Abstract
Indoleamine-2,3-dioxygenase (IDO) and arginase1 (ARG1) are immunosuppressive enzymes frequently overexpressed in cancer, with higher expression in colorectal cancer primary tumours paradoxically linked to favourable survival. Their significance in metastases remains unexplored. Employing multiplexed immunohistochemistry and supervised machine learning-based digital image analysis, we analysed the IDO and ARG1 expression in monocytic cells, granulocytes, mast-cells and tumour cells in 91 resected pulmonary metastases from 53 corresponding primary tumours. We also evaluated the spatial distribution of IDO+ and ARG1+ cells relative to tumour cells and compared these patterns between metastases and primary tumours. In metastases, higher IDO+ monocytic cell densities in the tumour centre were associated with favourable survival (adjusted HR 0.17, 95%CI 0.05–0.59, p = 0.005), while higher IDO– monocytic cell densities in the invasive margins were associated with worse survival (adjusted HR 4.19, 95%CI 1.45–12.05, p = 0.008). Moreover, higher densities of IDO+HLA-DR− immature monocytic cells and IDO+FCGR3+ monocytic cells in the invasive margin were also associated with poor survival, after adjusting for T-cell densities (adjusted HRs 26.4, 95%CI 4.95 − 140.42, p < 0.001 and 3.91 95%CI 0.97 − 15.76, p = 0.056, respectively). These findings provide detailed insights into the immunosuppressive myeloid cell landscape in colorectal cancer pulmonary metastases, highlighting key differences from primary tumours and potential implications for therapy development.
Similar content being viewed by others
Introduction
Indoleamine 2,3-dioxygenase (IDO) is a cytosolic enzyme involved in the catabolism of tryptophan in the first and rate-limiting step of the kynurenine pathway that is frequently overexpressed in cancer1. The catabolism of tryptophan, an essential amino-acid, leads to the functional and proliferative arrest of T-cells in the tumour microenvironment via kynurenine metabolites and tryptophan deprivation2. Another enzyme with immunosuppressive features is arginase-1 (ARG1), catalysing the hydrolysis of L-arginine into immunologically active metabolites3. ARG1 is also overexpressed in several solid tumours4. It takes part in the complex interplay of immunoediting and is associated with poor survival in several solid tumours5,6. IDO is expressed in tumour cells, as well as in monocytic cells, endothelial cells, and T-cells6, whereas ARG1 is usually expressed in granulocytes7. Given their immunosuppressive nature, ARG1 and IDO have been targets of immunotherapeutic development in recent clinical trials8,9,10.
Colorectal cancer (CRC) is one of the most common malignancies globally, ranking as the third leading cause of cancer mortality11. The prognostic significance of IDO and ARG1 expression in CRC have been contradictory12,13,14,15,16. This inconsistency may stem from the frequent use of qualitative, single-plex analysis methods that fail to distinguish between cell types contributing to their expression. Additionally, many studies have not differentiated the invasive margin and tumour centre in the analysis, thus overlooking the intricate complexity of the tumour microenvironment (TME). Applying multiplexed immunohistochemistry, we recently demonstrated a positive survival effect for both enzymes in CRC primary tumours17.
Of all CRC patients, 15 − 30% have synchronous metastases at the time of diagnosis and around 20% develop metachronous metastases within 5-years following primary tumour resection18,19. While the 5-year overall survival of CRC across all stages exceeds 60%, patients with stage IV disease have a 5-year survival of only 14%20. In pulmonary metastases, the myeloid cell distributions and the prognostic impact of the myeloid cell subsets remain largely unexplored.
The aim of this study was to evaluate the prognostic effect of myeloid cell densities, monocytic cell maturity (HLA-DR), myeloid cell FCGR3 expression, and immunosuppressive potential (IDO and ARG1) in resected CRC pulmonary metastases with a comparison to primary tumours. We hypothesized that higher expression of IDO and ARG1 would be associated with adverse survival outcomes, and the effect would be dependent on HLA-DR and FCGR3 expression. Additionally, we aimed to evaluate the spatial distribution of the myeloid cells in metastases and primary tumours according to their IDO/ARG1 expression.
Materials and methods
Study design
This retrospective population-based study included all patients who underwent pulmonary metastasectomy for histologically confirmed CRC metastases with curative intent at Oulu University Hospital and Central Finland Central Hospital between 2000 and 2020. These hospitals are the sole providers of thoracic surgical services within their respective regions. Patient identification was based on surgical and pathology records, and relevant clinical data were retrospectively gathered from electronic patient records. Tumour staging was updated to conform to the 8th edition of the Union for International Cancer Control (UICC) tumour-node-metastasis (TNM) classification system21. Survival data, 100% complete through December 31, 2021, were obtained from Statistics Finland. Haematoxylin and eosin (H&E) stained slides of primary tumours and pulmonary metastases were retrieved from pathology archives of Northern Finland Biobank Borealis and Central-Finland Biobank, and reviewed by a pathologist (V-M.P). For cases involving multiple metastatic nodules resected in a single procedure, a representative nodule was arbitrarily selected for further analysis. Digitalization of the slides was performed using either a NanoZoomer XR (Hamamatsu Photonics, Hamamatsu City, Japan) or Aperio AT2 (Leica Biosystems Imaging Inc., Wetzlar, Germany) scanner equipped with a 20× objective.
Immunohistochemical screening for DNA mismatch repair (MMR) deficiency (MLH1, MSH2, MSH6 and PMS2) and BRAF V600E mutation status (VE1) was conducted as described previously22. All patients were MMR proficient and only two exhibited BRAF V600E mutations. The densities of CD3+ and CD8+ T-cells in the invasive margin and tumour centre were analysed from whole section slides using methods outlined in prior studies22. A three-tiered immune cell score was generated based on CD3+ and CD8+ T-cell densities, following the principles of the consensus Immunoscore23.
Multiplex immunohistochemistry
Formalin-fixed paraffin-embedded tissue samples were used to construct tissue microarrays (TMAs) with 1-mm core diameter. Core selection was guided by H&E-stained slides, with two cores taken from the invasive margin and two from the tumour centre for both primary tumours and metastases. TMA-blocks were cut into 3.5-µm sections for staining.
A 10-plex multiplex immunohistochemistry assay was designed to characterize myeloid cell densities and maturity and expression of immunosuppressive enzymes. The panel included markers for myeloid cells (ITGAM [CD11b], CD33), monocytic cells (CD14), granulocytes (CEACAM8 [CD66b]), mast cells (TPSAB1 [mast cell tryptase]), and tumour cells (KRT [keratin]), along with HLA-DR (human leukocyte antigen, DR-isotype), FCGR3 (Fcγ receptor 3 A, [CD16]), IDO and ARG1. Haematoxylin was used to align cell nuclei. Examples of IDO and ARG1 expression in TMA cores are presented in Fig. S1. Standardized nomenclature was used for protein names24. Antibody optimization and assay validation have been described previously17. Staining was performed sequentially on a Bond-III automated stainer using a Bond Refine Detection kit (DS9800, Leica Biosystems), with 3,3’-dia-minobenzidine replaced with 3-amino-9-ethylcarbazole (AEC). All slides were stained in one batch ensuring uniform staining. The procedure included staining each marker individually, followed by slide scanning, heat-mediated antibody removal, and ethanol-based AEC removal to enable sequential staining of multiple markers.
Image analysis
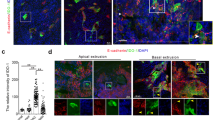
Tissue microarray cores were identified and extracted into single-core images using the TMA dearrayer function in QuPath25. Non-representative cores (e.g., those with minimal tumour cell content, necrosis, or procedural damage) were excluded. Representative cores were combined into pseudo-coloured multiplex immunohistochemistry images using Fiji ImageJ26. Staining patterns for each marker in a representative image are presented in Fig. 1A. The cell detection function in QuPath was used to identify cells, and the object classifier function to identify myeloid cells (ITGAM+/CD33+) and further classify them into three main phenotypes—monocytic cells (CD14+CEACAM8−TPSAB1−KRT−), granulocytes (CD14−CEACAM8+TPSAB1−KRT−), and mast cells (CD14−CEACAM8−TPSAB1+KRT−)—with the remaining cells categorized into tumour cells CD14−CEACAM8−TPSAB1−KRT+) and other cells (CD14−CEACAM8−TPSAB1−KRT−) (Fig. 1B)17. For tissue categorization, the pixel classifier function in QuPath was trained to identify tumour epithelium, stroma, and other regions (Fig. 1B), with other excluded from further analysis. Further data processing, quantification and spatial analysis was performed using packages tidyverse (2.0.0), cutpointr (1.1.2), purrr (1.0.2), ggplot2 (3.4.2), gtsummary (2.0.1), corrplot (0.92), ggpubr (0.6.0), patchwork (1.2.0) spatstat (3.0.5), survival (3.5–5.5) and survminer (0.4.9) in RStudio (version 2024.09.1).
Multiplex immunohistochemistry assay and machine learning-based image analysis. (A) Examples of each marker included in the assay separately and merged into pseudo-coloured multiplexed immunohistochemistry image. Haematoxylin (blue background staining) was used to align the cell nuclei. (B) Machine learning based image analysis for detecting and classifying tissue compartments and cells in QuPath. Tissue compartments were classified into tumour epithelium and stromal areas. Cells were classified into monocytic cells, granulocytes, mast cells, tumour cells, and other cells.
Cells were categorized according to their cytoplasmic staining intensities of FCGR3, HLA-DR, IDO and ARG1 by setting fixed cut-off values (positive/negative staining). Cell densities were calculated for each marker in regions of interest, with mean densities used for statistical evaluation. IDO expression in tumour cells was assessed by calculating the percentage of IDO+ tumour cells relative to all tumour cells; tumours with less than 1% of IDO+ tumour cells were categorized as negative. Myeloid cell densities were categorized into two categories (high vs. low) based on receiver-operating characteristics (ROC) curve analysis. The point closest to (0,1) coordinate was used as cut-point as suggested previous literature27. The density cutoffs based on ROC analysis are presented in Table S1.
To analyse spatial interactions between myeloid cells and tumour cells, we used the spatstat (3.0.5) package to calculate nearest neighbour distances (NNDs) from myeloid cells to their closest neighbour points of specific categories (e.g., tumour cells) (Fig. 2A,B). To visualize the results, scaled intensities of myeloid cell markers were plotted against NNDs from tumour cells using the ggplot2 (3.3.3) package and generalized additive model smoothing [formula y ~ s(x)] (Fig. 2C–E).
Nearest neighbour distance (NND) analysis for myeloid cells and tumour cells. (A) Example multiplex immunohistochemistry image representing granulocytes (CEACAM8+), monocytic cells (CD14+), mast cells (TPSAB1+) and tumour cells (KRT+), as well as IDO and ARG1 expression. (B) Cell phenotype map and NNDs from myeloid cells to the closest tumour cell. (C) Boxplots representing mean NNDs of myeloid cell phenotypes in metastases and primary tumours. (D) Boxplots representing mean NNDs of IDO+ monocytic cells and ARG1+ granulocytes between metastases and primary tumours. (E) Boxplot representing mean NNDs between IDO+ and IDO− monocytic cells between ARG1+ and ARG1− granulocytes in metastatic tumours. Wilcoxon rank-sum tests were applied for mean comparison. *** P < 0.0001.
Outcomes and definitions
Comorbidity was classified using the Royal College of Surgeons Comorbidity Score (RCSCC)28. Disease-free interval (DFI) was defined as the time between primary tumour resection and pulmonary metastasis detection. None of the patients had a lung-first approach. Pulmonary metastases diagnosed within 6 months post-primary treatment were classified as synchronous, while those detected later were labelled metachronous. The primary endpoint was 5-year overall survival following pulmonary metastasectomy. Given that only one death was unrelated to cancer, cancer-specific survival was not analysed.
Tumour regression grade (TRG) following neoadjuvant treatment was assessed by a pathologist (V-M.P) using a modified Dworak grading system29.
Statistical analysis
Categorical variables were compared using Chi-square or Fisher’s exact tests, while continuous variables were analysed using Student’s T-test or Mann-Whitney U-test. Normality and skewness of data distribution was visually inspected and confirmed with z-scores of skewness values for adequate use of statistical tests. Spearman coefficients were calculated for correlation analyses. Kaplan-Meier curves were used to visualize survival rates, and hazard ratios (HRs) with 95% confidence intervals (CIs) were computed using Cox proportional hazards regression models. The multivariable models were adjusted for sex (male/female), age (continuous variable), comorbidity (RCSCC 1/≥2), neoadjuvant therapy (no/yes), number of pulmonary metastases at diagnosis (1/≥2), size of the largest resected metastasis (continuous variable), former liver metastases (no/yes), synchronicity of pulmonary metastases (synchronous/metachronous), laterality of metastases (unilateral/bilateral), adjuvant therapy (no/yes), and time interval of surgery (2000–2010/2011–2020). The selection of adjustable variables was based on a recent meta-analysis30. Of all adjustable variables, missing values were detected in 7.7% (4 patients) of adjuvant therapy data and were imputed using the mode value (chemotherapy). The Schoenfeld residuals were analysed to ensure the proportionality of hazards assumption; no violations occurred (Fig. S2). Patients with an incomplete R1 resection or death within 30 days post-surgery were excluded from survival analysis. All statistical tests were two-tailed and the statistical significance was set at p < 0.05. Statistical analysis was performed using RStudio (version 2024.09.1) and R statistical programming (version 4.0.3).
Ethical aspects
The Oulu University Hospital Ethics Committee (EETMK 152/2008) approved the study. The Finnish National Authority of Medicolegal Affairs (VALVIRA) waived the need for informed consent due to the retrospective nature of the study (D.no 3916/06.01.03.01/2016). The study was performed in accordance with the Declaration of Helsinki.
Results
Baseline characteristics
This study examined 106 pulmonary metastasectomies performed on 74 patients, including 36 repeat procedures on 21 individuals. The median DFI following primary CRC surgery was 337 days (IQR 0–783). Bilateral pulmonary metastases were observed in 16% of patients (n = 12), and 34% (n = 25) presented with more than one pulmonary metastasis. One patient had microscopic tumour residuals in the surgical margin, and three underwent metastasis enucleation, all classified as R1 resections and excluded from survival analysis. A history of CRC liver metastasis treatment was reported in 43% of patients (n = 33). The median follow-up period was 26.5 months (IQR 18.6–48.48, range 1–209 months). An overall 5-year survival rate was 32%.
Neoadjuvant chemotherapy was given to 42% of patients (n = 45) for pulmonary metastases. For primary tumours, preoperative radiotherapy was given to 11% (n = 7), chemotherapy to 4.6% (n = 3), and chemoradiotherapy 23% (n = 15). Post-metastasectomy, only two patients did not receive any adjuvant treatment, while 93% (n = 54) received chemotherapy and 3% (n = 2) underwent radiotherapy. Tumour regression grading revealed minimal regression (TRG = 1) in 94% of neoadjuvant treated pulmonary metastases.
Myeloid cell densities
Analysis was successful for 291 TMA cores from 91 pulmonary metastases and 153 TMA cores from 54 primary tumours. Myeloid cells were predominantly located in the stromal areas of tumours, with higher densities in the invasive margins than in tumour centres (Table S2). Metastases had higher densities of invasive margin granulocytes and mast cells, as well as higher tumour centre granulocyte densities, compared to the primary tumours (Fig. S3). In metastases, monocytic cells in the invasive margin were more mature (HLA-DR+) compared to those in the tumour centre, a pattern not observed in primary tumours (Table S3). Monocytic cell maturity, based on HLA-DR expression, did not significantly differ between metastatic and primary tumours, though FCGR3 expression in monocytic cells was significantly higher in the invasive margin of metastases (p < 0.001) (Fig. 3A). No significant correlations were identified among different myeloid cell lineages or between their densities in primary tumours and metastases (Fig.s S4 and S5). Neoadjuvant chemotherapy was also not associated with myeloid cell densities or IDO/ARG1 expression of monocytic cells/granulocytes (Table S4).
Characteristics of CD14+ monocytic cells and CEACAM8+ granulocytic cells in colorectal cancer pulmonary metastases and corresponding primary tumours. Boxplots compare proportions of HLA-DR and FCGR3 expression in the monocytic cells (A), the densities and proportions of IDO+ monocytic cells (B) and ARG1+ granulocytes (C) in the invasive margins (IM) and tumour centres (TC) between first resected metastases (MET) and corresponding primary tumours (PRIM). Statistical significance was tested with the Wilcoxon signed rank test.
IDO and ARG1 expression and distribution
Of all CD14+ monocytic cells, 2.4% expressed IDO in metastases, and 1.8% in primary tumours. 2.9% of tumour cells also expressed IDO in the metastases. The cell density ranges of IDO expressing monocytic cell subtypes in metastases are presented in Fig. S6. IDO expression was higher in mature (HLA-DR+ 2.6% vs. HLA-DR− 1.6%) and FCGR3+ monocytic cells (FCGR3+ 3.1% vs. FCGR3− 1.2%) in the metastases, a same pattern also noticed in the primary tumours. In terms of cell densities, HLA-DR+IDO+ and FCGR3+IDO+ monocytic cells in the invasive margin in the metastases had the highest densities up to 184 cells/mm2 and 182 cells/mm2, respectively (Fig. S6). Densities of IDO expressing immature HLA-DR− monocytic cells ranged from 0 to 63 cells/mm2, with a median cell density of 0 cells/mm2, indicating their sparse presence. All IDO+ monocytic cell subtypes were significantly correlated with each other (invasive margin: rs=0.45–0.95, p < 0.001; Fig. S7) and with the proportion of IDO+ tumour cells, as well as with CD3+ and CD8+ cell densities. Invasive margin monocytic cell IDO expression was significantly higher in metastases compared to the primary tumours (Fig. 3B). In spatial analysis, IDO+ monocytic cells were located closer to the tumour cells compared to the IDO− counterparts (Fig. 2E).
Most of the granulocytes expressed ARG1 (72.3% in the metastases; 71.5% in the primary tumours). The expression was higher in FCGR3+ granulocytes compared to FCGR3− granulocytes (Fig. S8), with invasive margin median densities of 46 cells/mm2 and 8 cells/mm2, respectively (Table S3, Fig. S8). FCGR3+ARG1+ and FCGR3−ARG1+ granulocyte subtypes were significantly correlated (invasive margin: rs=0.63, p < 0.001; Fig. S7). The granulocyte ARG1 expression and monocytic cell IDO expression, however, were not intercorrelated, suggesting varying mechanisms of immunosuppression in the metastases. ARG1+ granulocytes, irrespective of FCGR3 expression status, had a weaker correlation with CD3+ and CD8+ T-cells compared to IDO+ monocytic cells (Fig. S7). The invasive margin ARG1 expression was higher in the metastases compared to the primary tumours (Fig. 3C). In the spatial analysis, ARG1− granulocytes were located closer to the tumour cells compared to the ARG1+ counterparts (Fig. 2E).
The proportion of IDO+ tumour cells was significantly correlated with IDO+ monocytic cells in the invasive margin and tumour centre, but not with ARG1+ granulocytes or mast cells (Fig. S7). The proportion of IDO+ tumour cells also positively correlated with the densities of CD8+ (tumour centre rs=0.49, p < 0.001; invasive margin rs=0.66, p < 0.001, Fig. S7) and CD3+ T-cells (tumour centre rs=0.42, p < 0.01; invasive margin rs=0.64, p < 0.001, Fig. S7).
Survival analysis
The prognostic impact of IDO expression differed between the invasive margin and tumour centre. Higher densities of IDO+ monocytic cells in the tumour centre were associated with longer 5-year overall survival (low 12% vs. high 63%, p < 0.001, Fig. 4; adjusted HR 0.17, 95% CI 0.05–0.59, p = 0.005, Table 1). This prognostic association was limited to mature and FCGR3+ monocytic cells (Fig. S9, Table S5). Conversely, in the invasive margin, overall IDO+ monocytic cell population was not prognostic, but higher densities of immature (HLA-DR−) IDO+ monocytic cells were associated with shorter survival (adjusted HR 5.60, 95% CI 1.70–18.48, p = 0.005, Table S5, Model 2). Similarly, IDO− monocytic cell densities in the invasive margin were associated with poorer outcomes (Fig. S10; adjusted HR 4.19, 95% CI 1.45–12.05, p = 0.008, Table 1) as did overall monocytic cell densities (Fig. S11, adjusted HR 2.87, 95% CI 1.10–7.50, p = 0.031, Table S5). The proportion of IDO expression in the tumour cells was not associated with prognosis (Table 1).
Kaplan-Meier 5-year survival curves after pulmonary metastasectomy stratified by overall (intraepithelial + stromal) CD14+ monocytic cell densities and their IDO expression and CEACAM8+ granulocyte cell densities and their arginase1 expression (ARG1) in the invasive margin (IM) and centre (TC) of the metastases. Log rank tests were applied.
Even after adjusting for CD3+ and CD8+ T-cell density-based immune cell scoring, higher densities of immature (HLA-DR−) IDO+ monocytic cells in the invasive margin remained associated with shorter survival (adjusted HR 26.37, 95%CI 4.95–140.42, p < 0.001, Table S5). Similarly, higher densities of FCGR3+IDO+ monocytic cells, a subtype significantly correlated with HLA-DR−IDO+ subtype, also had a suggestive adverse prognostic effect (adjusted HR 3.91, 95% CI 0.97–15.76, p = 0.056, Table S5). In the tumour centre, IDO+ monocytic cells remained associated with longer survival (adjusted HR 0.20, 95% CI 0.06–0.69, p = 0.011, Table S5).
Overall granulocyte densities, regardless of ARG1 expression, were not significantly associated with 5-year survival (Fig. 4; Table 1). However, a higher density of FCGR3−ARG1+ granulocyte subset in the tumour centre associated with shorter survival when adjusted for the immune cell score (adjusted HR 2.68, 95% CI 1.05–6.86, p = 0.039; Table S6). This cell subtype exhibited considerable variability in density, ranging from 0 to 525 cells/mm2, with a median of 6.21 cells/mm2 (Fig. S8). A similar, though non-significant trend toward poorer survival was observed for higher densities of ARG1+ granulocytes in the centre of metastasis (adjusted HR 2.30, 95% CI 0.89–5.96, p = 0.087, Table S6).
In the sensitivity analysis including only neoadjuvant-naïve patients (n = 41), higher densities of IDO− monocytic cells and immature HLA-DR−IDO+ monocytic cells in the invasive margin remained associated with worse survival (adjusted HRs 10.42, 95%CI 2.28–47.57, p = 0.002 and 41.19, 95% CI 2.31–735.46, p = 0.011, respectively; Table S7), whereas in the tumour centre, densities of monocytic cells, irrespective of their IDO expression status, were not significantly associated with survival after adjusting for confounders (Table S7). IDO+FCGR3+ monocytic cells were not associated with survival in the sensitivity analysis, but ARG1+FCGR3− granulocytes in the invasive margin and ARG1+ granulocytes in the tumour centre were also associated with worse survival (adjusted HRs 6.63 95% CI 1.10–39.78, p = 0.039, and 10.95, 95%CI 1.69–70.97, p = 0.012, respectively; Table S8).
Association with clinical parameters
Higher density of IDO+ monocytic cells in the tumour centre of metastases was associated with smaller metastasis size and lower number of resected metastases (Table 2), whereas higher ARG1+ granulocyte density in the invasive margin of metastases was associated with a history of liver metastasis (Table S9).
Discussion
Here, we conducted a comprehensive analysis of myeloid cell densities and IDO and ARG1 expression patterns in resected pulmonary metastases and their corresponding primary tumours. Our findings highlight the differences in myeloid cell profiles between the invasive margin and tumour centre of CRC pulmonary metastases. The invasive margin exhibits higher myeloid cell densities, greater maturity and FCGR3 positivity in monocytic cells, and increased IDO and ARG1 expression compared to the tumour centre. Prognostically, IDO+ monocytic cells seem to behave differently in these regions. After adjusting for confounders and T-cell densities, we found that in the invasive margin, higher densities of monocytic cells, including those expressing IDO (HLA-DR−IDO+ and FCGR3+IDO+) were associated with worse prognosis. Conversely, in the tumour centre, IDO+ monocytic cells (especially mature HLA-DR+ and FCGR3+) were associated with better prognosis. In neoadjuvant naïve patients, however, IDO expression had a negative prognostic impact only in immature monocytic cells in the invasive margin. In the CEACAM8+ granulocytic lineage, higher densities of FCGR3−ARG1+ cells in the tumour centre (and higher densities of FCGR3−ARG1+ cells in the invasive margin of neoadjuvant-naïve patients) were independently associated with worse survival, suggesting a potential role for ARG1 in adverse outcomes.
Few studies have examined the expression of immunosuppressive enzymes ARG1 and IDO at the single-cell level in specific cell types within the tumour microenvironment. Most evidence on the prognostic value of IDO expression derives from semiquantitative analysis without distinguishing between tumour regions or cell types13,14,15,31,32,33,34,35. Some studies have associated IDO with favourable outcomes14,15,31,32,33,35, while others link it to poor survival13,34. In studies that distinguish between the invasive margin and tumour centre, a TMA-based single-marker IHC study on stage I-III CRC demonstrated a negative survival effect of IDO expression in the invasive margin, but not in the tumour centre12, which was also demonstrated in melanoma and lung cancer36,37. Similarly, a recent multiplexed immunofluorescence study on lung cancer showed that higher IDO expression in peritumoural tertiary lymphoid structures was associated with worse survival38, whereas higher IDO expression in tumour cells was associated with better survival. On the other hand, a recent TMA-based study on oesophageal cancer demonstrated higher IDO expression in the invasive margin being associated with better survival, the difference being more significant in lower T-stage tumours39.
Concerning CRC, we previously published a comprehensive multiplexed IHC analysis on the myeloid cell densities in a cohort of primary tumours17, and found that higher IDO+ monocytic cell densities in both the invasive margin and the tumour centre were independently associated with favourable disease-specific survival, the association being stronger in stage I-III primary tumours compared to stage IV tumours. Higher ARG1+ granulocyte density was similarly associated with better survival in both the invasive margin and tumour centre, and the association was limited to stage I-III tumours. Supporting the notion of a weakening favourable survival effect of IDO expression alongside tumour progression, possibly developing towards a more negative survival effect in the invasive margin, a recent study incorporating single-cell sequencing and spatial transcriptomics in stage I-III CRC primary tumours revealed a stronger immunosuppressive milieu in the epithelial-to-mesenchymal transition (EMT) pseudotime-endpoint trajectories40, suggesting that the invasive margin—frequent site for EMT—might have a more immunosuppressive milieu compared to the tumour centre. EMT marker expression between metastases and primary tumours is also proposed to differ during metastatic dissemination41 and thus EMT might partly explain the prognostic differences of IDO/ARG1 in tumour types noted in our study. Additionally, myeloid derived suppressor cells and monocytes have a role in the immunosuppressive premetastatic niche formation even before the arrival of circulating tumour cells42, which could also account for the prognostic differences between the IDO and ARG1 expression in metastases and primary tumours17.
The strong correlation between CD8+ T-cell and IDO+ monocytic cell densities in the metastases in our study—noted also in the primary tumours—may appear contradictory, given the immunosuppressive nature of IDO. However, it aligns with a recent TCGA-based RNA sequencing study demonstrating a strong positive correlation between IDO1 expression and CD8A expression (a CD8+ T-cell biomarker) in most cancer types43. Functional assays also show the immunosuppressive mechanisms of IDO being mediated through TCR ζ-chain depletion and T-cell exhaustion, along with a reduction T-cells densities2. Similarly, the expression of other immune checkpoint molecules is generally positively correlated with CD8+ T-cells densities44,45. However, the prognostic differences of IDO between the primary tumours and metastases still remain unclear. Several studies report significant differences in immune composition not only between primary tumours and matched metastases46, but also between metastases in different tissue sites47,48. For instance, a study incorporating imaging mass cytometry and RNA sequencing found significantly higher macrophage and monocyte densities in brain metastases compared to primary brain gliomas, with differing interaction-avoidance scores between the tumour types46. Likewise, a recent meta-analysis comparing immunotherapy response−predictive biomarkers—such as PD-L1/PD-1 expression, T-cell infiltration and MSI-status—between primary tumours and corresponding metastases concluded significant discordance between the two49, which also could be seen to align with our findings. In our study, the IDO expression in the invasive margin was significantly higher in the metastases compared to the corresponding primary tumours. Still, the surgical patient selection in metastasectomy cohorts might also significantly contribute to the discordance in the comparative analyses between the metastases and primary tumours, and more rigorous comparison would require samples from more disseminated diseases.
FCGR3 (CD16), a surface receptor expressed on various myeloid cells, plays a critical role in antibody-dependent cytotoxicity50 and mediates antigen uptake, processing, and presentation51. Circulating CD14+ monocytes can transition from classical CD14+FCGR3− monocytes into FCGR3+ intermediate and non-classical monocytes52. An increase in circulating intermediate CD14+FCGR+ monocytes has been linked to inflammatory diseases such as atherosclerosis, rheumatoid arthritis and tuberculosis53. In tumour-infiltrating monocytic cells, the immunobiological role of FCGR3 expression is not completely understood. FCGR3 is shown to be highly expressed in immunosuppressive monocytes and TAM populations54. However, an ovarian cancer study found spatially co-localized enrichment of FCGR3 expression in CD68+ myeloid cells, T-cells and NK-cells, associated with improved survival55. In CRC, higher densities of tumour infiltrating FCGR3+ myeloid cells have been linked to improved survival56. In our study, the prognostic value of IDO+ monocytic cells in pulmonary metastases was dependent on FCGR3 expression, and CD14+FCGR3+IDO+ cells showed opposing prognostic effects in the invasive margin and tumour centre when including all patients in the analysis. However, this pattern was not observed in neoadjuvant naïve primary CRC tumours17, suggesting that the favourable prognostic effect of FCGR3+IDO+ monocytic cells in the tumour centre might reflect responses to neoadjuvant treatment.
Monocytic cell maturity can be evaluated based on HLA-DR expression and circulating immature HLA-DR−/low CD14+ monocytes have been linked to systemic immunosuppression and immunotherapy resistance57. A prior CRC study similarly reported that higher densities of CD14+HLA-DR− monocytic cells were associated with worse survival, whereas mature HLA-DR+ monocytic cells were associated with better survival58. In our study, higher density of IDO+ HLA-DR− immature monocytic cells in the invasive margin was strongly associated with worse survival, with this finding being even more pronounced in neoadjuvant naïve patients. This might suggest a role for IDO in immunosuppression associated with immature monocytic cells (with a monocytic myeloid derived suppressor cell-like phenotype), though interpretation requires caution due to low cell densities and lack of functional assays demonstrating immunosuppressive capacity. While HLA-DR−IDO+ monocytic cells were present at very low densities, their strong correlation with the more abundant FCGR3+IDO+ monocytic cells may help explain their unexpectedly pronounced association with poor survival, though this causality remains speculative.
This study has several limitations. First, the selection bias inherent in pulmonary metastasectomy cohorts—representing a highly selected subgroup of patients—restricts generalizability to all CRC patients with metastatic disease. The selection bias likely results in differing myeloid cell distribution compared to patients excluded from pulmonary metastasectomy. For example, myeloid cell densities in primary tumours of this study were lower compared to some earlier studies17,58, and primary tumours were of higher stages. Additionally, all patients had microsatellite stable tumours, and the results cannot be generalized to metastases from microsatellite unstable CRC. Second, TMAs only provide a snapshot of the tumour microenvironment, potentially limiting cell density estimates. However, numerous studies have shown reproducible results using TMAs. Third, data on RAS mutation status was not available. It was not used in clinical decision making at the beginning of the study period, and therefore post hoc determination of RAS status was not performed. Last, the study encompasses a 20-year period during which diagnostics and treatment might have evolved, however, this was addressed in the multivariable analysis. The strengths of this study include a reasonably sized, dual-institutional cohort. The machine learning based digital image analysis along with the multiplexed immunohistochemistry assay enabled detailed cell subtyping, as well as more precise estimates of cell densities compared to semi-quantitative visual estimates. The mortality data from Statistics Finland had full coverage, ensuring the reliability of the primary study outcome.
In conclusion, our results suggest distinct prognostic roles for IDO and ARG1 expression in CRC metastases compared to primary tumours. While higher overall IDO+ monocytic cell densities generally predict favourable survival, the adverse effect of IDO expression in FCGR3+HLA-DR− immature monocytic cells in metastases underscores the complexity of immune interactions in metastatic dissemination. Similarly, higher density of ARG1+FCGR3− granulocytes is an independent marker of poor survival. These findings provide novel insights into the tumour-immune dynamics in CRC metastasis, offering potential avenues for therapeutic targeting.
Data availability
Data generated and/or analysed during this study are not publicly available. The sharing of data will require approval from relevant ethics committees and/or biobanks. Further information including the procedures to obtain and access data from Finnish Biobanks are described at https://finbb.fi/en/fingenious-service.
References
Uyttenhove, C. et al. Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat. Med. 9(10), 1269–74 (2003).
Fallarino, F. et al. The Combined Effects of Tryptophan Starvation and Tryptophan Catabolites Down-Regulate T Cell Receptor ζ-Chain and Induce a Regulatory Phenotype in Naive T Cells. The J. Immunol. 176(11), 6752–61 (2006).
Munder, M. Arginase: An emerging key player in the mammalian immune system: Review. Br. J. Pharmacol. 158(3), 638–51 (2009).
Graboń, W., Mielczarek-Puta, M., Chrzanowska, A. & Barańczyk-Kuźma, A. L-arginine as a factor increasing arginase significance in diagnosis of primary and metastatic colorectal cancer. Clin. Biochem. 42(4–5), 353–7 (2009).
Grzywa TM, Sosnowska A, Matryba P, Rydzynska Z, Jasinski M, Nowis D, et al. Myeloid Cell-Derived Arginase in Cancer Immune Response. Front. Immunol. Front. Media 11 (2020).
Meireson A, Devos M, Brochez L. IDO Expression in Cancer: Different Compartment, Different Functionality? Front. Immunol. 11 (2020).
Munder, M. Arginase I is constitutively expressed in human granulocytes and participates in fungicidal activity. Blood 105(6), 2549–56 (2005).
Lorentzen CL, Martinenaite E, Kjeldsen JW, Holmstroem RB, Mørk SK, Pedersen AW, et al. Arginase-1 targeting peptide vaccine in patients with metastatic solid tumors – A phase I trial. Front Immunol. 13 (2022).
Naing A, Papadopoulos KP, Pishvaian MJ, Rahma O, Hanna GJ, Garralda E, et al. First-in-human phase 1 study of the arginase inhibitor INCB001158 alone or combined with pembrolizumab in patients with advanced or metastatic solid tumours. BMJ Oncol. 3(1) (2024).
Long, G. V. et al. Epacadostat plus pembrolizumab versus placebo plus pembrolizumab in patients with unresectable or metastatic melanoma (ECHO-301/KEYNOTE-252): a phase 3, randomised, double-blind study. Lancet Oncol. 20(8), 1083–97 (2019).
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74(3), 229–63 (2024).
Ferdinande, L. et al. Clinicopathological significance of indoleamine 2,3-dioxygenase 1 expression in colorectal cancer. Br. J. Cancer 106(1), 141–7 (2012).
Ma, W. J. et al. Indoleamine-2,3-dioxygenase 1/cyclooxygenase 2 expression prediction for adverse prognosis in colorectal cancer. World J. Gastroenterol. 24(20), 2181–90 (2018).
Lee, S. J. et al. CD274, LAG3, and IDO1 expressions in tumor-infiltrating immune cells as prognostic biomarker for patients with MSI-high colon cancer. J. Cancer Res. Clin. Oncol. 144(6), 1005–14 (2018).
Schollbach, J. et al. Prognostic value of tumour-infiltrating CD8 + lymphocytes in rectal cancer after neoadjuvant chemoradiation: is indoleamine-2,3-dioxygenase (IDO1) a friend or foe?. Cancer Immunol. Immunother. 68(4), 563–75 (2019).
Ma, Z. et al. Overexpression of Arginase-1 is an indicator of poor prognosis in patients with colorectal cancer. Pathol. Res. Pract. 215(6), 152383 (2019).
Elomaa H, Härkönen J, Väyrynen SA, Ahtiainen M, Ogino S, Nowak JA, et al. Quantitative Multiplexed Analysis of Indoleamine 2,3-Dioxygenase (IDO) and Arginase-1 (ARG1) Expression and Myeloid Cell Infiltration in Colorectal Cancer. Modern Pathol. 37(4) (2024).
Pugh, S. A. et al. Site and stage of colorectal cancer influence the likelihood and distribution of disease recurrence and postrecurrence survival data from the FACS randomized controlled trial. Ann. Surg. 263(6), 1143–7 (2016).
Väyrynen, V. et al. Incidence and management of patients with colorectal cancer and synchronous and metachronous colorectal metastases: a population-based study. BJS Open. 4(4), 685 (2020).
Colorectal Cancer Survival Rates | Colorectal Cancer Prognosis. https://www.cancer.org/cancer/colon-rectal-cancer/detection-diagnosis-staging/survival-rates.html (2021).
Brierley JD, Gospodarowicz MK, Wittekind C. TNM Classification of Malignant Tumours, 8th edition. Brierley JD, Gospodarowicz MK, Wittekind C, (ed). (2016).
Karjula, T. et al. CD3 + and CD8 + T-Cell-Based Immune Cell Score and PD-(L)1 Expression in Pulmonary Metastases of Microsatellite Stable Colorectal Cancer. Cancers (Basel). 15(1), 206–22 (2023).
Pagès, F. et al. International validation of the consensus Immunoscore for the classification of colon cancer: A prognostic and accuracy study. Lancet 391(10135), 2128–39 (2018).
Fujiyoshi K, Bruford EA, Mroz P, Sims CL, O’Leary TJ, Lo AWI, et al. Standardizing gene product nomenclature-a call to action. Proc Natl Acad Sci USA 118(3) (2021).
Bankhead P, Loughrey MB, Fernández JA, Dombrowski Y, McArt DG, Dunne PD, et al. QuPath: Open source software for digital pathology image analysis. Sci Rep. ;7(1) (2017).
Schindelin, J. et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 9, 676–82 (2012).
Rota, M. & Antolini, L. Finding the optimal cut-point for Gaussian and Gamma distributed biomarkers. Comput. Stat. Data Anal. 69, 1–14 (2014).
Armitage, J. N. & Van Der Meulen, J. H. Identifying co-morbidity in surgical patients using administrative data with the Royal College of Surgeons Charlson Score. British Journal of Surgery 97(5), 772–81 (2010).
Kim, S. H. et al. What is the ideal tumor regression grading system in rectal cancer patients after preoperative chemoradiotherapy?. Cancer Res Treat 48(3), 998 (2016).
Gkikas A, Kakos C, Lampridis S, Godolphin PJ, Patrini D. Preoperative prognostic factors for 5-year survival following pulmonary metastasectomy from colorectal cancer: a systematic review and meta-analysis. Eur. J. Cardio-Thoracic Surgery 63(3) (2023).
Ma, W. et al. High expression of indoleamine 2, 3-dioxygenase in adenosquamous lung carcinoma correlates with favorable patient outcome. J Cancer 10(1), 267–76 (2019).
Jacquemier, J. et al. High expression of indoleamine 2,3-dioxygenase in the tumour is associated with medullary features and favourable outcome in basal-like breast carcinoma. Int J Cancer. 130(1), 96–104 (2012).
Li, S. et al. Mechanism and prognostic value of indoleamine 2,3-dioxygenase 1 expressed in hepatocellular carcinoma. Cancer Sci. 109(12), 3726–36 (2018).
Inaba, T. et al. Role of the immunosuppressive enzyme indoleamine 2,3-dioxygenase in the progression of ovarian carcinoma. Gynecol Oncol 115(2), 185–92 (2009).
Ishio, T. et al. Immunoactivative role of indoleamine 2,3-dioxygenase in human hepatocellular carcinoma. Journal of Gastroenterology and Hepatology 19(3), 319–26 (2004).
Zeng Z, Yang F, Wang Y, Zhao H, Wei F, Zhang P, et al. Significantly different immunoscores in lung adenocarcinoma and squamous cell carcinoma and a proposal for a new immune staging system. Oncoimmunology 9(1) (2020).
Chevolet, I. et al. Peritumoral indoleamine 2,3-dioxygenase expression in melanoma: An early marker of resistance to immune control?. Br. J. Dermatol. 171(5), 987–95 (2017).
Bessede, A. et al. Upregulation of indoleamine 2,3-dioxygenase 1 in tumor cells and tertiary lymphoid structures is a hallmark of inflamed non–small cell lung cancer. Clin. Cancer Res. 29(23), 4883–93 (2023).
Loeser H, Kraemer M, Gebauer F, Bruns C, Schröder W, Zander T, et al. Indoleamine 2,3-Dioxygenase (IDO) Expression Is an Independent Prognostic Marker in Esophageal Adenocarcinoma. J. Immunol. Res. (2020).
Wang M, Deng C, Yang C, Yan M, Lu H, Zhang Y, et al. Unraveling temporal and spatial biomarkers of epithelial-mesenchymal transition in colorectal cancer: insights into the crucial role of immunosuppressive cells. J Transl Med. 21(1) (2023).
Yao, D., Dai, C. & Peng, S. Mechanism of the mesenchymal-epithelial transition and its relationship with metastatic tumor formation. Mol. Cancer Res. 9(12), 1608–20 (2011).
Liu, Y. & Cao, X. Characteristics and Significance of the Pre-metastatic Niche. Cancer Cell. 30, 668–81 (2016).
Panda A, Ganesan S. Genomic and Immunologic Correlates of Indoleamine 2,3-Dioxygenase Pathway Expression in Cancer. Front Genet. 12 (2021).
Lee, L. H. et al. Patterns and prognostic relevance of PD-1 and PD-L1 expression in colorectal carcinoma. Modern Pathol. 29(11), 1433–42 (2016).
Ko, Y. S. & Pyo, J. S. Clinicopathological significance and prognostic role of tumor-infiltrating lymphocytes in colorectal cancer. Int. J. Biol. Markers 34(2), 132–8 (2019).
Karimi, E. et al. Single-cell spatial immune landscapes of primary and metastatic brain tumours. Nature 614(7948), 555–63 (2023).
Zou Y, Ye F, Kong Y, Hu X, Deng X, Xie J, et al. The Single-Cell Landscape of Intratumoral Heterogeneity and The Immunosuppressive Microenvironment in Liver and Brain Metastases of Breast Cancer. Adv. Sci. 10(5) (2023).
Herrera-Rios D, Mughal SS, Teuber-Hanselmann S, Pierscianek D, Sucker A, Jansen P, et al. Macrophages/Microglia Represent the Major Source of Indolamine 2,3-Dioxygenase Expression in Melanoma Metastases of the Brain. Front Immunol. 11 (2020).
Zou Y, Hu X, Zheng S, Yang A, Li X, Tang H, et al. Discordance of immunotherapy response predictive biomarkers between primary lesions and paired metastases in tumours: a multidimensional analysis. EBioMedicine 63 (2021).
Yeap WH, Wong KL, Shimasaki N, Teo ECY, Quek JKS, Yong HX, et al. CD16 is indispensable for antibodydependent cellular cytotoxicity by human monocytes. Sci Rep. 6 (2016).
Junker F, Gordon J, Qureshi O. Fc Gamma Receptors and Their Role in Antigen Uptake, Presentation, and T Cell Activation. Front. Immunol. Front. Media 11 (2020).
Olingy, C. E., Dinh, H. Q. & Hedrick, C. C. Monocyte heterogeneity and functions in cancer. J. Leukoc Biol. 106(2), 309–22 (2019).
Kapellos TS, Bonaguro L, Gemünd I, Reusch N, Saglam A, Hinkley ER, et al. Human monocyte subsets and phenotypes in major chronic inflammatory diseases. Front. Immunol. (2019).
Yofe, I. et al. Anti-CTLA-4 antibodies drive myeloid activation and reprogram the tumor microenvironment through FcγR engagement and type I interferon signaling. Nat. Cancer 3(11), 1336–50 (2022).
Nersesian, S. et al. Improved overall survival in patients with high-grade serous ovarian cancer is associated with CD16a+ immunologic neighborhoods containing NK cells, T cells and macrophages. Front. Immunol. https://doi.org/10.3389/fimmu.2023.1307873 (2023).
Sconocchia, G. et al. Tumor infiltration by FcIγRIII (CD16)+ myeloid cells is associated with improved survival in patients with colorectal carcinoma. Int. J. Cancer 128(11), 2663–72 (2011).
Mengos, A. E., Gastineau, D. A. & Gustafson, M. P. The CD14 + HLA-DrlO/NEG monocyte: An immunosuppressive phenotype that restrains responses to cancer immunotherapy. Front. Immunol. https://doi.org/10.3389/fimmu.2019.01147 (2019).
Väyrynen JP, Haruki K, Väyrynen SA, Lau MC, Dias Costa A, Borowsky J, et al. Prognostic significance of myeloid immune cells and their spatial distribution in the colorectal cancer microenvironment. J. Immunother Cancer. 9(4) (2021).
Acknowledgements
The study benefited from samples/data from Northern Finland Biobank Borealis (Oulu, Finland) (https://oys.fi/biopankki/) and from Central Finland Biobank (Jyväskylä, Finland) (https://https://www.hyvaks.fi/sairaala-nova/biopankki).
Funding
This study was funded by Instrumentarium Science Foundation (O.H), Mary and Georg C. Ehrnrooth Foundation (O.H), Finnish State Research Funding (O.H, J-P.M, M.A, J.P.V, T.T.S), Cancer Foundation Finland (J.P.V, T.T.S), Sigrid Jusélius Foundation (J.P.V, T.T.S), J&A Erkko Foundation (J-P.M, T.T.S), Emil Aaltonen Foundation (J.P.V), Academy of Finland (T.T.S), iCAN Precision Medicine Flagship of the Academy of Finland (TTS), Relander Foundation (T.T.S). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization: T.Ka, H.E, S.A.V., O.H, F.Y, J.P.V Methodology: T.Ka., H.E, S.A.V., J.P.V. Formal Analysis: T.Ka, H.E, J.P. VInvestigation: All Authors Data Curation: All Authors Writing—Original Draft Preparation: All Authors Writing—Review and Editing: All Authors Supervision: O.H. F.Y. J.P.V. Funding Acquisition: O.H. J.P.V. J-P.M.
Corresponding author
Ethics declarations
Competing interests
T.T.S. declares consultation fee from Amgen Finland. T.T.S is CEO and co-owner of Healthfund Finland and Clinical Advisory board member of LS Cancer Diag. The other authors declare no conflict of interest.
Ethics approval and consent to participate
The Oulu University Hospital Ethics Committee (EETMK 152/2008) approved the study. The Finnish National Authority of Medicolegal Affairs (VALVIRA) waived the need for informed consent due to the retrospective nature of the study. The study was performed in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Karjula, T., Elomaa, H., Väyrynen, S.A. et al. Quantitative analysis of myeloid cell patterns and immunosuppressive enzyme (IDO, ARG1) expression in colorectal cancer pulmonary metastases and corresponding primary tumours. Sci Rep 16, 11770 (2026). https://doi.org/10.1038/s41598-026-42097-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-42097-8