1016/j.minpro.2007.08.007 (2007).
Uddin, S., Rao, S. R., Mirnezami, M. & Finch, J. A. Processing an ultramafic ore using fiber disintegration by acid attack. Int. J. Miner. Process. 102–103, 38–44. https://doi.org/10.1016/j.minpro.2011.09.015 (2012).
McCutcheon, J. & Power, I. M. Microbially mediated carbon dioxide removal for sustainable mining. PLoS Biol. 21, e3002026. https://doi.org/10.1371/journal.pbio.3002026 (2023).
Power, I. M. et al. Microbially mediated mineral carbonation: roles of phototrophy and heterotrophy. Environ. Sci. Technol. 45, 9061–9068. https://doi.org/10.1021/es201648g (2011).
Johnson, D. B. Biomining—biotechnologies for extracting and recovering metals from ores and waste materials. Curr. Opin. Biotechnol. 30, 24–31. https://doi.org/10.1016/j.copbio.2014.04.008 (2014). https://doi.org/https://doi.org/
Banerjee, I., Burrell, B., Reed, C., West, A. C. & Banta, S. Metals and minerals as a biotechnology feedstock: engineering biomining microbiology for bioenergy applications. Curr. Opin. Biotechnol. 45, 144–155. https://doi.org/10.1016/j.copbio.2017.03.009 (2017).
Valdés, J. et al. Acidithiobacillus ferrooxidans metabolism: from genome sequence to industrial applications. BMC Genom. 9, 597. https://doi.org/10.1186/1471-2164-9-597 (2008).
Dance, A. Microbial miners take on rare-earth metals. Nature 623, 876–878. https://doi.org/10.1038/d41586-023-03611-4 (2023).
Good, N. M. et al. Hyperaccumulation of gadolinium by Methylorubrum extorquens AM1 reveals impacts of lanthanides on cellular processes beyond Methylotrophy. Front. Microbiol. 13, 820327. https://doi.org/10.3389/fmicb.2022.820327 (2022).
Mattocks, J. A. et al. Enhanced rare-earth separation with a metal-sensitive Lanmodulin dimer. Nature 618, 87–93. https://doi.org/10.1038/s41586-023-05945-5 (2023).
Reed, D. W., Fujita, Y., Daubaras, D. L., Jiao, Y. & Thompson, V. S. Bioleaching of rare Earth elements from waste phosphors and cracking catalysts. Hydrometallurgy 166, 34–40. https://doi.org/10.1016/j.hydromet.2016.08.006 (2016).
Schmitz, A. et al. High efficiency rare earth element biomining with systems biology guided engineering of gluconobacter oxydans. (2023).
Schmitz, A. M. et al. Generation of a Gluconobacter oxydans knockout collection for improved extraction of rare Earth elements. Nat. Commun. 12, 6693. https://doi.org/10.1038/s41467-021-27047-4 (2021).
Harper, G. D. J. et al. Roadmap for a sustainable circular economy in lithium-ion and future battery technologies. J. Physics: Energy. 5 https://doi.org/10.1088/2515-7655/acaa57 (2023).
Sedlakova-Kadukova, J. et al. Comparison of three different bioleaching systems for Li recovery from lepidolite. Sci. Rep. 10, 14594. https://doi.org/10.1038/s41598-020-71596-5 (2020).
da Silva, G. A. R. et al. The industrial versatility of Gluconobacter oxydans: current applications and future perspectives. World J. Microbiol. Biotechnol. 38, 134. https://doi.org/10.1007/s11274-022-03310-8 (2022).
Ma, Y., Li, B., Zhang, X., Wang, C. & Chen, W. Production of gluconic acid and its derivatives by microbial fermentation: process improvement based on integrated routes. Front. Bioeng. Biotechnol. 10, 864787. https://doi.org/10.3389/fbioe.2022.864787 (2022).
Antonick, P. J. et al. Bio- and mineral acid leaching of rare Earth elements from synthetic phosphogypsum. J. Chem. Thermodyn. 132, 491–496. https://doi.org/10.1016/j.jct.2018.12.034 (2019).
Marecos, S. et al. Direct Genome-Scale screening of Gluconobacter oxydans B58 for rare Earth element bioleaching. (2024). https://doi.org/10.1101/2024.06.10.598312
Fausak, L. et al. Effect of wetting and drying processes on ultramafic and mafic tailing minerals amended with topsoil. Environ. Chem. 21 https://doi.org/10.1071/en23037 (2024).
Sánchez-Murillo, R. et al. Geochemical evidence for active tropical serpentinization in the Santa Elena ophiolite, Costa Rica: an analog of a humid early Earth? Geochem. Geophys. Geosyst. 15, 1783–1800. https://doi.org/10.1002/2013GC005213 (2014).
Vicca, S. et al. Is the climate change mitigation effect of enhanced silicate weathering governed by biological processes? Glob. Change Biol. 28, 711–726. https://doi.org/10.1111/gcb.15993 (2022).
Fuenzalida-Meriz, G. et al. Microbially-enhanced silicate weathering in agricultural soils to remove carbon dioxide. (2024).
Berghe, M. et al. Microbial catalysis for CO 2 sequestration: A Geobiological approach. Cold Spring Harb. Perspect. Biol. 16 (a041673). https://doi.org/10.1101/cshperspect.a041673 (2023).
Corbett, T. D. W. et al. Organic carbon source controlled microbial olivine dissolution in small-scale flow-through bioreactors, for CO2 removal. Npj Mater. Degrad. 8 https://doi.org/10.1038/s41529-024-00454-w (2024).
Klug, J. D. et al. Direct carbonation of olivine mediated by synthetic biology. Forthcoming (2024).
Kelemen, P. B. et al. Rates and mechanisms of mineral carbonation in peridotite: natural processes and recipes for enhanced, in situ CO2 capture and storage. Annu. Rev. Earth Planet. Sci. 39, 545–576. https://doi.org/10.1146/annurev-earth-092010-152509 (2011).
Strefler, J., Amann, T., Bauer, N., Kriegler, E. & Hartmann, J. Potential and costs of carbon dioxide removal by enhanced weathering of rocks. Environ. Res. Lett. 13 https://doi.org/10.1088/1748-9326/aaa9c4 (2018).
Mena, M. G. Dissolution kinetics of chromite in hydrochloric acid solutions. Phillippine Eng. J. 11, 39–52 (1990).
Thompson, V. S. et al. Techno-economic and life cycle analysis for bioleaching Rare-Earth elements from waste materials. ACS Sustain. Chem. Eng. 6, 1602–1609. https://doi.org/10.1021/acssuschemeng.7b02771 (2017).
Marecos, S. et al. Practical and thermodynamic constraints on Electromicrobially-Accelerated CO2 mineralization. (2022). https://doi.org/10.1101/2022.01.09.475503
Biddy, M. J. & Jones, S. B. Catalytic upgrading of sugars to hydrocarbons technology pathway United States (2013).
Tomas-Pejo, E., Morales-Palomo, S. & Gonzalez-Fernandez, C. Microbial lipids from organic wastes: outlook and challenges. Bioresour Technol. 323, 124612. https://doi.org/10.1016/j.biortech.2020.124612 (2021).
Khan, M. F. S., Akbar, M., Xu, Z. & Wang, H. A review on the role of pretreatment technologies in the hydrolysis of lignocellulosic biomass of corn Stover. Biomass Bioenerg. 155 https://doi.org/10.1016/j.biombioe.2021.106276 (2021).
Lips, D. Fuelling the future of sustainable sugar fermentation across generations. Eng. Biol. 6, 3–16. https://doi.org/10.1049/enb2.12017 (2022).
Dai, L. et al. Low pH stress enhances gluconic acid accumulation with enzymatic hydrolysate as feedstock using Gluconobacter oxydans. Fermentation 9 https://doi.org/10.3390/fermentation9030278 (2023).
Dai, L. et al. Multi-strategy in production of high titer gluconic acid by the fermentation of concentrated cellulosic hydrolysate with Gluconobacter oxydans. Ind. Crops Prod. 189 https://doi.org/10.1016/j.indcrop.2022.115748 (2022).
Zhou, X., Zhou, X., Huang, L., Cao, R. & Xu, Y. Efficient coproduction of gluconic acid and xylonic acid from lignocellulosic hydrolysate by Zn(II)-selective Inhibition on whole-cell catalysis by Gluconobacter oxydans. Bioresour Technol. 243, 855–859. https://doi.org/10.1016/j.biortech.2017.07.023 (2017).
Mills, T. Y., Sandoval, N. R. & Gill, R. T. Cellulosic hydrolysate toxicity and tolerance mechanisms in Escherichia coli. Biotechnol. Biofuels. 2, 26. https://doi.org/10.1186/1754-6834-2-26 (2009).
Zhang, M. et al. Genetic analysis of D-xylose metabolism pathways in Gluconobacter oxydans 621H. J. Ind. Microbiol. Biotechnol. 40, 379–388. https://doi.org/10.1007/s10295-013-1231-4 (2013).
Energy, U. D. & o Carbon Negative Shot, < (2024). https://www.energy.gov/fecm/carbon-negative-shot
Organization, I. S. About Sugar, < (2024). https://www.isosugar.org/sugarsector/sugar
Halford, N. G., Curtis, T. Y., Muttucumaru, N., Postles, J. & Mottram, D. S. Sugars in crop plants. Ann. Appl. Biol. 158, 1–25. https://doi.org/10.1111/j.1744-7348.2010.00443.x (2011).
Cheng, M. H., Wang, Z., Dien, B. S., Slininger, P. J. & Singh, V. Economic Analysis of Cellulosic Ethanol Production from Sugarcane Bagasse Using a Sequential Deacetylation, Hot Water and Disk-Refining Pretreatment. Processes (2019).
Slade, R., Bauen, A. & Gross, R. Global bioenergy resources. Nat. Clim. Change. 4, 99–105. https://doi.org/10.1038/nclimate2097 (2014).
Benjamin, M. M. Water chemistry. Second Edition edn, xxiii, 905 pages: illustrations ; 25 cmWaveland Press, Inc., (2015).
Ji, Y., Madhav, D. & Vandeginste, V. Kinetics of enhanced magnesium carbonate formation for CO2 storage via mineralization at 200°C. Int. J. Greenhouse Gas Control. 121 https://doi.org/10.1016/j.ijggc.2022.103777 (2022).
Ferrini, V., De Vito, C. & Mignardi, S. Synthesis of nesquehonite by reaction of gaseous CO2 with Mg chloride solution: its potential role in the sequestration of carbon dioxide. J. Hazard. Mater. 168, 832–837. https://doi.org/10.1016/j.jhazmat.2009.02.103 (2009).
Huang, H. P., Shi, Y., Li, W. & Chang, S. G. Dual alkali approaches for the capture and separation of CO 2. Energy Fuels - ENERG. FUEL. 15, 263–268. https://doi.org/10.1021/ef0002400 (2001).
Gadikota, G. Carbon mineralization pathways for carbon capture, storage and utilization. Commun. Chem. 4, 23. https://doi.org/10.1038/s42004-021-00461-x (2021).
Ncongwane, M. S., Broadhurst, J. L. & Petersen, J. Assessment of the potential carbon footprint of engineered processes for the mineral carbonation of PGM tailings. Int. J. Greenhouse Gas Control. 77, 70–81. https://doi.org/10.1016/j.ijggc.2018.07.019 (2018).
Addassi, M., Hoteit, H. & Oelkers, E. H. The impact of secondary silicate mineral precipitation kinetics on CO2 mineral storage. Int. J. Greenhouse Gas Control. 131 https://doi.org/10.1016/j.ijggc.2023.104020 (2024).
Kierczak, J., Pietranik, A. & Pedziwiatr, A. Ultramafic geoecosystems as a natural source of Ni, Cr, and Co to the environment: A review. Sci. Total Environ. 755, 142620. https://doi.org/10.1016/j.scitotenv.2020.142620 (2021).
ten Berge, H. F. et al. Olivine weathering in soil, and its effects on growth and nutrient uptake in ryegrass (Lolium Perenne L.): a pot experiment. PLoS One. 7, e42098. https://doi.org/10.1371/journal.pone.0042098 (2012).
Kelemen, P. B. et al. Engineered carbon mineralization in ultramafic rocks for CO2 removal from air: review and new insights. Chem. Geol. 550 https://doi.org/10.1016/j.chemgeo.2020.119628 (2020).
Buss, W., Hasemer, H., Sokol, N. W., Rohling, E. J. & Borevitz, J. Applying minerals to soil to draw down atmospheric carbon dioxide through synergistic organic and inorganic pathways. Commun. Earth Environ. 5, 602. https://doi.org/10.1038/s43247-024-01771-3 (2024).
Kulp, J. L. & Brobst, D. A. Notes on the Dunite and the geochemistry of vermiculite at the day book Dunite deposit, Yancey County, North Carolina. Econ. Geol. 49, 211–220. https://doi.org/10.2113/gsecongeo.49.2.211 (1954).
Al-Hakkani, M. F. Guideline of inductively coupled plasma mass spectrometry ICP–MS: fundamentals, practices, determination of the limits, quality control, and method validation parameters. SN Appl. Sci. 1, 791. https://doi.org/10.1007/s42452-019-0825-5 (2019).
Lee, J. J. & Barstow, B. Article 37 Release 1.01 for bioRxiv Submission, (2024). https://zenodo.org/account/settings/github/repository/barstowlab/article-037-ultramafic-bioleaching
Acknowledgements
This work was supported by Cornell University startup funds, a SPROUT award from the Cornell University College of Engineering, a Career Award at the Scientific Interface from the Burroughs-Welcome Fund to B.B., a gift from Mary Fernando Conrad and Tony Conrad to B.B., a gift from Nancy and Bob Selander to B.B. and E.G., and the U.S. Army Advanced Civil Schooling Program. J.J.L. was supported by a Cornell University graduate fellowship. S. Marecos was supported by a Link Foundation Graduate Fellowship. Daniel Schell at the National Renewable Energy Laboratory provided a sample of cellulosic hydrolysate. Lyndsey Fisher in the Department of Earth and Atmospheric Sciences at Cornell University processed ultramafic samples.
Author information
Authors and Affiliations
Contributions
Conceptualization, J.J.L., E.G., G.G., and B.B.; Methodology, J.J.L., S. Medin, S. Marecos., L.P, J.K., E.G.,M.C.R., and B.B.; Investigation, J.J.L, L.P., and B.P.; Writing—Original draft, J.J.L.; Writing—Review and editing, J.J.L. and B.B.; Funding acquisition, B.B., G.G., and E.G.; Resources, M.C.R., E.G., and B.B.; Supervision, B.B. and E.G; Data curation, J.J.L. and B.B.; Visualization, J.J.L.; Formal analysis, J.J.L.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, J.J., Plante, L., Pian, B. et al. Bio-accelerated weathering of ultramafic minerals with Gluconobacter oxydans. Sci Rep 15, 15134 (2025). https://doi.org/10.1038/s41598-025-99655-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-99655-9
Keywords
This article is cited by
-
Cross-species comparison of ultramafic rock bio-accelerated weathering performance
Scientific Reports (2025)