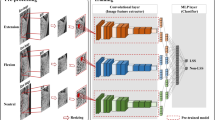
Abstract
Lumbar spine disorders represent a significant public health concern, with accurate diagnosis relying on vertebral segmentation and quantification. Traditional methods, such as Cobb angle measurement are constrained by two-dimensional projections, while cumulative segmentation and quantification errors limit automated CT analysis. To overcome these issues, this paper proposes a deep learning-based Boundary-Sensitive Network (BS-Net), integrating a Multi-Task Edge Processing (MEP) module and Contextual Bilateral Fusion (CBF) module to enhance vertebral edge feature extraction. The framework combines edge loss functions with morphological post-processing to achieve joint segmentation and quantification. Evaluations on 783 lumbar CT images from 379 patients and the public SPIDER MRI dataset demonstrate that BS-Net surpasses baseline models, achieving an MIoU of 96.56% and a Dice coefficient of 98.5%. Its spondylolisthesis quantification also shows strong agreement with manual assessment (ICC> 0.9). These results indicate that BS-Net provides an efficient and accurate solution for automated diagnosis of lumbar spondylolisthesis, with substantial clinical value.
Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Carragee, E. J. Clinical practice. persistent low back pain. N. Engl. J. Med. 352, 1891–1898. https://doi.org/10.1056/NEJMcp042054 (2005).
GBD 2021 Low Back Pain Collaborators. Global, regional, and national burden of low back pain, 1990–2020, its attributable risk factors, and projections to 2050: A systematic analysis of the global burden of disease study 2021. Lancet Rheumatol. 5, e316–e329. https://doi.org/10.1016/S2665-9913(23)00098-X (2023).
Kos, N., Gradisnik, L. & Velnar, T. A brief review of the degenerative intervertebral disc disease. Med. Arch. 73, 421–424. https://doi.org/10.5455/medarh.2019.73.421-424 (2019).
Wang, S. et al. Imaging comparison of paraspinal muscle degeneration in patients with degenerative and isthmic lumbar spondylolisthesis. Chin. J. Tissue Eng. Res. 29, 5869–5875. https://doi.org/10.12307/2025.804 (2025).
Iguchi, T. et al. Lumbar multilevel degenerative spondylolisthesis: Radiological evaluation and factors related to anterolisthesis and retrolisthesis. J. Spinal Disord. Tech. 15, 93–99. https://doi.org/10.1097/00024720-200204000-00001 (2002).
Zhang, Y. H., Gao, H. & Zhu, X. S. Correlation among CT attenuation value of cancellous bone in the lumbar vertebrae, age, and bone mineral density measured by dual-energy x-ray absorptiometry. Chin. J. Osteopor. 22, 695–699. https://doi.org/10.3969/j.issn.1006-7108.2016.06.008 (2016).
Kuklo, T. R., Potter, B. K., Schroeder, T. M. & O’Brien, M. F. Comparison of manual and digital measurements in adolescent idiopathic scoliosis. Spine 31, 1240–1246. https://doi.org/10.1097/01.brs.0000217774.13433.a7 (2006).
Aubin, C. E. et al. Reliability and accuracy analysis of a new semiautomatic radiographic measurement software in adult scoliosis. Spine 36, E780–E790. https://doi.org/10.1097/BRS.0b013e3181f0825a (2011).
Morrissy, R. T., Goldsmith, G. S., Hall, E. C., Kehl, D. & Cowie, G. H. Measurement of the cobb angle on radiographs of patients who have scoliosis: Evaluation of intrinsic error. J. Bone Jt. Surg. American 72, 320–327 (1990).
Harrison, D. E. et al. Cobb method or Harrison posterior tangent method: Which to choose for lateral cervical radiographic analysis. Spine 25, 2072–2078. https://doi.org/10.1097/00007632-200008150-00011 (2000).
Niu, S.-B., Yang, H., Yang, M.-Y., Chen, K. & Li, M. Imaging parameter measurement of spinal deformity: Research progress. Acad. J. Second Military Med. Univ. 41, 1177–1182 (2020) ((in Chinese with English abstract)).
Meyerding, H. W. Spondylolisthesis. Surg. Gynecol. Obstet. 54, 371 (1932).
Zheng, J., Wang, S., Zhang, X., Xiao, X. & Peng, S. Correlation of vertebral bone quality and paraspinal muscle changes with adjacent segment degeneration after transforaminal lumbar interbody fusion operation. Sichuan Da Xue Xue Bao Yi Xue Ban 55, 1301–1308. https://doi.org/10.12182/20240960105 (2024).
Boas, F. E. & Fleischmann, D. CT artifacts: Causes and reduction techniques. Imaging Med. 4, 229–240 (2004).
Zenk, M. et al. Comparative benchmarking of failure detection methods in medical image segmentation: Unveiling the role of confidence aggregation. Med. Image Anal. 101, 103392. https://doi.org/10.1016/j.media.2024.103392 (2025).
Qadri, S. F. et al. CT-based automatic spine segmentation using patch-based deep learning. Int. J. Intell. Syst. 2023, 2345835. https://doi.org/10.1155/2023/2345835 (2023).
Galbusera, F., Casaroli, G. & Bassani, T. Artificial intelligence and machine learning in spine research. JOR Spine 2, e1044. https://doi.org/10.1002/jsp2.1044 (2019).
Drozdzal, M. et al. Learning normalized inputs for iterative estimation in medical image segmentation. Med. Image Anal. 44, 1–13. https://doi.org/10.1016/j.media.2017.11.005 (2018).
Wang, W. et al. Multiview hyperedge-aware hypergraph embedding learning for multisite, multiatlas fMRI based functional connectivity network analysis. Med. Image Anal. 94, 103144. https://doi.org/10.1016/j.media.2024.103144 (2024).
Wang, Y. et al. Attention-enhanced u-net based network for cancerous tissue segmentation. Biomed. Signal Process. Control 106, 107728. https://doi.org/10.1016/j.bspc.2025.107728 (2025).
Wu, R. et al. SK-VM++: Mamba assists skip-connections for medical image segmentation. Biomed. Signal Process. Control 105, 107646. https://doi.org/10.1016/j.bspc.2025.107646 (2025).
Wang, H., Cao, P., Yang, J. & Zaiane, O. Narrowing the semantic gaps in u-net with learnable skip connections: The case of medical image segmentation. Neural Netw. 178, 106546. https://doi.org/10.1016/j.neunet.2024.106546 (2024).
Zhou, Z., Siddiquee, M. M. R., Tajbakhsh, N. & Liang, J. Unet++: A nested u-net architecture for medical image segmentation. In Deep Learning for Medical Image Analysis and Multimodal Learning for Clinical Decision Support 3–11 (Springer, 2018). https://doi.org/10.1007/978-3-030-00889-5_1.
Yadav, A. C., Kolekar, M. H. & Zope, M. K. Modified recurrent residual attention u-net model for MRI-based brain tumor segmentation. Biomed. Signal Process. Control 102, 107220. https://doi.org/10.1016/j.bspc.2024.107220 (2024).
Zhang, Q., Chen, M., Qin, Y. & Chen, X. Lung nodule segmentation based on 3D ResUNet network. Chin. J. Med. Phys. 36, 1356–1361. https://doi.org/10.1002/jy3rwl.2.0.tx;2-b (2019).
Kahal, S., Meddeber, L. & Zouagui, T. Enhancing skin lesions segmentation and classification through hierarchical attention ResUNet model. In Proceedings of the 2nd International Conference on Electrical Engineering, Automation and Control (ICEEAC 2024) (IEEE, 2024). https://doi.org/10.1109/ICEEAC61226.2024.10576576.
Abdani, S. R., Zulkifley, M. A. & Moubark, A. M. Pterygium tissues segmentation using densely connected deeplab. In Proceedings of the IEEE 10th Symposium on Computer Applications in Industrial Electronics (ISCAIE 2020) 229–232 (IEEE, 2020). https://doi.org/10.1109/iscaie47305.2020.9108822.
Kuang, X., Cheung, J. P. Y. & Wu, H. MRI-segflow: A novel unsupervised deep learning pipeline enabling accurate vertebral segmentation of MRI images. In 42nd Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 1633–1636. https://doi.org/10.1109/EMBC44109.2020.9175987 (2020).
Wang, J. & Liu, X. Medical image recognition and segmentation of pathological slices of gastric cancer based on deeplab v3+ neural network. Comput. Methods Prog. Biomed. 207, 106210. https://doi.org/10.1016/j.cmpb.2021.106210 (2021).
Atiya, S. U. & Ramesh, N. V. K. Enhancing non-small cell lung cancer radiotherapy planning: A deep learning-based multi-modal fusion approach for accurate gtv segmentation. Biomed. Signal Process. Control 92, 105987. https://doi.org/10.1016/j.bspc.2024.105987 (2024).
Douglas, L. et al. U-net breast lesion segmentations for breast dynamic contrast-enhanced magnetic resonance imaging. J. Med. Imaging 10, 064502. https://doi.org/10.1117/1.JMI.10.6.064502 (2023).
Ben Slama, A., Sahli, H., Amri, Y. & Labidi, S. V-net-vgg16: Hybrid deep learning architecture for optimal segmentation and classification of multi-differentiated liver tumors. Intell. Based Med. 11, 100210. https://doi.org/10.1016/j.ibmed.2025.100210 (2025).
Zeng, W., Fan, W., Zheng, Z. et al. Abdominal CT urography kidney segmentation using spatiotemporal fully convolutional network. In Medical Imaging 2021: Computer-Aided Diagnosis Vol. 11597, 115970L (SPIE, 2021). https://doi.org/10.1117/12.2581958.
Thakur, S. P., Doshi, J., Pati, S. et al. Skull-stripping of glioblastoma mri scans using 3d deep learning. In BrainLesion: Glioma, Multiple Sclerosis, Stroke and Traumatic Brain Injuries (BrainLes 2019), Part I, Vol. 11992 of Lecture Notes in Computer Science 57–68. https://doi.org/10.1007/978-3-030-46640-4_6 (2020).
Kamnitsas, K. et al. Deepmedic for brain tumor segmentation. In BrainLesion: Glioma, Multiple Sclerosis, Stroke and Traumatic Brain Injuries, of Lecture Notes in Computer Science Vol. 10154, 138–149. https://doi.org/10.1007/978-3-319-55524-9_14 (2016).
Zhou, T., Li, Q., Lu, H. L., Cheng, Q. & Zhang, X. X. Gan review: Models and medical image fusion applications. Inf. Fusion 91, 134–148. https://doi.org/10.1016/j.inffus.2022.10.017 (2023).
Deng, G., Sun, H. & Xie, W. Correlation-based switching mean teacher for semi-supervised medical image segmentation. Neurocomputing 633, 129818. https://doi.org/10.1016/j.neucom.2025.129818 (2025).
Kumaralingam, L. et al. Detsegdiff: A joint periodontal landmark detection and segmentation in intraoral ultrasound using edge-enhanced diffusion-based network. Comput. Biol. Med. 182, 109174. https://doi.org/10.1016/j.compbiomed.2024.109174 (2024).
Wu, H., Souedet, N., Jan, C., Clouchoux, C. & Delzescaux, T. A general deep learning framework for neuron instance segmentation based on efficient UNet and morphological post-processing. Comput. Biol. Med. 150, 106180. https://doi.org/10.1016/j.compbiomed.2022.106180 (2022).
Xu, G., Wu, X., Zhang, X. & He, X. Levit-unet: Make faster encoders with transformer for medical image segmentation. arXiv preprint. arXiv:2107.08623 (2021).
Chen, J. et al. Transunet: Rethinking the u-net architecture design for medical image segmentation through the lens of transformers. Med. Image Anal. 97, 103280. https://doi.org/10.1016/j.media.2024.103280 (2024).
Lv, C. et al. Ecm-transunet: Edge-enhanced multi-scale attention and convolutional mamba for medical image segmentation. Biomed. Signal Process. Control 107, 107845. https://doi.org/10.1016/j.bspc.2025.107845 (2025).
Cai, S., Jiang, Y., Xiao, Y., Zeng, J. & Zhou, G. Transumobilenet: Integrating multi-channel attention fusion with hybrid CNN-transformer architecture for medical image segmentation. Biomed. Signal Process. Control 107, 107850. https://doi.org/10.1016/j.bspc.2025.107850 (2025).
Goceri, E. An efficient network with CNN and transformer blocks for glioma grading and brain tumor classification from MRIs. Expert Syst. Appl. 268, 126290. https://doi.org/10.1016/j.eswa.2024.126290 (2025).
Vania, M., Mureja, D. & Lee, D. Automatic spine segmentation from CT images using convolutional neural network via redundant generation of class labels. J. Comput. Des. Eng. 6, 224–232. https://doi.org/10.1016/j.jcde.2018.05.002 (2019).
Janssens, R., Zeng, G. & Zheng, G. Fully automatic segmentation of lumbar vertebrae from CT images using cascaded 3d fully convolutional networks. In IEEE 15th International Symposium on Biomedical Imaging (ISBI 2018) 893–897. https://doi.org/10.1109/ISBI.2018.8363715 (IEEE, 2018).
Lessmann, N., van Ginneken, B. & Išgum, I. Iterative convolutional neural networks for automatic vertebra identification and segmentation in CT images. In Medical Imaging 2018: Image Processing Vol. 10574, 1057408. https://doi.org/10.1117/12.2293562 (2018).
Korez, R., Likar, B., Pernuš, F. & Vrtovec, T. Model-based segmentation of vertebral bodies from MR images with 3D CNNs. In Medical Image Computing and Computer Assisted Intervention – MICCAI 2016 Vol. 9901, 433–441 (Springer, 2016). https://doi.org/10.1007/978-3-319-46723-8_50.
Oktay, O., Schlemper, J., Le Folgoc, L. et al. Attention u-net: Learning where to look for the pancreas. arXiv preprint. arXiv:1804.03999 (2018).
Isensee, F. et al. nnU-Net: A self-configuring method for deep learning-based biomedical image segmentation. Nat. Methods 18, 203–210. https://doi.org/10.1038/s41592-020-01008-z (2021).
Gu, Z. et al. Ce-net: Context encoder network for 2d medical image segmentation. IEEE Trans. Med. Imaging 38, 2281–2292. https://doi.org/10.1109/TMI.2019.2903562 (2019).
Sekuboyina, A., Kukačka, J., Kirschke, J. et al. Attention-driven deep learning for pathological spine segmentation. In Computational Methods and Clinical Applications in Musculoskeletal Imaging - 5th International Workshop, MSKI 2017, Revised Selected Papers, Vol. 10734, 108–119 (Springer, 2018). https://doi.org/10.1007/978-3-319-74113-0_10.
Chen, J., Lu, Y., Yu, Q. et al. Transunet: Transformers make strong encoders for medical image segmentation. arXiv preprint. arXiv:2102.04306 (2021).
Wang, H., Cao, P., Wang, J. & Zaiane, O. Uctransnet: Rethinking the skip connections in u-net from a channel-wise perspective with transformer. arXiv preprint. arXiv:2109.04335 (2022).
Hatamizadeh, A., Nath, V., Tang, Y. et al. Swin unetr: Swin transformers for semantic segmentation of brain tumors in MRI images. arXiv preprint. arXiv:2201.01266 (2022).
Pu, Y. et al. Semi-supervised segmentation of coronary DSA using mixed networks and multi-strategies. Comput. Biol. Med. 156, 106493. https://doi.org/10.1016/j.compbiomed.2022.106493 (2023).
Athertya, J. & Saravana Kumar, G. Automatic segmentation of vertebral contours from CT images using fuzzy corners. Comput. Biol. Med. 72, 75–89. https://doi.org/10.1016/j.compbiomed.2016.03.009 (2016).
van der Graaf, J. et al. Lumbar spine segmentation in MR images: A dataset and a public benchmark. Sci. Data 11, 264. https://doi.org/10.1038/s41597-024-03090-w (2024).
Jeong, H., You, J., Sohn, H. & Park, S. Radiologic evaluation of degeneration in isthmic and degenerative spondylolisthesis. Asian Spine J. 7, 25–33. https://doi.org/10.4184/asj.2013.7.1.25 (2013).
Schwab, F., Lafage, V., Boyce, R., Skalli, W. & Farcy, J. Gravity line analysis in adult volunteers: Age-related correlation with spinal parameters, pelvic parameters, and sagittal balance. Spine 31, E959–E967. https://doi.org/10.1097/01.brs.0000248126.96737.0f (2006).
Jiang, H., Zeng, J., Zhu, S., Huo, S. & Ruan, K. Measurement of the morphological changes of the lumbar vertebral lamina gap in different body positions and its clinical significance. Chin. J. Bone Jt. Surg. 14, 10–15 (2025).
Samuvel, B.& Thomas, V. A mask based segmentation algorithm for automatic measurement of cobb angle from scoliosis x-ray image. In 2012 International Conference on Advances in Computing and Communications (ICACC) 110–113. https://doi.org/10.1109/ICACC.2012.24 (2012).
Yan, S., Constant, C., Ramazanian, T., Maradit Kremers, H. & Larson, A. Automated cobb angle measurement in adolescent idiopathic scoliosis: Validation of a previously-published deep learning method. In 2022 IEEE 10th International Conference on Healthcare Informatics (ICHI) 495–496. https://doi.org/10.1109/ICHI54592.2022.00085 (2022).
Vyas, D., Ganesan, A. & Meel, P. Computation and prediction of Cobb’s angle using machine learning models. In 2022 2nd International Conference on Intelligent Technologies (CONIT) 1–6. https://doi.org/10.1109/CONIT55038.2022.9847938 (2022).
Liu, Y. et al. A robust and efficient measurement method of lumbar lordosis based on three dimensional curvature line. In 2023 4th International Conference on Information Science, Parallel and Distributed Systems (ISPDS) 452–457. https://doi.org/10.1109/ISPDS58840.2023.10235615 (2023).
Kuang, X., Cheung, J., Wu, H., Dokos, S. & Zhang, T. Mri-segflow: A novel unsupervised deep learning pipeline enabling accurate vertebral segmentation of MRI images. In 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC) 1633–1636. https://doi.org/10.1109/EMBC44109.2020.9175987 (2020).
Cao, H. et al. Swin-unet: Unet-like pure transformer for medical image segmentation. arXiv preprint. arXiv:2105.05537 (2021).
Ronneberger, O., Fischer, P. & Brox, T. U-net: Convolutional networks for biomedical image segmentation. In International Conference on Medical Image Computing and Computer-Assisted Intervention 234–241. https://doi.org/10.1007/978-3-319-24574-4_28 (Springer, 2015).
Zhou, Z., Rahman Siddiquee, M., Tajbakhsh, N. & Liang, J. Unet++: A nested u-net architecture for medical image segmentation. In Deep Learning in Medical Image Analysis and Multimodal Learning for Clinical Decision Support, Vol. 11045 of Lecture Notes in Computer Science 3–11 (Springer, 2018). https://doi.org/10.1007/978-3-030-00889-5_1.
Zhou, N., Xu, M. & Shen, B. Vit-unet: A vision transformer based unet model for coastal wetland classification based on high spatial resolution imagery. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 17, 19575–19587. https://doi.org/10.1109/JSTARS.2024.3487250 (2024).
Cheng, P., Yang, Y. & Yu, H. Automatic vertebrae localization and segmentation in CT with a two-stage dense-u-net. Sci. Rep. 11, 1–13. https://doi.org/10.1038/s41598-021-01296-1 (2021).
Brianna, D., Kesuma, L., Geovani, D. & Sari, P. Combination of image enhancement and double u-net architecture for liver segmentation in CT-scan images. J. Electron. Electromed. Eng. Med. Inform. 7, 208–219 (2025).
He, K., Zhang, X., Ren, S. & Sun, J. Deep residual learning for image recognition. In Proc. IEEE Conference on Computer Vision and Pattern Recognition (CVPR), 770–778. https://doi.org/10.1109/CVPR.2016.90 (2016).
Funding
This work was supported by the Joint Scientific Research Fund (General Program) under Grant No. 23JRRA1484, titled “Research on Lumbar Imaging Knowledge Detection Based on Image Feature Understanding”, and the National Natural Science Foundation of China (Project No.: 62266031). The project was led by D. Ji.
Author information
Authors and Affiliations
Contributions
D.J. proposed and designed the research idea. D.J. and F.Q. jointly designed the study plan. F.Q. developed the model and conducted the experiments. F.Q. and Y.Z. analyzed the results and prepared the figures. D.J. and F.Q. wrote the first draft of the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ji, D., Qian, F. & Zong, Y. Boundary sensitive-net-based lumbar vertebra segmentation and spondylolisthesis measurement. Sci Rep (2026). https://doi.org/10.1038/s41598-026-38522-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-38522-7