Abstract
Chemotherapy / radiation induced oral mucositis (OM) poses significant clinical challenges, often leading to pain, malnutrition, treatment delays, and reduced quality of life. Regenerative medicine has been shown to have beneficial value in wound care. Hence, the current study was aimed at evaluating the safety and therapeutic efficacy of Mesenchymal Stem (Stromal) Cell derived Conditioned Media (MSC-CM) formulations (5%, 10%, and 15%) in preclinical models of 5-fluorouracil (5-FU)–induced OM and radiation wounds. The 28-day oral toxicity studies in rats/mice established that the formulations across treatment groups produced no treatment-related adverse effects. The NOAEL was established at 100% MSC-CM concentration. In the 5-FU based OM model, treatment with MSC-CM formulations demonstrated dose-dependent benefits, including significant reductions in ulcer size/ severity, regeneration of mucosal epithelium, and improvements in hematological, biochemical, hepatic, renal, and immunological parameters compromised by 5-FU. Histopathological evaluation revealed that chemotherapy-induced mucosal hyperplasia and blister formation were markedly reduced following MSC-CM treatment. These findings suggested enhanced tissue repair, possibly mediated through autophagy-associated mechanisms. The 15% formulation was most efficacious, yielding 100% survival, a mean ulcer healing score of 71.9%, and near-complete mucosal recovery, followed by 10% and 5% formulations. In the radiation wound healing model, administration of 10% and 15% MSC-CM formulations was well tolerated, as evidenced by stable body weights, and produced visible reductions in redness, hair loss, wound wetness, and oozing compared to controls. Notably, the 10% formulation promoted accelerated wound closure, with significant wound healing as early as day 4–7. Mechanistically, this could be attributed to induction of secretory IL-20 and IL-17 A/F in mice after wound healing gel (WHG) treatment at site of injury, which led to repair and regeneration of radiation-induced wounds. Collectively, these findings establish MSC-CM based topical formulations as safe and effective therapeutic candidates for mitigating chemotherapy and radiation induced toxicities, enhancing tissue regeneration, thereby supporting its translational potential in oral mucositis.
Similar content being viewed by others
Introduction
Mesenchymal Stem Cells (MSCs) are multipotent stromal cells derived from various sources such as bone marrow (BM-MSCs), adipose tissues (AT-MSCs), placenta (PMSCs), and umbilical cord (UC-MSCs). They are commonly employed stem cells in regenerative medicine and are considered multipotent adult stem cells that are capable of proliferation, self-regeneration, and multi-lineage differentiation1,2. Their isolation from bone marrow and adipose tissue includes invasive procedures, risk of pain, and complications of infection. In contrast, the isolation of MSCs from the umbilical cord offers distinct advantages. Isolation is non-invasive and painless, and the tissue is readily available as it is discarded during birth3. UC-MSCs often show fast self-renewing ability, which helps in tissue repair and in modulating immune response in damaged tissue4.
The therapeutic properties of MSCs are primarily attributed to their paracrine activity. Through secretion of growth factors, cytokines, and exosomes, MSCs exert potent effects on tissue repair, including enhanced cell migration, angiogenesis, and regeneration of injured tissues5,6,7,8,9. Despite their promise, cell-based therapies carry risks such as infection and immunogenic reactions following transplantation. As a safer alternative, cell-free therapy using MSC secretome (conditioned media) offers lower immunogenicity, reduced cost, and greater feasibility for long-term clinical use10.
The MSC conditioned media is rich in regenerative biomolecules such as hFGF, hHGF, hG-CSF, hIL-1Ra, hVEGF, and hIL-6, which are critical for tissue repair and wound regeneration11. Numerous studies have highlighted the translational potential of UC-MSC conditioned media (UC-MSC-CM), demonstrating its ability to accelerate skin repair, regeneration, and wound healing12,13,14,15,16. Some studies have also evidenced the use of exosomes for wound healing in full thickness wound models17,18 in hepatic regeneration via mitochondrial rescue19, and diabetic wound healing20. Both acute and chronic wound models have shown accelerated healing with MSC-CM treatment10,21.
Our earlier work also confirmed the potential of MSC-CM for wound healing11. Building upon these findings, the present study aimed to evaluate the in vivo safety and efficacy of a topical wound healing gel formulated with UCMSC-conditioned media, tested across different wound models in animals. The formulated gel is an improvement over conditioned media alone, as it eliminates the need for freezing and is a convenient format for direct application to the affected area of the wound.
Materials and methods
Preparation of topical wound healing gel
Wound healing gel (WHG) was prepared by incorporating sterile MSC-CM in a proprietary water-based gel. MSC-CM was prepared as previously described11. In brief, MSCs isolated from Wharton’s jelly, were cultured by xeno-free processes and characterized for morphology, flow cytometry for expression of CD90, CD105, CD34, CD45, and HLA-DR and karyotyping22. The MSCs were cultured from passage 2 (master cell bank), and expansion was carried out in T-500 flasks till passage 6 (working cell bank). Banked and cryopreserved MSCs were used for batch production of MSC-CM which was collected every 24–72 h cumulatively. The pooled MSC-CM was filter- sterilized with 0.2 μm filter and stored at − 20 °C till further use. Validated processes were followed for preparation of the WHG. The physico-chemical analyses included appearance, pH, sterility and heavy metals. WHG was visually examined for colour and form. The pH was determined using a pH meter. Microbiology assessment of WHG was done to check for total bacterial and fungal counts. The quantification of elemental impurities, like lead and arsenic, was carried out through analytical techniques such as Inductively Coupled Plasma–Optical Emission Spectrometry (ICP-OES) or Atomic Absorption Spectroscopy (AAS) and was done in a public testing laboratory. Both methods are widely employed in regulatory and quality-control settings for elemental impurity analysis in pharmaceutical and industrial materials (as per Indian Pharmacopoeia Sect. 2.4.26 and 2.4.2). The details of the reagents and consumables are provided in Supplementary Tables S1 and S2.
Various concentrations viz. 5%, 10% and 15% of WHG were analyzed for potency and bioactivity.
In vitro analysis of wound healing gels
Growth factor analysis by ELISA
Analysis of growth factors and cytokines such as HGF, G-CSF, TIMP-2 and IL-6 by antibody-based growth factor assay was performed as per manufacturer’s instructions (Raybiotech, elabscience). The OD was read at 450 nm (Thermo Labsystems; Ascent Software, US). Calibration curves were plotted based on increasing concentrations of standard growth factors provided in the kit and run simultaneously as positive control. Blank was kept as negative control. The test values were calculated from the standard curve.
Protein content and anti- oxidant activity
Protein content and antioxidant activity of MSC-CM formulations were analyzed by biuret and DPPH (2,2-diphenyl-1-picrylhydrazyl) assays, respectively as previously reported23. In brief, total protein content was estimated by the Biuret assay. The Biuret reagent was added to the test sample in the ratio 2:1. Duplicate samples were incubated in the dark for 30 min in 98-well plates. Bovine Serum Albumin was used as the reference standard. The plates were read at 570 nm in a spectrophotometer (Thermo Labsystems; Ascent Software, US). Assays were conducted as 3 independent experiments for validation. The data was expressed as a mean ± standard deviation (SD).
The free radical scavenging activity was assayed by the DPPH assay spectrophotometrically as per the methods described earlier24,25. A 1:1 solution of the 100% sample was mixed with a 9 × 10− 5 M solution of DPPH in methanol. After incubation at room temperature in the dark for 30 min, the OD was read at 570 nm (Ascent Software, Thermo Labsystems; US). Positive control L-Ascorbic acid, appropriate blanks, and controls were run concurrently.
The calculation was as follows:
Biocompatibility assay
For this experiment human keratinocyte (HaCaT) cell line was provided by ACTREC, Anti-cancer drug screening facility (ACDSF) and Normal Human Dermal Fibroblast (NHDF) cell line was procured from Promocell GmbH (Cat. no.: C-12302). The cells were cultured at 37 °C and 5% CO2 in DMEM and Fibroblast media respectively.
Cytocompatibility with human keratinocyte (HaCaT) and Normal Human Dermal Fibroblast (NHDF) cell lines was carried out using 3 different batches of 10% WHG and assessed by the Sulforhodamine B (SRB) method26. The assay was repeated for validation in 3 independent experiments. The effect of the samples was studied over 24 h in 96-well tissue culture plates using four concentrations ranging from 2.5% to 10%. The positive control Sodium dodecyl sulphate at concentrations ranging from 0.005% to 0.02% (HaCaT) and 0.001% to 0.004% (NHDF) were used. At the end of 24 h, cells were fixed, stained and the absorbance was measured at 570 nm using a plate reader (Ascent Software, Thermo Labsystems; US).
In vitro bacterial reverse mutation test
This was carried out as per the OECD 471-compliant Bacterial reverse mutation test using the plate incorporation method to assess the mutagenic potential of the 15% WHG across five Salmonella typhimurium bacterial strains (TA98, TA100, TA102, TA1535, TA1537) which are histidine auxotrophs. The study consisted of a Preliminary Cytotoxicity Assay (0.0499–5 µL/plate with ± 5% S9 rat liver mix) and a Confirmatory Mutation Test (0.3125–5 µL/plate with ± 10% S9 mix), both conducted in triplicate with appropriate vehicle and strain-specific positive controls (e.g., for absence of S9 - Sodium azide (20 µg/plate), 2-Nitrofluorene (25 µg/plate), 9-Aminoacridine (50 µg/plate), Mitomycin-C (0.25 µg/plate), and for presence of S9–2-Aminoanthracene (20 µg/plate)) with 48 h incubation. Acceptance criteria included valid control performance within historical ranges and predefined fold-increase thresholds (≥ 2-fold for TA98/TA100/TA102; ≥3-fold for TA1535/TA1537) to determine mutagenic potential.
In vitro mammalian chromosomal aberration test
This was carried out according to the in vitro mammalian chromosomal aberration test (OECD No. 473) in CHO-K1 (Chinese hamster ovary cell line) obtained from American Type Culture Collection (ATCC). The cells were cultured in RPMI – 1640 at 37 °C and 5% CO2. to evaluate the 15% WHG both with and without metabolic activation (rat liver S9). Concentrations up to 2 µL/mL were tested across Phase I (short-term treatment of 3 h exposure with recovery period of 20–21 h) and Phase II (continuous exposure for 24 h) before harvesting in absence of S9. Endpoints included Relative Increase in Cell Counts (RICC) as a measure of cytotoxicity and frequency of structural and numerical chromosome aberrations with appropriate positive controls (presence of S9 - Cyclophosphamide monohydrate; Absence of S9 – Mitomycin C) and vehicle controls (Distilled water). Approximately 300 metaphases per concentration were analyzed for structural aberrations (chromosomes and chromatids). Acceptability criteria for concurrent positive controls were the induction of responses that are compatible with those generated in the historical positive control data base and produced a statistically significant increase compared with the concurrent negative control.
In vivo evaluation of wound healing gels
The safety and efficacy study studies were approved by the Institutional Animal Ethics Committees. Animals were sourced from Global Bioresearch Solution Pvt. Ltd.(CCSEA reg no.−1899/PO/Bt/S/16/CCSEA). The animals were sacrificed humanely by euthanasia using carbon dioxide inhalation method at termination. Biosafety waste disposal methods were followed as per institutional biomedical waste management guidelines.
All surviving animals were used for the analyses except where stated.
In vivo repeated dose 28-day oral toxicity study
This was carried out by JDM Research Centre in compliance with the Principles of Good Laboratory Practices (GLP) as per the test guidelines of the ‘New Drugs and Clinical Trials Rules’, Ministry of Health and Family Welfare, Government of India and OECD guideline No. 407 in rats and mice. Animals were sourced from Global Bioresearch Solution Pvt. Ltd.(CCSEA reg no.−1899/PO/Bt/S/16/CCSEA). The animals were sacrificed humanely by euthanasia using carbon dioxide inhalation method at termination. Isoflurane (saturated chamber) was used for anesthesia. Biosafety waste disposal methods were followed as per institutional biomedical waste management guidelines.
The treatment groups included G1 vehicle-control (VC), G2 low dose (5%), G3 mid I dose (10%), G4 mid II dose (15%) and G5 high dose (100% active). The recovery groups included G1R (vehicle control recovery) and G5R (100% active recovery). During acclimatization, animals were given temporary animal number using permanent marker pen and cage cards. Animals were randomized on the basis of body weight using spread sheet and grouped, as mentioned in study design, ensuring that they were within ± 20% of the mean body weight of respective sex. After randomization, animals were allocated to different groups and given identification by permanent tattoo marking on tail using tattoo machine and study cage card detailing study no, cage no, sex, animal no, species, group, dose, start/end date and sign & date. All animals were dosed once daily for 28 days (i.e. G1 to G5, G1R and G5R) followed by 14 days recovery period for G1R and G5R group animals. Animals were observed twice daily for mortality and morbidity, throughout the study period and once daily for clinical observation such as ophthalmological examination, body weight, and feed consumption. Body weights were taken at the time of animal receipt, before randomization, on day 1 before dosing, weekly thereafter and on the day of necropsy. Body weights on day of necropsy were used to calculate relative organ weight. At the end of treatment and recovery periods, clinical parameters - hematology, biochemistry, urine analysis, and histopathological studies were performed.
In vivo repeated dose 28-day oral toxicity study in Wistar rats
For the study, 47 male and 47 female Wistar rats were selected for the study and acclimatized for 7 days at Research Animals Resources. The animals were 8 to 9 weeks at the time of dosing. Healthy young adult animals were selected. The females were nulliparous and non-pregnant. 42 males and 42 females were taken in the study after randomization and divided into seven groups with each group having 6 animals/sex. Two to three animals were housed during the acclimatization in polycarbonate cages of size 43 × 29 × 20 cm (LxBxH) with provision of feed and water bottle holding facility. During treatment and recovery period, maximum of two animals/cage were housed in polycarbonate cage with provision of feed and water bottle holding facility. Cage rotation was done weekly.
In vivo repeated dose 28-day oral toxicity study in Swiss albino mice
For the study, 92 male and 92 female mice were selected for the study and acclimatized for 6 days for male and 7 days for female animals at Research Animals Resources. The animals were 7 to 8 weeks at the time of dosing. Healthy young adult animals were selected. The females were nulliparous and non-pregnant. 84 males and 84 females were used in the study after randomization and divided into seven groups with each group having 12 animals/sex. Five animals/cage/sex (except last cage having two animals/cage/sex) were housed in polycarbonate cage with provision of feed and water bottle holding facility during acclimatization period. During treatment and recovery period, three animals/cage/sex were housed. Cage rotation was done weekly. The cage size was 29 × 22 × 14 cm (LxBxH).
Evaluation of efficacy of MSC-CM formulations for prevention/mitigation of chemotherapy induced oral mucositis
The study was carried out in Wistar rats at sa-FORD, Taloja, district Raigad, Navi Mumbai,
India. For Phase I pilot study, animals in-bred at sa-FORD.(CCSEA reg no.−1256/PO/Bt/Rc/S/09/CPCSEA) were used and for the Phase II efficacy study animals were sourced from Global Bioresearch Solution Pvt. Ltd.(CCSEA reg no.−1899/PO/Bt/S/16/CPCSEA). The animals were sacrificed humanely by euthanasia using carbon dioxide inhalation method at termination. Isoflurane (saturated chamber) was used for anesthesia. Biosafety waste disposal methods were followed as per institutional biomedical waste management guidelines.
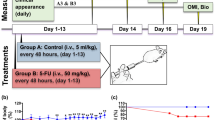
The animals were 8 to 9 weeks at the time of dosing. Healthy young male adult animals were selected. The pilot study in a rat model of OM was standardized using 5-fluorouracil (5-FU) a chemotherapy agent and acetic acid as previously reported27,28,29,30. In brief, the 5 groups included intravenous doses of either saline (control) 20 or 40 mg/kg 5-FU on days 1, 3, 5 and 7. Acetic acid at 20% or 25% was injected on Day 1 (in different combinations to groups G2-G5) in the right buccal mucosa. The animals were weighed daily from day 1 till day 8 (terminal sacrifice). Rats were observed daily for mortality/morbidity and clinical signs. At terminal sacrifice the sizes of ulcer were measured using digital vernier caliper and the severity was recorded. The final doses for inducing oral ulcer were selected as intravenous 20 mg/kg 5-FU dose at 2 ml/kg and buccal injections of 25% acetic acid at 60 µl/animal.
For the efficacy study, a total of 48 male rats were randomized into 6 groups of 8 animals each. Except G1, which was the normal control all the animals were induced with oral ulcers as described above. The groups included G1 (normal control), G2 (vehicle control), G3 (positive control - Placentrex gel), G4, G5 and G6 received 5%, 10% and 15% of WHG respectively at 0.1 ml/100gm body weight. Randomization was performed one day prior to the first day of treatment, based on the most recently recorded body weight. The animals were allocated to different test groups by a manual randomization method as per the internal SOP of sa-FORD. Individual body weights of allocated animals were within ± 20% of the group means. Details of the randomization were documented in the Raw Data. The rats were housed in groups (2–3 animals per cage). Rats were observed daily for mortality/morbidity and clinical signs. Blood was collected from all the surviving animals on days 0 (pre-treatment), 7 and 16 (terminal sacrifice) for hematology, serum biochemistry, and estimation of inflammatory markers Interleukin-10 (IL-10), Interleukin-17 (IL-17) and nuclear factor κB (NF-κB) by ELISA. On termination at Day 16, scoring of ulcers, gross observation was done and the sizes of ulcer were measured using digital Vernier caliper. The buccal mucosa (left and right) was collected for histopathological and microscopic evaluation. For all end-point analyses including serum analyses, ulcer measurements, histopathology and microscopic observations, samples were coded blind to avoid observer’s bias.
Evaluation of efficacy of MSC-CM formulations for prevention/mitigation of radiotherapy induced toxicity
This study was carried out at Advanced Centre for Treatment, Research and Education in Cancer (ACTREC), Navi Mumbai, India. All procedures involving mice were performed according to protocols approved by the Institutional Animal Ethics Committee, ACTREC, Tata Memorial Centre, Navi Mumbai (Proposal # 46/2022) and were adhered to CCSEA guidelines (Registration Number: 65/GO/ReBiBt/S/99/CCSEA). Six to eight-week-old, in-house bred BALB/c mice were included. Animals were purchased from Jackson Laboratories, USA and bred inhouse. Animals that were aged above and below six to eight weeks, body weight, and did not satisfy the wound size criteria were excluded from the study. Animals received humane care and all efforts were undertaken to minimize animal suffering before and during the experiments. During treatment and recovery period, upto six animals/cage/sex per group were housed. Cage rotation was done weekly or as and when required within a week. The cage size was 36.6 × 16 × 18 cm (LxBxH).
Humane endpoints were that mice will be sacrificed when (i) mice will have respiratory distress, (ii) mice have > 20% body weight loss and (iii) mice show difficulty in feeding and walking.
A pilot study was conducted using 7 BALB/c mice which are divided into 3 groups as Group1 (control, wounded, no radiation), Group2 (wounded, radiation, no treatment), Group3 (wounded, radiation, 10% WHG treatment). Group 2 and Group 3 mice were irradiated with 3 Gy/day/3 days (9 Gy/mice). WHG was applied to Group 3 twice/day during the experimental period. The body weights, wound measurement, scoring of the wound healing parameters like oozing, redness, hair loss were observed. Wound was also studied for 18FDG glucose uptake by PET-CT imaging.
A second pilot study was carried out for standardizing the radiation protocol and methods on 2 groups (n = 2/group), WHG treated and untreated and concluded to give 5 Gy as the highest dose.
For Wound Healing Efficacy testing, a sample size of eight animals per group was considered as per the NCI criteria, the minimum required to allow statistical significance. A total of 24 BALB/c male mice were used in this study. Before the start of the study, all mice were randomized into three desired groups (n = 8.) Group 1 control group, subjected to wounding, radiation, and receiving no treatment, while Group 2 underwent the same interventions but received 10% WHG treatment, and Group 3 received 15% WHG treatment. A circular wound of 15–20 mm diameter was inflicted by cutting the full thickness of the skin on the dorsum. All mice received a 5 Gy radiation dose using a calibrated Precision X-ray irradiation machine- xRAD 225 KV. Twenty-four hours post-irradiation, Groups 2 and 3 received 200 mg treatment of WHG twice-daily for 14 days.
Endpoint measurement
Comprehensive monitoring included body weights, wound measurements, PET-CT scans, and wound assessments of oozing, redness, and hair loss, was conducted at specified intervals. The wound size was measured through standardized photography and transparent paper tracing method. Analysis of soluble factors that modulate inflammation and wound repair including Thymus and activation-regulated chemokine (TARC), Interleukin-6 (IL-6), Interleukin-20 (IL-20), Interferon gamma (IFN-γ), Monocyte Chemoattractant Protein-1 (MCP-1) and Interleukin-17 (IL-17 A/F) in plasma samples from all groups was carried out using Luminex® Multiplex Assay (Bio-Plex 200 Systems, Bio-Rad).
Randomization and blinding
A simple randomization method of randomly grouping the animals was adopted. Study groups were assigned codes as Group 1, Group 2 and Group 3. The blind was maintained throughout the study and decoding was done after data analysis.
Post experimentation procedure
After the study period was over, the animals were sacrificed by euthanasia using carbon dioxide inhalation method. Further, all sacrificed animals were collected in biohazard bags and disposed as per institutional biomedical waste management guidelines.
Statistical analysis
The data was subjected to statistical analyses using Sigma Plot version 15 (2022) and Graph pad prism 10.4.1 (2024). The mean and standard deviations were calculated using the software. All continuous data were checked for their homogeneity using Shapiro-Wilk test.
All homogenous data were analyzed using ANOVA and data showing significance in their variances were subjected to Dunnett’s t-test and the data from different time points was analyzed using paired t-test. P values of less than or equal to 0.05 were accepted as being statistically significant.
Results
In vitro analysis of wound healing gels
WHG was characterized for physico-chemical properties viz. appearance, pH, sterility and presence of heavy metals. The appearance of WHG was colorless to yellowish, with pH ranging between 5 and 7. In microbiology assessment of WHG the microbial load was found to be within the specified limits for a topical pharmaceutical product. Heavy metal testing for lead and arsenic showed levels were below the limits of quantification.
Growth factor and cytocompatibility data
There was dose-dependent increase in the levels of growth factors (HGF and GCSF (Fig. 1a)) and cytokine (TIMP-2 and IL-6 (Fig. 1b)) of the WHG (10% and 15%). Further, there was also a dose-dependent increase in the protein content (Fig. 1c) of the formulations, however the anti-oxidant activity (Fig. 1d) of the 10% and 15% WHG remained the same. The biocompatibility evaluation of the 3 different batches of 10% WHG on HaCaT (Fig. 1e) and NHFD (Fig. 1f) cell line showed that the cell growth of these cells was not affected significantly at all the concentrations (2.5% to 10%) tested, thus indicating cytocompatibility.
In-vitro evaluation of 10% and 15% WHG. (a) Analysis of the 10% and 15% WHG revealed the presence of HGF and G-CSF. (b) IL-6 and TIMP-2. (c) Average protein content. (d) Average anti-oxidant activity. (e-f) No IC50 could be obtained in HaCaT and NHDF cell lines for the 10% WHG.
Genotoxicity analysis
Bacterial reverse mutation test (ames test)
Cytotoxicity (lawn inhibition) was observed at the maximum tested concentration (5 µL/plate) across all bacterial strains under both metabolic activation conditions. Neither the Preliminary Cytotoxicity Assay nor the Confirmatory Mutation Test showed biologically significant increases in revertant colonies, with fold-increases remaining consistently low (1.01–1.46) across all strains and conditions. Vehicle controls performed within acceptable historical ranges while positive controls yielded expected robust increases, confirming assay sensitivity, S9 functionality, and sterility checks were satisfactory. Thus the 15% WHG did not induce point gene mutations at the histidine locus in Salmonella typhimurium tester strains, either directly or after metabolic activation. (Supplementary Table S3)
In vitro mammalian chromosomal aberration test
Chromosomal aberration test conducted on CHO-K1 cells treated with 15% WHG demonstrated minimal toxicity, with RICC values remaining in acceptable ranges. A single statistically significant rise was observed at the highest concentration in the presence of S9 during Phase I and was not consistently observed in Phase II or under other conditions (Supplementary Table S4). Positive controls validated the assay. No significant increase in the mean percent aberrant cells in relation to vehicle control was observed across Phase II concentrations.
Based on the results, 15% WHG did not induce chromosomal aberrations up to the concentration of 2 µL/mL in CHO-K1 cells under the specified experimental conditions. This suggests a low potential for mutagenicity.
Safety studies (in vivo evaluation of wound healing gel)
In vivo repeated dose 28-day oral toxicity study in wistar rats
There were no mortality, morbidity, or abnormal clinical observations either in the treatment groups G1 to G5 or the recovery groups G1R and G5R. (Supplementary Tables S5 and S6)
There was no statistically significant difference in the body weight, percentage change in body weights and feed consumption observed in both sexes of treatment groups as compared to the control group.
Ophthalmological examination, urine analysis and Functional Observation Battery including neurobehavioral observation, sensory observation and activity measurement were normal.
Grip Strength assessment and Foot Splay measurement were performed in the last week of treatment phase for treatment groups and recovery phase for recovery groups. There was no statistically significant difference observed in the Grip Strength assessment and Foot splay measurement of all animals of both sexes as compared to respective control group animals, except a statistically significant decrease in foot splay measurement was observed in female (G4) in comparison with control group female (G1) However, in Grip Strength assessment, there was statistically significant increase in Hindlimb observed in mid Il dose treatment group female (G4) and similarly in hindlimb and forelimb was observed in high dose recovery group female (G5R) in comparison with respective control group female animals. However, all these observations were considered incidental and not attributed to test item.
Although some statistically significant changes were noted in hematology, biochemistry, and organ weights, these findings were deemed incidental, and no dose response could be established (Supplementary Tables S7-S10). Gross and microscopic observations showed that some changes were spontaneous and within expected biological variation (Supplementary Tables S11-S12).
In organ weight, there was no statistically significant in absolute and relative organ weights except statistically significant decrease in absolute organ weight of heart in high dose (G5R) males.
However, all the above observations were not treatment related and lacked consistency between both the sexes. Therefore, all findings are considered incidental or spontaneous in nature, and the test item was well tolerated under the conditions of this study.
In vivo repeated dose 28-day oral toxicity study in Swiss albino mice
Detailed evaluations across the different treatment groups (G1–G5) and recovery groups (G1R, G5R) revealed no mortality/morbidity or abnormal clinical signs (Supplementary Tables S13-S14). Administration of the test item produced no treatment-related effects on body weight, feed consumption, ophthalmology, neurobehavioral parameters, hematology, clinical chemistry, organ weights, gross pathology, histopathology, or estrus cycle evaluation in either sex across all treatment and recovery groups. Although some statistically significant changes were noted in hematology, biochemistry, and organ weights, these findings were deemed incidental, and no dose response could be established (Supplementary Tables S15-S18). Gross and microscopic observations showed that some changes were spontaneous and within expected biological variation (Supplementary Tables S19-S24). Overall, there were no treatment-related adverse effects.
Efficacy studies (in vivo evaluation of wound healing gel)
Evaluation of efficacy of MSC-CM formulations for prevention/mitigation of chemotherapy induced oral mucositis
There was successful induction of chemo-induced oral ulcers in severely immunosuppressed model. Regarding mortality/morbidity, no deaths occurred until day 7 (post-ulcer induction). Thereafter, mortality was observed in G2 (1), G3 (2), G4 (2), and G5 (2), with additional moribund animals in G4 and G5 in which the induced ulcers resulted in inflammation of the entire cheek, exposing the oral cavity and hence were sacrificed on ethical grounds. No mortality was noted in G6 (15% wound healing gel) (Supplementary Tables S25 and S26). Administration of 5-FU induced oral and gastric ulcers with reduced feed intake resulting in significant body weight loss. Higher feed intake was noted in G2, G5, and G6 compared to G3 and G4. The body weight was stable in early induction phase, but significant reduction occurred in all 5-FU–treated groups (G2–G6) compared with normal control (G1) from induction onwards. (Supplementary Table S27 and Fig. S1). During treatment, weight loss persisted but was less pronounced in G2 and G6 than in G3, G4, and G5 (Supplementary Table S28, Fig. S2).
Figure 2 shows the ulcer sizes (Fig. 2a) and healing potential (Fig. 2b) of the MSC-CM formulations. The microscopic lesions of buccal mucosa in all treatment groups along with their severity in percentage are given in Table 1. The observed lesions were moderately severe in G2 and G3 group animals compared to other groups. The treatment groups with 5% (G4), 10% (G5), and 15% (G6) group animals showed regeneration of tissue. However, G6 showed the maximum healing effect where the ulcers of 3 animals were found completely healed on day 16 (terminal sacrifice) (Fig. 3; Supplementary Table S29 and Fig. S3 & S4). Histopathological comparison across Groups 1–6 confirmed that , G1 (normal control) had a normal healthy mucosal epithelium (Fig. 3a). Progressive transition from epithelial and connective tissue disruption was evident in G2 which was the untreated control (Fig. 3b). These sections also demonstrated mucosal hyperplasia and blisters caused due to chemotherapy agent as an irritant. G3 which was the positive control demonstrated active fibroblastic repair and early collagen deposition in (Fig. 3c). This was followed by organized collagen remodeling and neovascularization in G4 which was the 5% WHG treatment group (Fig. 3d), restoration of normal epithelial architecture in G5, 10% WHG (Fig. 3e), and finally mature connective tissue reinforcement with surface protective adaptation in G6, 15% WHG (Fig. 3f). The findings indicated dose-dependent enhancement of mucosal healing without evidence of pathological fibrosis.
Efficacy of WHG in chemo-induced oral ulcer healing. (a) Shows the severity of ulcers recorded was markedly reduced in the 10% WHG (G5) and 15% WHG (G6) treatment groups in a dose dependent manner. (b) The healing potential of WHG.
Representative H & E stained photomicrographs of rat buccal mucosa from G1 to G6. . (a) G1 (control group) showing normal histological structure of stratified squamous epithelium (red arrow) with broad, regular and short epithelial ridges (green arrow), densely packed collagen fibers of lamina propria, and uniform layer of stratum corneum (black arrow). (b) G2 (Vehicle group) showing hyperplasia of stratum spinosum (red arrow). The lamina propria consists of blood vessels (black arrow). (c) G3 (positive control- Placentrex gel) showing immature fibroblasts (black arrow), various sized collagen bands (green arrow) and a band of thick collagen replacing a lesion (yellow arrow). (d) G4 (5% WHG) showing intact uniform layered mucosal epithelium (black arrow), strong vasculature (white arrow), immature fibroblasts generation (yellow arrow), invading the thick band of collagen of a lesion (green arrow), bands of thin collagen formation (yellow arrow). (e) G5 (10% WHG) showing stratified squamous epithelial lining (white arrow) of the buccal pouch with prominent rete ridges (dual arrow) extending into the lamina propria. Single basal layer of cuboidal to columnar cells adjacent to the basement membrane (green arrow). (f) G6 (15% WHG) showing normal histological structure of stratified squamous epithelium (black arrow), with broad epithelial ridges (white arrow), densely packed collagen fibers of lamina propria (yellow arrow), and increased layer of stratum corneum (green arrow). (Magnification, 100X).
Biochemical changes which showed elevated levels of ALT, ALP, GGT, altered urea, BUN, electrolytes, and albumin, indicated 5-FU–induced hepatic and renal toxicities, while hematological findings confirmed anemia and thrombocytopenia. These toxicities showed recovery by Day 16, particularly in G5 and G6 and indicates mitigation of systemic toxicity. Serum cytokine analysis for inflammatory markers revealed downregulation IL-17 in treated groups (G3, G5, G6) by Day 7 (Fig. 4a), demonstrating an anti-inflammatory effect, while IL-10 remained unchanged (Fig. 4b) and NF-κB was below detectable levels (data not shown).
Serum cytokine analysis in rats with chemotherapy-induced oral ulcers. (a) IL-17 showed no significant difference on days 0 and 16 between the groups. However, on day 7 of treatment period, there was significantly lower expression in group 3 (p < 0.05), group 5 (p < 0.001) and group 6 (p < 0.0001) animals. (b) IL-10 showed no significant difference across groups or time-points.
Other observations included hepatotoxicity, renal toxicity, anemia, and thrombocytopenia in ulcer-induced groups (G2–G6). There was increased severity in oral ulcers in G2–G4, but wound size and severity were significantly reduced in G5 and G6, proving a protective role of MSC-CM based formulations.
Overall, MSC-CM based formulations demonstrated a dose-dependent protective and therapeutic effect in reducing chemotherapy-induced oral mucositis and associated systemic toxicities.
Evaluation of efficacy of MSC-CM formulations for prevention/mitigation of radiotherapy induced toxicity
The results highlighted the efficacy of 10% WHG at Day 7 in promoting wound healing (Fig. 5a). Stability in body weight indicated that WHG at 10% and 15% were safe for topical administration over period of experimental duration (Fig. 5b). Later, mice in all groups achieved healing. The WHG-treated groups (both 10% and 15%) appeared to show improvements in reducing redness, hair loss, wound wetness, and oozing compared to the control group over the 14-day period (Fig. 6). In vivo studies showed dose dependent increase in serum levels of IL-20 and IL-17/A/F in the treatment groups compared to others (Fig. 7). The levels of cytokines TARC, IL-6, IFN-γ and MCP-1 were not statistically significant (data not shown). Figure 8 shows the healing of representative animals from the 3 groups over the study period (Supplementary Fig. S5). The PET-CT scans corroborated with visual wound closure kinetics data (Supplementary Fig. S6).
Efficacy of WHG in radiation-induced wounds. (a) Shows the wound healing kinetics. (b) Animal body weight.
Effect of WHG on clinical score improvement in wound healing. (a) Redness, (b) hair loss, (c) wound wetness, and (d) oozing score were reduced compared to the control group over the 14-day period.
Plasma cytokine analysis in mice with radiation-induced wounds. (a) Cytokine profiling for IL-20 and (b) Cytokine profiling for IL-17 A/F.
Progression of healing for the 3 groups. Shows healing of representative animals from the 3 groups over the 14-day study period.
Discussion
Wound healing modalities can be classified into either conventional or regenerative approaches. The conventional treatments include wound dressing or use of split thickness skin grafts, while the regenerative methods include the use of bioactive biomaterial, stem cell therapy, gene therapy or bioengineered skin graft for skin healing31. The regenerative approach has shown good results in accelerating the process of wound healing and reducing scar tissue formation32but wound healing for OM has not been reported. In this context, we attempted to create chemo induced ulcers and radiation wound models to evaluate the efficacy of conditioned media based WHG for wound healing.
The growth factor profiling of the WHG formulations revealed the presence of critical factors such as HGF, G-CSF, IL-6 and TIMP-2, all of which are implicated in wound healing and tissue regeneration via immunomodulation33,34,35,36. Soluble mouse IL-20 and IL-17 A/F were distinctly found in plasma of WHG treated mice with radiation injury and their increase was dose dependent. These cytokines are known to be associated with repair and regeneration as per previous reports37,38,39. IL-20 is known to play crucial role in repair of epithelial tissues, including the oral mucosa, by enhancing immune responses and supporting the regeneration of keratinocytes. There is emerging evidence for interplay between IL-17 and IL-20 signaling to maintain oral epithelial surface integrity and promote wound healing for maintaining homeostasis37. Separately we had observed distinct levels of angiogenic growth factors, predominantly angiopoietins, VEGF, PlGF and bFGF in main ingredient of UC-MSC-CM (data not shown, unpublished observations). All these factors could have been mechanistically responsible for healing properties observed in WHG treated wounds. The in vitro cytotoxicity testing did not achieve inhibitory concentrations and the genotoxicity testing of the 15% WHG showed no clastogenic effect, strongly supporting a favorable genotoxicity safety profile for the intended therapeutic use.
The 28-day repeated oral toxicity studies in rats and mice established that administration of the MSC-CM formulations across treatment and recovery groups produced no treatment-related adverse effects in clinical, functional, hematological, biochemical, urinalysis, organ weight, gross, or microscopic evaluations. Observed findings were minimal, lacked consistency or dose-response relationships, and were considered incidental or spontaneous in nature.
Accordingly, the NOAEL (No Observed Adverse Effect Level) was observed at the highest dose tested (G5) which was the 100% MSC-CM and the LOAEL (Lowest Observed Adverse Effect Level) could not be identified, as no treatment-related adverse effects were observed at any dose.
The efficacy studies in the OM model showed dose-dependent wound healing of the formulations at 5%, 10% and 15%, which included reduced ulcer size and lesion severity, regeneration of mucosal epithelium, and improvement of hematological, biochemical, hepatic, renal, and immune parameters disrupted by 5-FU. The 15% formulation achieved superior efficacy, with 100% survival and a mean ulcer healing score of 71.9%.
In the radiation wound model, both the 10% and 15% WHG were safe and well tolerated, reducing inflammation and accelerating wound closure. Notably, the 10% gel supported early wound healing, with significant improvements by Days 4 and 7.
Chemotherapy-induced oral mucositis (OM) and impaired wound healing due to radiation are major clinical complications that compromise quality of life and limit treatment adherence in cancer patients. No approved regenerative treatments exist for OM beyond the use of Palifermin, entailing a single growth factor, for HSCT patients40and is also restricted by cost and parenteral administration. Current options such as mouthwashes and analgesics offer only transient relief without disrupting disease prevention or progression41.
While there are reports about the use of MSCs and MSC-CM for wound healing42,43,44,45, to the best of our knowledge this is the first instance of a topical gel being developed specifically for OM. A 10% topical formulation containing the secretome of human umbilical cord mesenchymal stem cells was shown to be efficacious in promoting healing in a phase 2 clinical trial for chronic ulcers caused by diabetes and leprosy. This study showed that the gel used did not cause any local or systemic adverse effects46. Our findings support the preventive and ameliorative potential of MSC-CM based topical formulations, particularly 10% and 15%, as promising candidates for management of chemotherapy and radiation induced toxicities by promoting tissue repair. Importantly, the safety profile was favourable, with no genotoxic, tumorigenic, or systemic toxic effects observed. Pharmacokinetic studies could not be carried out due to the biological complexity of MSC-CM. Another limitation was that the molecular mechanisms of wound healing via gene expression studies were not investigated as they were beyond the scope of the study.
Conclusion
WHG revealed concentration-dependent efficacy in chemotherapy-induced oral mucositis and radiation wound models. This is the first topical MSC-CM gel developed specifically for oral mucositis, delivering a regenerative secretome with multiple growth factors rather than a single growth factor, with demonstrated efficacy across both chemotherapy- and radiation-induced tissue injuries. The formulation offers a safe, non-invasive, and clinically feasible regenerative therapy that may improve healing, possibly through cytokine-mediated immune-assisted wound repair, reduce symptom burden, and enhance treatment compliance in cancer patients, addressing a major unmet clinical need.
Future work is warranted on elucidating the molecular mechanisms driving MSC-CM–mediated wound healing and optimizing dose and treatment regimens. Our data has provided interesting leads that MSC-CM based WHG offers a physiologically relevant, cell-free alternative to MSC transplantation, overcoming regulatory and logistical challenges associated with live cell therapies. Its topical gel formulation provides a convenient and patient-friendly delivery format. This makes a strong case for the advancement of MSC-CM formulations for clinical testing.
Data availability
The data that support the findings of this study are available upon reasonable request.
References
Saeed, H. et al. Mesenchymal stem cells (MSCs) as skeletal therapeutics–an update. J. Biomed. Sci. 23 (1), 41 (2016).
Dominici, M. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy 8 (4), 315–317 (2006).
Mebarki, M., Abadie, C., Larghero, J. & Cras, A. Human umbilical cord-derived mesenchymal stem/stromal cells: a promising candidate for the development of advanced therapy medicinal products. Stem Cell. Res. Ther. 12 (1), 152 (2021).
Nagamura-Inoue, T. Umbilical cord-derived mesenchymal stem cells: their advantages and potential clinical utility. World J. Stem Cells. 6 (2), 195 (2014).
Song, N., Scholtemeijer, M. & Shah, K. Mesenchymal stem cell immunomodulation: mechanisms and therapeutic potential. Trends Pharmacol. Sci. 41 (9), 653–664 (2020).
Kolios, G. & Paspaliaris, V. Mesenchyme stem cell-derived conditioned medium as a potential therapeutic tool in idiopathic pulmonary fibrosis. Biomedicines 10 (9), 2298 (2022).
Liang, X., Ding, Y., Zhang, Y., Tse, H. F. & Lian, Q. Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell. Transpl. 23 (9), 1045–1059 (2013).
Ivanova, G., Pereira, T., Caseiro, A. R., Georgieva, P. & Maurício, A. C. Metabolomic and Proteomic Analysis of the Mesenchymal Stem Cells’ Secretome (InTech eBooks, 2016).
Linero, I. & Chaparro, O. Paracrine effect of mesenchymal stem cells derived from human adipose tissue in bone regeneration. PLoS ONE 9(9), e107001 (2014).
Li, X., Zhang, D., Yu, Y., Wang, L. & Zhao, M. Umbilical cord-derived mesenchymal stem cell secretome promotes skin regeneration and rejuvenation: from mechanism to therapeutics. Cell. Prolif. 57(4), e13586 (2023).
Mathen, C., Sawant, M. G., Gupta, R., Dsouza, W. & Krishna, S. G. Evaluation of potential application of wharton’s jelly-derived human mesenchymal stromal cells and its conditioned media for dermal regeneration using rat wound healing model. Cells Tissues Organs. 210 (1), 31–44 (2021).
Md Fadilah, N. I. et al. Cell secretomes for wound healing and tissue regeneration: next generation acellular based tissue engineered products. J. Tissue Eng. 13, 20417314221114273 (2022).
Jo, H. et al. Applications of mesenchymal stem cells in skin regeneration and rejuvenation. Int. J. Mol. Sci. 22 (5), 2410 (2021).
Yin, S., Ji, C., Wu, P., Jin, C. & Qian, H. Human umbilical cord mesenchymal stem cells and exosomes: bioactive ways of tissue injury repair. Am. J. Transl Res. 11 (3), 1230–1240 (2019).
Al-Masawa, M. E. et al. Efficacy and safety of small extracellular vesicle interventions in wound healing and skin regeneration: A systematic review and meta-analysis of animal studies. Theranostics 12 (15), 6455–6508 (2022).
Kusindarta, D. L. et al. Human umbilical mesenchymal stem cells conditioned medium promote primary wound healing regeneration. Vet. World. 9 (6), 605–610 (2016).
Wang, F. et al. Skin-inspired elastomer-hydrogel Janus fibrous membrane creates a superior pro-regenerative microenvironment toward complete skin regeneration. Biomater. Adv. 170 (11), 214227 (2025).
Xu, L. et al. Double-sided protector Janus hydrogels for skin and mucosal wound repair: applications, mechanisms, and prospects. J. Nanobiotechnol. 23 (1), 387 (2025).
Shen, P. et al. Genetically engineered MSC-derived hybrid cellular vesicles for ROS-scavenging and mitochondrial homeostasis in hepatic ischemia-reperfusion injury. Mater. Today Bio. 34, 102215 (2025).
Lu, H. et al. Protein-based functional hydrogel improves cutaneous nerve repair and diabetic wound healing. Nano Res. 18 (1), 94907012 (2025).
Hendrawan, S. et al. Wound healing potential of human umbilical cord mesenchymal stem cell conditioned medium: an in vitro and in vivo study in diabetes-induced rats. Vet. World. 14 (8), 2109–2117 (2021).
Mathen, C. & Dsouza, W. Xeno-free human wharton’s jelly mesenchymal stromal cells maintain their characteristic properties after long-term cryopreservation. Cell. J. 23 (2), 145–153 (2021).
Mathen, C. & Dsouza, W. In vitro and clinical evaluation of umbilical cord-derived mesenchymal stromal cell‐conditioned media for hair regeneration. J. Cosmet. Dermatol. 21 (2), 740–749 (2021).
Blois, M. S. Antioxidant determinations by the use of a stable free radical. Nature 181 (4617), 1199–1200 (1958).
Nia, R. et al. Evaluation of the anti-oxidant and anti-angiogenic effects of Sphenocentrum Jollyanum Pierre. Afr J. Biomed. Res 8(1) (2006).
Skehan, P. et al. New colorimetric cytotoxicity assay for Anticancer-Drug screening. JNCI J. Natl. Cancer Inst. 82 (13), 1107–1112 (1990).
Reithofer, M. & Jahn-Schmid, B. Allergens with protease activity from house dust mites. Int. J. Mol. Sci. 18 (7), 1368 (2017).
Takeuchi, I., Kawamata, R. & Makino, K. A rat model of oral mucositis induced by cancer chemotherapy for quantitative experiments. Anticancer Res. 40 (5), 2701–2706 (2020).
Idrus, E. et al. An experimental model of chemically-induced ulceration of the buccal mucosa of Mus musculus. Makara J. Health Res. 23, 181–187 (2019).
Shimamura, Y., Takeuchi, I., Terada, H. & Makino, K. A mouse model for oral mucositis induced by cancer chemotherapy. Anticancer Res. 38 (1), 307–312 (2018).
Kolimi, P., Narala, S., Nyavanandi, D. & Youssef, A. A. A. Dudhipala, N. Innovative treatment strategies to accelerate wound healing: trajectory and recent advancements. Cells 11 (15), 2439 (2022).
Liu, L. et al. Human umbilical cord mesenchymal stem cells transplantation promotes cutaneous wound healing of severe burned rats. PLoS ONE. 9 (2), e88348 (2014).
Bevan, D., Gherardi, E., Fan, T. P., Edwards, D. & Warn, R. Diverse and potent activities of HGF/SF in skin wound repair. J. Pathol. 203 (3), 831–838 (2004).
Huang, H. et al. Granulocyte-Colony stimulating factor (G-CSF) accelerates wound healing in hemorrhagic shock rats by enhancing angiogenesis and attenuating apoptosis. Med. Sci. Monit. 23, 2644–2653 (2017).
McFarland-Mancini, M. M. et al. Differences in wound healing in mice with deficiency of IL-6 versus IL-6 receptor. J. Immunol. 184 (12), 7219–7228 (2010).
Gill, S. E. & Parks, W. C. Metalloproteinases and their inhibitors: regulators of wound healing. Int. J. Biochem. Cell. Biol. 40 (6–7), 1334–1347 (2008).
Beute, J. E. et al. The IL-20RB receptor and the IL-20 signaling pathway in regulating host defense in oral mucosal candidiasis. Front. Cell. Infect. Microbiol. 26 (12), 979701 (2022).
Barada, O. et al. IL-20 cytokines are involved in the repair of airway epithelial barrier: implication in exposure to cigarette smoke and in COPD pathology. Cells 12 (20), 2464 (2023).
Saul-McBeth, J. et al. Tissue damage in radiation-induced oral mucositis is mitigated by IL-17 receptor signaling. Front. Immunol. 17 (12), 687627 (2021).
Panjwani, M. Efficacy of palifermin in the hematopoietic stem cell transplant setting. J. Adv. Pract. Oncol. 4 (2), 89–100 (2013).
Harris, D. J. Cancer treatment-induced mucositis pain: strategies for assessment and management. Ther. Clin. Risk Manag. 2 (3), 251–258 (2006).
Ayavoo, T., Murugesan, K. & Gnanasekaran, A. Roles and mechanisms of stem cell in wound healing. Stem Cell. Investig. 8, 4 (2021).
Kucharzewski, M. et al. Novel trends in application of stem cells in skin wound healing. Eur. J. Pharmacol. 843, 307–315 (2018).
Li, M. Mesenchymal stem cell-conditioned medium accelerates wound healing with fewer scars. Int. Wound J. 14 (1), 64–73 (2015).
Bormann, D., Gugerell, A., Ankersmit, H. J. & Mildner, M. Therapeutic application of cell secretomes in cutaneous wound healing. J. Investig Dermatol. 143 (6), 893–912 (2023).
Tan, S. T. et al. Effectiveness of secretome from human umbilical cord mesenchymal stem cells in gel (10% SM-hUCMSC Gel) for chronic wounds (diabetic and trophic ulcer) – phase 2 clinical trial. J. Multidiscip Healthc. 16, 1763–1777 (2023).
Acknowledgements
The authors wish to thank the infrastructural support from Laboratory Animal house and Small animal imaging facility. Authors acknowledge Aastha Singh for extending help with manuscript writing.
Funding
This work was supported by funding from OCT Therapies and Research Pvt Ltd. AK received salary from OCT TRPL funds.
Author information
Authors and Affiliations
Contributions
Caroline Mathen: Conceptualization, Study design, Methodology, Investigation, Supervision, Data curation, Validation, Writing – original draft, review & editingWilfrid Dsouza: Formal analysis, Methodology, Investigation, Supervision, Data curation, Validation, Writing –original draft, review & editingDeepali Sharma: Supervision, Investigation, Formal analysis, Data curation, review & editingTrupti Ninad Pradhan: Investigation, Data curation, review & final approvalApeksha Kesarwani: Methodology, Investigation, Data curation, review & final approvalDivya Gujar: Methodology, Investigation, Data curation, review & final approvalMangi Lal Choudhary: Investigation, Data curation, review & final approvalAishwarya Nithyanand: Investigation, Data curation, review & final approvalRutika Ghosalkar: Investigation, Data curation, review & final approvalArvind Ingle: Animal house support, review & final approvalPradip Chaudhari: Small animal radiation and imaging, review & final approvalJayant Sastri Goda: Small animal radiation, Methodology, Investigation, review & final approvalJyoti Kode: Conceptualization, Study design, Methodology, Investigation, Supervision, Data curation, Writing – original draft, review & editing.
Corresponding authors
Ethics declarations
Competing interests
Caroline Mathen is the Director of OCT Therapies and Research Pvt Ltd.The other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethics statements
1. The study approved by Institutional Animal Ethics Committee (IAEC) of the test facility under the project title "Repeated Dose 28 Day Oral Toxicity Study in Rat" on July 06, 2023 (JDM/IAEC/2022/03/115). 2. The study was approved by Institutional Animal Ethics Committee (IAEC) of the test facility under the project title "Repeated Dose 28 Day Oral Toxicity Study in Mice" on July 06, 2023 (JDM/IAEC/2023/02/087 and JDM/IAEC/2023/02/088). 3. The study was approved by the Institutional Animals Ethics Committee [(IAEC Proposal No.SF_23_55_001) (23_657)] under the project title “Evaluation of efficacy of an indigenously prepared topical formulation containing Mesenchymal Stromal Cell Conditioned Media for prevention/mitigation of chemotherapy induced oral mucositis”. 4. The study was approved by the Institutional Animals Ethics Committee (IAEC) project # 46/2022 under the project title “Evaluation of efficacy of an indigenously prepared topical formulation containing mesenchymal stem cell conditioned media for prevention/ mitigation of radiotherapy induced toxicity”. 5. Authors confirm that the study was conducted and reported in accordance with ARRIVE guidelines.
Patent granted
This work is protected under PCT/IN2018/050078.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mathen, C., Dsouza, W., Sharma, D. et al. Preclinical efficacy and safety evaluation of a topical mesenchymal stem cell derived conditioned media gel for oral mucositis management. Sci Rep 16, 10326 (2026). https://doi.org/10.1038/s41598-026-40193-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-40193-3