Abstract
There are individual differences in the efficacy of glucocorticoid therapy for hepatitis B virus-related acute-on-chronic liver failure (HBV-ACLF). Annexin A3 (ANXA3), as a potential biomarker, may be associated with the clinical outcomes of glucocorticoid therapy in HBV-ACLF patients. We enrolled 108 HBV-ACLF patients receiving glucocorticoid therapy to assess the effects of glucocorticoid therapy on ANXA3 mRNA and methylation levels in peripheral blood mononuclear cells. At the same time, clinical parameters during therapy were collected. In HBV-ACLF patients who survived, ANXA3 expression significantly decreased after glucocorticoid therapy, while methylation levels increased. No significant changes were observed in non-survivors. Logistic regression analysis of 90-day follow-up further indicated that ANXA3 methylation levels could serve as an independent predictor of glucocorticoid treatment response, with its elevation significantly correlating with favorable clinical outcomes. In HBV-ACLF patients, ANXA3 methylation levels were associated with clinical outcomes following glucocorticoid therapy, suggesting its potential as a candidate therapy-associated prognostic biomarker.
Similar content being viewed by others
Introduction
Hepatitis B-related acute-on-chronic liver failure (HBV-ACLF) is one of the major types of liver failure worldwide, characterized by high morbidity and mortality1. Its complex pathogenesis and limited therapeutic means have been a pressing clinical challenge2. Although liver transplantation remains the definitive and most effective therapy, its widespread application is limited by factors such as donor shortage, high costs, and the risk of graft rejection3. Currently, in addition to conventional antiviral therapy and symptomatic supportive therapy, the application of glucocorticoids in the therapy of HBV-ACLF has been shown to improve prognosis4. The early response to glucocorticoid therapy within the initial 4-week therapy period is crucial for subsequent therapeutic decision-making and prognosis management in patients with HBV-ACLF. However, accurately predicting patient responsiveness to glucocorticoid therapy before therapy initiation remains an unmet clinical need.
Annexin A3 (ANXA3) is an important member of the annexin family. In recent years, it has attracted much attention for its key role in a variety of diseases5. Early research focuses on its role in the development of tumors, including liver cancer, breast cancer, prostate cancer, and gastric cancer. It involves tumor pathogenesis, drug resistance, as well as diagnosis and prognostic assessment. Subsequent studies have found that ANXA3 is also significantly differentially expressed in a variety of inflammatory diseases, making it an important target for inflammation-related research. Studies have shown that downregulation of ANXA3 can regulate inflammation-related signaling pathways, which in turn affect disease progression6,7. Notably, ANXA3 is predominantly highly expressed in neutrophils and monocytes. Several studies have confirmed that it is closely related to neutrophils8,9. Neutrophils, as the core immune cells of the early response to inflammation, play a key role in the pathogenesis of HBV-ACLF. Therefore, ANXA3 may play an important role in the immune response and is associated with clinical outcomes to glucocorticoid therapy for HBV-ACLF.
In this study, based on the HBV-ACLF-related dataset in the Gene Expression Omnibus (GEO) database, the potential association between ANXA3 and response to glucocorticoid in HBV-ACLF was further analyzed. To further verify the potential impact of glucocorticoid therapy on the transcription levels of ANXA3 in peripheral blood mononuclear cells (PBMCs), we measured the mRNA and methylation levels of ANXA3 in PBMCs of patients with HBV-ACLF. Additionally, this study also investigated the predictive value of the epigenetic characteristics of ANXA3 for outcomes of glucocorticoid therapy.
Materials and methods
Dataset collection
In this study, we obtained the Gene Set Enrichment 168,048 (GSE168048) dataset from the GEO database (https://www.ncbi.nlm.nih.gov/geo/), which is based on high-throughput sequencing data of PBMCs transcriptome from 16 HBV-ACLF patients10.
Study population
165 patients with HBV-ACLF in the Department of Hepatology, Qilu Hospital of Shandong University from December 2021 to October 2024 were retrospectively included according to the consensus recommendations of the Asian Pacific Association for the Study of the Liver (APASL)2, and finally 156 cases were analyzed in this study. The diagnostic criteria for HBV-ACLF were as follows: (1) Positive hepatitis B surface antigen (HBsAg) for > 6 months. (2) Serum total bilirubin≥5 mg/dL (85µmol/L) with coagulopathy [International normalized ratio (INR) ≥ 1.5 or prothrombin activity (PTA) < 40%]. (3) Development of clinical ascites and/or encephalopathy within 4 weeks. (4) Completed 28-day medical therapy. Participants were excluded for: (1) Other viral hepatitis (HCV, HDV, HEV, HIV). (2) Other types of liver diseases, such as hepatic vascular disorders and liver cancer or other malignancies. (3) Severe cardiac, pulmonary, neurological, or renal dysfunction. (4) Patients with liver transplantation. (5) Pregnancy. (6) Declined informed consent. At the same time, 24 healthy volunteers were matched as controls. Specifically, healthy controls (HCs) were individuals with normal liver function, negative for all viral hepatitis markers, and without a history of malignancy or chronic inflammatory diseases such as ulcerative colitis.
This research was reviewed and approved by the Medical Ethics Committee of Qilu Hospital of Shandong University in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants before the start of the study.
Patient allocation and therapy protocols
All patients with HBV-ACLF were treated with nucleos(t)ide analogue antiviral therapy and had not received glucocorticoid therapy for at least 6 months before the start of the study. According to the consensus recommendations of APASL, all patients also received conventional medical therapy, including nutritional support, hepatoprotective therapy, and complication management.
Patients with HBV-ACLF were divided into the glucocorticoid therapy group (GC group) and the conventional medical group (CM group) based on the use of glucocorticoids. Glucocorticoid (prednisolone) intervention protocol (in addition to conventional medical therapy): initial phase (day1-3): 0.75 mg/kg/day. Dose reduction phase 1 (day4-6): 0.5 mg/kg/day. Dose reduction phase 2 (day7-9): 0.25 mg/kg/day. Depending on the recovery of liver function, the dose of prednisolone is reduced by 5 to 10 mg every 4 to 7 days. Finally, it was maintained at 10 mg daily until 28 days11. Patients were considered for treatment if they had a MELD score < 35 and hepatic encephalopathy grade < 4, after excluding those with absolute contraindications (e.g., systemic infection, active gastrointestinal bleeding, uncontrolled severe psychiatric history, or diabetes)12. The treatment process was ultimately determined by the clinician’s thorough evaluation of individual risks and benefits.
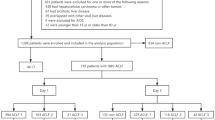
The CM group consisted of patients meeting the same inclusion criteria but who did not receive glucocorticoid therapy. Primary reasons for not receiving glucocorticoid therapy included: (1) The presence of contraindications (primarily for active gastrointestinal ulcer/bleeding, uncontrolled infection, diabetes, and history of psychosis); (2) Severe baseline condition at admission; (3) Refusal by the patient or family. The patient allocation process was shown in Fig. 1.
Flowchart of patient enrollment and treatment allocation. HBV-ACLF hepatitis B virus-related acute-on-chronic liver failure, HCs: healthy controls, GC: glucocorticoid, CM: conventional medical.
Clinical data collection
General information included age and gender. Clinical indicators included alanine transaminase (ALT), aspartate transferase (AST), total bilirubin (TBIL), albumin (ALB), creatinine (Cr), International normalized ratio (INR), prothrombin activity (PTA), white blood cell (WBC), neutrophil ratio (NEUT%), platelet (PLT), and procalcitonin (PCT). Virological indicators included hepatitis B virus surface antigen (HBsAg), hepatitis B e antigen (HBeAg), hepatitis B virus deoxyribonucleic acid (HBV DNA). Complications included hepatic encephalopathy (HE) and ascites. The model for end-stage liver disease (MELD) score was obtained as follows13:
Study endpoints
Patients were followed for 90 days to record survival time, with dynamic liver function changes and clinical outcomes systematically evaluated. In this study, the clinical response of patients receiving glucocorticoid therapy was evaluated as follows. The primary endpoint was 90-day liver-transplant-free survival. Secondary endpoints included post-treatment: (1) Early biochemical response: ≥ 50% reduction from baseline in TBIL or PTA; (2) Clinical-score improvement ≥ 5-point decrease in MELD score from baseline; (3) ACLF reversal reduction of at least one ACLF grade from baseline; and (4) Complication-resolution: resolution of HE and ascites.
PBMCs collection
Peripheral blood (5 mL) was collected from all subjects. PBMCs were separated by density gradient centrifugation with Ficoll-Paque (GE Healthcare, Uppsala, Sweden). All samples were stored at −80 °C for later use.
Real-time quantitative reverse transcription PCR (RT-qPCR)
Total RNA and DNA were extracted from PBMCs using the Trizol (Invitrogen, Carlsbad, CA, United States). The total RNA collected subsequently was reverse transcribed into cDNA with the First-Strand cDNA Synthesis Kit (Fermentas, Vilnius, Lithuania). β-actin was used as an endogenous control, and the mRNA expression levels of ANXA3 and PPAR-γ were quantitatively detected by the 2^-∆∆CT method. The RT-qPCR amplification mixture (10µL) consisted of 5µL SYBR Green (Toyobo, Osaka, Japan), 3µL nuclease-free water, 0.5µL forward and reverse primer, and 1µL of cDNA and performed under the following condition: the initial step was 95℃ for 30 s, followed by 50 cycles of 95℃ for 5 s, 55℃ for 30 s, and a final step of 72℃ for 30s.
Taqman probe-based quantitative methylation-specific polymerase chain reaction (Methylight)
The DNA was modified with EZ DNA Methylation-Goldkit (ZymoResearch, Orange, CA, United States) to obtain the modified DNA template. Two sets of primers and probes targeting bisulfite-converted DNA were designed using Oligo 7 software (OLIGO 1267 Vondelpark ColoradoSprings, CO 80,907, USA), as shown in Table 1. One set targets the ANXA3 gene for methylation detection, and the other targets the ACTB gene, which serves as an internal control to standardize input DNA quantity. Methylight was conducted with the EpiTect MethyLight PCR + ROX Vial Kit (QIAGEN, Hilden, Germany) to detect DNA methylation levels. The methylight amplification mixture (10µL) consisted of 5µL MethyLight Master Mix consisting of HotStarTaq Plus DNA Polymerase, 2µL nuclease-free water, 0.4µL forward and reverse primers, 0.2µL Taqman probe, 2µL modified DNA, and performed under the following conditions: 95℃ for 15 min, followed by 50 cycles of 95℃ for 15 s and 60℃ for 1 min. Results are expressed in percent methylated reference (PMR)14.
Enzyme-linked immunosorbent assay (ELISA)
The ELISA kits (Lengton Bioscience, Shanghai, China) were used to quantify the concentration of the serum Interleukin-6 (IL-6) and Reactive Oxygen Species (ROS). The absorbance of each well was read at 450 nm. The absolute concentration of IL-6 and ROS was determined from a standard curve.
Statistical analysis
Data were analyzed via SPSS 26.0 (SPSS Inc., Chicago, IL, USA), GraphPad Prism 10.1.2 (GraphPad Software Inc., La Jolla, CA, USA), and R 4.4.1 (R Project for Statistical Computing, Vienna, Austria). Quantitative and categorical variables were reported as median (centile 25; centile 75) and number (%), respectively. Chi-square test and Mann-Whitney U test were employed to compare differences. Spearman’s test further assessed the correlation of ANXA3 with peroxisome proliferator-activated receptor γ (PPAR-γ), IL-6, and ROS. Multivariable Cox regression analysis was performed to adjust for known key baseline variables that could potentially influence both treatment allocation and prognosis. Logistic regression analysis was employed to identify independent factors influencing the response to glucocorticoid therapy. Receiver operating characteristic (ROC) was utilized to evaluate the predictive performance of ANXA3 methylation levels for clinical response to glucocorticoid therapy. Kaplan-Meier (KM) survival curves were employed to identify the optimal cut-off value for distinguishing responders from non-responders among HBV-ACLF patients receiving glucocorticoid therapy. All tests used a two-tailed P < 0.05 as the significance threshold.
Results
Transcriptomic analysis
Differential expression analysis was performed using the criterion of |log2 Fold Change|>1.2 and p-value < 0.05. The Gene Ontology enrichment analysis (GO) was conducted. Biological processes showed that ANXA3 was associated with the response to glucocorticoid (Fig. 2A). Moreover, Fig. 2B and Table 2 displayed that ANXA3 was also associated with neutrophil activation, epithelial cell migration, and regulation of DNA-binding transcription factor activity and angiogenesis. Next, the samples were divided into a high-expression group (≥ 50%) and a low-expression group (< 50%) based on the expression level of ANXA3. Figure 2C showed that the high expression group was associated with the PPAR and P53 signaling pathway.
Transcriptomic analysis of PBMCs from patients with HBV-ACLF. GSE168048: Differential expression analysis was performed using the criterion of |log2 Fold Change| > 1.2 and p-value < 0.05. (A,B) Genetic Ontology (GO) enrichment analysis: Circular Diagram and Bubble Chart of biological processes. ANXA3 was associated with response to glucocorticoids, neutrophil activation, epithelial cell migration, and regulation of DNA-binding transcription factor activity, as well as angiogenesis. (C) Based on the expression level of ANXA3, the samples were divided into a high-expression group (≥ 50%) and a low-expression group (< 50%). Gene Set Enrichment Analysis (GSEA): The high expression group was associated with the PPAR and P53 signaling pathway. GSE168048: Differential expression analysis was performed using the criterion of |log2 Fold Change| > 1.2 and p-value < 0.05.
General characteristics
A total of 165 patients with HBV-ACLF were recruited in this study. According to the predefined inclusion/exclusion criteria, a total of 156 patients with HBV-ACLF were included in the 90-day follow-up, of which 108 patients with HBV-ACLF received glucocorticoid therapy, and 48 received conventional medical treatment. Table 3 presented the baseline clinical characteristics of HBV-ACLF patients in the GC and CM groups compared with HCs. No significant differences were observed between the GC and CM groups. Multivariate Cox regression analysis revealed that after adjusting for key baseline variables, glucocorticoid therapy remained an independent protective factor for improved 90-day survival (HR = 0.333, 95% CI: 0.117–0.945, P = 0.039) (Supplementary Table 1).
Expression profiles of ANXA3 and PPAR-γ and their interrelationship in patients with HBV-ACLF
Previous research has demonstrated the critical role of PPAR-γ in the pathogenesis of HBV-ACLF15. To explore the potential association between ANXA3 and the PPAR signaling pathway during the progression of HBV-ACLF, we examined the expression levels of ANXA3, PPAR-γ, IL-6, and ROS in HBV-ACLF patients. As shown in Fig. 3, HBV-ACLF patients exhibited significantly higher ANXA3 mRNA expression but lower methylation levels compared to HCs patients (all p < 0.0001), along with significantly reduced PPAR-γ mRNA expression (p = 0.0014). Spearman correlation analysis further revealed that ANXA3 mRNA was negatively correlated with PPAR-γ mRNA (Spearman’s r=−0.4193, p < 0.0001) but positively correlated with serum IL-6 (Spearman’s r = 0.2815, p = 0.0004) and ROS levels (Spearman’s r = 0.2293, p = 0.0050). Conversely, the PMR of ANXA3 showed a positive association with PPAR-γ mRNA (Spearman’s r = 0.3462, p < 0.0001) and an inverse relationship with both serum IL-6 (Spearman’s r=−0.3452, p < 0.0001) and ROS levels (Spearman’s r=−0.2071, p = 0.0095).
The expression profiles of ANXA3 and PPAR-γ and their interrelationship in patients with HBV-ACLF. (A) mRNA levels of ANXA3 in PBMCs from HCs and HBV-ACLF groups. (B) Methylation levels of ANXA3 in PBMCs from HCs and HBV-ACLF groups. (C) mRNA levels of PPAR-γ in PBMCs from HCs and HBV-ACLF groups. (D) Correlation between the mRNA levels of ANXA3 and PPAR-γ, IL-6, and ROS. (E) Correlation between the PMR value of ANXA3 and PPAR-γ, IL-6, and ROS. *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
Effect of glucocorticoid therapy on ANXA3 expression in patients with HBV-ACLF
To characterize the effect of glucocorticoid therapy on ANXA3 expression, we completed a dynamic follow-up of 54 HBV-ACLF patients with received glucocorticoid therapy. As shown in Fig. 4A-C,E-G, ANXA3 mRNA levels in the non-survival group were significantly higher than in the survival group [1.12 (0.90–1.65) vs. 0.65 (0.28–0.85), p < 0.0001]. On the 7th and 28th days of therapy, the expression levels of ANXA3 decreased in the survival group compared with those on the 0th day [day7: 0.52 (0.14–0.63), p = 0.0317; day28: 0.33 (0.15–0.41), p = 0.0001]. The methylation levels of ANXA3 in the non-survival group were significantly lower than in the survival group [8.02% (5.14%−9.41%) vs. 14.26% (10.59%−16.96%), p < 0.0001]. At day 28 of therapy, the survival group expressed higher methylated levels of ANXA3 [18.30% (14.06%−22.38%), p = 0.0025]. In contrast, there was no significant change in the non-survival group. We also had a dynamic follow-up of 48 HBV-ACLF patients who received conventional medical treatment. We found that the surviving patients also exhibited decreased ANXA3 expression levels by day 28 of therapy (p > 0.05). However, no significant changes in methylation levels were observed between survivors and non-survivors at day 28 post-therapy (p > 0.05) (Fig. 4G,H, Supplementary Table 2).
Dynamic changes for mRNA and methylation levels of ANXA3 in survivor and non-survivor groups of HBV-ACLF patients who received glucocorticoid therapy and conventional medical treatment. (A–C) The mRNA expression levels of ANXA3 in the survivor and non-survivor groups receiving GC therapy at days 0, 7, and 28. (E-G) The methylation levels of ANXA3 in the survivor and non-survivor groups receiving GC therapy at days 0, 7, and 28. (D) The mRNA expression levels of ANXA3 in the survivor and non-survivor groups receiving CM treatment at days 0, 7, and 28. (H) The methylation levels of ANXA3 in the survivor and non-survivor groups receiving CM treatment at days 0, 7, and 28. *p < 0.05. GC: glucocorticoid, CM: conventional medical.
Impact of glucocorticoid therapy on clinical parameters in patients with HBV-ACLF in the survival and non-survival groups
The therapeutic effect of glucocorticoids was determined by influencing the relevant clinical indicators of patients with HBV-ACLF with different outcomes. We dynamically evaluated the therapy’s impact on both liver function and viral loads (Table 4). Compared with HCs, the ALT, TBIL, and INR of HBV-ACLF patients in the survival and non-survival groups were significantly increased, and the ALB and PTA were significantly decreased. After therapy, ALT, TBIL, and INR in the survival group decreased, and ALB and PTA increased. In contrast, the non-survival group showed no significant improvement in liver function indices except for ALT. Furthermore, our results demonstrated that 28-day glucocorticoid therapy had no significant impact on viral replication.
The MELD score was used to evaluate the severity of HBV-ACLF. Spearman’s test showed that MELD score was positively correlated with ANXA3 mRNA expression levels (pre-treatment Spearman’s r = 0.3302, p = 0.0147, post-treatment Spearman’s r = 0.6415, p < 0.0001) but negatively correlated with ANXA3 methylation levels (pre-treatment Spearman’s r=−0.4259, p = 0.0013, post-treatment Spearman’s r=−0.6150, p < 0.0001) (Fig. 5A-D).
Predictive value of ANXA3 methylation for GC therapy outcomes in HBV-ACLF patients. (A,B) Correlation between the mRNA levels of ANXA3 and MELD score (pre-GC treatment and post-GC treatment). (C,D) Correlation between the PMR of ANXA3 and MELD score (pre-treatment and post-treatment). (E) The Kaplan-Meier (KM) survival analysis compares survival outcomes between the GC group (n = 108) and the CM group (n = 48). (F) Receiver operating characteristic (ROC) curves for predicting 90-day mortality in HBV-ACLF patients receiving glucocorticoid therapy. The areas under the curve (AUC) for each predictor are: MELD score (0.79), mRNA (ANXA3) (0.83), PMR (ANXA3) (0.88), and the combined model of MELD score and PMR (ANXA3) (0.89). (G) The KM survival curves for HBV-ACLF patients receiving GC therapy stratified by PMR (ANXA3) cut-off values. GC: glucocorticoid, CM: conventional medical.
Independent predictors of prognosis in HBV-ACLF patients with glucocorticoid therapy identified by multivariable logistic regression analysis
A logistic regression analysis was employed to identify independent factors influencing the prognosis of glucocorticoid therapy (Table 5). Multivariable logistic analysis demonstrated that PMR (ANXA3) (OR = 0.662, 95% CI: 0.536–0.818, p < 0.001), MELD score (OR = 1.123, 95% CI: 1.014–1.243, p = 0.026), and HE (OR = 4.734, 95% CI: 1.263–17.746, p = 0.021) were independent factors influencing glucocorticoid therapy response.
Correlation between glucocorticoid therapy and improved survival rates in patients with HBV-ACLF
To assess the long-term effects of different therapies, we monitored the survival status of all enrolled patients with HBV-ACLF during the 90-day follow-up. Follow-up results showed the survival outcome was significantly more favorable in the GC group compared to the CM group. Specifically, the survival rates were 67.59% (73/108) and 56.25% (27/48) for the GC and CM groups, respectively. KM survival analysis confirmed a statistically significant survival advantage for the GC group (p = 0.0205; Fig. 5E). These findings indicated that improved survival rates in patients with HBV-ACLF may be associated with glucocorticoid therapy.
Higher ANXA3 methylation level as a predictor for a favorable outcomes to glucocorticoid therapy in HBV-ACLF patients
The MELD score typically reflects the severity of liver dysfunction and may influence the efficacy of glucocorticoid therapy. We evaluated the predictive value of ANXA3 expression levels and MELD score for 90-day survival in patients receiving glucocorticoid therapy, reflecting the primary endpoint of glucocorticoid efficacy. The ROC and KM survival curves were performed. The PMR of ANXA3 demonstrated significantly superior predictive performance compared with the MELD score and mRNA (ANXA3). Using the optimal cutoff value of 9.54%, the ANXA3 methylation (PMR) model demonstrated robust diagnostic performance with an Area Under the Curve (AUC) of 0.88. In comparison, the mRNA (ANXA3) model achieved a maximum AUC of 0.83, and the MELD score model yielded an AUC of 0.79. Combining the MELD score with PMR (ANXA3) further improved the AUC to 0.89. at the optimized cutoff value of −0.67 (Fig. 5F; Table 6). The combined model [MELD score + PMR (ANXA3)] was expressed as R = 1.528 + 0.097*MELD score-0.412*PMR (ANXA3). Additionally, we stratified HBV-ACLF patients and plotted KM survival curves based on the PMR (ANXA3) cutoff values of 9.54%. The results indicated that patients with HBV-ACLF in the high methylation group were associated with a more favorable prognosis for glucocorticoid therapy. (p < 0.0001, Fig. 5G).
Subsequently, we further examined the predictive performance of baseline PMR (ANXA3) for improvements in various secondary endpoints following glucocorticoid therapy. As shown in Fig. 6 and Table 6, PMR (ANXA3) was significantly higher in responders than in non-responders across all secondary endpoints. ROC analysis demonstrated favorable predictive accuracy: AUC 0.79/0.81 for early biochemical response of TBIL/PTA (≥ 50% decline), AUC 0.82 for MELD score improvement (≥ 5-point decline), AUC 0.84 for reversal of ACLF grade (reduction of ≥ 1 grade), AUC 0.82/0.83 for resolution of HE and ascites. These results indicated that PMR (ANXA3) correlated with improvement in secondary endpoints after glucocorticoid treatment in patients with HBV-ACLF.
Predictive value of baseline PMR (ANXA3) for improvements in secondary endpoints after glucocorticoid therapy. (A,C,E,G,I,K) Box plots comparing PMR (ANXA3) between responders (R) and non-responders (NR) for different secondary endpoints (early biochemical response of TBIL/PTA, MELD score improvement, ACLF grade reversal, HE and ascites resolution, respectively). ****p < 0.0001. (B,D,F,H,J,L) Receiver operating characteristic (ROC) curves for different endpoints. The areas under the curve (AUC) values are as follows: 0.79/0.81 for early biochemical response of TBIL/PTA, 0.82 for improvement of MELD score (≥ 5-point decline), 0.84 for reversal of ACLF grade (reduction of ≥ 1 grade), 0.82/0.83 for resolution of HE and ascites. TBIL: total bilirubin, PTA: prothrombin activity, HE: hepatic encephalopathy.
Discussion
In this study, we preliminarily investigated the relationship between ANXA3 and glucocorticoid therapy response in HBV-ACLF. The study revealed that glucocorticoid therapy significantly reduced ANXA3 expression levels while increasing its methylation in the survival group, with no such changes observed in the non-survival group. Moreover, glucocorticoid therapy has further confirmed its efficacy in improving liver function in patients with HBV-ACLF. After a 90-day follow-up, we found that the PMR value of ANXA3 may serve as an effective predictive indicator to assess the clinical outcomes of HBV-ACLF patients to glucocorticoid therapy. Additionally, biological analysis and correlation studies suggested that ANXA3 may be associated with the PPAR signaling pathway.
Inflammation and oxidative stress serve as pivotal pathological mechanisms driving the progression of HBV-ACLF, with IL-6 and ROS acting as crucial effector molecules mediating injury. Studies have demonstrated significantly elevated levels of IL-6 and ROS in patients with HBV-ACLF16,17. As an inflammation-associated protein, ANXA3 exhibits characteristic upregulation in various inflammatory diseases. The suppression of ANXA3 expression can effectively mitigate inflammatory responses and promote tissue repair18,19,20. In this study, we observed that the expression of ANXA3 was significantly negatively correlated with PPAR-γ and positively correlated with serum IL-6 and ROS. Interestingly, the methylation levels of ANXA3 showed an opposite trend. This discovery suggested that ANXA3 may influence the activity of the PPAR signaling pathway through epigenetic regulatory mechanisms, thereby modulating inflammatory and oxidative stress responses. The latest study also found that ANXA3 and the PPAR signaling pathway played a crucial role in neutrophil activation during the inflammatory response in patients with cerebral hemorrhage, further supporting the critical role of ANXA3 in regulating inflammation21. However, it should be clarified that this association is based on bioinformatics analysis and observational data, which needs further experimental validation.
Glucocorticoids, known for their remarkable anti-inflammatory and immunomodulatory effects, are recognized as pivotal therapies for ACLF management. Previous studies have demonstrated that glucocorticoids exert synergistic anti-inflammatory effects by inhibiting IL-6 and ROS production while upregulating PPAR-γ expression22,23. Furthermore, ANXA1 has been shown to have immunomodulatory functions. ANXA1 expression and function are regulated by glucocorticoids and mediate immunosuppressive effects24. To clarify the effect of glucocorticoids on ANXA3 expression, we analyzed the expression of ANXA3 in patients with HBV-ACLF before and after glucocorticoid therapy. Interestingly, the expression levels of ANXA3 decreased in the survival group, while there was no significant difference in the expression levels in the non-survival group. This difference was more pronounced compared to the CM group. Although selection bias may be present. Furthermore, changes in ANXA3 expression before and after treatment were closely related to the MELD score. These results indicated that ANXA3 may be associated with disease severity and the prognosis of glucocorticoid treatment.
Previous study confirmed that glucocorticoid therapy significantly improved the clinical prognosis of patients with HBV-ACLF25. Our findings also supported this conclusion, with the GC group showing significantly higher survival rates than the CM group. This study further found that glucocorticoid therapy was correlated with improvements in liver function parameters, while short-term therapy appeared to have no significant impact on viral replication. This finding aligns with the results reported by Lin Jia et al.4. However, there is still a lack of precise quantitative indicators for individual differences in response to glucocorticoid therapy26. Studies have shown that glucocorticoids can mediate epigenetics. DNA methylation can be used as a pharmacodynamic marker of glucocorticoid response27,28. Therefore, in our study, we focused on exploring the potential association between ANXA3 methylation levels and outcomes of glucocorticoid therapy. The aim was to establish a biomarker that could predict the clinical efficacy of glucocorticoids by analyzing the epigenetic modification characteristics of ANXA3. In our study, multivariable logistic regression analysis identified PMR (ANXA3) as an independent predictor of prognosis in HBV-ACLF patients receiving glucocorticoid therapy. The ANXA3 methylation levels in surviving patients after glucocorticoid treatment were significantly higher than that in non-surviving patients. Longitudinal monitoring revealed that glucocorticoid therapy downregulated ANXA3 mRNA expression while upregulating its methylation levels. As DNA methylation represents a fundamental epigenetic mechanism for gene expression regulation29,30, these findings suggest that epigenetic modulation may constitute a key mechanism underlying glucocorticoid efficacy.
This study has several limitations. First, due to the non-random allocation of glucocorticoid therapy, patient selection may be subject to indication bias, although we excluded for known confounders through multivariable statistical models. Second, the study cohort included only HBV-related ACLF patients and was designed as a single-center study, lacking coverage of other etiologies (such as alcohol- or NAFLD-related ACLF). Additionally, the model lacks independent external validation, which may affect the generalizability of the results. Third, HBV genotyping was not performed, preventing an assessment of the potential impact of viral genotypes on treatment efficacy. Fourth, all molecular analyses were conducted using PBMCs and have not yet been validated in liver tissue. Future studies will enroll patients with ACLF of varying etiologies in multicenter prospective randomized trials to systematically evaluate their impact on treatment response and independently validate the ANXA3 biomarker. Further investigation into the specific mechanistic role of ANXA3 in glucocorticoid-mediated inflammatory responses through in vitro and in vivo experiments is also under consideration.
Conclusion
In summary, this study identified a correlative link between glucocorticoid therapy and ANXA3 expression patterns in HBV-ACLF patients with differing outcomes. Furthermore, higher ANXA3 methylation levels were associated with improved clinical outcomes following glucocorticoid therapy. These findings suggest that ANXA3 methylation may serve as a prognostic biomarker for patients receiving glucocorticoid therapy in HBV-ACLF. Prospective validation in larger cohorts and mechanistic studies are needed to confirm these observations.
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Wu, D. et al. Plasminogen as a prognostic biomarker for HBV-related acute-on-chronic liver failure. J. Clin. Invest. 130(4), 2069–2080. https://doi.org/10.1172/jci130197 (2020).
Sarin, S. K. et al. Acute-on-chronic liver failure: consensus recommendations of the Asian Pacific association for the study of the liver (APASL): an update. Hepatol. Int. 13(4), 353–390. https://doi.org/10.1007/s12072-019-09946-3 (2019).
Burra, P. et al. Limitations of current liver donor allocation systems and the impact of newer indications for liver transplantation. J. Hepatol. 75(Suppl 1), S178–s190. https://doi.org/10.1016/j.jhep.2021.01.007 (2021).
Jia, L. et al. The efficacy and safety of Methylprednisolone in hepatitis B virus-related acute-on-chronic liver failure: a prospective multi-center clinical trial. BMC Med. 18(1), 383. https://doi.org/10.1186/s12916-020-01814-4 (2020).
Mussunoor, S. & Murray, G. I. The role of Annexins in tumour development and progression. J. Pathol. 216(2), 131–140. https://doi.org/10.1002/path.2400 (2008).
Toufiq, M. et al. Annexin A3 in sepsis: novel perspectives from an exploration of public transcriptome data. Immunology 161(4), 291–302. https://doi.org/10.1111/imm.13239 (2020).
Huang, Z. et al. Integrated bioinformatics exploration and preliminary clinical verification for the identification of crucial biomarkers in severe cases of COVID-19. J. Inflamm. Res. 17, 1561–1576. https://doi.org/10.2147/jir.S454284 (2024).
Jiang, J. et al. Upregulated of ANXA3, SORL1, and neutrophils May be key factors in the progressionof ankylosing spondylitis. Front. Immunol. 13, 861459. https://doi.org/10.3389/fimmu.2022.861459 (2022).
Zhu, Q. et al. Annexin A3 upregulates the infiltrated neutrophil-lymphocyte ratio to remodel the immune microenvironment in hepatocellular carcinoma. Int. Immunopharmacol. 89(Pt A), 107139. https://doi.org/10.1016/j.intimp.2020.107139 (2020).
Ma, S. et al. Identification of a potential miRNA-mRNA regulatory network associated with the prognosis of HBV-ACLF. Front. Mol. Biosci. 8, 657631. https://doi.org/10.3389/fmolb.2021.657631 (2021).
Li, F. et al. SOCS1 methylation level is associated with prognosis in patients with acute-on-chronic hepatitis B liver failure. Clin. Epigenetics. 15(1), 79. https://doi.org/10.1186/s13148-023-01495-9 (2023).
Ye, C. et al. Glucocorticoid treatment strategies in liver failure. Front. Immunol. 13, 846091. https://doi.org/10.3389/fimmu.2022.846091 (2022).
Malinchoc, M. et al. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology 31(4), 864–871. https://doi.org/10.1053/he.2000.5852 (2000).
Gao, S. et al. Aberrant GSTP1 promoter methylation predicts short-term prognosis in acute-on-chronic hepatitis B liver failure. Aliment. Pharmacol. Ther. 42(3), 319–329. https://doi.org/10.1111/apt.13271 (2015).
Zhao, Z. H. et al. Promoter methylation status and expression of PPAR-γ gene are associated with prognosis of acute-on-chronic hepatitis B liver failure. Clin. Epigenetics. 7, 115. https://doi.org/10.1186/s13148-015-0149-2 (2015).
Li, J. H. et al. SOCS3 and IL-6 mRNA levels in PBMC enhance the early prediction for patients with acute-on-chronic hepatitis B liver failure receiving glucocorticoid therapy. Front. Cell. Infect. Microbiol. 15, 1571443. https://doi.org/10.3389/fcimb.2025.1571443 (2025).
Wang, Y. et al. Altered PGE2-EP2 is associated with an excessive immune response in HBV-related acute-on-chronic liver failure. J. Transl Med. 17(1), 93. https://doi.org/10.1186/s12967-019-1844-0 (2019).
Zhang, Z. et al. Microglial Annexin A3 downregulation alleviates ischemic injury by inhibiting NF-κB/NLRP3-mediated inflammation. Inflammation https://doi.org/10.1007/s10753-025-02287-4 (2025).
Liang, J. et al. ANXA3 interference inactivates ERK/ELK1 pathway to mitigate inflammation and apoptosis in sepsis-associated acute lung injury. Mol. Immunol. 167, 25–33. https://doi.org/10.1016/j.molimm.2024.01.006 (2024).
Meng, H. et al. Annexin A3 gene Silencing promotes myocardial cell repair through activation of the PI3K/Akt signaling pathway in rats with acute myocardial infarction. J. Cell. Physiol. 234(7), 10535–10546. https://doi.org/10.1002/jcp.27717 (2019).
Durocher, M. et al. Inflammatory, regulatory, and autophagy co-expression modules and hub genes underlie the peripheral immune response to human intracerebral hemorrhage. J. Neuroinflammation. 16(1), 56. https://doi.org/10.1186/s12974-019-1433-4 (2019).
Li, A. et al. Methylprednisolone promotes Mycobacterium smegmatis survival in macrophages through NF-κB/DUSP1 pathway. Microorganisms 11(3). https://doi.org/10.3390/microorganisms11030768 (2023).
Marino, J. S. et al. Glucocorticoid receptor β induces hepatic steatosis by augmenting inflammation and Inhibition of the peroxisome Proliferator-activated receptor (PPAR) α. J. Biol. Chem. 291(50), 25776–25788. https://doi.org/10.1074/jbc.M116.752311 (2016).
Perretti, M. & D’Acquisto, F. Annexin A1 and glucocorticoids as effectors of the resolution of inflammation. Nat. Rev. Immunol. 9(1), 62–70. https://doi.org/10.1038/nri2470 (2009).
Gao, S. et al. Early prediction model for prognosis of patients with hepatitis-B-virus-related acute-on-chronic liver failure received glucocorticoid therapy. Eur. J. Med. Res. 27(1), 248. https://doi.org/10.1186/s40001-022-00891-w (2022).
Xue, R. & Meng, Q. The management of glucocorticoid therapy in liver failure. Front. Immunol. 10, 2490. https://doi.org/10.3389/fimmu.2019.02490 (2019).
Wiencke, J. K. et al. DNA methylation as a pharmacodynamic marker of glucocorticoid response and glioma survival. Nat. Commun. 13(1), 5505. https://doi.org/10.1038/s41467-022-33215-x (2022).
Zen, M. et al. The kaleidoscope of glucorticoid effects on immune system. Autoimmun. Rev. 10(6), 305–310 (2011).
Kim, H. et al. Differential DNA methylation and mRNA transcription in gingival tissues in periodontal health and disease. J. Clin. Periodontol. 48(9), 1152–1164. https://doi.org/10.1111/jcpe.13504 (2021).
Westin, I. M. et al. DNA methylation changes and increased mRNA expression of coagulation proteins, factor V and thrombomodulin in Fuchs endothelial corneal dystrophy. Cell. Mol. Life Sci. 80(3), 62. https://doi.org/10.1007/s00018-023-04714-x (2023).
Funding
This work was supported by the National Natural Science Foundation of China (82472260), the Natural Science Foundation of Shandong Province (ZR2022MH006), and the Crosswise Project of Shandong University (6010123188).
Author information
Authors and Affiliations
Contributions
Z.-H. W. designed the study, performed the research, analyzed the data, and drafted the manuscript. F. Z. analyzed the data and supervised this study. H.-X. Z. performed the research and collected the data. Z.-Z. T. collected the data. M.-M. X. analyzed the data. Y.-C. F. revised the manuscript. S. G. supervised this study. K. W. designed the study and revised the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This research protocol was reviewed and approved by the Medical Ethics Committee of Qilu Hospital of Shandong University in accordance with the principles of the Declaration of Helsinki (approval number “KYLL-202111-244-1”). Written informed consent was obtained from all participants prior to the start of the study. The ethical approval was exempted for the use of the GEO database by the Medical Ethics Committee of Qilu Hospital of Shandong University because the database was public and could be reused without restriction via an open license.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Z., Zhang, F., Zhu, H. et al. Association of ANXA3 methylation with clinical outcomes of glucocorticoid therapy in patients with hepatitis B virus-related acute-on-chronic liver failure. Sci Rep 16, 7235 (2026). https://doi.org/10.1038/s41598-026-37687-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-37687-5