Abstract
Salinity stress is a major abiotic factor limiting crop productivity. This study investigated the efficacy of nano-silicon (NSi) and conventional silicon (Si) in mitigating salinity effects on cluster bean. Plants subjected to salinity stress (0, 6, and 12 dS m− 1) were treated with foliar applications of NSi or Si (50, 100, 200 mg L− 1). While both forms of silicon ameliorated salt-induced damage, NSi was significantly more effective. The 50 mg L− 1 NSi treatment emerged as the optimal concentration, markedly enhancing antioxidant enzyme activities, reducing oxidative stress markers (MDA and H2O2), and restoring ion homeostasis by decreasing Na+ and increasing K+ accumulation. These physiological improvements translated into agronomic benefits, with a 2.8-fold increase in seed weight under high salinity. Crucially, NSi at 50 mg L− 1 superiorly improved seed oil quality by significantly increasing the proportion of nutritionally valuable unsaturated fatty acids (C18:1, C18:2), a change strongly correlated with enhanced antioxidant capacity. The results demonstrate that NSi, particularly at 50 mg L− 1, outperforms conventional Si by more effectively bolstering the plant’s antioxidant system and ion regulation, leading to significantly higher yield and better seed quality under salt stress. This positions NSi as a potent nano-biostimulant for sustainable crop production in saline conditions.
Similar content being viewed by others
Introduction
Cluster bean (Cyamopsis tetragonoloba L.) is an important leguminous crop known for its seeds rich in galactomannan having vast industrial applications, but at the same time suffers from salinity stress. Reductions in yield due to moderate and high salinity (depending on cultivar and growing conditions) have been previously reported as 23–29% [1]. Even though cluster bean is known for its tolerance to drought and the capability of growing in arid areas, both its yield potential and efficient production are greatly reduced under salinity conditions indicating that there is a need to developing salt-tolerant varieties and construct sustainable cropping systems2.
Salinity stress initiates a convoluted suite of physiological and metabolic disruptions involving osmotic stress, oxidative damage, and ionic imbalance3,4. While associated oxidative stress activates overproduction of reactive oxygen species (ROS), excessive accumulation of sodium (Na+) in plant tissues disrupts cellular balance. As indicated by changes to chlorophyll fluorescence characteristics, these ROS further cause membrane lipid peroxidation, loss of photosynthetic pigments, and negation of photosynthetic efficiency. In addition, being a constituent of plant defense response, salinity stress injures K+/Na+ ratio, a crucial determinant that influences cellular metabolic function, and activates accumulation of defensive secondary metabolites involving phenolics, flavonoids, and anthocyanins5,6,7. Salinity stress induces a complex array of physiological, biochemical, and growth-related disturbances in plants, including cluster bean, through osmotic stress, oxidative damage, and ionic imbalance3,4. Excessive sodium (Na+) accumulation in plant tissues disrupts ion homeostasis, particularly the K+/Na+ ratio, which is a critical determinant of enzymatic activity and cellular metabolism. Concurrently, salinity triggers oxidative stress, leading to overproduction of reactive oxygen species (ROS) that cause membrane lipid peroxidation, degradation of photosynthetic pigments, impaired chlorophyll fluorescence, and a decline in photosynthetic efficiency. These biochemical perturbations are often accompanied by reduced growth, decreased biomass, and lower seed and yield. Moreover, salinity stress activates the accumulation of defensive secondary metabolites, such as phenolics, flavonoids, and anthocyanins, as part of the plant’s adaptive response5,6,7. In cluster bean, these physiological and biochemical disruptions compromise both vegetative growth and reproductive performance, highlighting the importance of developing strategies, such as silicon and nanosilicon supplementation, to mitigate salinity-induced damage and preserve crop productivity.
In this context, silicon (Si) and nanosilicon (NSi) have emerged as highly promising agents, and numerous studies in different plant species have shown that Si alleviates salt stress for several major reasons2,8,9. It improves ionic homeostasis under stress conditions by decreasing the uptake of Na⁺ into roots cells and increasing the accumulation of potassium (K⁺), therefore preserving cellular processes10,11,12. In addition, Si enhances the activity of superoxide dismutase (SOD), catalase (CAT) and peroxidase (POD) as well as non-enzyme antioxidants such as phenolics and flavonoids in resistance to plant’s antioxidant defense system13. This coordinated regulation serves to stabilize photosynthesis and reduce oxidative damage in response to stress. The effectiveness of silicon is also enhanced in its nano-form. Nanoscale dimensions of nanosilicon contribute to an enhanced bioavailability and stress reduction derived from the better uptake and translocation to the plant tissues. Studies of various species reveal that NSi induce the synthesis of defensive secondary metabolites more efficiently, decrease lipid peroxidation as well as ROS scavenging and nutrient uptake in addition to growth and yield under salinity14,15.
Despite promising findings on silicon and nanosilicon, a significant knowledge gap remains regarding the precise mechanisms and regulatory networks through which they confer salt tolerance in cluster bean (Cyamopsis tetragonoloba L.). We hypothesize that the application of silicon and nanosilicon enhances salinity tolerance in cluster bean by maintaining ion homeostasis, protecting photosynthetic machinery, reducing oxidative stress, and promoting the accumulation of beneficial secondary metabolites. Therefore, this study aimed to investigate the effects of silicon and nanosilicon on the physiological and biochemical responses of cluster bean plants under salinity stress. Specifically, we assessed photosynthetic pigments, chlorophyll fluorescence, ion balance, oxidative stress markers (ROS and MDA), antioxidant potential, secondary metabolite yield, and seed oil quality. This work provides innovative insights into the mechanisms by which nanosilicon mitigates salinity stress and underscores its potential as an eco-friendly strategy for managing soil salinity.
Materials and methods
Plant material
Uniform and healthy cluster bean seeds (cv. BR-2017) were obtained from Ardakan University, Iran. This early-maturing genotype, released in 2017, was selected for its high gum and protein content, short-term vegetative growth with high yield potential, and resistance to common pests and diseases, making it suitable for controlled experimental studies and for evaluating responses to salinity stress.
Determination of soil physico-chemical properties
The physical and chemical properties of the soil were characterized through a series of standardized analyses. Soil pH, electrical conductivity and organic matter were quantified according to the method of Huang et al.16. Particle size distribution (soil texture) was determined using the protocols established by Ryżak and Bieganowski17 and Hardie et al.18. Furthermore, the concentrations of major macro-elements were analyzed following procedures described by Huang et al.16 and Ojewumi et al.19.
Soil samples were collected from a depth of 0–30 cm for analysis. The soil was classified as a clay loam with the following properties: pH of 8.0, electrical conductivity (EC) of 0.59 dS m− 1, organic matter content of 0.85%, and available cation concentrations of 6 meq L− 1 calcium (Ca), 10.5 meq L− 1 potassium (K), 3.3 meq L− 1 magnesium (Mg), and 3.4 meq L− 1 sodium (Na).
Selection of salinity levels
Based on the preliminary experiment results20 and considering the salinity tolerance threshold of cluster bean, three salinity levels (0, 6, and 12 dS m− 1) were selected and induced using sodium chloride (NaCl) as the salt source. The required salinity concentrations were obtained by dissolving 3.84 g L− 1 and 7.68 g L− 1 of NaCl in distilled water to obtain the 6 and 12 dS m− 1 treatments, respectively, while the control treatment (0 dS m− 1) was maintained using distilled water only.
Silicon oxide nanoparticles (SiO2-NPs) and silicon (Si) preparation
Silicon dioxide nanopowder (SiO2-NPs; NSi) (US3438) was purchased from US Research Nanomaterials, Inc., USA. The powder had a purity of 99.5%, an average particle size (APS) of 20–30 nm, a bulk density of < 0.10 g cm− 3, and a molecular weight of 60.08 g mol− 1 (CAS No.: 7631-86-9) (Table 1). To prepare the treatment solutions, the necessary amount of this nanopowder was first dissolved in 5 mL of ethanol to create a stock solution, which was then diluted with deionized water in a volumetric flask. This stock solution was subsequently diluted to yield a working concentration of 50, 100 and 200 mg L− 1 SiO2-NPs.
The target silicon (Si) concentrations were achieved by dissolving sodium metasilicate (Na2SiO3; Sigma-Aldrich, Burlington, MA, USA) in distilled water at rates of 50, 100, and 200 mg L− 1. A control treatment was maintained using distilled water only.
Characterization of SiO2-NPs
The structural properties and particle size of the NSi (SiO2-NPs) were characterized using X-ray diffraction (XRD) and transmission electron microscopy (TEM). XRD analysis was conducted at room temperature using a Siemens D8 Advance spectrometer, with patterns recorded in the 2θ range of 10° to 70°. Phase identification was further confirmed using a Panalytical XPert-PRO diffractometer. Particle size and morphology were examined using a Philips CM200 EFG transmission electron microscope (FEI Company, Eindhoven, Netherlands). The chemical functional groups in both the raw materials and the synthesized nanoparticles were characterized by Fourier transform infrared (FTIR) spectroscopy. Spectra were acquired in the range of 500 to 3500 cm⁻¹ using a Bruker TENSOR II spectrometer (Bruker, Ettlingen, Germany).
TEM imaging of the nanoparticles in dry powder form was performed using a Philips CM200 EFG instrument (FEI Company, Eindhoven, Netherlands), confirming that the particles were smaller than 100 nm (Fig. 1a). The XRD patterns of the NSi (SiO2-NPs) are presented in Fig. 1b. The pattern confirms an amorphous structure, with a broad hump typically observed in the 2θ range of 10–20°, rather than sharp peaks. Analysis of the FTIR spectrum of the NSi (SiO2-NPs) composite revealed the presence of key functional groups associated with its components and their interactions. (Fig. 1c).
Characterization of NSi (SiO2-NPs). (a) TEM image, (b) XRD pattern and (c) FTIR peaks of NSi (SiO2-NPs).
Experimental design and treatments
The experiment was carried out at the School of Agriculture, Shiraz University, located at an elevation of 1810 m above sea level. A factorial arrangement based on a completely randomized design with three replications was used to evaluate the effects Si and NSi on cluster bean plants. Cluster bean seeds were surface-sterilized in a 3% NaClO solution for three minutes and then thoroughly rinsed with deionized water. Seven seeds were sown on June 26, 2024, in pots that were 20 cm in height and 18 cm in diameter. Each pot was filled with 4.5 kg of a 1:1 soil-to-cocopeat mixture (on a dry weight basis). At approximately 7 DAS (V0 Zadok’s scale), each pot was thinned to three uniform and healthy plants, resulting in a final plant density of three plants per pot. Plants were grown under controlled conditions with a temperature of 29 ± 2 °C, 80% relative humidity, and a 14-h photoperiod with a photosynthetically active radiation (PAR) intensity of 800 µmol m− 2 s− 1. The pots were arranged in a completely randomized design and were rotated periodically (every three days) to minimize the effect of any potential environmental gradients. In total, 21 treatments and 63 pots were used in the experiment. Seedlings were irrigated with fresh water during the initial growth period. Foliar applications of silicon (Si, as sodium metasilicate) and nano-silicon (NSi, as silicon dioxide nanopowder) were conducted twice. The treatment including the specific concentrations (50, 100, and 200 mg L− 1) and application timings was selected based on a preliminary dose-response optimization study that identified this protocol as the most effective for enhancing salt stress tolerance in cluster bean. The first foliar application was performed at 24 days after sowing (V4 Zadok’s scale), followed by a second foliar application 22 days later (at 46 DAS; V13 Zadok’s scale). An application rate of 10 ml per plant was used for all treatments. No additional surfactant was added to the spray solution, as the suspension itself demonstrated satisfactory wetting properties. Control plants were sprayed with an equivalent volume of distilled water. Salinity stress was initiated 31 DAS (V6 Zadok’s scale), which was after the first foliar application had been completed. Saline irrigation was applied based on the plants’ water requirements until harvest. Pots were irrigated with sufficient water to maintain moisture near field capacity, allowing limited leaching (≈ 10–20% of total water applied) to prevent salt accumulation in the root zone. The electrical conductivity of the leachate (ECe) was systematically monitored after each irrigation, and the salinity of irrigation water was adjusted accordingly: if ECe exceeded the target level, irrigation salinity was reduced, and if ECe was below the target, salinity was increased. This approach ensured that the average salinity in the root zone remained at the intended treatment levels throughout the study. The pot capacity had been predetermined prior to the experiment by saturating pots with water, allowing them to drain, and then weighing the soil before and after oven-drying at 105 °C until a constant weight was achieved. The weight difference was used to calculate the water required to reach pot capacity.
Sampling dates
Plant sampling for the evaluation of all traits was carried out at 60 DAS (V19 Zadok’s scale). One plant was randomly selected from each pot (experimental replicate). Since each treatment had three pots (replications), this resulted in three independent biological replicates (n = 3) per treatment for all biochemical assays. Each plant sample was processed and analyzed individually; no pooling of samples was performed. Immediately after collection, samples were flash-frozen in liquid nitrogen and subsequently stored at -80 °C for preservation. Upon completion of the stress period, pods and grains from individual plants were separately collected for subsequent evaluation of yield-related parameters (135 DAS; R7 Zadok’s scale).
Determination of chlorophyll fluorescence parameters
Chlorophyll fluorescence parameters (F0 and Fm) were measured in fully expanded young leaves 10 days after salinity imposition, between 9:00 and 11:00 a.m., using dark-adapted leaves (30 min) and an OS-30p Chlorophyll Fluorometer (OptiSciences, Hudson, NH, USA), following the method of Alinia et al.9. The maximum quantum efficiency of photosystem II (Fv/Fm) was then calculated as (Fm – F0)/Fm.
Content of photosynthetic pigments
The contents of photosynthetic pigments were quantified from 500 mg of fresh third-leaf tissue. After an overnight dark incubation, pigments were extracted from the plant material using 80% acetone, following the protocol established by Lichtenthaler and Wellburn21. The homogenized mixture was centrifuged at 3000 rpm for 10 min to separate the extract. The supernatant was then analyzed using a UV-Vis spectrophotometer (7315 UV/visible Spectrophotometer, Jenway, UK). Absorbance was measured at 470 nm, 646 nm, and 663 nm for carotenoids, chlorophyll b (Cb), chlorophyll a (Ca) and chlorophyll t, respectively.
Determination of antioxidant enzymes activity
The activities of peroxidase (POD), superoxide dismutase (SOD), catalase (CAT) and ascorbate peroxidase (APX) were analyzed. For each enzyme, 0.5 g of cluster bean leaf tissue was ground to a fine powder under liquid nitrogen. The powder was homogenized in 10 ml of ice-cold phosphate buffer (50 mM, pH 7.0). The homogenate was then centrifuged at 15,000 × g for 20 min at 4 °C. The resulting supernatant was collected and used as the crude enzyme extract for subsequent spectrophotometric analyses. Enzyme activity assays were performed at 25 °C using a UV-Vis spectrophotometer (7315 UV/visible Spectrophotometer, Jenway, UK), following the methodology outlined by Sairam et al.22. The specific activity for each enzyme was determined based on the change in absorbance per minute under defined assay conditions.
Peroxidase (POD) activity
POD activity was determined based on the oxidation of pyrogallol, following the procedure of Chance and Maehly23. The assay was performed by mixing 3 mL of pyrogallol phosphate buffer solution with 0.5 mL of 1% H2O2 and 0.1 mL of enzyme extract in a cuvette. The increase in absorbance at 420 nm was recorded at 20-second intervals for 3 min using a spectrophotometer. A control reaction, prepared by replacing the enzyme extract with buffer, was subtracted from the sample readings.
Superoxide dismutase (SOD) activity
The Beauchamp and Fridovich24 approach was used to determine SOD activity by measuring its capacity to prevent the photochemical reduction of nitroblue tetrazolium (NBT). The 3 mL reaction mixture was made up of 1 mL of 50 mM phosphate buffer (pH 7.8), 0.5 mL of 13 mM methionine, 0.5 mL of 75 µM NBT, 0.1 mL of the enzyme extract, 0.5 mL of 2 µM riboflavin, and 0.5 mL of 0.1 mM EDTA. The response was started in one set of tubes by lighting it under a 15 W fluorescent lamp for 10 min, with an unlit combination serving as the blank. The reaction was stopped by taking away the light source, and the absorbance was measured at 560 nm. Under the assay conditions, one unit of SOD activity was defined as the quantity of enzyme necessary to produce 50% inhibition of NBT reduction.
Catalase (CAT) activity assay
CAT activity was determined by monitoring the decomposition of hydrogen peroxide (H2O2), measured as a decrease in absorbance at 240 nm25. The assay mixture contained 2.0 ml of 0.1 M phosphate buffer (pH 6.5) and 100 µl of 269 mM H2O2. The reaction was initiated by adding 50 µl of the enzyme extract to the mixture. The change in absorbance at 240 nm was recorded using a spectrophotometer.
Ascorbate peroxidase (APX) activity
APX activity was assayed according to the method of Yoshimura et al.26. The reaction mixture consisted of 25 mM phosphate buffer (pH 7.0), 0.25 mM ascorbic acid, 0.1 mM EDTA, and 1 mM hydrogen peroxide. The reaction was initiated by adding 0.2 ml of the enzyme extract. The decrease in absorbance at 290 nm, corresponding to the oxidation of ascorbate, was monitored spectrophotometrically for 1 min. Enzyme activity was calculated based on the rate of absorbance change.
Anthocyanin
To assay anthocyanins, as much as 20 mg of fresh leaves was homogenized with 0.5 mL of methanol/HCl (99/1, v/v) at the 4 °C temperature for a 24 h period and then subjected to centrifuging at 6000 rpm for 10 min. Absorbance related to the considered supernatant was subjected to measurement at 530 as well as 657 nm. Anthocyanin’s amount was then subjected to the related calculation by employing these formulas27.
FW: fresh weight (g); E: extract (mL); ε: extinction coefficient (L.mol− 1.cm− 1).
The anthocyanin concentration was determined using an extinction coefficient of 26,900 L.mol− 1.cm− 1 for cyanidin-3-glucoside and expressed as micrograms per gram fresh weight (µg g− 1 FW).
Total phenol content (TPC)
TPC was quantified according to the method of Narayan et al.28. Fresh leaf tissue (1 g) was homogenized in 5 mL of an ice-cold extraction solvent (methanol: acetone: HCl, 90:8:2 v/v) and centrifuged at 4000 × g for 20 min. For the assay, 0.1 mL of the supernatant was reacted with 0.2 mL of Folin-Ciocalteu reagent. After 3–5 min, 0.8 mL of sodium carbonate solution (700 mM) was added. The mixture was vortexed and allowed to stand for 60 min before measuring the absorbance at 765 nm. TPC was calculated from a gallic acid standard curve and expressed as milligrams of gallic acid equivalents per gram of fresh weight (mg GAE g− 1 FW).
Total flavonoid content (TFC)
TFC was determined using the aluminum chloride colorimetric method as described by Fattahi et al.29. Dried leaf powder (1 g) was extracted with 10 mL of methanol overnight on an orbital shaker. The methanolic supernatant was collected and evaporated to dryness. The resulting residue was weighed and reconstituted in methanol to a final concentration of 1 mg mL− 1. For the assay, 100 µL of this extract was mixed with 400 µL of methanol, 100 µL of 10% aluminum chloride, and 100 µL of 1 M sodium acetate. The reaction mixture was vortexed, incubated in the dark for 40 min, and its absorbance was read at 415 nm. TFC was determined using a quercetin standard curve and expressed as milligrams of quercetin equivalents per gram of dry weight (mg QE g− 1 DW).
Hydrogen peroxide (H2O2) content
The hydrogen peroxide (H2O2) content was quantified using a protocol adapted from Islam et al. Briefly, 0.5 g of leaf tissue was homogenized in a 0.1% trichloroacetic acid (TCA) solution and centrifuged at 12,000 × g for 20 min. The supernatant was then used to prepare a reaction mixture for the spectrophotometric (7315 UV/visible Spectrophotometer, Jenway, UK) assay at 390 nm30.
Malondialdehyde (MDA) assay
To evaluate lipid peroxidation, the thiobarbituric acid (TBA) method was employed to determine the content of MDA, based on the work of Heath and Packer31. Briefly, 0.5 g of plant material was homogenized in 10 ml of 0.1% (w/v) trichloroacetic acid (TCA). The resulting homogenate was centrifuged at 15,000 × g for 10 min and filtered to obtain a clear supernatant. For the reaction, 2 ml of the supernatant was mixed with 2 ml of 0.67% TBA prepared in 20% TCA. The mixture was heated in a water bath at 95–100 °C for 30 min, then rapidly cooled in an ice bath for 5 min before equilibrating to room temperature. The absorbance of the solution was measured at 532, 600 and 450 nm using a UV-Vis spectrophotometer (7315 UV/visible Spectrophotometer, Jenway, UK). The MDA concentration was calculated based on the absorbance difference at these wavelengths.
Leaf nutrient concentrations and K+/Na+ ratio
Oven-dried shoot samples were ground and then ashed in a muffle furnace at 450 °C for 4 h. The resulting ash was dissolved in 1 N HCl. Potassium (K+) and sodium (Na+) concentrations were determined by flame photometry. Calibration was performed using standard solutions of KCl and NaCl, with concentrations ranging from 5 to 250 mg L− 1. The K+/Na+ ratio was then calculated from the measured ion concentrations.
Fatty acid composition
Fatty acid composition was analyzed in seeds at the full-ripening stage (90 days after sowing; R7 Zadok’s scale). Following the method of Golmakani et al.32, powdered seeds (500 mg) were esterified in 10 ml of methanol: acetyl chloride (95:5, v/v) at 80 °C for 1 h. After cooling, 5 mL of deionized water was added, and the mixture was vortexed. Subsequently, 2 mL of hexane was added, and the sample was centrifuged at 5,000 rpm for 5 min. The hexane layer was analyzed by gas chromatography (SP-3420 A, Beijing Beifen-Ruili Analytical Instrument Group, China) equipped with a BPX70 capillary column (120 m × 0.25 mm × 0.25 μm). The oven temperature was set to 90 °C, with injector and detector temperatures at 250 and 300 °C, respectively, using hydrogen as the carrier gas. Fatty acids were identified by comparing peak retention times to those in the Wily 7n and NIST05a standard libraries.
Statistical analysis
Data were subjected to analysis of variance (ANOVA) using SAS software (v. 9.1). Normality was verified through Shapiro-Wilk tests, supported by skewness and kurtosis evaluation. Treatment means were compared using the Least Significant Difference (LSD) test at 5% and 1% probability levels. Graphical representations were generated in Microsoft Excel. A heatmap of Pearson correlations, radar plot, stack graph and principal component analysis (PCA) biplot were constructed using OriginPro software (version 2022).
Results
Maximum quantum efficiency of photosystem II (Fv/Fm) and photosynthetic pigments
Based on the analysis of variance results, the interaction effect of the biostimulants and salinity stress on Fv/Fm, total chlorophyll, and carotenoid content was significant. With an increase in salinity from 0 to 12 dS m− 1, all three parameters showed a decreasing trend across all treatments. The highest Fv/Fm value (0.729) was obtained at a concentration of 50 mg L− 1 NSi under non-stress conditions (Fig. 2a, b and c).
The application of NSi and Si treatments, compared to the control, did not show a significant difference in the Fv/Fm parameter at both salinity levels of 6 and 12 dS m− 1 across all three concentrations. However, the application of NSi at a concentration of 50 mg L− 1 was able to mitigate the negative effects of salt stress on total chlorophyll and carotenoid parameters. Specifically, it increased these parameters by 1.5 and 2.4 times, and 2.4 and 11.3 times, respectively, at salinity levels of 6 and 12 dS m− 1 compared to the control treatment (Fig. 2a, b and c).
Effects of biostimulants and salinity levels on Fv/Fm (a), chlorophyll t (b) and carotenoid (c) of cluster bean. NSi1, NSi2 and NSi3 treatments: foliar applications of 50, 100 and 200 mg L-1 SiO2-NPs; Si1, Si2 and Si3 treatments: foliar applications of 50, 100 and 200 mg L-1 Na2SiO3; C treatment: foliar applications of distilled water, S1, S2 and S3 treatments: 0, 6, and 12 dS m-1 NaCl. Different letters above the columns indicate statistically significant differences at p ≤ 0.05 according to LSD test. * and **: significant at p ≤ 0.05 and p ≤ 0.01, respectively and ns: non-significant. Data shown are means of three replicates per treatment.
Antioxidant enzymes activity
The activity of antioxidant enzymes was significantly influenced by the interaction between the biostimulants and salinity stress. As salinity levels increased from 0 to 6 and 12 dS m− 1, a notable rise in antioxidant enzyme activity was observed across all applied treatments. This increase was particularly remarkable at a concentration of 50 mg L− 1 NSi compared to other treatments. Specifically, the application of this concentration of NSi enhanced the activity of peroxidase, superoxide dismutase, catalase, and ascorbate peroxidase by 48.9 and 62.6%, 76 and 86.7%, 92.7 and 89.1%, and 49.6 and 32%, respectively, at salinity levels of 6 and 12 dS m− 1 compared to the control (Fig. 3a-d).
Effects of biostimulants and salinity levels on antioxidant enzymes activity (POD (a), SOD (b), CAT (c) and APX (d)) of cluster bean. NSi1, NSi2 and NSi3 treatments: foliar applications of 50, 100 and 200 mg L-1 SiO2-NPs; Si1, Si2 and Si3 treatments: foliar applications of 50, 100 and 200 mg L-1 Na2SiO3; C treatment: foliar applications of distilled water; S1, S2 and S3 treatments: 0, 6, and 12 dS m-1 NaCl. Different letters above the columns indicate statistically significant differences at p ≤ 0.05 according to LSD test. * and **: significant at p ≤ 0.05 and p ≤ 0.01, respectively and ns: non-significant. Data shown are means of three replicates per treatment.
Anthocyanin, total phenol and flavonoid contents
The interaction between biological stimulants and salt stress on anthocyanin, phenol, and flavonoid content was significant at the 5% probability level (Fig. 4a-c). Anthocyanin content increased as salinity rose from 0 to 12 dS m− 1. The highest anthocyanin content was observed at the highest salinity level and a concentration of 50 mg L− 1 NSi, which showed no significant difference with other treatments at salinity levels of 6 and 12 dS m− 1 (Fig. 4a).
The highest phenolic content was also obtained at a concentration of 50 mg L− 1 NSi, which was not significantly different from the concentrations of 100 and 200 mg L− 1 NSi or 50 mg L− 1 Si. The application of 50 mg L− 1 NSi increased phenolic content by 55.8%, 10.1%, and 48.9% at salinity levels of 0, 6, and 12 dS m− 1, respectively, compared to the control (Fig. 4b).
Flavonoid content was also highest at a concentration of 50 mg L− 1 NSi and showed no significant difference with other treatments at this salinity level. Compared to the control, the 50 mg L− 1 NSi treatment increased flavonoid content by 16.6%, 13.8%, and 15.7% at salinity levels of 0, 6, and 12 dS m− 1, respectively compared to the control (Fig. 4c).
Effects of biostimulants and salinity levels on anthocyanin (a), total phenol content (b) and total flavonoid content (c) of cluster bean. NSi1, NSi2 and NSi3 treatments: foliar applications of 50, 100 and 200 mg L− 1 SiO2-NPs; Si1, Si2 and Si3 treatments: foliar applications of 50, 100 and 200 mg L− 1 Na2SiO3; C treatment: foliar applications of distilled water; S1, S2 and S3 treatments: 0, 6, and 12 dS m− 1 NaCl. Different letters above the columns indicate statistically significant differences at p ≤ 0.05 according to LSD test. * and **: significant at p ≤ 0.05 and p ≤ 0.01, respectively and ns: non-significant. Data shown are means of three replicates per treatment.
Hydrogen peroxide and malondialdehyde contents
The contents of malondialdehyde and hydrogen peroxide was significantly affected (at the 5% probability level) by the interaction of biostimulants and salinity stress. Salinity increased the amount of malondialdehyde and hydrogen peroxide. The highest malondialdehyde content (91.731 nmol g− 1 FW) was observed in the control treatment under the highest salinity level (12 dS m− 1), which showed no significant difference with the Si treatments at concentrations of 50 and 100 mg L− 1 under the same salinity level. Meanwhile, the highest hydrogen peroxide content (2.25 nmol g− 1 FW) was obtained at 100 mg L− 1 Si under the highest salinity level, which was not significantly different from the 50 mg L− 1 Si treatment and the control at the same salinity level. The application of NSi at 50 mg L− 1 significantly reduced the malondialdehyde and hydrogen peroxide contents at salinity levels of 6 and 12 dS m− 1 by 52.9 and 57.2%, and 59.1 and 51.4%, respectively, compared to the control treatment (Fig. 5a and b).
Effects of biostimulants and salinity levels on MDA (a) and H2O2 (b) of cluster bean. NSi1, NSi2 and NSi3 treatments: foliar applications of 50, 100 and 200 mg L− 1 SiO2-NPs; Si1, Si2 and Si3 treatments: foliar applications of 50, 100 and 200 mg L− 1 Na2SiO3; C treatment: foliar applications of distilled water; S1, S2 and S3 treatments: 0, 6, and 12 dS m− 1 NaCl. Different letters above the columns indicate statistically significant differences at p ≤ 0.05 according to LSD test. * and **: significant at p ≤ 0.05 and p ≤ 0.01, respectively and ns: non-significant. Data shown are means of three replicates per treatment.
Na+ and K+ concentrations and K+/Na+ ratio
The concentration of sodium, potassium, and the potassium to sodium ratio were significantly influenced (at the 1% probability level) by the interaction of biostimulants and salinity stress. As salinity increased, the sodium concentration exhibited an upward trend, whereas the potassium concentration and the potassium to sodium ratio showed a decreasing trend. At the highest salinity level (12 dS m− 1), the control treatment recorded the highest sodium concentration (287.42 mg g− 1 DW), which was not significantly different from the Si treatment at a concentration of 50 mg L− 1 under the same salinity level. The application of NSi at a concentration of 50 mg L− 1 significantly reduced the sodium concentration by 53.3 and 60.5% at salinity levels of 6 and 12 dS m− 1, respectively, compared to the control treatment. Furthermore, at salinity levels of 6 and 12 dS m− 1, the application of 50 mg L− 1 NSi significantly increased the potassium concentration by 87.7 and 100.1%, and elevated the potassium to sodium ratio by 4- and 5.1-fold, respectively, compared to the control (Fig. 6a-c).
Effects of biostimulants and salinity levels on Na+ (a) and K+ (b) concentrations and K+/Na+ ratio (c) of cluster bean. NSi1, NSi2 and NSi3 treatments: foliar applications of 50, 100 and 200 mg L− 1 SiO2-NPs; Si1, Si2 and Si3 treatments: foliar applications of 50, 100 and 200 mg L− 1 Na2SiO3; C treatment: foliar applications of distilled water; S1, S2 and S3 treatments: 0, 6, and 12 dS m− 1 NaCl. Different letters above the columns indicate statistically significant differences at p ≤ 0.05 according to LSD test. * and **: significant at p ≤ 0.05 and p ≤ 0.01, respectively and ns: non-significant. Data shown are means of three replicates per treatment.
Pod number and grain weight
The number of pods and seed weight were significantly affected (at the 5% probability level) by the biostimulants and salinity stress. Increasing salinity levels led to a reduction in both parameters across all biostimulant treatments. The application of NSi treatments resulted in a significant increase in both the number of pods and seed weight compared to conventional Si at all stress levels. The highest number of pods (11) and the greatest seed weight (2.89 g pot− 1) were obtained with the 50 mg L− 1 NSi treatment under non-stress conditions. Regarding seed weight, no significant difference was observed between the 50 mg L− 1 and 100 mg L− 1 NSi concentrations under non-stress conditions. At salinity levels of 6 and 12 dS m− 1, the application of 50 mg L− 1 NSi increased the number of pods by 2.2- and 3.2-fold, and seed weight by 1.3- and 2.8-fold, respectively, compared to the control treatment (Fig. 7a and b).
Effects of biostimulants and salinity levels on pod number (a) and grain weight (b) of cluster bean. NSi1, NSi2 and NSi3 treatments: foliar applications of 50, 100 and 200 mg L− 1 SiO2-NPs; Si1, Si2 and Si3 treatments: foliar applications of 50, 100 and 200 mg L− 1 Na2SiO3; C treatment: foliar applications of distilled water; S1, S2 and S3 treatments: 0, 6, and 12 dS m− 1 NaCl. Different letters above the columns indicate statistically significant differences at p ≤ 0.05 according to LSD test. * and **: significant at p ≤ 0.05 and p ≤ 0.01, respectively and ns: non-significant. Data shown are means of three replicates per treatment.
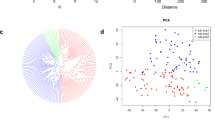
Pearson correlations and principal component analysis
The goal of PCA is to identify the number of major factors that can be extracted to minimize the number of effective parameters. In this study, two elements accounted for 100% of the observed variability. Table 2 shows the proportion variance for the primary components. Two components accounted for 73.42% and 18.42% of the variation, respectively. With loading coefficients greater than 0.234, the principal component 1 (PC1) mostly accounted for Fv/Fm, Cht, Car, APX, Ant, Fla, MDA, Na+, K+, K+/Na+, and GW (Table 2). The primary component 2 (PC2) accounts for POD, SOD, CAT, Ph, H2O2, and PN (loading coefficient > 0.302, Table 2).
The relationships between the variables assessed during various growth stages were shown to be partially comparable by PCA. As a result, a clear distinction between the various NaCl treatments in the three analyzed genotypes was seen. Consequently, the treatments with 0, 50, and 100 mM NaCl were located in the right, middle, and left of the PCA biplot, respectively (Fig. 8b). Although a link between S0NSi1 and S0NSi2 can be seen with PN, K, GW, K/Na, Cht, Car, and Fv/Fm, as illustrated in Fig. 8b, CAT, The physiological characteristics of CAT, POD, SOD, APX, Fla, Ph, Ant, Na, MDA, and H2O2 were linked to S2NSi2, S2NSi3, S2Si3, S2Si2, and S2Si1. The chemicals SOD, APX, Fla, Ph, Ant, Na, MDA, and H2O2 were grouped together (in the upper and bottom right quadrant), indicating a similarity in the way they are controlled. under enforced stress circumstances for all treatments. As mentioned earlier, the biplots (Fig. 8b) and PCA results (Table 2) demonstrated a negative correlation between axis 1 PC1 and Cht, Car, and Fv/Fm. K+, K+/Na+, PN, and GW. In reality, salinity causes these parameters to decrease in the treatments examined, which may be useful as a measure of stress tolerance. Put another way, choosing therapies based on their negative coordinates along PC1 might lead to therapies with higher Cht, Car, Fv/Fm, K+, K+/Na+, and PN. and GW in saline stress compared to the control group.
A heat map of the Pearson correlations (a) and an analysis PCA-Biplot (b) of the principal component analysis for the linked biochemical responses related to biostimulants treated cluster bean seedlings subjected to salinity stress. Fv/Fm: Maximum quantum efficiency of photosystem II, Cht: Total chlorophyll, Car: Carotenoid, CAT: Catalase, POD: Peroxidase, SOD: Superoxide dismutase, APX: Ascorbate peroxidase, Ant: Anthocyanin, Ph: Total phenol content, Fla: Total flavonoid content, MDA: Malondialdehyde, H2O2: Hydrogen peroxide, Na: Sodium, K: Potassium, K/Na: K+/Na+ ratio, PN: Pod number, and GW: Grain weight .
Fatty acid composition
Based on the peaks obtained from the GC-MS, the fatty acid compositions of the samples under different treatments were identified (Figs. 9, 10 and 11) and are presented in Table 3. The results indicated that the fatty acid compositions were influenced by the various treatments of biological stimulants and salinity (Fig. 12). Only C18:1 and C18:2 compounds, which are unsaturated fatty acids, played a significant role in improving the quality of the oil obtained from cluster bean. These compounds were affected by the application of NSi, with the highest levels observed under different salinity conditions resulting from various concentrations of NSi (Fig. 12a). Conversely, the lowest levels of saturated fatty acids were also obtained from the NSi treatments (Fig. 12a). Based on the radar plot results, it was found that the application of NSi treatments contributed the most to the improvement of fatty acid compositions (Fig. 12b). Additionally, correlation results revealed a significant positive relationship between unsaturated fatty acid compounds and the activity of antioxidant enzymes (Fig. 12c). see Table 3.
GC-MS profile of cluster bean extract treated with biostimulants under salinity stress. The peak assignments are listed in Table 1.
GC-MS profile of cluster bean extract treated with biostimulants under salinity stress. The peak assignments are listed in Table 1.
GC-MS profile of cluster bean extract under salinity stress. The peak assignments are listed in Table 1.
Effects of biostimulants and salinity levels on fatty acid composition of cluster bean (a). NSi1, NSi2 and NSi3 treatments: foliar applications of 50, 100 and 200 mg L− 1 SiO2-NPs; Si1, Si2 and Si3 treatments: foliar applications of 50, 100 and 200 mg L− 1 Na2SiO3; C treatment: foliar applications of distilled water; S1, S2 and S3 treatments: 0, 6, and 12 dS m− 1 NaCl. Different letters above the columns indicate statistically significant differences at p ≤ 0.05 according to LSD test. * and **: significant at p ≤ 0.05 and p ≤ 0.01, respectively and ns: non-significant. Data shown are means of three replicates per treatment. A radar plot (b) and a heat map of the Pearson correlations (c) for the linked biochemical responses related to biostimulants treated cluster bean seedlings subjected to salinity stress.
Discussion
The present study demonstrates that the application of NSi, particularly at a concentration of 50 mg L− 1, effectively mitigates the detrimental effects of salinity stress on cluster bean plants (Cyamopsis tetragonoloba L.). Significant improvements were observed across a broad range of physiological, biochemical, and yield parameters, underscoring the potential of NSi as a superior biostimulant compared to conventional Si under saline conditions. The initial characterization confirmed the successful synthesis of amorphous SiO2 nanoparticles with sizes below 100 nm. This nano-size is critical, as it potentially facilitates easier uptake and translocation within plant tissues compared to bulk silicon, thereby contributing to more efficient alleviation of abiotic stress (Fig. 1).
Salinity stress is known to affect photosynthetic productivity due to disruption in biochemical and biological activities that are fundamental to photosynthesis. This can be explained mechanistically by a dose-dependent decline in the Fv/Fm ratio (indicating maximum quantum yield of photosystem II is decreasing) and a concomitant dip in the overall chlorophyll and carotenoid content2,14. The inhibition of specific pathways involved in chlorophyll biosynthesis, along with the degradation of pigment protein complexes in the thylakoid membranes, are well-known toxic effects. These processes are often triggered by ionically and osmotically hypertonic conditions in the cytosol. Similar results were reported by Alinia et al.2 who found that salinity stress reduced photosynthetic pigments by enhancing chlorophyll degradation in Cyamopsis tetragonoloba L. seedlings. However, they also demonstrated that applying silicon at an appropriate concentration alleviated these detrimental effects. Salt stress photochemical efficiency (Fv/Fm parameter) is standardized to the control which are non-saline and photoinhibition is moderated under saline conditions. For instance, while supplementation of NSi at lower concentrations such as 1 and 10 mg L− 1 doesn’t have a significant effect on the Fv/Fm parameter, it is a strong mitigator of photodegradation of pigments. Salt induced pigment loss where 50 mg L− 1 NSi is shown to substantially add to the overall and carotenoid concentrations that are stored at elevated salinity conditions (Fig. 2). This is to say that under conditions that are osmotically stressful, NSi is well known to maintain the breakdown of the photosynthetic apparatus by photoinhibition counteracting the loss in photosynthetic productivity which is then subsequently transformed to cellular energy33,34. Monv et al.33 and Morshedloo et al.34 highlighted this positive effect of silicon and nano-silicon on the photosynthetic productivity.
To counter the dominant oxidative tension caused by salinity, plants use a complex system of enzymatic antioxidants. Our results revealed a consistent increase in the activity of key ROS-scavenging enzymes, SOD, CAT, POD, and APX, with increasing salinity levels2,10. This increase in activity represents the fundamental biochemical response to the excessive production of cytotoxic ROS. Notably, 50 mg L− 1 NSi treatment acted as biostimulant agents and caused super-induction of these enzymatic activities, which exceeded the control as well as conventional silicon plant treated responses (Fig. 3). This implicates a role of NSi in pre-activating or sensitizing the pathways of transcription and/or post-translation control of the antioxidant system. By strengthening the intrinsic antioxidant defense system, NSi markedly enhances ROS detoxification capacity. This primed defense system is evidenced as the sharp decrease in oxidative stress parameters: MDA, the dominant phenom of lipid peroxidation, as well as H2O2. The decline in these oxidative damage indicators provides clear evidence that the NSi-induced enhancement of antioxidant enzyme activities effectively mitigates membrane and cellular injury under salinity stress35,36,37.
Additionally, NSi use caused a gathering of secondary metabolites including anthocyanins, phenols, and flavonoids. Apart from being non-enzymatic antioxidants, these compounds also have important roles in membrane stabilization and signaling38,39. The method underlying this accumulation is likely complex. Silicon nanoparticles could serve as elicitors, stimulating defense-related gene expression in the phenylpropanoid pathway by upregulating essential enzymes such Phenylalanine Ammonia-Lyase (PAL) Chalcone Synthase (CHS) also supports the production of phenolic chemicals, flavonoids, and anthocyanins by means of this increased enzymatic activity40,41,42. By storing in cell walls, NSi also reinforces the plant’s physical barrier, hence lowering sodium (Na+) intake and relieving ionic stress. Rather than being used for damage repair, this stored cellular energy can then be diverted toward the manufacture of these protective metabolites. The highest concentrations of these metabolites at 50 mg L− 1 NSi, even under extreme salinity, further demonstrate how NSi can activate many, synergistic Pathways of defense in cluster bean plants include physiological (ionic homeostasis) as well as biochemical (antioxidant production).
The disturbance of ion homeostasis especially the accumulation of toxic Na+ ions and the accompanied by a reduction in vital K+ ions, leads to a decreased the K+/Na+ ratio. Our data clearly show that 50 mg L− 1 NSi was quite successful at managing this imbalance. It considerably lowered sodium absorption and enhanced potassium retention, therefore greatly improving the K+/Na+ ratio. This points to NSi as being possibly important for controlling the expression and activity of critical ion transporter genes. Specifically, silicon can raise the expression of genes coding high-affinity potassium transporters (HKTs), especially those in the HKT1 family, which are known to preferentially unload Na+ from the xylem sap, thereby restricting its movement to the shoots43,44,45.
In addition, NSi may stimulate the activity of plasma membrane and vacuolar H⁺-ATPases and H⁺-PPases, generating the proton motive force required to drive SOS1 (Salt Overly Sensitive 1) Na⁺ efflux transporters that extrude Na⁺ from the cytoplasm into the apoplast46. For potassium homeostasis, silicon likely promotes the activation of K⁺ channels (e.g., AKT1) and transporters that facilitate K⁺ uptake and sequestration into vacuoles, preventing its leakage under salt stress47. Moreover, the deposition of silica in the apoplast and its interaction with cell wall components enhance membrane integrity and selectivity, forming a physical barrier that reduces the apoplastic flow of Na+ and maintains a more negative membrane potential, which is crucial for K+ uptake and Na+ exclusion. As suggested for other crops, this multifaceted mechanism efficiently excludes Na+ and promotes K+ absorption, thereby restoring the critical K+/Na+ balance required for enzyme activity and cellular homeostasis.
The application of NSi mitigates salinity stress through a synergistic combination of key physiological mechanisms. Primarily, it enhances photosynthetic efficiency by protecting chloroplasts and improving stomatal conductance, resulting in optimal CO2 capture in the stomata, which drives growth6,14,33. Simultaneously, NSi strengthens the plant’s antioxidant defenses and minimizes the oxidative damage caused by reactive oxygen species (ROS) to membranes, proteins, and DNA36,42. It is also important in ion balance because it suppresses the influx of toxic sodium ions and facilitates the accumulation of beneficial potassium ions36,43,45. By reducing the metabolic stress of defense and repair, NSi supports more favorable mobilization of energy and resources towards reproduction rather than survival. This is the key cause of the recovery in pod number and seed weight which explains the enhanced yield during salinity stress.
Improved oxidative state of the plant directly results from the enhanced seed oil quality under NSi treatment. The important process is the strong activation of the antioxidant enzyme system (e.g., superoxide dismutase, catalase, peroxidase) by NSi15,35,37. This increases antioxidant capacity creates developing seeds have a protected, low-stress cellular environment that is very important for the enzymes that make lipids work well and stay healthy. Particularly, the main desaturase enzymes, including convert saturated fatty acids (SFAs) into unsaturated ones (UFAs) by double Highly sensitive to oxidative damage are bonds to the carbon chain. The NSi enhanced antioxidant system protects these fragile bonds by successfully scavenging reactive oxygen species (ROS). It also activates enzymes that allow the desaturation process to proceed more rapidly. This causes a metabolic change towards the manufacture of a greater ratio of oleic (C18:1) and other economically and nutritionally valuable unsaturated fatty acids. While lowering the ratio of less desirable saturated fats, linoleic (C18:2) acid also works. This cause-effect link is strongly supported by the clear positive correlation between antioxidant activity and UFA content, which means NSi is not only a yield-enhancing agent but also a critical modulator of seed quality and biochemical composition38,48.
The Principal Component Analysis (PCA) effectively consolidated these findings, showing that the majority of the positive traits (photosynthetic pigments, antioxidant enzymes, yield, and ion homeostasis) were strongly associated with the NSi treatments and loaded heavily on PC1.
Conclusion
This study demonstrates that nanosilicon (NSi), particularly at 50 mg L− 1, is more effective than bulk silicon in alleviating salinity stress in cluster bean. Its superior performance is attributed to enhanced bioavailability and a broader defensive action due to its high surface area to volume ratio. NSi application improved physiological traits (chlorophyll content, chlorophyll fluorescence, anthocyanin), enhanced biochemical responses (increased antioxidants, phenolics, and flavonoids, along with reduced oxidative stress markers), and restored ionic balance (lower Na+ and higher K+), collectively contributing to maintained yield and improved seed oil quality. While these results highlight the potential of NSi as a nano enabled strategy for sustainable crop production in salt-affected soils, the study is limited by its use of a single cultivar and controlled experimental conditions. Future research should explore different genotypes, field trials, and underlying molecular mechanisms to fully elucidate NSi mediated salinity tolerance.
Data availability
Data is provided within the manuscript or supplementary information files.
References
Jukanti, A. K., Bhatt, R., Sharma, R. & Kalia, R. K. Morphological, agronomic, and yield characterization of cluster bean (Cyamopsis Tetragonoloba L.) germplasm accessions. J. Crop Sci. Biotechnol. 18 (2), 83–88. https://doi.org/10.1007/s12892-014-0092-3 (2015).
Alinia, M., Kazemeini, S. A., Meftahizadeh, H. & Mastinu, A. Alleviating salinity stress in Cyamopsis Tetragonoloba L. seedlings through foliar application of silicon or melatonin in arid and semi-desert environments. S Afr. J. Bot. 174, 347–359. https://doi.org/10.1016/j.sajb.2024.09.024 (2024).
Alinia, M., Kazemeini, S. A., Sabbaghi, S., Sayahi, S. & Abolghasemi, A. Asgari Lajayer, B. The effect of silicon–melatonin nanoparticles on improving germination parameters and reducing salinity toxicity by maintaining ion homeostasis in Cyamopsis Tetragonoloba L. seedlings. Agronomy 15 (2), 427. https://doi.org/10.3390/agronomy15020427 (2025).
Soni, S., Jha, A. B., Dubey, R. S. & Sharma, P. Nanowonders in agriculture: unveiling the potential of nanoparticles to boost crop resilience to salinity stress. Sci. Total Environ. 925, 171433. https://doi.org/10.1016/j.scitotenv.2024.171433 (2024).
Javed, S. A. et al. Optimization of nitrogen regulates the ionic homeostasis, potassium efficiency, and proline content to improve the growth, yield, and quality of maize under salinity stress. Environ. Exp. Bot. 226, 105836. https://doi.org/10.1016/j.envexpbot.2024.105836 (2024).
Kamal, M. Z. U. et al. Manure-biochar compost mitigates the soil salinity stress in tomato plants by modulating the osmoregulatory mechanism, photosynthetic pigments, and ionic homeostasis. Sci. Rep. 14 (1), 21929. https://doi.org/10.1038/s41598-024-73093-5 (2024).
Singh, A. et al. Zinc oxide nanoparticles influence on plant tolerance to salinity stress: insights into physiological, biochemical, and molecular responses. Environ. Geochem. Health. 46 (5), 148. https://doi.org/10.1007/s10653-024-01921-8 (2024).
Xiong, J. et al. Mitigation effect of exogenous nano-silicon on salt stress damage of rice seedlings. Int. J. Mol. Sci. 26 (1), 85. https://doi.org/10.3390/ijms26010085 (2024).
Naz, T., Iqbal, M. M., Ullah, I., Mubeen, M. A. & Ditta, A. Nano-silicon application enhances salt stress tolerance in Lens culinaris L.: a comparative study with bulk silicon. J. Plant. Nutr. 1–17. https://doi.org/10.1080/01904167.2025.2513404 (2025).
Ali, M. et al. Silicon mediated improvement in the growth and ion homeostasis by decreasing Na+ uptake in maize (Zea Mays L.) cultivars exposed to salinity stress. Plant. Physiol. Biochem. 158, 208–218. https://doi.org/10.1016/j.plaphy.2020.10.040 (2021).
Sarkar, M. M., Mukherjee, S., Mathur, P. & Roy, S. Exogenous nano-silicon application improves ion homeostasis, osmolyte accumulation and palliates oxidative stress in Lens culinaris under NaCl stress. Plant. Physiol. Biochem. 192, 143–161. https://doi.org/10.1016/j.plaphy.2022.10.001 (2022).
He, S. et al. Nano silicon dioxide reduces cadmium uptake, regulates nutritional homeostasis and antioxidative enzyme system in barley seedlings (Hordeum vulgare L.) under cadmium stress. Environ. Sci. Pollut Res. 30 (25), 67552–67564. https://doi.org/10.1007/s11356-023-27130-x (2023).
Patel, M., Fatnani, D. & Parida, A. K. Silicon-induced mitigation of drought stress in peanut genotypes (Arachis Hypogaea L.) through ion homeostasis, modulations of antioxidative defense system, and metabolic regulations. Plant. Physiol. Biochem. 166, 290–313. https://doi.org/10.1016/j.plaphy.2021.06.003 (2021).
Mahmoud, A. W. M. et al. Nanopotassium, nanosilicon, and Biochar applications improve potato salt tolerance by modulating photosynthesis, water status, and biochemical constituents. Sustainability 14 (2), 723. https://doi.org/10.3390/su14020723 (2022).
Fan, N. et al. Nanosilicon alters oxidative stress and defence reactions in plants: A meta-analysis, mechanism and perspective. Environ. Sci. : Nano. 9 (10), 3742–3755. https://doi.org/10.1039/D2EN00478J (2022).
Huang, T., Li, Z., Long, Y., Zhang, F. & Pang, Z. Role of desorption-adsorption and ion exchange in isotopic and chemical (Li, B, and Sr) evolution of water following water–rock interaction. J. Hydrol. 610, 127800. https://doi.org/10.1016/j.jhydrol.2022.127800 (2022).
Ryżak, M. & Bieganowski, A. Methodological aspects of determining soil particle-size distribution using the laser diffraction method. J. Plant. Nutr. Soil. Sci. 174 (4), 624–633. https://doi.org/10.1002/jpln.201000255 (2011).
Hardie, M., Clothier, B., Bound, S., Oliver, G. & Close, D. Does Biochar influence soil physical properties and soil water availability? Plant. Soil. 376 (1), 347–361. https://doi.org/10.1007/s11104-013-1980-x (2014).
Ojewumi, A. W., Ayoola, S. A., Abdullahi, M. H., Adeola, Z. B. & Olubunmi, S. M. Bio-fortification of agronomic attributes and biochemical molecules in black night shade as influenced by two forms of Guinea grass Biochar. Zanco J. Pure Appl. Sci. 36 (6), 77–86 (2024).
Kazemeini, S. A., Rahimi, H. & Alinia, M. Effect of salinity stress on germination and tolerance threshold of Guar (Cyamopsis Tetragonoloba l.) seed.In: 18th Iranian National & 4th International Crop Science Congress, 10–12. (2024).
Lichtenthaler, H. K. & Wellburn, A. R. Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 11 (5), 591–592 (1983).
Sairam, R. K., Rao, K. V. & Srivastava, G. C. Differential response of wheat genotypes to long term salinity stress in relation to oxidative stress, antioxidant activity and osmolyte concentration. Plant. Sci. 163 (5), 1037–1046. https://doi.org/10.1016/S0168-9452(02)00278-9 (2002).
Chance, B. & Maehly, A. C. [136] assay of catalases and peroxidases. Methods Biochem. Anal. 1, 357–424 (1955).
Beauchamp, C. & Fridovich, I. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 44 (1), 276–287 (1971).
Aebi, H. [13] Catalase in vitro. In Methods in enzymology 105, 121–126. (Academic press, 1984).
Yoshimura, K., Yabuta, Y., Ishikawa, T. & Shigeoka, S. Expression of spinach ascorbate peroxidase isoenzymes in response to oxidative stresses. Plant. Physiol. 123 (1), 223–234. https://doi.org/10.1104/pp.123.1.223 (2000).
Havaux, M. & Kloppstech, K. The protective functions of carotenoid and flavonoid pigments against excess visible radiation at chilling temperature investigated in Arabidopsis Npq and Tt mutants. Planta 213 (6), 953–966. https://doi.org/10.1007/s004250100572 (2001).
Narayan, S., Sharma, R. K., Kumar, V., Sanyal, I. & Shirke, P. A. Alterations in plant anatomy and higher lignin synthesis provides drought tolerance in cluster bean [Cyamopsis Tetragonoloba (L.) Taub]. Plant. Physiol. Biochem. 201, 107905. https://doi.org/10.1016/j.plaphy.2023.107905 (2023).
Fattahi, S. et al. Total phenolic and flavonoid contents of aqueous extract of stinging nettle and in vitro antiproliferative effect on Hela and BT-474 cell lines. IJMCM 3 (2), 102 (2014).
Velikova, V., Yordanov, I. & Edreva, A. J. P. Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant. Sci. 151 (1), 59–66. https://doi.org/10.1016/S0168-9452(99)00197-1 (2000).
Heath, R. L. & Packer, L. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 125 (1), 189–198. https://doi.org/10.1016/0003-9861(68)90654-1 (1968).
Golmakani, M. T., Mendiola, J. A., Rezaei, K. & Ibáñez, E. Expanded ethanol with CO2 and pressurized Ethyl lactate to obtain fractions enriched in γ-Linolenic acid from Arthrospira platensis (Spirulina). J. Supercrit Fluids. 62, 109–115. https://doi.org/10.1016/j.supflu.2011.11.026 (2012).
Mony, C. et al. Nanomaterials for enhancing photosynthesis: interaction with plant photosystems and scope of nanobionics in agriculture. Environ. Sci. : Nano. 9 (10), 3659–3683. https://doi.org/10.1039/D2EN00451H (2022).
Morshedloo, M. R., Saeidi, S., Zahedi, S. M., Jahantab, E. & Ghorbanpour, M. Foliar-applied silicon and nano-silicon alter the product quality and defense system in ginger mint (Mentha gracilis R. Br.) under drought stress by inducing essential oil accumulation and antioxidant activity. J. Soil. Sci. Plant. Nutr. 25 (1), 1913–1926. https://doi.org/10.1007/s42729-025-02245-x (2025).
Farhangi-Abriz, S. & Torabian, S. Nano-silicon alters antioxidant activities of soybean seedlings under salt toxicity. Protoplasma 255 (3), 953–962. https://doi.org/10.1007/s00709-017-1202-0 (2018).
Sohby, M. K., Khalil, H. A., Eissa, A. M. & Fekry, W. M. Influence of nano-silicon and nano-chitosan on growth, ion content, and antioxidant defense enzyme of two citrus rootstocks under salinity conditions. Mesop. J. Agric. 51 (2), 147–166 (2023).
Abd-El-Aty, M. S., Kamara, M. M., Elgamal, W. H., Mesbah, M. I., Abomarzoka, E. A.,Alwutayd, K. M. Abdelaal, K. Exogenous application of nano-silicon, potassium sulfate,or proline enhances physiological parameters, antioxidant enzyme activities, and agronomic traits of diverse rice genotypes under water deficit conditions. Heliyon 10 (5). https://doi.org/10.1016/j.heliyon.2024.e26077 (2024).
Abdi, M. J., Ghanbari Jahromi, M., Mortazavi, S. N., Jari, K., Nazarideljou, M. J. & S. & Foliar-applied silicon and selenium nanoparticles modulated salinity stress through modifying yield, biochemical attribute, and fatty acid profile of Physalis Alkekengi L. Environ. Sci. Pollut Res. 30 (45), 100513–100525. https://doi.org/10.1007/s11356-023-29450-4 (2023).
Hajihashemi, S. & Kazemi, S. The potential of foliar application of nano-chitosan-encapsulated nano-silicon donor in amelioration the adverse effect of salinity in the wheat plant. BMC Plant. Biol. 22 (1), 148. https://doi.org/10.1186/s12870-022-03531-x (2022).
Xiao, Z., Fan, N., Yue, L., Chen, F., Ji, H., Shu, Y. Wang, Z. Dose-dependent effects of CeO2 nanomaterials on tomato plant chemistry and insect herbivore resistance. Environ. Sci. Nano. 8 (12), 3577–3589. https://doi.org/10.1039/D1EN00837D (2021).
Sepasi, M., Iranbakhsh, A., Saadatmand, S., Ebadi, M. & Oraghi Ardebili, Z. Silicon nanoparticles (SiNPs) stimulated secondary metabolism and mitigated toxicity of salinity stress in Basil (Ocimum Basilicum) by modulating gene expression: a sustainable approach for crop protection. Environ. Sci. Pollut Res. 31 (11), 16485–16496. https://doi.org/10.1007/s11356-024-32260-x (2024).
Mostafavi, S., Tavallali, V., Asadi-Gharneh, H. A. & Rowshan, V. Modulation of polyphenolic Content, essential Oils, and antioxidant activity in Zataria multiflora by Silicon-Based nanocomplexes. BioNanoSci 15 (3), 493. https://doi.org/10.1007/s12668-025-01953-z (2025).
Waters, S., Gilliham, M. & Hrmova, M. Plant high-affinity potassium (HKT) transporters involved in salinity tolerance: structural insights to probe differences in ion selectivity. Int. J. Mol. Sci. 14 (4), 7660–7680. https://doi.org/10.3390/ijms14047660 (2013).
Liu, B., Soundararajan, P. & Manivannan, A. Mechanisms of silicon-mediated amelioration of salt stress in plants. Plants 8 (9), 307. https://doi.org/10.3390/plants8090307 (2019).
Wang, Q., Shan, C., Zhang, P., Zhao, W., Zhu, G., Sun, Y.Rui, Y. The combination of nanotechnology and potassium: applications in agriculture. Environ. Sci. Pollut. Res. 31 (2), 1890–1906. https://doi.org/10.1007/s11356-023-31207-y (2024).
Gharbi, P., Amiri, J., Mahna, N., Naseri, L. & Sadaghiani, M. R. Silicon-induced mitigation of salt stress in GF677 and GN15 rootstocks: insights into physiological, biochemical, and molecular mechanisms. BMC Plant. Biol. 25 (1), 719. https://doi.org/10.1186/s12870-025-06753-x (2025).
Alves, D. M. R., de Mello Prado, R. & Barreto, R. F. & Da Silva Carvalho, L. T. Nano-silicon and sodium mitigate Damage by potassium deficiency in Chicory. Sci. Rep. 14 (1), 16841. https://doi.org/10.1038/s41598-024-67875-0 (2024).
Rezghiyan, A., Esmaeili, H. & Farzaneh, M. Nanosilicon application changes the morphological attributes and essential oil compositions of hemp (Cannabis sativa L.) under water deficit stress. Sci. Rep. 15 (1), 3400. https://doi.org/10.1038/s41598-025-87611-6 (2025).
Acknowledgements
The authors would like to gratefully acknowledge the Agricultural and Natural Resources Engineering Organization of Fars Province for their financial support of this research.
Author information
Authors and Affiliations
Contributions
Conceptualization H.R., and S.A.K.; data curation, S.A.K., H.R., and M.A.; formal analysis and methodology, S.A.K., H.R., M.A., S.M.H.H., and M.J.A.; project administration, S.A.K.; visualization, H.R., S.A.K., and S.M.H.H.; writing original draft, S.A.K., M.A., and M.J.A.; writing-review and editing. S.A.K., and M.A. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All procedures were conducted following the relevant institutional, national, and international guidelines and legislations.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rahimi, H., Kazemeini, S.A., Alinia, M. et al. Silicon nanoparticles ameliorate salt stress in cluster bean by improving antioxidant defense and ion homeostasis. Sci Rep 16, 10057 (2026). https://doi.org/10.1038/s41598-026-39447-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-39447-x