Abstract
Aedes aegypti, the primary vector of dengue, has developed widespread resistance to insecticides, posing a threat to the efficacy of vector control programs. This study assessed the susceptibility status of Ae. aegypti across Malaysian dengue hotspots and characterized knockdown resistance (kdr) mutations, including the first detection of T1520I. Adult female mosquitoes were collected from seven localities across five Malaysian states. Susceptibility to deltamethrin (0.03%), permethrin (0.4%), pirimiphos-methyl (60 mg/m2), and malathion (5%) was evaluated using bioassay protocols. Knockdown times (KT50, KT95) and resistance ratios (RR) were estimated using probit analysis, and the voltage-gated sodium channel gene was sequenced to detect kdr mutations. All field populations remained susceptible to malathion, with mortality ≥ 95%, though one Johor strain (ABS) showed suspected resistance (95%). Pirimiphos-methyl resistance was widespread, with mortality as low as 6% (ABS). Whereas resistance to pyrethroids was pronounced, with deltamethrin mortality ranging from 22% (ABS) to 95% (AA), and permethrin from 0% (TMB, ABS) to 95% (AA). Knockdown assays revealed high resistance ratios, particularly for permethrin (TMB: RR50=9.98, RR95=14.98; ABS: RR95=41.46). Sequencing identified multiple kdr mutations, including S989P, V1016G, F1534C, A1007G, and the novel detection of T1520I in Malaysian populations. F1534C was strongly associated with permethrin resistance, while V1016G and S989P predominated under deltamethrin exposure. Aedes aegypti populations in Malaysia exhibit widespread pyrethroid resistance and emerging organophosphate resistance, underpinned by multiple kdr mutations. The first detection of T1520I underscores the evolving genetic basis of resistance. These findings highlight the urgent need for integrated resistance management, including molecular surveillance and insecticide rotation, to sustain effective dengue control.
Similar content being viewed by others
Introduction
Aedes aegypti is one of the most widespread and medically important mosquito species, inhabiting tropical and subtropical regions worldwide20. It serves as the primary vector of several arboviruses, including dengue virus (DENV), chikungunya virus (CHIKV), yellow fever virus, and Zika virus1,2,3. Dengue has emerged as the most significant arboviral disease affecting humans, with nearly 390 million infections estimated annually, of which approximately 96 million are clinically apparent4,5.
In Malaysia, dengue poses an escalating public health burden, characterized by the hyperendemic circulation of all four DENV serotypes and recurrent outbreaks, resulting in substantial morbidity and economic costs6,7,8. In 2025 alone, 32,238 cases were recorded nationwide despite advances in dengue vaccines, including a tetravalent formulation that has demonstrated partial efficacy, large-scale implementation remains limited due to variable performance across serostatus groups and regulatory concerns9,10. As a result, vector control continues to serve as the primary method of dengue prevention in Malaysia.
Vector control strategies largely rely on environmental management to reduce larval habitats and the extensive use of chemical insecticides to suppress adult mosquito populations11,12. Pyrethroids (e.g., deltamethrin, permethrin) and organophosphates (e.g., malathion, pirimiphos-methyl) are the most widely deployed insecticides in Malaysia’s dengue control programs13. However, this sustained reliance on chemical interventions has exerted strong selective pressure on mosquito populations, leading to the emergence and spread of insecticide resistance.
Insecticide resistance in Ae. aegypti has been increasingly documented across Malaysia. A nationwide study by Asgarian et al.14 revealed heterogeneous but widespread resistance to pyrethroids in major dengue hotspots, including Penang, Johor, and Selangor. While Ae. aegypti populations remain largely susceptible to malathion, localized resistance has been detected in Kuala Lumpur and Selangor15. At the molecular level, resistance is frequently associated with knockdown resistance (kdr) mutations in the voltage-gated sodium channel (VGSC) gene. Several key mutations—S989P, V1016G, F1534C, and more recently A1007G—have been reported in Malaysian Ae. aegypti populations16,17,18,19, reflecting the cumulative effects of insecticide selection pressure and highlighting complex, region-specific resistance dynamics.
While insecticides remain indispensable in Malaysia’s vector control programs, their effectiveness is increasingly undermined by resistance. The over-reliance on chemical interventions as a primary control strategy has inadvertently accelerated the selection of resistant phenotypes, threatening the long-term sustainability of dengue control. Understanding the prevalence and geographic distribution of kdr mutations provides valuable insights into resistance dynamics, enabling vector control programs to adapt their interventions and maintain the efficacy of chemical-based measures. Therefore, the study aims to assess the susceptibility status of Ae. aegypti populations from diverse Malaysian locations and to characterize the frequency of key kdr mutations associated with pyrethroid resistance.
Materials and methods
Mosquito collections
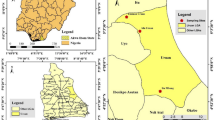
Aedes aegypti populations were collected from seven dengue hotspot localities distributed across five states in Malaysia (Fig. 1). The sampling sites were selected based on dengue hotspot designations provided by the Ministry of Health Malaysia (MOH), following an analysis of national dengue case distributions. The selected sites represented geographically distinct regions of Malaysia, including, (1) Northern Malaysia (Penang): Apartment Asoka (AA), Penang Island (5°21′N, 100°18′E); Taman Machang Bubuk (TMB), Penang Mainland (5°30′02″N, 100°25′70″E); and Flat Sri Pauh (FSP), Penang Mainland (5°33′92″N, 100°50′46″E), (2) East Coast Malaysia (Kelantan): Taman Desa Rahmat (TDR), Kelantan (6°11′49″N, 102°28′09″E), (3) Central Malaysia: Taman Dahlia (TD), Negeri Sembilan (2°84′37″N, 101°82′65″E); and PPR Raya Permai (PRP), Kuala Lumpur (3°06′17″N, 101°70′56″E), and (4) Southern Malaysia (Johor): Apartment Bukit Saujana (ABS), Johor (1°47′44″N, 103°74′87″E).
The locations of Aedes aegypti mosquito sampling sites in Malaysia. The map was generated in QGIS 3.30.1 using a geospatial open boundary dataset from the Department of Survey and Mapping Malaysia (JUPEM).
At each locality, 60 ovitraps were randomly deployed for five consecutive days. Hardwood paddles served as oviposition substrates and were subsequently collected and transported to the Medical Entomology Laboratory, Universiti Sains Malaysia. Eggs were reared to adults under controlled insectary conditions (27 ± 2 °C; 70–80% relative humidity). Only the dominant Aedes species obtained from each locality were subjected to subsequent insecticide susceptibility assays.
Adult mosquito bioassay
Adult susceptibility bioassays were performed following the World Health Organization (WHO) standard protocol20. Four replicates of 25 non-blood-fed female Ae. aegypti (2–5 days old) with a total of 100 individuals from each field population were exposed to insecticide-impregnated filter papers prepared by the Vector Control Research Unit (VCRU), Universiti Sains Malaysia. The insecticides tested were: (1) 0.03% deltamethrin, (2) 0.4% permethrin, (3) pirimiphos-methyl at 60 mg/m2, and (4) 5% malathion. Resistance intensity testing using 5× and 10× WHO diagnostic dosages was not conducted, as the study aimed to assess baseline susceptibility, knockdown dynamics, and the distribution of kdr mutations rather than to quantify operational resistance intensity, in accordance with the primary objectives of the investigation.
Mosquitoes were initially introduced into holding tubes and acclimatized for one hour. Any damaged, injured, or dead individuals were replaced prior to testing. A total of 25 females were then transferred into exposure tubes lined with insecticide-impregnated papers. Knockdown data was recorded at 5-minute intervals for one hour. Following exposure, mosquitoes were transferred into clean paper cups supplied with 10% sucrose solution and maintained at 27 ± 2 °C and 60–80% relative humidity. Mortality was assessed 24 h post-exposure, and four replicates were performed for each insecticide. Two control replicates were prepared with silicone oil for pyrethroids, olive oil for malathion, and acetone for pirimiphos-methyl. Laboratory-susceptible Ae. aegypti VCRU strain maintained at VCRU (Penang, Malaysia; 5°21′N, 100°18′E) which were reared for over 300 generations since the 1960s was used as the reference baseline.
Genomic DNA extraction
Genomic DNA was extracted from individual adult Ae. aegypti mosquitoes that survived the susceptibility bioassays using the PrimeWay Genomic II DNA Extraction Kit (Apical Scientific Sdn Bhd, Malaysia), following the manufacturer’s protocol with slight modifications to optimize yield. Prior to extraction, the elution buffer was preheated to 60 °C to improve recovery efficiency. Each mosquito was homogenized in 100 µl of GL1 buffer using a sterile pestle, which was subsequently rinsed with an additional 100 µl of GL1 buffer to maximize tissue recovery. Protein digestion was carried out by adding 20 µl of Proteinase K solution to the homogenate, followed by incubation at 60 °C for 3 h with occasional inversion to ensure complete lysis of cellular and nuclear membranes. After incubation, the lysate was centrifuged at 12,000 × g for 2.5 min at room temperature, and the supernatant was carefully transferred to a new 1.5 mL microcentrifuge tube to prevent disruption of the pellet.
The supernatant was mixed with 200 µl of GL2 buffer and vortexed thoroughly. Afterward, 4 µL of RNase A solution was added. The mixture was vortexed again and incubated at room temperature for 5 min to degrade RNA contaminants. DNA precipitation was induced by adding 200 µl of absolute ethanol, and the lysate–ethanol mixture was vortexed until homogeneous. A total of 750 µL of the resulting mixture was applied to a PrimeWay silica spin column inserted into a clean collection tube and centrifuged at 12,000 x g for 1.2 min. Afterward, the flow-through was discarded. To remove residual impurities, 400 µl of Wash Buffer G1 was added to the column, followed by centrifugation at 12,000 x g for 35 s; the flow-through was then discarded. This was followed by a second wash step with 600 µl of Wash Buffer G2 and centrifugation at 12,000 × g for 35 s. To ensure complete removal of residual ethanol and drying of the membrane, the column was subjected to an additional centrifugation step at 12,000 x g for 3.5 min.
For DNA elution, the spin column was transferred into a new 1.5 ml microcentrifuge tube, and 50 µl of preheated (60 °C) elution buffer was added directly to the center of the membrane to maximize contact. After incubating at room temperature for at least 3 min, the column was centrifuged at 12,000 x g for 35 s to obtain the final DNA yield. The eluted DNA was quantified and assessed for purity using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, USA) at an absorbance of 260 nm. The extracted DNA was subsequently stored at − 20 °C until further molecular analysis.
Detection of knockdown resistance mutation (kdr) in domains II and III
Two fragments of the voltage-gated sodium channel (VGSC) coding region from domains II and III, encompassing positions 989, 1007, 1011, 1016, 1520, and 1534, were amplified and sequenced to detect potential kdr mutations. Each 30 µl polymerase chain reaction (PCR) consisted of 15 µl of OneTaq Quick-Load 2× Master Mix (for domain II) or 15 µl of EconoTaq Plus Green 2× Master Mix (for domain III), combined with 3 µl of forward primer, 3 µl of reverse primer, and 3 µl of genomic DNA template. Prior to thermal cycling, the mixtures were briefly centrifuged (10 s) in a mini centrifuge to ensure homogeneity.
Amplification of domain II was performed using primers AaSCF1 (AGACAATGTGGATCGCTTCC) and AaSCR4 (GGACGCAATCTGGCTTGTTA), while domain III was amplified using primers AaSCF7 (GAGAACTCGGCCGATGAACTT) and AaSCR7 (GACGACGAAATCGAACAGGT) (Kawada et al., 2014). Thermal cycling conditions comprised an initial denaturation at 94 °C for 5 min, followed by 36 cycles of 94 °C for 30 s (denaturation), 50 °C for 30 s (annealing, domain II) or 60.4 °C for 30 s (annealing, domain III), and 72 °C for 1 min (extension), with a final elongation at 72 °C for 10 min, and a subsequent hold at 4 °C.
PCR amplicons were resolved by electrophoresis on 1.7% agarose gels prepared with FluoroSafe nucleic acid stain in 1× TAE buffer. Electrophoresis was conducted at 140 V for 50 min, and DNA fragments were visualized under UV illumination. PCR products were subsequently purified and submitted to MyTACG DNA Sequencing Services (Malaysia) for sequencing. Sequencing was carried out using primers AaSCR8 (CGACTTGATCCAGTTTGGAGA) for domain II and AaSCR9 (TAGCTTTCAGCGGCTTCTTC) for domain III21.
Data analysis
The susceptibility status of Ae. aegypti mosquitoes were determined according to World Health Organization (WHO) criteria, based on 24-hour post-exposure mortality. Populations exhibiting 98–100% mortality were classified as susceptible, those with 90–97% mortality as possibly resistant, and those with < 90% mortality as resistant20. Statistical comparisons of insecticide susceptibility among mosquito populations from different geographical localities were conducted using one-way analysis of variance (ANOVA).
Knockdown times for 50% (KT50) and 95% (KT95) of exposed mosquitoes were estimated using probit regression analysis in SPSS (IBM Corp., USA) version 30.0 after log-transformation of time data to satisfy model assumptions. Resistance ratios (RR) were calculated as the ratio of KT50 and KT95 values for field-collected populations in comparison to the laboratory-susceptible VCRU strain. Resistance was interpreted as follows: susceptible (RR < 1), low resistance (1 ≤ RR < 10), moderate resistance (10 ≤ RR ≤ 30), high resistance (30 < RR < 100), and very high resistance (RR ≥ 100)22,23.
DNA sequence data generated by MyTAGC DNA Sequencing Services were aligned using ClustalW, and the nucleotide sequences were translated into amino acid sequences with the protein translation function implemented in MEGA X software24. The six unique DNA haplotype sequences were deposited in the GenBank database under accession numbers PX559884-PX559849. The heatmaps were generated using Heatmapper 1.0, an online software25, and further analyzed using correlation analysis in SPSS version 30 (IBM Corp., USA).
Results
Bioassay of insecticides
The susceptibility status of Ae. aegypti populations varied markedly across insecticides and collection sites (Fig. 2). For pirimiphos-methyl (60 mg/m2), only the laboratory reference strain (VCRU) remained fully susceptible (100% mortality), whereas all field populations demonstrated resistance, with mortality ranging from 6% (ABS) to 88% (TD). TMB strains showed significantly lower mosquito mortality than other strains (p < 0.05).
Susceptibility status of Aedes aegypti laboratory and field populations against four types of insecticides (pirimiphos-methyl, malathion, deltamethrin, and permethrin). The same small letter represents no significant differences between strains for each insecticide. ** S-Susceptibility, PR-Possible Resistance, R-Resistance; VCRU-Vector Control Research Unit, AA-Apartment Asoka, TMB-Taman Machang Bubuk, FSP-Flat Sri Pauh, TDR-Taman Desa Rahmat, TD-Taman Dahlia, PRP-PPR Raya Permai, ABS-Apartment Bukit Saujana.
In contrast, malathion exposure produced consistently high mortality across all tested populations, demonstrating that, Ae. aegypti populations remained largely susceptible to this organophosphate insecticide. One-way ANOVA revealed significant differences among strains in Ae. mortality. aegypti mosquitoes (F = 0.000, df = 7, p < 0.001; Table 1). The VCRU strain and six field populations (AA, TMB, FSP, TDR, TD, and PRP) were fully susceptible (100% mortality), while only one site (ABS) exhibited suspected resistance, with slightly reduced mortality (95%) but showed significant differences between strains (p < 0.001).
For deltamethrin, susceptibility was limited to the laboratory reference strain (VCRU), which achieved complete mortality. Among field-collected populations, only the AA strain (95%) showed signs of possible resistance (95%), whereas other sites demonstrated resistance, with mortality as low as 22% (ABS). All field strains were found to have significantly lower mortality than the VCRU strain (P < 0.01). Whereas, permethrin also displayed the same pattern as deltamethrin. The Negeri Sembilan population (TD strain) showed significantly higher resistance with lower mortality compared to AA (p = 0.011), FSP (p = 0.005), and VCRU (p < 0.001) strains. Thus indicates, the pyrethroid group is more resistant than organophosphate group in almost all of the localities.
Knockdown time
Knockdown time (KT) assays revealed clear differences in susceptibility patterns of Ae. aegypti across insecticides and localities, with resistance ratios (RR) providing insight into the relative speed of knockdown compared to the laboratory reference strain (VCRU), as in Table 2. For malathion (5%), VCRU recorded KT50 and KT95 values of 64.40 and 177.75 min, respectively, while most field populations displayed shorter knockdown times, reflected in RR50 and RR95 values below 1.0 (e.g., ABS: RR50 = 0.26, RR9 = 0.22; TDR: RR50 = 0.34, RR95 = 0.26), indicating that malathion was more effective in field populations than in the susceptible reference strain.
In contrast, exposure to 0.03% deltamethrin revealed delayed knockdown among several populations, with FSP and TDR exhibiting markedly elevated values (KT50 = 34–35 min, KT95 = 80–100 min; RR50> 3.4, RR95 > 4.2), consistent with strong resistance. Interestingly, PRP and ABS demonstrated discordant patterns, where KT50 values were close to or below the reference (RR50 = 0.62–1.15), but KT95 values were substantially prolonged (RR95 = 9.50–11.94), suggesting heterogeneous responses within these populations. For 0.4% permethrin, pronounced resistance was evident in TMB, which exhibited extremely high knockdown times (KT50 = 92.91 min, KT95 = 265.47 min) with RR50 and RR95 values of 9.98 and 14.98, respectively, indicating nearly ten- to fifteen-fold slower knockdown relative to VCRU. The ABS strain also displayed variable responses, with a moderate KT50 (26.30 min; RR50 = 2.82) but an exceptionally prolonged KT95 (734.61 min; RR95 = 41.46), pointing to substantial intra-population variability and delayed knockdown in a subset of individuals. In contrast, the AA strain showed more moderate resistance to permethrin (RR50 = 2.23; RR95 = 2.50).
The results highlight a consistent trend of susceptibility to malathion across populations, but widespread and variable resistance to pyrethroids, with resistance ratios quantifying both moderate (2–5 fold slower knockdown) and extreme (> 10-fold slower knockdown) resistance phenotypes among field-collected Ae. aegypti. The results for pirimiphos-methyl could not be computed due to no knockdown in Ae. aegypti mosquitoes during the one-hour observation.
Detection of kdr mutations
The distribution of kdr genotypes differed between populations exposed to permethrin and deltamethrin, with the resistant alleles present at multiple loci, but their frequency and fixation levels varied by insecticide (Fig. 3). For the S989P mutation, resistant alleles were detected under both insecticide exposures, with all of Ae. aegypti mosquitoes TDR strain was detected with homozygous resistance (PP) for both pyrethroids exposure (frequency allele = 1.0). At the same time, the AA strain showed a high frequency of heterozygous resistance (SP) to both pyrethroids exposure (frequency allele = 1.0). At the V1016G locus, resistance alleles (VG and GG) were widespread under both insecticides. At the V1016G locus, resistant allele frequencies (V/G) were high under both insecticides, but were consistently greater with deltamethrin (0.67–1.0) compared to permethrin (0.34–0.8). The frequency of homozygous resistant individuals (GG1016) was higher in deltamethrin-exposed groups (e.g., Asoka Apartment, AA; TMB; PRP) compared to permethrin, where heterozygotes were more common. The TD strain exhibited homozygous resistance to all tested mosquitoes when exposed to both permethrin and deltamethrin (frequency allele = 1.0). No detection of the I1011M mutation was found for insecticide exposure (Fig. 3). In the case of A1007G, homozygous resistance alleles were found in the TDR strain only when exposed to permethrin.
Heatmap of amino acid proportions of the six principal insecticide resistance–associated mutations for (A) deltamethrin and (B) permethrin across all seven sampling locations.
While, for T1520I, resistance was nearly fixed in deltamethrin-exposed populations, with all individuals carrying heterozygous resistance genotypes (TI). The F1534C locus showed the most variation between insecticides. For permethrin, resistant heterozygotes and homozygotes (FC and CC) were frequently observed, with resistance allele frequencies ranging from 0.17 (TD) to 0.83 (TMB). Under deltamethrin exposure, however, several populations (e.g., AA, TDR) retained higher proportions of the wild-type (FF) allele at F1534C, and resistance allele frequencies were generally lower (0.17–0.67), whereas they were higher in permethrin. This suggests that F1534C contributes more to permethrin resistance than to deltamethrin resistance.
Strong positive associations were detected among resistance-associated alleles under both pyrethroid exposures. Under permethrin, significant correlations were observed between homozygous PP989 and GG1016 (r = 0.950, p = 0.001). Similarly, under deltamethrin exposure, multiple high-magnitude correlations were evident, including PP989 with GG1016 (r = 0.881, p = 0.009) and SS989 with CC1534 (r = 0.871, p = 0.011).
Discussion
This study revealed Ae. aegypti in Malaysia are responding differently to the insecticides used in control programs, being more impacted by pyrethroids, which have caused resistance. Malathion still works well in most sites, but resistance to pirimiphos-methyl was already widespread for the organophosphate group. Pyrethroids such as deltamethrin and permethrin were the least effective. In many places, survival rates were far higher than the WHO limit for resistance, as in the WHO20 protocol. These findings confirm what has been reported in Malaysia before, that the pyrethroids are losing their effectiveness in dengue-prone regions because of long and repeated use17.
The present findings reveal notable spatial heterogeneity in insecticide resistance across Malaysia. Populations from Johor (ABS) exhibited particularly high levels of pyrethroid resistance, whereas populations from Penang (TMB and FSP) demonstrated only moderate resistance. Localized ecological and operational pressures most likely shape such spatial variation in resistance. These include differences in human population density, variation in the frequency and intensity of fogging activities, and the extent of household insecticide use. Previous studies in Malaysia have reported similar trends, where areas subjected to intensive vector control interventions showed elevated frequencies of resistance alleles compared to less-treated localities16,19. Sustained reliance on chemical interventions in densely populated dengue hotspots, such as Penang, Johor, and Selangor, has been shown to impose strong selective pressure on mosquito populations, accelerating the emergence and spread of resistance alleles14. Although malathion remains largely effective in most surveyed sites, the early indications of reduced susceptibility in Johor raise concerns that continued reliance on this compound may precipitate resistance development analogous to that observed with pyrethroids.
Knockdown assays provided additional insight from the present study, revealing that mosquitoes exposed to pyrethroids exhibited markedly prolonged knockdown times compared to the laboratory-susceptible strain, in some cases requiring over tenfold longer durations to achieve comparable knockdown. This phenomenon of extended knockdown times not only underscores the growing insecticide resistance among mosquito populations but also raises concerns about the effectiveness of current vector control strategies27. This delay points to a combination of reduced insecticide absorption and stronger metabolic defenses, especially the activity of detoxifying enzymes such as cytochrome P450s. These mechanisms have been reported previously in Malaysian mosquito populations16,17. A notable observation was the pronounced heterogeneity in knockdown responses within certain populations. For example, in Johor, individual mosquitoes displayed highly variable susceptibility, with some exhibiting rapid knockdowns. In contrast, others remained active for several hours, highlighting that resistance dynamics can be heterogeneous and complex even within a single geographic population.
Molecular analysis provided insight into the resistance mechanisms underlying the phenotypic variation. Several established knockdown resistance (kdr) mutations, including S989P, V1016G, and F1534C, were detected, together with more recently reported substitutions in this study, such as A1007G and T1520I, within Malaysia. In certain populations, resistant alleles were found at near-fixation levels, indicating that almost all individuals carried these mutations27. Notably, the F1534C mutation appeared to confer a stronger association with permethrin resistance than with deltamethrin resistance, a pattern consistent with reports from other Southeast Asian populations5,29. Plernsub et al. (2016)29 demonstrated that the F1534C mutation is particularly important for type I pyrethroid (permethrin) resistance, while other combinations (S989P + V1016G) contribute more to type II pyrethroid (deltamethrin) resistance. These findings underscore that individual kdr mutations differ in their functional impact across pyrethroid compounds, emphasizing the importance of monitoring the distribution and frequency of specific alleles to guide the continued efficacy of vector control strategies29.
When placed in a global context, the resistance profile observed in Malaysia reflects broader patterns documented elsewhere. Multiple mutations were detected in the voltage-gated sodium channel (VGSC) gene in this study, including S989P, V1016G, F1534C, A1007G, and the newly observed T1520I. The present study reveals a complex kdr resistance profile in Malaysian Ae. aegypti, shaped by prolonged pyrethroid use. The consistent co-occurrence of mutations at positions S989P, V1016G, and F1534C indicates a jointly structured resistance profile, supporting the interpretation that pyrethroid resistance in Malaysian Ae. aegypti is mediated by multilocus allelic architecture rather than isolated single-locus effects. The co-occurrence and high frequencies of these mutations indicate a multilocus resistance architecture, consistent with resistance profiles reported in other regions subjected to sustained pyrethroid selection pressure30. The widespread occurrence of S989P and V1016G, often in combination, is consistent with their established role in conferring resistance to type II pyrethroids such as deltamethrin, whereas F1534C was more strongly associated with resistance to type I compounds such as permethrin, supporting earlier reports from Southeast Asia that different alleles exhibit compound-specific effects28.31. In Asia, particularly in Thailand and Vietnam, the combined presence of S989P and V1016G mutations has been linked to significantly reduced efficacy of pyrethroid-based interventions32,33.
We hypothesize that this pattern reflects the emergence of novel mutations under sustained selection pressure, as evidenced by the localized detection of A1007G and the near fixation of T1520I under deltamethrin exposure, mirroring resistance trends previously reported in populations from India and Myanmar, where these alleles often occur in combination with F1534C31,34. In Latin America, widespread pyrethroid resistance has been strongly associated with V1016I/G and F1534C mutations, which have been directly implicated in operational control failures112,35. The coexistence of multiple kdr alleles across different regions suggests that Malaysian populations are evolving heterogeneous but increasingly robust resistance phenotypes, posing a substantial challenge to vector control.
The first detection of T1520I in Malaysian Ae. aegypti broadens the regional picture of VGSC evolution under pyrethroid selection. In our study, the T1520I mutation was present at a high frequency under deltamethrin exposure, with TI heterozygotes approaching fixation in several populations. Comparable occurrences of mutation have been reported initially in India, where it co-segregated with F1534C in pyrethroid-resistant populations and was associated with reduced permethrin susceptibility5. Myanmar documented T1520I alongside S989P, V1016G, and F1534C, including a quadruple haplotype (S989P/V1016G/T1520I/F1534C) indicative of strong multi-locus selection34. A study by Fan et al. (2020)36 indicates that the F1534C mutation alone confers a 7- to 16-fold resistance to pyrethroids, while the addition of T1520I further elevates this resistance specifically against Type I compounds. The S989P and V1016G mutations have been widely reported across Asia, occurring at a frequency of 18.27% in Ae. aegypti populations from Bengaluru, India5, forming a fixed haplotype in Taiwanese populations37, and being prevalent in Sri Lankan populations, collectively indicating the widespread establishment of pyrethroid resistance in the region38. Regionally, reviews and multi-country syntheses have noted that T1520I, typically linked with F1534C, has now been detected across parts of South and Southeast Asia, reinforcing its relevance as a surveillance marker for operational programs21.
These findings highlight a divergence in susceptibility within the organophosphate class, where malathion continues to serve as a viable option in most settings, whereas pirimiphos-methyl efficacy is severely compromised. In India and Sri Lanka, Ae. aegypti have already developed reduced susceptibility to malathion through enzyme-based detoxification mechanisms like esterases and GSTs5,39. The overall profile emphasizes that although organophosphates remain more effective than pyrethroids, localized resistance is emerging, and continued surveillance is critical to prevent further erosion of their utility in dengue control programs. In short, this study shows that Ae. aegypti in Malaysia are already resistant to pyrethroids and are showing the signs of resistance to organophosphates. The observed resistance patterns have critical implications for vector control and pest management in Malaysia. The widespread resistance to pyrethroids, combined with the emergence of resistance to organophosphates such as pirimiphos-methyl, indicates that relying solely on chemical insecticides will increasingly undermine the effectiveness of dengue control programs15,16,18.
This study focuses exclusively on target-site resistance by characterising kdr mutations in the voltage-gated sodium channel; metabolic resistance mechanisms were not assessed, despite evidence that organophosphate resistance has begun to emerge and should be investigated in future studies. Keeping track of the spread of resistance mutations, including metabolic resistance, can help predict where and when insecticides are likely to fail. In Malaysia, embedding routine monitoring of kdr mutations into dengue control programs would provide a strong early-warning system, similar to how malaria vector programs already use genetic tools to track resistance trends12,28. Importantly, molecular surveillance of kdr mutations, particularly the novel T1520I identified in this study, should be incorporated into routine monitoring frameworks to provide early warnings of resistance spread and to guide evidence-based interventions. To address this challenge, integrated resistance management strategies should be prioritized, including insecticide rotation with compounds of different modes of action20, the strengthening of larval source reduction and environmental management, and also the integration of biological control measures where feasible40. Additionally, community education and the regulation of domestic insecticide use are crucial in reducing household-level selection pressures that accelerate the development of resistance. Collectively, these strategies will strengthen the resilience of pest management programs and help sustain the long-term efficacy of dengue control efforts in Malaysia.
Conclusion
In conclusion, this study demonstrates that, Ae. aegypti populations in Malaysia exhibit a complex resistance landscape characterized by widespread pyrethroid resistance and emerging, though more localized, resistance to organophosphates. The detection of multiple kdr mutations, including well-established alleles (S989P, V1016G, F1534C) and newer variants (A1007G, T1520I), underscores the adaptive capacity of vector populations under sustained chemical pressure and highlights the compound-specific nature of resistance mechanisms. While malathion remains broadly effective, the compromised efficacy of pirimiphos-methyl signals that even organophosphates are not invulnerable to resistance development. Collectively, these findings underscore the urgent need for integrated resistance management strategies that incorporate molecular surveillance, insecticide rotation, and non-chemical interventions to sustain the long-term effectiveness of vector control programs and mitigate the risk of operational failure in dengue prevention. In this context, broader integrated vector management approaches should be prioritized, including the use of insect growth regulators (IGRs), biological control agents, and Wolbachia-infected mosquitoes as essential alternative or complementary strategies to reduce reliance on conventional chemical insecticides and enhance sustainable dengue control.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Leta, S. et al. Global risk mapping for major diseases transmitted by Aedes aegypti and Aedes albopictus. Int. J. Infect. Dis. 67, 25–35. https://doi.org/10.1016/j.ijid.2017.11.026 (2018).
Simmons, C. P., Farrar, J. J., Nguyen, V. V. C., Wills, B. & Dengue N Engl. J. Med. ;366(15):1423–1432. https://doi.org/10.1056/NEJMra1110265 (2012).
Woon, Y. L. et al. A Two-Year Review on Epidemiology and Clinical Characteristics of Dengue Deaths in Malaysia, 2013–2014. PLOS Negl. Trop. Dis. 10 (5), e0004575. https://doi.org/10.1371/journal.pntd.0004575 (2016).
Bhatt, S. et al. The global distribution and burden of dengue. Nature 496, 504–507. https://doi.org/10.1038/nature12060 (2013).
Sumitha, M. K. et al. Status of insecticide resistance in Aedes aegypti in India: A review. J. Vector Borne Dis. 60 (2), 116–124. https://doi.org/10.4103/0972-9062.361174 (2023).
Mohd-Zaki, A. H., Brett, J., Ismail, E. & L’Azou, M. Epidemiology of dengue disease in Malaysia (2000–2012): A systematic literature review. PLoS Negl. Trop. Dis. 8 (11), e3159. https://doi.org/10.1371/journal.pntd.0003159 (2014).
Shepard, D. S., Undurraga, E. A., Halasa, Y. A. & Stanaway, J. D. The global economic burden of dengue: A systematic analysis. Lancet Infect. Dis. 16 (8), 935–941. https://doi.org/10.1016/S1473-3099(16)00146-8 (2016).
Mulligan, K., Dixon, J., Sinn, C. L. J. & Elliott, S. J. Is dengue a disease of poverty? A systematic review. Pathog Glob Health. 109 (1), 10–18. https://doi.org/10.1179/2047773214Y.0000000168 (2015).
Wichmann, O. et al. Live-attenuated tetravalent dengue vaccines: The needs and challenges of post-licensure evaluation of vaccine safety and effectiveness. Vaccine 35 (42), 5535–5542. https://doi.org/10.1016/j.vaccine.2017.08.066 (2017).
Biswal, S. et al. Efficacy of a tetravalent dengue vaccine in healthy children and adolescents. N Engl. J. Med. 381 (21), 2009–2019. https://doi.org/10.1056/NEJMoa1903869 (2019).
World Health Organization (WHO). Pesticides and Their Application for the Control of Vectors and Pests of Public Health Importance 6th edn (World Health Organization, 2006).
Moyes, C. L. et al. Contemporary status of insecticide resistance in the major Aedes vectors of arboviruses infecting humans. PLoS Negl. Trop. Dis. 11 (7), e0005625. https://doi.org/10.1371/journal.pntd.0005625 (2017).
Kraemer, M. U. G. et al. The global distribution of the arbovirus vectors Aedes aegypti and Ae. albopictus. eLife 4, e08347. https://doi.org/10.7554/eLife.08347 (2015).
Asgarian, F., Vatandoost, H., Hanafi-Bojd, A. A. & Nikpoor, F. Worldwide status of insecticide resistance of Aedes aegypti and Ae. albopictus, vectors of arboviruses of Chikungunya, Dengue, Zika and Yellow Fever. J. Athrapod-Borne Dis. 17 (1), 1–27. https://doi.org/10.18502/jad.v17i1.13198 (2023).
Sabar, N. N. et al. Insecticide resistance status of Aedes aegypti and Aedes albopictus in Malaysia (2010 to 2022): a review. Asian Pac. J. Trop. Med. 16 (10), 434–445. https://doi.org/10.4103/1995-7645.378561 (2023).
Ishak, I. H., Jaal, Z., Ranson, H. & Wondji, C. S. Contrasting patterns of insecticide resistance and knockdown resistance (kdr) in the dengue vectors Aedes aegypti and Aedes albopictus from Malaysia. Parasites Vectors. 8, 181. https://doi.org/10.1186/s13071-015-0797-2 (2015).
Amelia-Yap, Z. H., Chen, C. D., Sofian-Azirun, M. & Low, V. L. Pyrethroid resistance in the dengue vector Aedes aegypti in Southeast Asia: Present situation and prospects for management. Parasites Vectors. 11, 332. https://doi.org/10.1186/s13071-018-2899-0 (2018).
Zuharah, W. F. & Sufian, M. Discovery of a novel knockdown resistance (kdr) mutation A1007G in Aedes aegypti (Diptera: Culicidae) from Malaysia. Sci. Rep. 11, 5180. https://doi.org/10.1038/s41598-021-84669-w (2021).
Akhir, M. A. M. et al. Knockdown resistance (kdr) gene of Aedes aegypti in Malaysia with the discovery of a novel regional specific point mutation A1007G. Parasites Vectors. 15, 122. https://doi.org/10.1186/s13071-022-05192-z (2022).
World Health Organization (WHO). Manual for Monitoring Insecticide Resistance in Mosquito Vectors and Selecting Appropriate Interventions (World Health Organization, 2022).
Kawada, H. et al. Co-occurrence of point mutations in the voltage-gated sodium channel of pyrethroid-resistant Aedes aegypti populations in Myanmar. PLoS Negl. Trop. Dis. 8 (7), e3032. https://doi.org/10.1371/journal.pntd.0003032 (2014).
Khan, H. A. A., Akram, W., Shehzad, K. & Shaalan, E. A. S. First report of field evolved resistance to agrochemicals in dengue mosquito, Aedes albopictus (Diptera: Culicidae), from Pakistan. Parasites Vectors. 4, 146. https://doi.org/10.1186/1756-3305-4-146 (2011).
Solis-Santoyo, F. et al. Insecticide resistance in Aedes aegypti from Tapachula, Mexico: Spatial variation and response to historical insecticide use. PLoS Negl. Trop. Dis. 15 (9), e9746. https://doi.org/10.1371/JOURNAL.PNTD.0009746 (2021).
Kumar, S., Stecher, G., Li, M., Knyaz, C. & Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 35 (6), 1547–1549. https://doi.org/10.1093/molbev/msy096 (2018).
Babicki, S. et al. Heatmapper: web-enabled heat mapping for all. Nucleic Acids Res. 44 (W1), W147–W153. https://doi.org/10.1093/nar/gkw419 (2016).
Owusu, H. F., Jančáryová, D., Malone, D. & Müller, P. Comparability between insecticide resistance bioassays for mosquito vectors: time to review current methodology? Parasites Vectors. 8, 357. https://doi.org/10.1186/S13071-015-0971-6 (2015).
Chang, C., Huang, X. Y., Chang, P. C., Wu, H. H. & Dai, S. M. Inheritance and stability of sodium channel mutations associated with permethrin knockdown resistance in Aedes aegypti. Pestic. Biochem. Physiol. 104 (2), 136–142. https://doi.org/10.1016/J.PESTBP.2012.06.003 (2012).
Kawada, H. et al. Discovery of point mutations in the voltage-gated sodium channel gene of Aedes aegypti populations in Myanmar, suspected to confer pyrethroid resistance. PLoS Negl. Trop. Dis. 10 (7), e0004780. https://doi.org/10.1371/journal.pntd.0004780 (2016).
Plernsub, S. et al. Additive effect of knockdown resistance mutations, S989P, V1016G, F1534C, in a heterozygous genotype conferring pyrethroid resistance in Aedes aegypti in Thailand. Parasites Vectors. 9, 417. https://doi.org/10.1186/s13071-016-1713-0 (2016).
Li, T. et al. Multiple mutations and mutation combinations in the sodium channel of permethrin resistant mosquitoes, Culex quinquefasciatus. Sci. Rep. (2012). https://doi.org/10.1038/SREP00781
Kushwah, R. B. S., Dykes, C. L., Kapoor, N., Adak, T. & Singh, O. P. Pyrethroid-resistance and presence of two knockdown resistance (kdr) mutations, F1534C and a novel mutation T1520I, in Indian Aedes aegypti. PLoS Negl. Trop. Dis. 9 (1), e3332. https://doi.org/10.1371/journal.pntd.0003332 (2015).
Stenhouse, S. A. et al. Detection of the V1016G mutation in the voltage-gated sodium channel gene of Aedes aegypti (Diptera: Culicidae) by allele-specific PCR assay, and its distribution and effect on deltamethrin resistance in Thailand. Parasites Vectors. 6, 253. https://doi.org/10.1186/1756-3305-6-253 (2013).
Hirata, K. et al. A Single Crossing-Over Event in Voltage-Sensitive Na+ Channel Genes May Cause Critical Failure of Dengue Mosquito Control by Insecticides. PLOS Negl. Trop. Dis. 8 (8), e3085. https://doi.org/10.1371/journal.pntd.0003085 (2014).
Naw, H. et al. Knockdown resistance mutations in the voltage-gated sodium channel of Aedes aegypti (Diptera: Culicidae) in Myanmar. Insects 13 (4), 322. https://doi.org/10.3390/insects13040322 (2022).
Saavedra-Rodriguez, K. et al. A mutation in the voltage-gated sodium channel gene associated with pyrethroid resistance in Aedes aegypti from Latin America. Insect Mol. Biol. 16 (6), 785–798. https://doi.org/10.1111/j.1365-2583.2007.00774.x (2007).
Fan, Y. & Scott, J. G. The F1534C voltage-sensitive sodium channel mutation confers 7- to 16-fold resistance to pyrethroid insecticides in Aedes aegypti. Pest Manag Sci. 76 (6), 2251–2259. https://doi.org/10.1002/ps.5763 (2020).
Chung, H. H. et al. First detection of the S989P+V1016G+D1763Y haplotype and expansion of voltage-gated sodium channel mutations in Aedes aegypti in Taiwan in 2016–2023. PLoS Negl. Trop. Dis. 19 (1), e0012768. https://doi.org/10.1371/journal.pntd.0012768 (2025).
Fernando, S. D. et al. First report of V1016G and S989P knockdown resistant (kdr) mutations in pyrethroid-resistant Sri Lankan Aedes aegypti mosquitoes. Parasites Vectors. 11 (1), 1–6. https://doi.org/10.1186/S13071-018-3113-0 (2018).
Vontas, J. et al. Insecticide resistance in the major dengue vectors Aedes albopictus and Aedes aegypti. Pestic. Biochem. Physiol. 104 (2), 126–131. https://doi.org/10.1016/j.pestbp.2012.05.008 (2012).
Becker, N. et al. Integrated control of Aedes albopictus in Southwest Germany supported by the sterile insect technique. Parasites Vectors. (2022). https://doi.org/10.1186/s13071-021-05112-7
Acknowledgements
We would like to express our sincere gratitude to the School of Biological Sciences, Universiti Sains Malaysia, for their invaluable support throughout this research. The institution’s resources and facilities greatly contributed to the successful completion of this work. We want to acknowledge the collaborative efforts of the Ministry of Health Malaysia.
Funding
Declaration.
This research/project was funded by the Ministry of Higher Education Malaysia under the Fundamental Research Grant Scheme, grant number [FRGS/1/2023/STG03/USM/02/4]. The funding agency had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript/report.
Author information
Authors and Affiliations
Contributions
The authors’ contributions are as follows: WFZ: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Validation, Supervision, Writing—original draft; Writing—review & editing, MT: Project administration, Methodology, Data curation, Formal analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
No ethical approval is needed for this project.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ma, T., Zuharah, W.F. The susceptibility status of Aedes aegypti (Diptera: Culicidae) mosquitoes in Malaysia on pyrethroid and organophosphate insecticides with first detection of T1520I mutation. Sci Rep 16, 10375 (2026). https://doi.org/10.1038/s41598-026-41000-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-41000-9