Abstract
Quantitative studies of cortical development in preterm infants are needed. Until now, magnetic resonance imaging (MRI) has been the only technique capable of providing these 3D measurements. However, bedside ultrasound may offer a more accessible, repeatable alternative for both clinical and research applications. To assess the feasibility and reproducibility of three-dimensional transfontanellar ultrasound (3D US) for the semi-automated quantification of central sulcus (CS) morphology in very preterm infants, we performed a prospective study acquiring 109 serial 3D US volumes from 13 infants born < 32 weeks’ gestation. Twelve CS morphometric parameters were extracted with a semi-automated pipeline; reliability was assessed with intra-class correlation coefficients (ICC), and maturational trends were described using group comparisons across PMA categories and complemented with linear mixed-effects models reporting fixed-effect estimates and variance components. Single-sweep ICCs ranged from 0.15 (length) to 0.71 (mean opening), while averaging three sweeps improved reproducibility to 0.34–0.88 (highest ICC 0.88, 95% CI 0.74–0.95). CS length increased from 25.9 ± 9.8 mm at < 28 weeks to 34.3 ± 8.9 mm at 36 weeks post-menstrual age (PMA) (p = 0.006), and mean sulcal opening from 4.0 ± 1.8 mm to 5.7 ± 1.9 mm (p = 0.023). In mixed-effects models, CS length increased with PMA (β = 0.96 mm/week; p = 0.008), and maximum opening also increased (β = 0.27 mm/week ; p = 0.002).The right hemisphere consistently showed larger openings than the left (5.4 ± 1.3 mm vs. 3.2 ± 1.5 mm; p < 0.0001). 3D US-based CS metrics thus capture lateralised, maturational changes and, when multiple sweeps are averaged, achieve good reproducibility (ICC up to 0.88), supporting their use for serial monitoring of cortical folding in the neonatal intensive care unit (NICU).
Similar content being viewed by others
Introduction
In addition to the high risk of early mortality, preterm infants (PT) are vulnerable to a range of short- and long-term complications. These complications often include a wide spectrum of neurodevelopmental impairments, which manifest throughout infancy and childhood. The most severe outcome is cerebral palsy, which remains a leading motor outcome of very preterm birth1. However, motor impairments represent only part of the spectrum. A significant proportion of preterm survivors also exhibit cognitive, behavioral, and sensory deficits, including executive dysfunction, attention difficulties, impaired visuospatial processing, and language delays. These outcomes may occur even in the absence of obvious brain injury2,3.
The neurodevelopmental sequelae of prematurity are closely linked to structural and microstructural alterations of the developing brain. Diffuse white matter injury is recognized as a predominant pattern in very preterm infants. This injury disrupts axonal growth, myelination, and oligodendrocyte maturation during a critical window of vulnerability4,5. In addition to white matter pathology, prematurity has been associated with abnormal development of key gray matter structures, including the thalamus, basal ganglia, cerebellum, and cerebral cortex6.
Preterm birth disrupts typical cortical folding trajectories due to the premature transition from the intrauterine to the extrauterine environment, leading to alterations in gyral and sulcal morphology. These alterations, notably reduced cortical surface area and decreased gyrification index, are especially evident in regions involved in sensorimotor integration, such as the central sulcus (CS) and the adjacent pre- and postcentral gyri7,8. Consistent findings from neuroimaging studies reveal that preterm infants exhibit simplified cortical folding and reduced sulcal complexity compared to term-born peers, likely resulting from both direct brain injury and secondary effects of inflammation and altered sensory input. The central sulcus, in particular, is of interest due to its early development and its fundamental role in motor function, making it a relevant marker for studying the impact of prematurity on brain structure and subsequent neurodevelopmental outcomes9,10.
The CS undergoes critical morphological development during the late second and early third trimesters. In preterm infants, the sulcus becomes visible and begins to exhibit interindividual morphological variability as early as 30 weeks postmenstrual age (PMA). Recent findings suggest that many of its defining morphological features are already established by this time, underscoring the susceptibility of sulcal development to the extrauterine environment in the context of prematurity7. The early maturation of the CS is particularly important, because it may have long-term implications for functional outcomes, such as fine motor abilities and manual dexterity. Several studies have used magnetic resonance imaging (MRI) as the main tool to describe the morphology of CS in newborns, particularly in the preterm population. Through quantitative and longitudinal analyses, parameters such as sulcus depth, surface area, and curvature have been characterized, as well as its hemispheric variability and its predictive value on long-term motor and cognitive functions7,8,11,12. However, despite the extensive use of MRI in this field, our study introduces a novel approach by employing 3D ultrasound (US) to characterize CS.
When applied to the CS evaluation, 3D US combines the advantages of traditional 2D US with the additional benefits of multiplanar reconstruction, volume rendering, and anatomical segmentation13. These features enable a comprehensive visualization of the sulcal morphology, including volume, tortuosity, surface, length, depth, and sulcal opening. If 3D US is to play a meaningful role in the early assessment of cortical folding and neuromotor prognosis, robust and efficient segmentation tools must be used. This pilot study aimed to verify the feasibility of transfontanellar 3D US for semi-automatic quantification of the CS in PT infants. Two secondary objectives were also established: to quantify its reproducibility and to describe the maturation trajectory, including hemisphere analysis. In this pilot study, our aim was to assess bedside feasibility and to obtain preliminary evidence that the measurements are reproducible and sensitive to developmental trends. Given the bedside-oriented nature of the protocol, we designed this work as a single-center pilot feasibility study. Accordingly, it was not powered for subgroup analyses or for linking imaging metrics to neurodevelopmental outcomes, and all inferential analyses should be interpreted as exploratory.
Results
Study population
Patients and 3D US
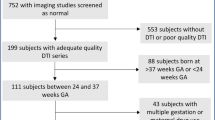
We included 13 preterm infants born at less than 32 weeks of gestational age (GA). The mean GA was 28.6 weeks (± 2.97), and the mean birth weight was 1023.1 g (± 363.4). One infant (7.7%) was classified as small for gestational age (SGA). Eight patients were male (61.5%) and ten (77%) were born by cesarean Sect. (76.9%). Most mothers received a complete course of antenatal steroids (83.3%). The mean maternal age was 29.1 years (SD 4.76), with a predominance of intermediate educational level (75%). According to the GMH-IVH classification, 5 patients (45.5%) showed no hemorrhage, 3 (27.3%) had grade I GMH, 2 (18.2%) had grade III GMH, and 1 patient (9.1%) presented with intraparenchymal hemorrhage (IPH). A more detailed description of the perinatal variables is shown in Table 1. The 13 patients had a median of 8 3D US, with a minimum of 4 and a maximum of 10. The total number of 3D US included in the study was 109.
Reliability
We assessed reproducibility across three independent 3D ultrasound acquisitions during the same day, which enabled us to quantify reproducibility. Using a two-way random effects intraclass correlation coefficient (ICC), single acquisitions did not yield robust reproducibility for any parameter (none reached good reliability). Averaging the three same-day acquisitions improved reliability to 0.53, 0.49, 0.38, 0.34, 0.36, 0.45, 0.39, 0.55, 0.78, 0.88, 0.64, and 0.81, respectively. These findings indicate that most variability stems from the acquisition step, particularly for volumetric and depth measures, whereas combining the three sweeps yields moderate repeatability for most CS parameters.
Bland-Altman analysis of mean sulcal opening
Bland-Altman plots for the three pair-wise combinations of same-day acquisitions (Fig. 1) showed minimal bias (< 0.35 mm) and symmetric 95% limits of agreement of approximately ± 3 mm. In each plot, more than 95% of points lay within these limits, and no trend toward greater dispersion at larger opening values was observed. Together with the ICC results, these findings confirm that averaging three sweeps greatly reduces acquisition noise and delivers an absolute error for mean sulcal opening that is acceptable for clinical use Table 2.
Pair-wise Bland–Altman plots comparing the three same-day acquisitions for mean sulcal opening: Acquisition 1 vs. Acquisition 2 (A), Acquisition 1 vs. Acquisition 3 (B), and Acquisition 2 vs. Acquisition 3 (C). The solid line indicates the bias; dashed lines represent the 95% limits of agreement.
Central sulcus analysis according to postmenstrual age, sex, and hemisphere
We quantitatively analyzed the segmented central sulcus (CS) in 3D ultrasound studies using twelve morphological parameters: volume, tortuosity, surface area, length, mean and maximum depth, sulcal opening, and fold opening (Fig. 2). Sulci were categorized by postmenstrual age (PMA) at the time of the scan into three groups: <28, 28–32, and 32–36 weeks.
A significant increase in sulcal length was observed with advancing PMA, being significantly greater in the 32–36-week group.
(34.35 ± 8.94 mm) compared to the < 28-week group (25.86 ± 9.77 mm; p = 0.006). Similarly, maximum lineal depth was higher in the 32–36-week group (9.30 ± 2.41 mm) than in the < 28-week group (8.87 ± 2.23 mm; p = 0.014). Notably, mean sulcal opening also increased significantly with PMA, reaching 5.70 ± 1.92 mm in the 32–36-week group vs. 3.98 ± 1.76 mm in the < 28-week group (p = 0.023). (Table 3).
Linear mixed-effects models showed significant positive associations with PMA for most central sulcus metrics, including volume, tortuosity, surface, length, and depth-related measures (Table 4). Variance components and σ²between/σ²within ratios are reported in Table 4.
Analysis by hemisphere revealed consistently greater opening values for the right CS vs. the left CS, with significant differences in mean sulcal opening (5.41 ± 1.27 mm vs. 3.23 ± 1.47 mm; p < 0.0001), maximum opening (8.93 ± 2.14 mm vs. 5.99 ± 2.74 mm; p < 0.0001), and maximum fold opening (9.17 ± 2.19 mm vs. 6.36 ± 2.80 mm; p < 0.0001). These findings suggest rightward asymmetry in sulcal development during early maturation (Table A1).
3D reconstructions of the central sulcus obtained using 3D ultrasound in a preterm infant born at 28 weeks of gestational age. The images illustrate the longitudinal development of the CS at different postmenstrual ages: at birth (A), 30 weeks (B), 32 weeks (C), and 38 weeks (D) postmenstrual age.
Discussion
This study demonstrates the feasibility of semi-automated quantification of CS morphology using 3D US in very preterm infants. Importantly, the reproducibility analysis showed that single acquisitions yielded low intraclass correlation coefficients (ICC ≤ 0.40) for most metrics, indicating limited reliability for individual clinical prognostic assessment. However, averaging the three acquisitions substantially improved reliability, particularly for the sulcal opening parameters (ICC ≥ 0.75), suggesting potential for clinical application in certain morphometric domains. Other measures, such as volume and depth, showed moderate reliability (ICC 0.45–0.55), which may still support use in longitudinal or research settings. These findings reinforce the value of 3D-US for characterising early alterations in CS morphology.
Recent MRI work that applied manifold-learning techniques to detailed shape reconstructions of the central sulcus has shown that several geometric descriptors are already well defined by 30 weeks PMA and that a right-greater-than-left asymmetry emerges early and persists into later childhood, potentially related to laterality development, including handedness7. Our method complements these high-resolution but episodic MRI assessments by enabling high-frequency monitoring during this critical developmental window because 3D US can be repeated at the bedside without sedation.
Neonatal intensive care unit (NICU) typical Clinical exposures(low birth-weight, multiple gestation and prolonged mechanical ventilation) selectively blunt early growth of the CS and other primordial folds; these alterations are associated with less favorable fine-motor and overall cognitive scores at two years9. MRI studies in very PT cohorts have reported associations between early cortical folding features and later neurodevelopmental performance at toddler age, including motor and broader cognitive domains. Because mean opening was the only parameter that reaches good reproducibility after averaging three sweeps (ICC 0.88), it could constitute a robust ultrasound signature of motor vulnerability that could be combined with MRI-based cortical metrics to refine early prognostic models while reducing the need for multiple transportations to the MRI scan. Serial 3D US could extend this developmental trajectory back into the early life exposure after preterm birth, yielding a continuous view of how early extrauterine factors sculpt the morphology of the CS. Changes in the depth of the CS motor representations continue over the first three years of life and run in parallel with the achievement of major gross- and fine-motor milestones14.
Advanced MRI imaging studies have shown that extrauterine stressors as nutritional deficits, inflammation, and white-matter injury, can leave lasting imprints on cortical folding and motor circuitry15. Because 3D US is innocuous, quick, and easily repeatable, it represents a practical tool for real-time monitoring of brain growth and development with the potential to evaluate how neuroprotective strategies influence CS development during the NICU stay.
This study has several methodological strengths that enhance the reliability of its findings. The cohort was prospectively examined with a standardized 3D US protocol that acquired three independent sweeps per session, enabling a true within-subject.
assessment of reproducibility under real NICU conditions. The workflow captured the full 3D contour of the CS rather than relying on single planar slices, yielding a comprehensive set of quantitative metrics for each examination. Each infant underwent several serial scans (median = 8 per patient), resulting in a dense longitudinal dataset that could be analyzed to account for individual growth trajectories.
This preliminary single-center study has some limitations. First, the number of infants is small and all participants were recruited from a single NICU. Therefore, confidence intervals are expected to be wide, subgroup analyses are underpowered, and external validity is limited. These results should be considered hypothesis-generating and require confirmation in larger cohorts. Second, reproducibility was assessed only intra-day with one operator and scanner; inter-observer, cross-platform, and longer-term reliability still need to be established. Future reliability studies should include inter-observer segmentation variability, and inter-day acquisitions. Third, the absence of concurrent MRI data precluded direct cross-modal validation of our US metrics. Because MRI was not acquired concurrently, we cannot quantify systematic bias or directly assess measurement accuracy against an established cortical morphometry reference. A planned next step is to acquire paired 3D-US and MRI within a short time window to enable cross-modal agreement analyses and calibration of US-derived opening metrics.
Conclusions
3D ultrasound enables bedside quantification of CS morphology in very preterm infants. Averaging three sweeps yields highly reproducible sulcal opening measurements (ICC ≥ 0.75). The portability and repeatability of this method offer advantages over MRI for longitudinal monitoring of cortical development. Integration of 3D US morphometry into neonatal care may improve understanding of CS maturation and support early neuroprotective interventions.
Methods
Patients
Eligible participants were very-low-birth-weight infants (VLBWI) admitted to the NICU of Hospital Puerta del Mar, defined as a birthweight ≤ 1,500 and/or a gestational age ≥ 32 weeks. Infants with congenital or chromosomal anomalies, metabolic disorders, or central nervous system infections were excluded. The Research and Ethics Committee approved the study, and.
written informed consent was obtained from all parents or guardians.
We reviewed the 109 3D US scans serially performed at different time points, with a minimum of 1 day apart. This was a feasibility pilot study; therefore, the sample size was determined by convenience and operational constraints (available eligible infants and scanning windows), with no formal a priori power calculation. All inferential analyses, including sex-stratified comparisons, were considered exploratory. Transfontanellar 3D US images of the brain were acquired using the 4D option in the 3D/4D Voluson i portable ultrasound system (GE Healthcare, Milwaukee, WI, USA). First, the transducer (SVNA5-8B, 5–8 MHz), using a center frequency of 6.5 MHz, was situated in the third coronal plane, and the scan angle was settled to 90º. With the transducer fixed in that position, the beam moves from them anterior to posterior planes with a maximum total scan time of 40–50 s.
Ethics approval and consent to participate
The study protocol was approved by the Research and Ethics Committee of Hospital Universitario Puerta del Mar (Cádiz, Spain). All procedures were performed in accordance with relevant institutional and national guidelines and regulations and with the Declaration of Helsinki. Written informed consent was obtained from the parents or legal guardians of all participants prior to inclusion.
Image analysis
Image segmentation and post-processing were conducted using a semi-automated pipeline. Manual slice-by-slice delineation of the CS was performed by trained raters using multiplanar 3D US reconstructions. Morphometric parameters were extracted using custom-developed software. Each segmentation was subjected to visual inspection and manual correction to ensure anatomical fidelity before final metric extraction.
Assisted segmentation of cerebral sulci in 3D ultrasound using a graphical interface
An interactive tool was developed using MATLAB (R2024a Update 5) for the semi-automatic segmentation of cerebral sulci from 3D ultrasound volumes. The volume is represented as a three-dimensional matrix, and users can freely explore axial, coronal, and sagittal planes through a graphical user interface (GUI). (Fig. 3)
Screenshot of the custom MATLAB-based graphical user interface (GUI) for semi-automatic sulcal segmentation.
The left panel displays the 3D ultrasound volume as navigable orthogonal planes, with the central sulcus manually refined (yellow trace). The right panel shows the corresponding 3D reconstruction of the segmented sulcus. The interface provides interactive tools for slice navigation, labeling, and visualization, and automatically outputs quantitative morphometric metrics.
Sulcus segmentation is performed on selected 2D slices, starting from a manually marked point of interest. From this point, Frangi filters16are applied to enhance sulcus-like structures, and closed level-set contours are identified around the region. The initial mask is then refined using an active contour model17,18to better capture the sulcus geometry. The segmentation can be progressively propagated to adjacent slices. The quality of the segmentation depends on the resolution and contrast of the ultrasound images, as well as on the technique employed. The developed software supports a flexible approach, offering different segmentation modes ranging from manual tracing to local region-adaptive active contours. The system allows labeling of anatomical sulci, visualization and manual editing of their contours, and the calculation of morphometric features such as volume, surface area, depth, width, and tortuosity. Results can also be exported in Excel format. This tool is particularly suited for clinical and research settings that require personalized, guided segmentation of cortical relief in neonatal ultrasound. Once segmentation is completed, various morphometric measurements are computed directly from the segmented volume to characterize the geometry and spatial configuration of the central sulcus. These include:
Volume. The volume is calculated as the total number of segmented voxels multiplied by the unit voxel volume defined by the acquisition spacing. (Fig. 4)
Definition of the volume metric (mm3) for the central sulcus.
Surface Area. The surface area is estimated by extracting the 3D contour of the segmented sulcus and generating a surface mesh. The total area is obtained as the sum of the triangle areas in this mesh. (Fig. 5)
Definition of the surface area metric (mm²) for the central sulcus (grid = measured surface).
Sulcal opening. The sulcal opening is estimated from equally spaced cross-sections along the sulcus’s main axis. In each slice, the 2D binary section is reconstructed and the number of active voxels per row is computed. This results in a local width matrix representing the sulcus opening along its length. A localized fold opening measure is also defined as the maximum width among the uppermost active rows, providing a clinically relevant index of cortical compression or expansion near the.
surface. (Fig. 6)
Definition of opening measures (mm) for the central sulcus. Arrows indicate orthogonal distances: opening and fold opening.
Tortuosity. From the 3D skeleton of the segmented volume, a spatial graph is constructed that represents the sulcus trajectory. Tortuosity is defined as the ratio between the actual path length and the straight-line Euclidean distance between its endpoints, quantifying the deviation from an ideal linear path. (Fig. 7)
Tortuosity of the central sulcus (unitless): defined as Lcurved/LstraightLcurved/Lstraight between endpoints A and B.
Depth. Sulcal depth is estimated from the segmented volume following a conceptually similar approach to that proposed by Coulon et al. (2006)19, adapted to volumetric data. Instead of solving the heat equation for surface parameterization, this method uses equally spaced transverse slices along the main sulcal axis, previously aligned by PCA-based rotation. In each slice, equidistant points are interpolated along the sulcal contour, and a 3D graph is built connecting consecutive points between adjacent slices. Depth is calculated as the sum of shortest-path distances from each central point to the outer sulcal edges, following the actual surface geometry. A complementary linear depth is also computed as the direct Euclidean distance between those endpoints. (Fig. 8)
Depth metrics of the central sulcus (mm): semiperimeter depth and linear depth.
Statistical analysis
We performed a descriptive analysis, and the results were described as frequency and percentage if they were categorical, mean and standard deviation (sd), or median and interquartile range [IQR] according to their distribution if they were quantitative. Bivariate analysis was performed using Pearson’s chi-square test or Fisher’s exact test for categorical data and Student’s t-test or the Mann–Whitney U test for two-group comparisons, and one-way ANOVA across the three PMA groups. Differences in morphometric parameters across the three PMA groups (< 28, 28–32, and 32–36 weeks) were assessed using one-way ANOVA. Reliability was assessed with two-way random-effects ICCs. Bland–Altman plots assessed agreement. Longitudinal associations between PMA and each CS metric were assessed using linear mixed-effects models with PMA as a continuous fixed effect and a subject-specific random interceptto account for repeated measurements. For each model, we report the PMA slope (β, units/week) with 95% confidence interval and variance components (σ² between-subject and σ² within-subject residual variance), including their ratio (σ²between/σ²within).Statistical analysis was performed using Stata 16.0 (Stata Statistical Software: Release 16. College Station, TX: StataCorpLP). A result was considered statistically significant with a p < 0.05. All the methods were carried out in accordance with relevant guidelines and regulations.
Data availability
The data that support the findings of this study are available from the corresponding author on reasonable request.
References
Samantha Johnson and Neil Marlow. Early and long-term outcome of infants born extremely preterm. In: Archives of Disease in Childhood 102.1 97–102. ISSN: 0003-9888, 1468–2044. https://doi.org/10.1136/archdischild-2015-309581 (2017). (visited on 06/30/2025).
Elisabeth, C., McGowan & Vohr, B. R. Neurodevelopmental Follow-up of Preterm Infants. In: Pediatric Clinics of North America 66.2Apr. 509–523. ISSN: 00313955. (2019). https://linkinghub.elsevier.com/retrieve/pii/S0031395518302025 (visited on 06/30/2025).
Abdul, R. et al. Temporal trends in severe brain injury and associated outcomes in very preterm infants. Neonatology 121, 4 (2024). https://karger.com/article/doi/10.1159/000537801 visited on 05/02/2025). 440–449. ISSN: 1661–7800, 1661–7819. DOI: 10.1159/000537801. URL.
Joseph, J. & Volpe Dysmaturation of Premature Brain: Importance, Cellular Mechanisms, and Potential Interventions. In: Pediatric Neurology 95June pp. 42–66. ISSN: 08878994. https://doi.org/10.1016/j.pediatrneurol.2019. 02.016. URL: (2019). https://linkinghub.elsevier.com/retrieve/pii/S0887899419301079 (visited on 06/30/2025).
Shannon, E. G. et al. Nov. Trends in severe brain injury and neurodevelopmental outcome in premature newborn infants: The role of cystic periventricular leukomalacia. In: The Journal of Pediatrics 145.5. 593–599. ISSN: 00223476. https://doi.org/10.1016/j.jpeds.2004.05.042. (2004). https://linkinghub.elsevier.com/retrieve/pii/S002234760400455X (visited on 06/30/2025).
Terrie, E., Inder, J. J., Volpe & Anderson, P. J. (eds). Defining the Neurologic Consequences of Preterm Birth. In: New England Journal of Medicine 389.5 Ed. by Julie R. Ingelfinger, 441–453. ISSN: 0028-4793, 1533- 4406. DOI: 10.1056/NEJMra2303347. http://www.nejm.org/doi/ (2023). https://doi.org/10.1056/NEJMra2303347 (visited on 06/24/2025).
de Vareilles, H. et al. Shape variability of the central sulcus in the developing brain: A longitudinal descriptive and predictive study in preterm infants. NeuroImage 251, 118837. https://doi.org/10.1016/j.neuroimage.2021.118837 (2022).
Julia, E. K. et al. Sept. Early cortical maturation predicts neurodevelopment in very preterm infants. In: Archives of Disease in Childhood - Fetal and Neonatal Edition 105.5 460–465. ISSN: 1359–2998, 1468–2052. https://fn.bmj.com/lookup/doi/ (2020). https://doi.org/10.1136/archdischild-2019-317466 (visited on 05/02/2025).
Kersbergen, K. J. et al. Relation between clinical risk factors, early cortical changes, and neurodevelopmental outcome in preterm infants. NeuroImage 142, 301–310. https://doi.org/10.1016/j.neuroimage.2016.07.010 (2016).
Erin, E. et al. Jan. Regional impairments of cortical folding in premature infants. In: Annals of Neurology 77.1 pp. 154–162. ISSN: 0364–5134, 1531–8249. https://onlinelibrary.wiley.com/doi/10.1002/ana.24313. (2015). (visited on 05/02/2025).
Shimony, J. S. et al. Comparison of cortical folding measures for evaluation of developing human brain. NeuroImage 125, 780–790. https://doi.org/10.1016/j.neuroimage.2015.11.001 (2016).
Norio, H. et al. Jan. Development of identification of the central sulcus in brain magnetic resonance imaging. In: Radiological Physics and Technology 4.1 pp. 53–60. ISSN: 1865 – 0333, 1865 – 0341. : http://link.springer.com/ (2011). https://doi.org/10.1007/s12194-010-0104-z (visited on 05/02/2025).
Riccabona, M. Potential role of 3DUS in infants and children. In: Pediatric Radiology 41S1 May pp. 228–237. ISSN: 0301–0449, 1432–1998. http://link.springer.com/ (2011). https://doi.org/10.1007/s00247-011-2051-1 (visited on 05/02/2025).
Niharika, G. et al. Central sulcus development in early childhood. In: 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). Seogwipo: IEEE, July 2017, 161–164. ISBN: 978-1-5090-2809-2. 10.1109/EMBC.2017.8036787. (2017). https://ieeexplore.ieee.org/document/8036787/ (visited on 07/03/2025).
Petra, S. & Hüppi Cortical Development in the Fetus and the Newborn: Advanced MR Techniques. In: Topics in Magnetic Resonance Imaging 22.1Feb. 33–38. ISSN: 0899–3459. (2011). https://journals.lww.com/00002142-201102000-00005 (visited on 05/02/2025).
Multiscale vessel enhancement filtering. In: Alejandro F. Frangi et al. Lecture Notes in Computer Science. ISSN: 03029743, 1611–3349 & Berlin Heidelberg: Springer Berlin Heidelberg, pp. 130–137. ISBN: 978-3-540-65136-9 978-3- 540-49563-5. https://doi.org/10.1007/bfb0056195. URL: (1998). http://link.springer.com/10.1007/BFb0056195 (visited on 08/14/2025).
Kass, M. & Terzopoulos, D. rew Witkin, and Snakes: Active contour models. In: International Journal of Computer Vision 1.4Jan. Publisher: Springer Science and Business Media LLC, 321–331. ISSN: 0920–5691, 1573 – 1405. https://doi.org/10.1007/bf00133570. URL: (1988). http://link.springer.com/10.1007/BF00133570 (visited on 08/14/2025).
Wang, X. F., Huang, D. S. & Xu, H. An efficient local Chan–Vese model for image segmentation. In: Pattern Recognition 43.3 (Mar. 2010). Publisher: Elsevier BV, 603–618. ISSN: 0031-3203. DOI: 10. 1016/j.patcog.2009.08.002. URL: https://linkinghub.elsevier.com/retrieve/pii/ S0031320309002982 (visited on 08/14/2025).
Coulon, O. et al. Cortical localization via surface parameterization: a sulcus-based approach. In: NeuroImage 31.Suppl 1 S46. (2006).
Funding
This study has been funded by Instituto de Salud Carlos III (ISCIII) through the project “DTS22/00142” and co-funded by the European Union and the I + D+i PID2023-149783OB-I00 funded by MCIN/AEI/https://doi.org/10.13039/501100011033/ FEDER. This study was funded by the Cádiz integrated territorial initiative for biomedical research, European Regional Development Fund (ERDF) 2014–2020 (ITI-0019-2019).
Author information
Authors and Affiliations
Contributions
C.R.B., I.B.F., J.P. and S.P.L.L. made substantial contributions to conception and design of the study. S.P.L.L., M.L.G. and IBF performed all the ultrasounds. C.R.B, I.B.F., S.P.L.F and J.P. contributed with the acquisition of data, analysis, and interpretation of data. I.G.R. and M.L.G. contributed with acquisition of data. S.L.L.P. contributed substantially to data interpretation. C.R.B. and I.B.F. drafted the article and all the authors reviewed it critically for important intellectual content and have approved this final version to be submitted.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Barrios, C.R., Rosa, I.G., Fernández, S.P.L. et al. 3D ultrasound assessment of the central sulcus in very preterm infants: feasibility and reproducibility of opening metrics study. Sci Rep 16, 10199 (2026). https://doi.org/10.1038/s41598-026-40148-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-40148-8