Abstract
About 80% of neonates with hypoxic-ischaemic encephalopathy (HIE) have cardiovascular dysfunction and are vulnerable to late-onset injury due to disrupted cerebral blood flow and metabolism. We previously reported the neuroprotective effect of hydrogen (H2) gas. This study investigated the effects of H2 gas inhalation on cardiac function in piglets after hypoxia-ischaemia (HI). Seventeen piglets ≤ 24 h old were subjected to HI insult for approximately 40 min and, after 10 min of resuscitation, were divided into an HI group (n = 10) and an HI-H2 gas inhalation group (2.2%–2.7% H2 gas for 6 h; n = 7). We examined biventricular single stroke volume (SV) and cardiac output (CO) within 6 h after HI insult using transthoracic echocardiography. The HI group (n = 10) showed a transient increase in SV and CO followed by a decline. In contrast, the HI-H2 group (n = 7) experienced a increase in right ventricular SV and CO, with a mild decrease in left ventricular parameters. Notably, right CO were significantly higher at 5 h after insult in the HI-H2 group (p = 0.023). Hydrogen gas inhalation may help preserve right ventricular performance after asphyxia.
Similar content being viewed by others
Introduction
A hypoxic-ischaemic (HI) insult occurring around the time of birth may result in an encephalopathic state characterised by the need for resuscitation at birth, neurological depression, seizures and electroencephalographic abnormalities1,2. Any interruption of the placental blood or oxygen supply may cause multi-organ dysfunction3. Cardiovascular dysfunction has been increasingly recognised in these infant patient groups as a result of HI damage to the myocardium4,5. For example, neonates with asphyxia have a low cardiac output (CO) with decreased myocardial contractility, systemic hypotension and pulmonary hypertension.
Right heart failure has recently been reported to be one of the factors impacting the neurological prognosis of HI encephalopathy (HIE)6,7. These neonates have a low CO with decreased myocardial contractility, systemic hypotension and pulmonary hypertension8. Hence, the cardiovascular management of infants with HIE has recently had to consider right heart failure, but there are few approaches to cardiovascular management that can be performed from the resuscitation period.
Molecular hydrogen is a very small molecule and is very easily transferred to tissues. The concentration at which the effect appears has been reported to be about 3%, and the effect appears without ignition at that concentration9. Ohsawa et al. were the first to report that molecular hydrogen selectively scavenges free radicals and exerts its neuroprotective and cardiomyocyte-protective effects through its antioxidant properties10. Using a neonatal piglet model, we have also reported that therapeutic hypothermia combined with hydrogen gas therapy improved cerebral hemodynamic and oxygenation changes within 24 h after HI insult, compared with therapeutic hypothermia alone and accelerated the recovery of neuromotor function up to day 5 11. However, the effects of hydrogen gas on organs other than the brain, especially the heart, after HI insult are not clear. In addition, it has been suggested that molecular hydrogen may be useful in the treatment of persistent pulmonary hypertension by lowering pulmonary vascular resistance without suppressing NO production in the vascular endothelium. However, no studies have revealed the direct protective and ameliorative effects of hydrogen gas on cardiac heart function after HI exposure. In this study, we investigated the effects of hydrogen gas for 6 h on biventricular CO after HI insult using a neonatal piglet model.
Results
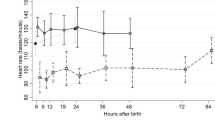
The mean (± SD) duration of LAEEG after insult was not significantly different between the two groups (HI, 21.6 [± 12.5] min; HI-H2, 16.9 [± 6.3] min). HR and MABP recovered to baseline at 1 h after HI insult and were not significantly different between the two groups (Figs. 1A–D and 2). For blood gas data, their values after insult were not significantly different between the two groups. For haemoglobin, the HI-H2 group showed significantly higher values at 3 and 6 h after insult than baseline and versus the HI group (Table 1, p value at 3, 6 h: 0.047, 0.038).
In the HI group, both CO values reached their minimum levels just before resuscitation and then increased afterwards. The left CO gradually decreased from 1 h and became significantly lower than baseline at 3 h after insult (Fig. 1E: p value at 3, 4, 5, 6 h: 0.04, 0.01, 0.003, 0.001). At the same time, the right CO decreased from 0.5 h, reached its minimum at 2 h, and then showed partial recovery towards 6 h ((Fig. 1G: p value at 2, 5 h: 0.004, 0.02). In the HI-H₂ group, the left CO remained relatively stable after resuscitation, showing no significant decrease compared with baseline (Fig. 1F). The right CO tended to increase after resuscitation and exceeded baseline levels from around 2 h after insult, reaching its highest mean value at 5 h (Fig. 1H). Although the left SV and CO did not differ at 6 h after insult (Fig. 3A, C), the right SV, RVOT VTI (Figs. 3B and 4) and right CO were higher in the HI-H2 group than in the HI group, significantly so for CO at 5 h (Fig. 4D, p = 0.023). The mean (± SD) AUC of RV CO was significantly greater in the HI-H₂ group (298.115 ± 82.015 mL·min− 1·h) than in the HI group (224.161 ± 70.544 mL·min− 1·h; p = 0.041), corresponding to a large effect size (d = 1.02).
To evaluate myocardial injury, serum cardiac troponin T was measured at baseline and 3, 6 h after insult. Mean (± ng/mL) troponin T levels at 6 h after insult were significantly lower in the HI–H₂ group than in the HI group (HI: 0.16 ± 0.04; HI–H₂: 0.10 ± 0.02, p = 0.048) (Fig. 5). Because of limited serum volume, troponin T could be measured in 9 of 10 piglets in the HI group and 4 of 7 piglets in the HI–H₂ group.
Discussion
In this study, piglets in the HI group showed a clearly decreased left CO after insult, with the right CO remaining lower than baseline. In contrast, in the HI-H2 group, although the left CO decreased, as in the HI group, these decreases were moderate and the right CO increased gradually, especially at 5 h after insult, with the right CO significantly higher than in the HI group. Hydrogen inhalation significantly increased the overall AUC of right ventricular cardiac output across the 0–6 h observation window (mean difference,74.0 mL/min/h; p = 0.041), corresponding to a large effect size (Cohen’s d = 1.02). This indicates that the significant difference observed at the 5-h time point reflects a sustained hemodynamic improvement throughout the entire post-resuscitation period, rather than an isolated fluctuation.This is first report showing the ability of hydrogen gas to enhance right cardiac function, and There are several possible reasons why this H2 gas inhalation increased right CO.
Both groups exhibited marked metabolic acidosis and hyperlactatemia immediately after the HI insult, followed by a rapid recovery of pH, base excess, and lactate within 1 h after resuscitation (Table 1). These results confirm that comparable hypoxic–ischemic stress was applied and that the resuscitation protocol effectively restored systemic oxygenation and circulation. The absence of intergroup differences in blood gas parameters throughout the experiment also indicates that hydrogen gas inhalation did not impair respiratory or metabolic recovery. A slight but significant increase in hemoglobin concentration was observed in the HI–H₂ group at 3 and 6 h after insult. This variation is likely attributable to transient hemoconcentration or redistribution of intravascular fluid during resuscitation and ventilation rather than a true hematologic change. Given the relatively small sample size and inter-animal variability inherent to this model, minor baseline differences could have contributed to this finding. Since other blood gas and metabolic parameters were comparable between groups, this difference is considered physiologically minimal and unlikely to have affected systemic oxygen delivery or cardiac output.
First, we believe that pulmonary vascular resistance would be the one of the factors affecting right CO. Pulmonary vascular resistance generally increases after HI 12,13, and neonatal asphyxia is often complicated by persistent pulmonary hypertension14. Hypoxia induces disequilibrium in the ratio between endothelin and nitric oxide (NO), in favour of the former, causing vasoconstriction through endothelial dysfunction and decreased NO production. In addition, if the hypoxia is persistent, upregulation of NO synthase is blocked and the NO required for pulmonary vasculature relaxation is lacking4. This explains why treatment with NO inhalation is so efficient. In this study, RVOT VTI was higher in HI-H2 group than HI group. Previous studies have reported that the RVOT VTI can reflect pulmonary vascular resistance (PVR) in the patients with acute heart failure15 and, patients with lower RVOT VTI had greater PVR15,16. This finding may suggest the lower pulmonary arterial pressure in the HI-H2 group. Several mechanisms have been proposed to explain how H₂ gas may reduce pulmonary vascular resistance. Kishimoto et al. have demonstrated that molecular hydrogen decreased monocrotaline-induced pulmonary hypertension in rats by suppressing macrophage accumulation, reducing oxidative stress, and modulating the activators of the transcription-3 and nuclear factor of activated T-cells axis17. Various types of ROS are produced in vivo, including hydroxyl free radicals, peroxynitrite anion, peroxide and superoxide anions, but it is unclear which ROS actually plays a role in the development and progression of pulmonary hypertension. Previous related studies focused mainly on the use of antioxidants, and few studies have focused on which ROS or how many ROS participate17. Oral hydrogen water has a protective effect on pulmonary hypertension due to its antioxidative ability and reduced pulmonary inflammatory response18, although differences are evident in the timing of the pathomechanism of this phenomenon. In our study, pulmonary artery acceleration time (PAAT) was evaluated as a surrogate marker of pulmonary vascular resistance, and no significant difference was observed between the HI and HI-H₂ groups (Suppl. Figure 1). Although the pulmonary artery acceleration time (PAAT) did not differ significantly between groups, this negative result suggests that the increase in right ventricular output cannot be explained solely by pulmonary vasodilation or reduced pulmonary hypertension. Rather, hydrogen gas may have contributed to maintaining right ventricular contractility or microcirculatory function under hypoxic–ischemic conditions. Although CK-MB and other cardiomyocyte-specific markers were not measured, cardiac troponin T was assessed and showed significantly lower levels in the HI–H₂ group. Combined with previous reports demonstrating hydrogen-mediated cardioprotection, these findings support a potential direct myocardial protective effect of hydrogen gas. Hayashida et al. 19 reported that low-concentration hydrogen inhalation significantly reduced infarct size in a rat myocardial ischemia–reperfusion model. Similarly, Sakai et al. showed that hydrogen inhalation protected against myocardial stunning and infarction in a porcine model20, and Chen et al. demonstrated that hydrogen activates the PI3K/Akt signaling pathway to prevent ischemic myocardial injury21. Collectively, these findings support the possibility that hydrogen gas exerts a direct protective effect on cardiomyocytes, which may explain the observed improvement in right ventricular cardiac output at 5 h in our study, even in the absence of changes in pulmonary vascular indices.
Hypothermia has the potential to induce pulmonary vasoconstriction and may lead to or intensifyPPHN22. The diminished heart rate resulting from the attenuation of cardiac function associated with hypothermia may also decrease pulmonary blood flow, thereby potentially precipitating PPHN.
In the present study, an effect of hydrogen gas inhalation on left ventricular cardiac output was not evidenced, as the difference between groups was not statistically significant. Although a slight, non-significant decrease in left ventricular output was observed, this finding is unlikely to have contributed to the increase in right ventricular output. Previous studies have demonstrated that neonatal myocardium possesses inherently greater tolerance to ischemia compared with the adult heart, attributable to metabolic adaptations such as lower energy demand and enhanced anaerobic glycolysis. Interestingly, ischemic preconditioning (IPC), a well-known protective phenomenon in adults, is absent in the immediate neonatal period and only becomes evident later in postnatal development, suggesting that the neonatal myocardium is already in a “preconditioned-like” state23,24. In addition, recent evidence indicates that this innate resistance of neonatal hearts is nitric oxide (NO) dependent: NOS inhibition by L-NAME significantly reduced ischemic tolerance of 1-day-old neonatal rat hearts, while NO donors improved post-ischemic recovery25. These findings strongly support the concept that neonatal cardioprotection is mediated by NO and differs fundamentally from adult mechanisms. Taken together, these developmental and molecular characteristics may partly explain why left ventricular function in our piglet model showed no significant changes, whereas the right ventricle, known to be more vulnerable to ischemia, tended to show a greater improvement with H₂ inhalation, although statistical significance was observed only at 5 h after insult. Therefore, this finding should be interpreted cautiously given the variability and small sample size of this study. Heart rate variability could have contributed to the observed difference in right cardiac output at 5 h. However, in neonatal piglets, heart rate is often unstable due to immature autonomic regulation and may not accurately reflect stress or myocardial contractile state. To minimize this confounding factor, we primarily used stroke volume–derived cardiac output rather than heart rate alone. Although RVOT VTI did not reach statistical significance, the combined trends of RVOT VTI and right CO suggest potential hemodynamic improvement. These considerations represent important limitations of our echocardiographic assessment. Hayashida et al. already reported that H2 gas inhalation improved left ventricular function at 30 min after resuscitation in an ischaemia-reperfusion rat model19 and noted that the IL-6 elevation at 2 h after return of spontaneous circulation was markedly suppressed by H2 gas inhalation. Thus, the anti-inflammatory effect of hydrogen gas inhalation may have been effective from an early period in this study as well and may have contributed to the stabilization or slight improvement of left ventricular cardiac output during the early phase after resuscitation.
Molecular hydrogen has been recognized for its ability to exert an antioxidant effect by eliminating oxygen radicals, which are a form of free radicals. Through this antioxidant effect, molecular hydrogen mitigates damage to myocardial cells and helps to enhance cardiac function10. To further evaluate myocardial involvement, serum cardiac troponin T was analyzed as a biomarker of cardiac injury. The HI–H₂ group showed significantly lower troponin T levels at 6 h after insult compared with the HI group, suggesting that hydrogen gas inhalation attenuated myocardial injury after HI insult. This biochemical evidence supports a potential cardioprotective effect of hydrogen, which may have contributed to the maintenance of cardiac output observed in this study.
Hydrogen gas therapy can help to not only reduce brain injuries, but also improve cardiac function in HIE neonates. In addition, hydrogen inhalation therapy can simultaneously treat PPHN by decreasing pulmonary vascular resistance. Hydrogen inhalation therapy can be easily administered by connecting it to the respiratory circuit. The hydrogen concentration administered is low (under 4%), and the risk of explosion is zero, so the treatment can be initiated safely and quickly.
There are some limitations in this study. Firstly, we were only able to assess the short-term effects of hydrogen gas inhalation because it was performed for only 6 h after insult. On the other hand, in this neonatal piglet model, H₂ gas inhalation showed a trend toward increased right CO on echocardiography from 1 to 2 h after insult, with statistical significance confirmed at 5 h. In clinical practice, early initiation of hydrogen gas inhalation after resuscitation may potentially improve cardiovascular function, as suggested by previous preclinical and pilot clinical studies. Furthermore, right cardiac dysfunction and neurological prognosis in neonates with HIE have recently been the topic of increased research interest7,26. Thus, H2 gas therapy from an early period after birth in the HIE neonate may lead to a stabilisation of cardiovascular function, which, in turn, may contribute to long-term neuroprotective effects. We have already reported that 24-h H2 gas inhalation and therapeutic hypothermia ameliorated the changes in cerebral circulatory oxygen metabolism at 24 h27, reduced the seizure burden28 and exerted a neuroprotective effect on the fifth day after insult in piglets11,27,29 Secondly, there are differences in myelination patterns between piglets and humans after birth30 and, given that myelination is more precocious in piglets, differences may appear in the EEG patterns that emerge when hypoxia occurs.
The third, we were unable to confirm the presence of a shunt. The difference in cardiac output between the left and right ventricles may have been due to the influence of a shunt. Additionally, a bubble study echocardiogram was not performed to evaluate potential intracardiac or extracardiac shunts. This was due to technical limitations in the neonatal piglet model; therefore, subtle shunting effects could not be fully excluded.Regarding other echocardiographic parameters, we acknowledge that TAPSE or tissue Doppler indices would have provided useful information. However, due to the prone positioning of the piglets during ultrasound examination, there were technical limitations in probe access, and TAPSE could not be measured under these experimental conditions.
The fourth, inhaled anesthetics or opioids have cardioprotective effects and affect pulmonary vascular resistance31,32,33. Therefore, they may affect the experimental results. To minimize this effect, the same anesthesia induction and maintenance methods were used in the HI and HI-H₂ groups. Finally, the relatively small sample size, particularly in the HI-H₂ group (n = 7), limits the statistical power of our analyses. Therefore, the observed trends in cardiac output and stroke volume should be interpreted with caution, and future studies with larger sample sizes are needed to confirm these findings.
Methods
Animal procedures
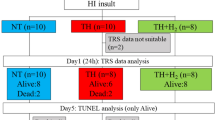
Seventeen newborn piglets (Camborough®; Daiwa Chikusan, Kagawa, Japan) within 24 h of birth and weighing 1.5–2.1 kg were obtained for this study.
Anaesthesia, ventilation and monitoring of physiological variables
Anaesthesia, ventilation, and physiological monitoring were performed as previously described in our established neonatal piglet hypoxic–ischaemic model, with minor modifications11,27,29. The piglets were initially anaesthetised with 1%–2% isoflurane in air using a facemask. Each piglet was then intubated and mechanically ventilated using an infant ventilator. The umbilical vein and artery were cannulated using a neonatal umbilical catheter for drip infusion and blood pressure monitoring/blood sampling, respectively. After cannulation, pancuronium bromide was used at an initial dose of 0.1 mg/kg, followed by infusion at 0.1 mg/kg/h to induce paralysis. Fentanyl citrate was then administered at an initial dose of 10 µg/kg followed by infusion at 5 µg/kg/h for anaesthesia. A maintenance solution of electrolytes plus 2.7% glucose (KN3B; Otsuka Pharmaceutical Co., Tokyo, Japan) was infused continuously at a rate of 4 mL/kg/h via the umbilical vein. Arterial blood samples were taken throughout the experiment at critical time points and when clinically indicated. Each piglet was then placed under a radiant warmer to maintain a mean (standard deviation [± SD]) rectal temperature of 39.0 (± 0.5) °C. The inspired gas was prepared by mixing O2 and N2 gases to obtain the oxygen concentrations required for the experiment. Ventilation was adjusted to maintain the arterial oxygen tension (PaO2) and arterial carbon dioxide tension within their normal ranges. To ensure consistent respiratory management, all piglets were mechanically ventilated in synchronized intermittent mandatory ventilation (SIMV) mode using an infant ventilator. Ventilator settings were standardized as follows: peak inspiratory pressure (PIP) 20 cmH₂O, positive end-expiratory pressure (PEEP) 4 cmH₂O, respiratory rate 12–15 /min, inspiratory time 0.5 s, and FiO₂ 0.21–0.30 during the stabilization and post-insult periods. Arterial CO₂ levels were monitored through serial arterial blood gas analyses, and hypoxia was induced under normocapnic conditions, with PaCO₂ maintained within the physiological range (approximately 40–45 mmHg) to avoid the confounding effects of hypercapnia or hypocapnia on vascular tone and hemodynamic measurements.
Echocardiographic evaluation
Echocardiographic evaluations were performed using Xario100S (Cannon Medical Systems Corporation, Otawara, Japan) with a 12-MHz multifrequency sector probe. The left ventricular and right ventricular outflow velocity patterns were measured from the parasternal short-axis view and long-axis view with the pulsed-wave Doppler sample volume positioned just above the aortic or pulmonary valve. We calculated right ventricle stroke volume (SV) and left ventricle SV, right ventricular outflow tract velocity time integral (RVOT VTI) by tracing the Doppler flow in offline mode. Left and right ventricle cardiac output (CO) was calculated by multiplying SV by heart rate.
Measurement of serum cardiac troponin T
Serum cardiac troponin T concentrations were measured to evaluate myocardial injury 6 h after resuscitation. Blood samples were centrifuged at 3000 rpm for 10 min, and the separated serum was stored at − 80 °C until analysis. Troponin T levels were determined by an electrochemiluminescence immunoassay (ECLIA) using a high-sensitivity Troponin T kit (Roche Diagnostics Japan), and measurements were performed by Nikken Seil Co., Ltd. (Tokyo, Japan) according to the manufacturer’s instructions.
Hypoxic-ischaemic insult protocol
The hypoxic–ischaemic (HI) insult protocol was conducted as previously described in our earlier studies11,27,28,29 (Supplementary Fig. 2A, B) ; therefore, only a brief outline is provided here. Hypoxia was induced by reducing the inspired oxygen concentration of the ventilator to 4% after at least 120 min of stabilisation from the initial anaesthetic induction. To obtain a low-amplitude integrated EEG (LAEEG) pattern (< 5 µV), the inspired oxygen concentration was further reduced if necessary, with adjustments as required to avoid cardiopulmonary arrest. From the beginning of the LAEEG, the insult was continued for 30 min. FiO2 was decreased (1% decrements) or increased (1% increments) during the insult to maintain the LAEEG, heart rate (HR) (> 130 beats/min) and mean arterial blood pressure (MABP) (> 70% of baseline). LAEEG was maintained for 20 min. For the final 10 min of the 30-min insult, if the MABP exceeded 70% of the baseline, hypotension was induced by decreasing the FiO2. Resuscitation was performed when the cerebral blood volume value dropped below 30% and/or the MABP declined below 70% of baseline. Hypoxia was terminated by resuscitation with 100% oxygen. NaHCO3 was used to correct a base deficit (base excess below − 5.0 mEq/L) to maintain a pH of 7.3–7.5. After 10 min of 100% FiO2, the ventilator rate and FiO2 were gradually reduced to maintain an SpO2 of 95%–98%. We measured blood gas, glucose, lactate and haemoglobin levels using a blood gas analyser (ABL90 FLEX PLUS; Radiometer Co., Ltd., Copenhagen, Denmark).
Post-insult treatment
After the HI insult, the 17 piglets were randomised into two groups: HI insult without any therapy (HI; n = 10) and HI insult with H2 gas (HI-H2; 2.1%–2.7% H2 for 6 h, n = 7). For H2 inhalation, two types of cylinders were used, one containing a gas mixture comprising 3.8% H2 and 96.2% N2, and the other containing 100% O2, as in a previous study11,27,29. The H2 concentration depended on the oxygen requirement of each piglet. Therefore, the H2 concentration was usually between 2.1 and 2.7 (FiO2 range, 0.21–0.4) during the therapy. H2 gas was delivered through the ventilator for 6 h. The concentration of H2 gas was measured by a portable gas monitor (TP-70D; Riken Keiki Co., Ltd., Tokyo, Japan). For all piglets, the rectal temperature was monitored continuously to maintain a normal range (38.5–39.5 °C) under the radiant warmer under anaesthesia-ventilation for 6 h after the insult.
Data analysis
GraphPad Prism 9.3.1 (GraphPad Software, La Jolla, CA) was used for all statistical analyses. All values are expressed as the mean ± SD for physiological and blood gas data after the insult in the HI and HI-H2 groups. Physiological data, blood gas data and measurement of HR, MABP, SV, CO, RVOT VTI and serum cardiac troponin T levels were compared between the two groups at each time point using two-way repeated-measures analysis of variance (ANOVA) followed by Tukey’s post hoc analysis. A p value < 0.05 was considered significant. To complement time-point comparisons, we calculated the area under the curve (AUC) for RV CO from 0 to 6 h after insult using the trapezoidal rule. Group differences in AUC were evaluated with Welch’s t test, and effect sizes were expressed as Cohen’s d.
Ethical approval
The study protocol was approved by the Kagawa University Animal Care and Use Committee (15070-1) and was conducted in accordance with the Animal Research: Reporting In Vivo Experiments (ARRIVE) guidelines and all other applicable guidelines and regulations.
Conclusion
In this study, we examined the effect of hydrogen gas inhalation on right and left ventricular CO and SV within 6 h after HI insult using a translational piglet model of HIE. Hydrogen gas inhalation significantly increased right CO at 5 h after insult. The RVOT VTI increased by hydrogen gas, suggesting that hydrogen gas may reduce pulmonary vascular resistance and ameliorate right ventricular dysfunction.
Time course of changes in the physiological parameters MBP, HR and Lt. and Rt.CO before and within 6 h after HI insult in HI and HI-H2 groups. The shaded area indicates HI insult and dotted lines indicate baselines. Closed circle (●), HI group (n = 10); open circle (〇), HI-H2 group (n = 7). #p < 0.05, ##p < 0.01, ####p < 0.0005 vs. baseline. Mean blood pressure (MBP) in HI (A) and HI-H2 (B) groups. Heart rate (HR) in HI (C) and HI-H2 (D) groups. Lt. and Rt. cardiac output (CO) in HI (E, G) and HI-H2 (F, H) groups. Value indicated mean (SD).
Comparison of MBP (A) and HR (B) in HI and HI-H2 groups before and within 6 h after insult. The shaded area indicates HI insult and dotted lines indicate baselines. Closed circle (●), HI group (n = 10); open circle (〇), HI-H2 group (n = 7). Value indicated mean (SD).
Comparison of ventricular SV (A, B) and CO (C, D) in HI and HI-H2 groups before and within 6 h after insult. The shaded area indicates HI insult and dotted lines indicate baselines. Closed circle (●), HI group (n = 10); open circle (〇), HI-H2 group (n = 7). *p < 0.01 vs. HI group. Value indicated mean (SD).
Comparison of RVOT VTI in HI and HI-H2 groups before and within 6 h after insult. The shaded area indicates HI insult and dotted lines indicate baselines. Closed circle (●), HI group (n = 10); open circle (〇), HI-H2 group (n = 7). Value indicated mean (SD).
Comparison of serum cardiac troponin T levels in HI and HI-H2 groups before, at 3, 6 h after insultSerum cardiac troponin T concentrations were measured to assess myocardial injury. Due to sample availability, troponin T could be measured in 9 of 10 piglets in the HI group and 4 of 7 piglets in the HI–H₂ group.Troponin T levels at 6 h were significantly lower in the HI–H₂ group than in the HI group (HI: 0.16 ± 0.04 ng/mL; HI–H₂: 0.10 ± 0.02 ng/mL, p = 0.048). Closed circle (●), HI group (n = 9); open circle (〇), HI-H2 group (n = 4). Data are presented as mean ± SD. p < 0.05 vs. HI group.
Data availability
All data relevant to the study are included in the article or have been uploaded as supplemental information.
Change history
06 March 2026
A Correction to this paper has been published: https://doi.org/10.1038/s41598-026-42659-w
References
Gluckman, P. D. et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial. Lancet (London England). 365, 663–670 (2005).
Azzopardi, D. et al. Implementation and conduct of therapeutic hypothermia for perinatal asphyxial encephalopathy in the UK–analysis of National data. PloS One. 7, e38504 (2012).
LaRosa, D. A., Ellery, S. J., Walker, D. W. & Dickinson, H. Understanding the full spectrum of organ injury following intrapartum asphyxia. Frontiers Pediatrics 5 (2017).
Popescu, M. R. et al., Getting an early start in understanding perinatal Asphyxia impact on the cardiovascular system. Front. Pediatrics 8 (2020).
Evans, N. Which inotrope for which baby? Archives of disease in childhood. Fetal and neonatal edition 91, F213-220 (2006).
Giesinger, R. E. & McNamara, P. J. Hemodynamic instability in the critically ill neonate: an approach to cardiovascular support based on disease pathophysiology. Semin Perinatol. 40, 174–188 (2016).
Giesinger, R. E. et al. Neurodevelopmental outcome following hypoxic ischaemic encephalopathy and therapeutic hypothermia is related to right ventricular performance at 24-hour postnatal age. Archives Disease Child. - Fetal Neonatal Ed. 107, 70–75 (2022).
Joynt, C. & Cheung, P. Y. Cardiovascular supportive therapies for neonates with Asphyxia - A literature review of Pre-clinical and clinical studies. Front. Pead. 6, 363 (2018).
Aokage, T. et al., The effects of inhaling hydrogen gas on macrophage polarization, fibrosis, and lung function in mice with bleomycin-induced lung injury. BMC Pulmonary Medicine 21 (2021).
Ohsawa, I. et al. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat. Med. 13, 688–694 (2007).
Htun, Y. et al. Hydrogen ventilation combined with mild hypothermia improves short-term neurological outcomes in a 5-day neonatal hypoxia-ischaemia piglet model. Sci. Rep. 9, 4088 (2019).
Lapointe, A. & Barrington, K. J. Pulmonary hypertension and the asphyxiated newborn. J. Pediatr. 158, e19–24 (2011).
Wojciak-Stothard, B. & Haworth, S. G. Perinatal changes in pulmonary vascular endothelial function. Pharmacol. Ther. 109, 78–91 (2006).
Liu, X., Tooley, J., LØberg, E. M., Suleiman, M. S. & Thoresen, M. Immediate hypothermia reduces cardiac troponin i after hypoxic-ischemic encephalopathy in newborn pigs. Pediatr. Res. 70, 352–356 (2011).
Koestenberger, M., Schweintzger, S., Pocivalnik, M., Kurath-Koller, S. & Grangl, G. Relevance of right ventricular outflow tract velocity time integral (RVOT VTI) and tricuspid regurgitation velocity/RVOT VTI ratio determination in children following heart transplantation. Pediatr. Cardiol. 39, 642–643 (2018).
Arkles, J. S. et al. Shape of the right ventricular doppler envelope predicts hemodynamics and right heart function in pulmonary hypertension. Am. J. Respir Crit. Care Med. 183, 268–276 (2011).
Kishimoto, Y. et al. Hydrogen ameliorates pulmonary hypertension in rats by anti-inflammatory and antioxidant effects. J. Thorac. Cardiovasc. Surg. 150, 645–654e643 (2015).
He, B. et al. Protection of oral hydrogen water as an antioxidant on pulmonary hypertension. Mol. Biol. Rep. 40, 5513–5521 (2013).
Hayashida, K. et al. H(2) gas improves functional outcome after cardiac arrest to an extent comparable to therapeutic hypothermia in a rat model. J. Am. Heart Association. 1, e003459 (2012).
Sakai, K. et al. Inhalation of hydrogen gas protects against myocardial stunning and infarction in swine. Scandinavian Cardiovasc. J. 46, 183–188 (2012).
Chen, O. et al. High-concentration hydrogen protects mouse heart against ischemia/reperfusion injury through activation of thePI3K/Akt1 pathway. Sci. Rep. 7, 14871 (2017).
Lakshminrusimha, S., Konduri, G. G. & Steinhorn, R. H. Considerations in the management of hypoxemic respiratory failure and persistent pulmonary hypertension in term and late preterm neonates. J. Perinatol. 36, 12–S19 (2016).
Ostadalova, I., Ostadal, B., Kolár, F., Parratt, J. R. & Wilson, S. Tolerance to ischaemia and ischaemic preconditioning in neonatal rat heart. J. Mol. Cell. Cardiol. 30, 857–865 (1998).
Ostádalová, I., Ostádal, B., Jarkovská, D. & Kolár, F. Ischemic preconditioning in chronically hypoxic neonatal rat heart. Pediatr. Res. 52, 561–567 (2002).
Doul, J., Minaříková, M., Charvátová, Z. & Maxová, H. Nitric oxide is involved in the cardioprotection of neonatal rat hearts, but not in neonatal ischemic postconditioning. Physiological Rep. 12, e16147 (2024).
Giesinger, R. E. et al. Impaired right ventricular performance is associated with adverse outcome after hypoxic ischemic encephalopathy. Am. J. Respir. Crit Care Med. 200, 1294–1305 (2019).
Nakamura, S. et al. Impact of hydrogen gas inhalation during therapeutic hypothermia on cerebral hemodynamics and oxygenation in the asphyxiated piglet. Sci. Rep. 13, 1615 (2023).
Tsuchiya, T. et al. Hydrogen gas can ameliorate seizure burden during therapeutic hypothermia in asphyxiated newborn piglets. Pediatr. Res. 95, 1536–1542 (2024).
Htun, Y. et al. Conflicting findings on the effectiveness of hydrogen therapy for ameliorating vascular leakage in a 5-day post hypoxic-ischemic survival piglet model. Sci. Rep. 13, 10486 (2023).
Sweasey, D., Patterson, D. S. P. & Glancy, E. M. Biphasic myelination and the fatty acid composition of cerebrosides and cholesterol esters in the developing central nervous system of the domestic pig. J. Neurochem. 27, 375–380 (1976).
Cohen, M. V. et al. bradykinin, opioids, and phenylephrine, but not adenosine, trigger preconditioning by generating free radicals and opening mitochondrial K(ATP) channels. Circ. Res. 89, 273–278 (2001).
Ma, L. L. et al. Hypertrophied myocardium is refractory to sevoflurane-induced protection with alteration of reperfusion injury salvage kinase/glycogen synthase kinase 3β signals. Shock (Augusta Ga). 40, 217–221 (2013).
Harisseh, R. et al. Cyclophilin D modulates the cardiac mitochondrial target of Isoflurane, Sevoflurane, and desflurane. J. Cardiovasc. Pharmacol. 69, 326–334 (2017).
Acknowledgements
We thank Mr. Ryosuke Kusunoki for support with echography (Canon Medical Systems Corporation, Otawara, Japan) and Mrs. Akiko Takeshima for her assistance with experiments at the Faculty of Medicine, Kagawa University (Takamatsu, Japan), Mr. Hideo Masuda for suppling piglet in his farm (Daiwa Chikusan, Kagawa, Japan).
Funding
This study was financially supported by JSPS KAKENHI grants 22K15923 (Y.N.), 22K07822 (T.K.), 22K15922 (A.M.), 22K18171 (T.M.), 23K07332 (S.N.) and 25K19202 (T.W.). Kagawa University Faculty of Medicine School of Medicine Alumni Association Sanjukai Research Aid grants R5-1 (S.N.).
Author information
Authors and Affiliations
Contributions
K.S., S.N., and T.K. were involved in the initial study design and wrote the main text. S.N., T.M., Y.N., T.K. T.W. and A.M. obtained the necessary financial support for this project and provided study materials. T.T., Y.N. T.W. and Y.H. were primarily responsible for evaluating the aEEG findings. K.S., T.T., T.M, Y.N., E.I., K.I., and A.M. carried out the animal experiments and recorded blood gas and physiological data. T.Y., K.K., Y.K., T.I. and T.H. contributed to data analysis and performed the statistical analysis. All members drafted the article and critically revised it.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The original version of this Article contained an error in Figures 1-2 and 4-5. In addition, author Eri Inoue was incorrectly affiliated. Full information regarding the corrections made can be found in the correction for this Article.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sakamoto, K., Nakamura, S., Tsuchiya, T. et al. Association between hydrogen gas inhalation and cardiac output in an asphyxiated piglet model. Sci Rep 16, 5262 (2026). https://doi.org/10.1038/s41598-026-35115-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-35115-2