Abstract
Infectious mononucleosis (IM), a common disease in children, was often accompanied by excessive inflammatory response and organ damage. This study aimed to explore the CD26/ADA axis in the pathogenesis of acute Epstein-Barr virus (EBV)-associated IM. Thirty children with acute IM and thirty age-matched controls from Children’s Hospital of Soochow University were enrolled in this study. The CD26 and adenosine deaminase (ADA) mRNA were detected by real-time PCR in the peripheral blood. Meanwhile, the levels of plasma interferon gamma (IFN-γ), interleukin (IL)-4 and IL-10 were measured by ELISA, and the frequency of CD26-positive cells in CD4+ T cells and CD8+ T cells was analyzed by flow cytometry. Children with acute IM had significantly higher expression levels of CD26 mRNA and ADA mRNA than control group in peripheral blood (P < 0.05). Plasma IL-4, IL-10 and INF-γ level were markedly elevated compared to the control group (P < 0.05). Furthermore, the plasma CD26 protein level was positively correlated with ADA (r = 0.610, P<0.01) and INF-γ (r = 0.417, P = 0.022), and negatively correlated with the percentage of CD3+ CD4+ T cell (r = -0.446, P = 0.014) and CD4+ /CD8+ ratio (r = -0.414, P = 0.023) in acute IM group. In addition, the frequency of CD26-positive cells was elevated on CD8+ T cells but markedly decreased on CD4+ CD39+ regulatory T cells (P < 0.05). The peripheral blood CD26 and ADA mRNA levels were elevated in acute IM. Moreover, CD26 exhibited cell-specific alterations, being increased on CD8+ T cells but reduced on CD4+ CD39+ Tregs.
Similar content being viewed by others
Introduction
Epstein-Barr virus (EBV) belongs to the herpesvirus family, with a positivity rate of antibody in serum exceeded 90%1. Primary EBV infection can cause infectious mononucleosis (IM), which is typically characterized by fever, cervical lymphadenopathy, and pharyngotonsillitis2. Although acute IM is a self-limiting disease, 5–10% of pediatric patients may still progress to severe illness such as acute hepatic failure3 and hemophagocytic lymphohistiocytosis (HLH), of which the mortality rate reaches as high as 50%4. However, the immunopathogenic mechanisms underlying EBV infection remain unclear.
Numerous previous studies have shown that the cytokines IL-4, IL-6, IL-10 and IFN-γ were significantly elevated in acute IM5. In addition, the EBV-specific CD8+ T-cell responses undergo significant expansion, potentially driving immunopathological processes in acute EBV infection6. During the persistent phase of infection, EBV-specific CD8+ T cells appear to be vital for controlling viral replication. However, the immune response was excessively robust, which can lead to the production of a large number of inflammatory cytokines such as TNF-α and IL-67, and caused organ damage. Our previous studies8,9 have revealed that serum adenosine deaminase (ADA) enzymatic activity was significantly elevated in children with EBV-IM and was positively correlated with the number of CD8+ T cells. Moreover, serum ADA also served as a biomarker for disease severity in children with IM10.
ADA, a key enzyme in the adenosine metabolic pathway, can bind to CD26 and anchor on the cell surface as membrane-bound ADA, thereby efficiently regulating adenosine concentration in the local microenvironment. Additionally, the ADA-CD26 complex also delivers co-stimulatory signals that activate lymphocytes11.CD26 is a type II transmembrane glycoprotein expressed on activated T cells, B cells, and NK cells, with its expression level closely correlating with cellular activation status and inflammatory response intensity12. Previous research have demonstrated that ADA/CD26 axis was involved in the pathogenesis of a variety of immune-related diseases such as metabolic diseases, autoimmune diseases, inflammatory infectious diseases, and tumors, and was closely associated with disease severity, therapeutic efficacy, and prognosis13,14. Notably, CD39 serves as a key marker of functionally stable regulatory T cells (Treg cells) and exerts immunosuppressive effects by hydrolyzing ATP to produce adenosine15. Furthermore, CD39-derived adenosine can be metabolized by ADA, while CD26-mediated co-stimulatory signals may also influence the activity and stability of CD39+ Treg cells. However, the role of ADA/CD26 axis in the pathogenesis of EBV-IM has not yet been explored. This study aims to investigate the CD26/ADA axis expression in peripheral blood of children with acute IM and its association with T cell subsets and cytokines, thereby providing a basis for elucidating IM pathogenesis and identifying potential biomarkers.
Materials and methods
Participants
This prospective study was conducted at the Children’s Hospital of Soochow University from May 2023 to August 2023. The study population comprised children aged > 1 month to ≤ 16 years with a confirmed diagnosis of IM. Exclusion criteria included: (1) immunodeficiency disorders; (2) chronic or metabolic diseases; and (3) use of immunosuppressive agents, antiviral drugs, or immunomodulators within the preceding month. Thirty children with acute IM were enrolled without severity stratification. There were eighteen males and twelve females, with an average age of (5.5 ± 2.4) years. Thirty healthy children served as control group in the same period, twelve males and eighteen females, with an average age of (5.7 ± 3.2) years. The inclusion criteria of the control group were no history of immune system diseases, chronic diseases and infectious diseases within the last month. There were no statistical difference in gender and age between the two groups. This study, which was approved by the Ethics Committee of Children’s Hospital of Soochow University (No.2023CS059), meets the ethical principles of the Declaration of Helsinki. The informed consent was obtained from the children’s parents.
Definition of IM
The criteria of IM were16,17: (1) the presence of at least three clinical manifestations: fever, cervical lymphadenopathy, pharyngitis, splenomegaly, hepatomegaly and eyelid edema; (2) presence of immunoglobulin IgM and IgG to EBV viral capsid antigen (VCA), and without immunoglobulin IgG to EB nuclear antigen (EBNA); and (3) exclusion of other viral infections such as cytomegalovirus, human immunodeficiency virus, hepatotropic virus and herpes simplex virus.
Real-time quantitative PCR (RT-qPCR) for CD26 and ADA mRNA
2 ml peripheral venous blood was collected from participants into the EDTA tube within 24 h after admission, then used the Ficoll separation medium (Dakewei Biotechnology Co., Ltd. Guangdong, China) to obtain peripheral blood mononuclear cells (PBMCs). Total RNA was extracted using Trizol reagent according to the manufacturer’s protocol. Next, using the Reverse Transcription kit (Yisheng Biotechnology Co., Ltd, Shanghai, China) to perform reverse transcription PCR. The qRT-PCR was performed using a Universal Blue qPCR SYBR Master Mix kit (Applied Biosystems, Foster City, CA, USA) and human RPS18 was used as an endogenous control for CD26 and ADA (Table 1). The reactions were performed under the following conditions:95 °C for 60 s, followed by 40 cycles of 10 s at 95 °C and 60 °C for 30 s, and concluded with 1 min at 60 °C. The average values of the cycle threshold (Ct) of the reactions in triplicate were determined and use the 2-ΔΔCt method to analysis data.
Enzyme-linked immunosorbent assay (Elisa) assay for IL-4, IL-10, INF-γ, CD26 and ADA
Plasma was separated from venous blood, and IL-4, IL-10, INF-γ, CD26 and ADA levels were measured by enzyme-linked immunosorbent assay kits (double antibody sandwich method, Jiangsu Jingmei Biotechnology Co., Ltd, Jiangsu, China) in a microplate reader.The standard diluent was used as the zero standard.
Flow cytometry
Peripheral whole blood samples were labeled with antibodies including anti-CD26-allophycocyanin (APC, clone BA5b, BioLegend, Inc., San Diego, United States), anti-CD39-phycoerythrin (PE, clone A1, BioLegend, Inc., San Diego, United States), anti-CD3-brilliant violet (clone OKT3, BioLegend, Inc., San Diego, United States), anti-CD4-fluorescein isothiocyanate (FITC, clone RPA-T4, BioLegend, Inc., San Diego, United States) and anti-CD8-peridinin chlorophyll protein-cyanine 5.5 (PerCP-Cy5.5, clone SK1, BioLegend, Inc., San Diego, United States), and then incubated in the dark for 15 min. Afterward, 2 mL of erythrocyte lysis buffer was added and the sample was incubated for 10 min. The mixture was then centrifuged to remove the supernatant. Subsequently, PBS was added to the pellet, followed by centrifugation and resuspension. Data were acquired on a multi-color flow cytometer (Cytek Northern Light 3 laser 16 V-14B-8R, Shanghai, China). For each sample, a minimum of 20,000 lymphocytes were collected. The acquisition rate was maintained at 500–600 events per second to ensure data quality. Data were analyzed using FlowJo software (version 10.10, Tree Star, Ashland, OR, USA).The gating strategy was as follows: (1) lymphocytes were identified by FSC-A vs. SSC-A; (2) single cells were selected using FSC-H vs. FSC-A; (3) CD3⁺ T cells were identified; (4) within CD3⁺ cells, CD4⁺ and CD8⁺ subsets were distinguished; (5) The percentage of CD39+ cells among CD4⁺ T cell and CD8⁺ T cell populations; (6) The percentage of CD26+ cells among CD4⁺ T cell and CD8⁺ T cell populations.
Statistical analysis
The continuous variables were presented as mean±standard deviation or interquartile ranges, and categorical variables as percentages (%), respectively. The T-Test (normally distributed) and Mann-Whitney U test (normally distributed) for continuous variables. Categorical variables were analyzed using the chi-square test. Pearson’s correlation analysis was used to calculate the correlation between two continuous variables. All statistical analyses were conducted using IBM SPSS Statistics version 25.0 (IBM Corp., New York, USA), and the figures were constructed using GraphPad Prism version 9.0 (GraphPad Software, LLC, San Diego, USA). A P-value of less than 0.05 was deemed statistically significant.
Results
Demographic and clinical characteristics of children with acute IM
A total of 30 children with acute IM and 30 control children were enrolled in this study. As shown in Table 2, peripheral blood white blood cell (WBC) count, lymphocyte count, IgA, IgG, IgM, CD3+ T cell (% and count) and CD3+ CD8+ T cell (% and count) in children with acute IM were all significantly increased compared with controls (all p < 0.05). However, the CD3+ CD4+ T cell (% and count) and CD4+/CD8+ ratio was significantly decreased in acute IM group (p < 0.05).
The expression of CD26 and ADA mRNA and plasma cytokines levels in acute IM patients
As shown in Fig. 1A and B, the levels of CD26 and ADA mRNA expression were higher in children with acute IM compared with controls (P<0.05). As shown in Fig. 2, it showed that the plasma ADA, IL-4, IL-10 and INF-γ level were higher in acute IM patients than control group (P<0.05). However, there was no significant difference in plasma CD26 level between the two groups (P = 0.5161).
Comparisons of CD26 mRNA and ADA mRNA expression between IM patients and controls. (A) CD26 mRNA in peripheral blood; (B) ADA mRNA in peripheral blood. Statistically significant differences (statistical t-test, *: P<0.05, ****༚P<0.0001).
The expression level of plasma CD26, ADA and cytokines between IM patients and controls. (A) The expression level of plasma CD26; (B) The expression level of plasma ADA; (C) The expression level of plasma IL-4; (D) The expression level of plasma IL-10; (E) The expression level of plasma IFN-γ. ns: not significant; Statistically significant differences (statistical t-test, *༚P<0.05, **༚P<0.01).
The correlation between plasma CD26 level and other indicators
As shown in Table 3, the CD26 was positively correlated with ADA (r = 0.610, P<0.01) and INF-γ (r = 0.417, P = 0.022), and negatively correlated with the percentage of CD3+ CD4+ T cell (r = −0.446, P = 0.014) and CD4+/CD8+ ratio (r = −0.414, P = 0.023) in acute IM group. However, no correlation was found between plasma CD26 levels and other parameters, including lymphocyte count, levels of IgA, IgG, IgM, IL-4, IL-10, and the frequencies of CD3⁺ T cells and CD3⁺ CD8⁺ T cells (P > 0.05).
CD26 expression on lymphocyte subsets by flow cytometry
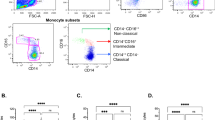
We analyzed the percentage of CD26-positive cells on lymphocyte subsets between patients with IM and healthy controls. First, we explored the percentage of CD26+ cells in the CD3+ CD4+ T cell and CD3+ CD8+ T cell in peripheral blood between two groups (Fig. 3A and B). The frequency of CD8⁺ CD39⁺ T cells showed no difference between the two groups, while the frequency of CD4⁺ CD39⁺ T cells was markedly increased in children with acute IM (P < 0.01) (Fig. 4A). After gating on CD39-positive (CD39⁺) and CD39-negative (CD39⁻) CD8⁺ T cells (Fig. 3C and D), the frequencies of CD26-positive (CD26⁺) cells in these T cell subsets were higher (P < 0.05) in the acute IM group than in the control group (Fig. 4B). After gating on CD39-positive (CD39⁺) and CD39-negative (CD39⁻) CD4⁺ T cells (Fig. 3E and F), the frequency of CD4⁺ CD39⁺ CD26⁺ T cells and CD4⁺ CD39⁺ CD26- T cells showed no significant difference between the acute IM and control groups (Fig. 4C). However, the acute IM group exhibited a lower frequency of CD4⁺ CD39⁻ CD26⁺ T cells but a higher frequency of CD4⁺ CD39⁻ CD26⁻ T cells compared to the control group.
The CD26 expression in CD8+T cells and CD4+ T cells. PBMC obtained from IM and control group were stained with relevant antibodies and analyzed by flow cytometry. (A) The percentage of CD39-positive cells in CD3⁺ CD8⁺ T cells. (B) The percentage of CD39-positive cells in CD3⁺ CD4⁺ T cells. (C) The percentage of CD26-positive cells in CD8+ CD39+ T cells. (D) The percentage of CD26-positive cells in CD8+ CD39− T cells. (E) The percentage of CD26-positive cells in CD4+ CD39+ T cells. (F) The percentage of CD26-positive cells in CD4+ CD39− T cells.
Comparison of the frequency of CD26-positive cells in CD8⁺ T cells and CD4+T cells between IM and control group. (A) Comparison of the frequency of CD39-positive cells in CD8⁺ T cells and CD4+T cells. (B) Comparison of the frequency of CD26-positive cells in CD8+ CD39+T cells and CD8+ CD39−T cells. (C) Comparison of the frequency of CD26-positive cells in CD4+ CD39+T cells and CD4+ CD39−T cells. ns: not significant; Statistically significant differences (statistical Mann-Whitney U test, *P<0.05, **P<0.01,***P<0.001,****P<0.0001).
Discussion
To explore the pathogenesis of EBV-infectious mononucleosis (EBV-IM) in children, we focused on the CD26 molecule in immune responses mediated by CD8+ T cells and CD4+ T cells. Our findings shed light on the potential significance of the further elucidating the pathogenesis mechanisms during primary EBV infection.
Following the entry of EBV into the human body, the host immune system played a critical role in recognizing and eliminating EBV infection. EBV-infected B lymphocytes undergo surface antigen modification, prompting activated T cells to secrete inflammatory mediators. Nevertheless, the dysregulation of immune response contributed to the initiation and advancement of disease5. In this study, the level of IgA, IgM, IgG, IL-4, IL-10 and INF-γ in peripheral blood were elevated in acute EBV-IM, which implied that humoral and cellular immunity were activated in the acute EBV infection8,9. In addition, the high level of the pro-inflammatory factor such as IL-2 and INF-γ and anti-inflammatory factor IL-4 in peripheral blood elucidated that the acute phase of EBV infection manifested as a dynamic equilibrium between pro-inflammatory and anti-inflammatory responses. Moreover, the peripheral blood lymphocytes, particularly CD8+ T cells, were significantly increased in children with acute EBV-IM compared to the control group, with an inverted CD4+/CD8+ lymphocyte ratio. It was thought to be due to excessive proliferation of CD8+ T lymphocytes with a typical form, which was crucial for eliminating EBV infection9,18,19.
Previous studies have indicated the proliferation of effector T cells and the production of cytokines were dependent on the activity of ADA in malignancy-associated diseases20. Our previous study demonstrated that ADA enzymatic activity was markedly elevated in the peripheral blood of children with EBV-IM and positively correlated with the CD8+ T cell fraction while negatively correlating with CD4+ T lymphocytes10. In this study, it show that both ADA mRNA and protein levels were significantly increased. The CD26 molecule, which is a key player in T-cell activation through its interaction with ADA to modulate the adenosine metabolic pathway, showed increased mRNA expression in children with EBV-IM in this study. Concurrently, it was found that plasma CD26 protein level correlated with ADA, CD4+ T lymphocyte percentage, CD4+/CD8+ lymphocyte ratio and INF-γ. Collectively, suggesting that CD26 and ADA play significant roles in the immunopathogenesis of EBV-IM. Moreover, it was demonstrated that ADA/CD26 axis can enhance CD8+ T cell function within the tumor microenvironment14.
The ectoenzymes CD39, another pivotal enzyme in adenosine metabolism, can degrade extracellular ATP to adenosine and have been implicated in T-cell exhaustion21. Moreover, the absence of surface CD26 along with CD39 expression can be regarded as a useful biomarker for identifying and isolating functionally active human Treg cells22,23. In contrast, CD26 was overexpressed on most of effector T cells24. In this study, the proportion of CD39-positive CD8⁺ T cells in the peripheral blood of children with acute IM showed no significant difference compared to healthy children. However, the proportion of CD26-positive was significantly increased in both CD8+ CD39+ T cells and CD8+ CD39− T cells. As a crucial co-stimulatory molecule for T cell activation, CD26 synergistically potentiates CD3-mediated signaling. In CD39⁺CD8⁺ T cells, CD26 upregulation functions as a compensatory mechanism that sustains effector function by neutralizing adenosine (ADO)-mediated suppression via the ADA/CD26 axis25. Notably, even in CD39⁻ cells, elevated CD26 enhances microenvironmental ADO clearance and metabolic fitness. Previous studies have demonstrated that the CD26 positive T cells were closely associated not only with the inflammatory progression of coronary syndrome26 but also with the tumor resistant microenvironment14. It was of note that the proportion of CD39-expressing CD4⁺ T cells was markedly higher, which indicated that regulatory CD4+ T cells were predominant among CD4+ T cells during the acute phase of EBV infection, which may be responsible for increasing IL-10 in peripheral blood and preventing excessive inflammation. Previous studies have established CD4⁺ CD39⁻ T cells as functionally active effector helper T cell subsets, including Th1, Th17, and Th2, which characteristically express high levels of CD26 on their surface27. However, in infectious mononucleosis, the proportion of CD26-expressing CD4⁺ CD39⁻ T cells is significantly decreased, thereby impairing helper T cell proliferation and inflammatory cytokine secretion. In this study, the plasma CD26 protein exhibited no alteration, whereas the frequency of CD26-expressing CD8⁺T lymphocytes was markedly elevated and a significant decrease in CD4+ CD39+ T cells in EBV-IM. This discrepancy suggests that CD26 regulation in IM may be primarily at the transcriptional level in immune cells, with the protein remaining membrane-bound rather than being shed into circulation. This contrasts with findings in some autoimmune diseases. Previous research found that the serum CD26 was significantly decreased and increased CD26 expresses on immune cells in rheumatoid arthritis, systemic lupus erythematosus and multiple sclerosis28,29,30,31, which suggested CD26 acted in different ways in different diseases.
Conclusions
In acute EBV-IM, peripheral blood CD26 and ADA mRNA levels were elevated. CD26 exhibited cell-specific alterations, being increased on CD8+ T cells but reduced on CD4+ CD39+ Tregs, while plasma CD26 protein correlated with ADA, IFN-γ and CD4 + T cell parameters.
Limitations
This study has not yet explored the CD26/ADA axis in mild versus severe IM or its correlation with cytokine profiles. Subsequent studies will recruit patients with severe IM for further investigation and will single-cell RNA sequencing to delineate the activation pathways in which CD26 is implicated.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- IM:
-
Infectious mononucleosis
- IFN-γ,:
-
Interferon gamma
- IL-4:
-
Interleukin-4
- EBV:
-
Epstein-Barr virus
- HLH:
-
Hemophagocytic lymphohistiocytosis
- ADA:
-
Adenosine deaminase
- VCA:
-
Capsid antigen
- EBNA:
-
Nuclear antigen
- APC:
-
Allophycocyanin
- PE:
-
phycoerythrin
- FITC:
-
Fluorescein isothiocyanate
- WBC:
-
Peripheral blood white blood cell
References
Thompson, M. P. & Kurzrock, R. Epstein-Barr virus and cancer. Clin. Cancer Res. 10 (3), 803–821 (2004).
Luzuriaga, K. & Sullivan, J. L. Infectious mononucleosis. N Engl. J. Med. 362 (21), 1993–2000 (2010).
Mellinger, J. L. et al. Epstein-Barr virus (EBV) related acute liver failure: a case series from the US acute liver failure study group. Dig. Dis. Sci. 59 (7), 1630–1637 (2014).
Zhang, W. et al. Clinical significance and different strategies for re-elevation of plasma EBV-DNA during treatment in pediatric EBV-associated hemophagocytic lymphohistiocytosis. J. Pediatr. (Rio J) 100(5), 505–511 (2024).
Zhang, Y. et al. Characteristics of immunological events in Epstein-Barr virus infection in children with infectious mononucleosis. Front. Pediatr. 11, 1060053 (2023).
Silva, J. M., Alves, C. E. C. & Pontes, G. S. Epstein-Barr virus: the mastermind of immune chaos. Front. Immunol. 15, 1297994 (2024).
Wada, T. et al. Cytokine profiles in children with primary Epstein-Barr virus infection. Pediatr. Blood Cancer. 60 (7), E46–E48 (2013).
Shi, T. et al. Diversity of adenosine deaminase in children with EBV-related diseases. Ital. J. Pediatr. 48 (1), 148 (2022).
Shi, T. et al. Concordance of adenosine deaminase with Immunoglobulins and lymphocyte subsets in EBV-related diseases. Ital. J. Pediatr. 49 (1), 49 (2023).
Shi, T., Li, J., Miao, Y., Huang, L. & Tian, J. Adenosine deaminase as a marker for the severity of infectious mononucleosis secondary to EBV in children. BMC Infect. Dis. 22 (1), 164 (2022).
Cordero, O. J. et al. Distinctive CD26 expression on CD4 T-Cell subsets. Biomolecules 11 (10), 1446 (2021).
Ishii, T. et al. CD26-mediated signaling for T cell activation occurs in lipid rafts through its association with CD45RO. Proc. Natl. Acad. Sci. U S A. 98 (21), 12138–12143 (2001).
Jedrzejewska, A. et al. Kutryb-Zając B. Changes in adenosine deaminase activity and endothelial dysfunction after mild coronavirus Disease-2019. Int. J. Mol. Sci. 24 (17), 13140 (2023).
Franzese, O. et al. ADA/CD26 axis increases intra-tumor PD-1 + CD28+ CD8 + T-cell fitness and affects NSCLC prognosis and response to ICB. Oncoimmunology 13 (1), 2371051 (2024).
Liu, X. et al. The CD39-CD73-adenosine axis: master regulator of immune evasion and therapeutic target in pancreatic ductal adenocarcinoma. Biochim. Biophys. Acta Rev. Cancer. 1880 (5), 189443 (2025).
Ming, Y. et al. Infectious mononucleosis in children and differences in biomarker levels and other features between disease caused by Epstein-Barr virus and other pathogens: a single-center retrospective study in China. PeerJ 11, e15071 (2023).
Shi, T., Huang, L., Chen, Z. & Tian, J. Characteristics of primary Epstein-Barr virus infection disease spectrum and its reactivation in children, in Suzhou, China. J. Med. Virol. 93 (8), 5048–5057 (2021).
Wang, Y. et al. HLA-DR expression level in CD8 + T cells correlates with the severity of children with acute infectious mononucleosis. Front. Immunol. 12, 753290 (2021).
Tomkinson, B. E., Wagner, D. K., Nelson, D. L. & Sullivan, J. L. Activated lymphocytes during acute Epstein-Barr virus infection. J. Immunol. 139 (11), 3802–3807 (1987).
Yang, Y. et al. The progress and prospects of targeting the adenosine pathway in cancer immunotherapy. Biomark. Res. 13 (1), 75 (2025).
Tsai, A. K. & Stromnes, I. M. CD39 deletion in TCR-engineered T cells enhances antitumour immunity. Gut 73 (5), 716–717 (2024).
Mandapathil, M. et al. CD26 expression and adenosine deaminase activity in regulatory T cells (Treg) and CD4(+) T effector cells in patients with head and neck squamous cell carcinoma. Oncoimmunology 1 (5), 659–669 (2012).
Deaglio, S. et al. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J. Exp. Med. 204 (6), 1257–1265 (2007).
Mandapathil, M. et al. Generation and accumulation of immunosuppressive adenosine by human CD4 + CD25highFOXP3+ regulatory T cells. J. Biol. Chem. 285 (10), 7176–7186 (2010).
Timperi, E. & Barnaba, V. CD39 regulation and functions in T cells. Int. J. Mol. Sci. 22 (15), 8068 (2021).
Moro, P. J. et al. Mononuclear cell adenosine deaminase and CD26/dipeptidylpeptidase-IV activities are sensitive markers of reperfusion during percutaneous transluminal angioplasty. Int. J. Cardiol. 166 (1), 225–229 (2013).
Antonioli, L., Blandizzi, C., Pacher, P. & Haskó, G. Immunity, inflammation and cancer: a leading role for adenosine. Nat. Rev. Cancer. 13 (12), 842–857 (2013).
Grujic, M. et al. Activity and expression of dipeptidyl peptidase IV on peripheral blood mononuclear cells in patients with early steroid and disease modifying antirheumatic drugs naïve rheumatoid arthritis. Clin. Chem. Lab. Med. 55 (1), 73–81 (2017).
Zhao, Y. CD26 in autoimmune diseases: the other side of moonlight protein. Int. Immunopharmacol. 75, 105757 (2019).
Hatano, R. et al. An abnormal increase in CD26(-)CD28(-) cytotoxic effector CD4 and CD8 T cell populations in patients with systemic lupus erythematosus. Int. Immunol. 37 (3), 153–172 (2025).
Tejera-Alhambra, M. et al. Low DPP4 expression and activity in multiple sclerosis. Clin. Immunol. 150 (2), 170–183 (2014).
Funding
This study was supported by the Science and Technology Project of Suzhou, China (2023ZDPY03).
Author information
Authors and Affiliations
Contributions
TS and WS collected data; LH analyzed and interpreted data; TS wrote the manuscript; WS prepared figures and tables; LH and JT revised critically the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
This study adheres to the ethical principles of the Declaration of Helsinki. It was approved by the Ethics Committee of Children’s Hospital of Soochow University (No.2023CS059). Given the pediatric population, special protections for child participants were implemented: Written informed consent was obtained from the legal guardians of all children, and additional verbal assent was obtained from children aged ≥ 7 years. The informed consent form was specifically revised to include: (i) explicit statements that only residual clinical specimens/routine data would be used to avoid additional procedures; (ii) clear emphasis on the child’s right to refuse participation or withdraw at any time without affecting clinical care; (iii) detailed description of data anonymization and privacy protection measures; (iv) contact information for an independent pediatrician (unaffiliated with the research team) for consultation regarding child participants’ rights.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shi, T., Shi, W., Tian, J. et al. Altered expression of the CD26/ADA axis in immune-mediated inflammation of infectious mononucleosis. Sci Rep 16, 9316 (2026). https://doi.org/10.1038/s41598-026-38891-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-38891-z