Abstract
Iron deficiency anemia (IDA) is a major contributor to global morbidity and disease burden. While observational studies have suggested associations between autoimmune diseases (ADs) and IDA, the causal nature of these relationships remains unclear. This study aimed to explore the potential causal effects of ADs on IDA. A bidirectional two-sample Mendelian randomization (MR) analysis was conducted using summary-level data from genome-wide association studies (GWAS). Genetic variants associated with exposures at genome-wide significance (P < 5 × 10⁻⁸) were selected as instrumental variables. Steiger filtering was applied to exclude SNPs explaining more variance in the outcome than in the exposure. Radial regression was used to detect outliers, and the inverse variance weighted (IVW) method served as the primary analytical approach. Sensitivity analyses were performed using MR-Egger regression, Cochran’s Q test, and the MR-PRESSO global test. IVW analysis revealed significantly increased risks of IDA associated with genetically predicted rheumatoid arthritis, inflammatory bowel disease, and ulcerative colitis after excluding variants in the MHC region. Suggestive associations were observed for ankylosing spondylitis, celiac disease, Crohn’s disease, and systemic lupus erythematosus, while the association with membranous nephropathy was no longer significant after MHC removal. This study provides evidence supporting potential causal relationships between several ADs and IDA, with the strongest evidence observed for rheumatoid arthritis, inflammatory bowel disease, and ulcerative colitis. Further investigations are warranted to elucidate the underlying biological mechanisms and to perform gender-specific and ancestry-stratified analyses.
Similar content being viewed by others
Introduction
Iron deficiency anemia (IDA) is the most common form of anemia caused by an iron deficiency in the body1. Patients afflicted by IDA often endure a number of serious health implications, including, profound fatigue, attention deficiency, restricted exercise capacity, impaired work performance, and poor maternal outcomes in pregnant women2. IDA is considered a major cause of morbidity and burden of disease worldwide, mostly in developing nations3. Statistics show that anemia exerts its influence on approximately a third of the world’s population, while women of child bearing age are particularly at risk of its development4.
Although the understanding of etiology and pathogenesis of IDA are not fully understood, reduced iron intake, defective iron absorption, increased menstrual flow, and occult intestinal bleeding play a crucial role in their development5,6. In addition, growing evidence suggests that autoimmune diseases (ADs) are closely related to IDA. For instance, various studies have reported the occurrence of IDA in systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), and ankylosing spondylitis (AS)7,8,9. Dube et al. showed that celiac disease (CeD) patients with digestive symptoms had an increased prevalence of IDA10.
Many investigations assessing the association between ADs and IDA are cross-sectional, leaving the causal nature of these relationships ambiguous. However, establishing causal connections not only enhances comprehension of the pathogenesis of autoimmune-derived IDA pathogenesis but also provides valuable insights for implementing autoimmune-orientated interventions against IDA in clinical settings. Therefore, there exists an imperative demand to elucidate the causative relationship between diverse categories of ADs and IDA.
Randomized controlled trials (RCTs) are acknowledged as the gold standard design for deducing causality. However, due to inadequate long-term compliance and ethical constraints, RCTs are often unfeasible to implement in most cases. Mendelian randomization (MR) analysis emerges as a statistical methodology that can help overcome these limitations. It uses genetic variants, such as single-nucleotide polymorphisms (SNPs), to assess the causal associations between an exposure and an outcome. However, to date, only a limited number of investigations utilizing Mendelian randomization to estimate the causal effects of multiple potential exposures on IDA have been reported11.
In this study, with the objective of exploring the causal relationship between a diverse array of ADs and IDA, we conducted a comprehensive two-sample MR analysis of eight ADs, including SLE, RA, inflammatory bowel disease (IBD), Crohn’s disease (CD), ulcerative colitis (UC), CeD, membranous nephropathy (MN), and AS. The primary goal of this study was to furnish a comprehensive overview of potential variable risk factors for IDA and to offer new insights into the etiology of IDA.
Materials and methods
Study design and framework
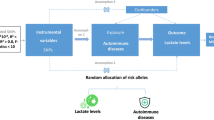
This investigation employs a two-sample MR framework, utilizing SNPs as IVs to explore the causal effects of ADs on IDA. Briefly, we selected common ADs such as SLE, RA, IBD, CD, UC, CeD, MN, and AS as exposure, and IDA as outcome. Three assumptions must be met to confirm the validity of MR analysis: (1) genetic variants directly affect the risk factor; (2) genetic variants are not associated with any known or unknown confounders; and (3) genetic variants affect the outcome through only the risk factor and not through other pathways. For a detailed discussion of these assumptions, refer to Greenland12. Our analysis utilized publicly available Genome Wide Association Study (GWAS) summary statistics; thus, we did not collect any new data, nor was further ethical approval required. Figure 1 and Supplementary Fig. 1 presents a schematic diagram summarizing the MR assumptions and a flowchart illustrating our bidirectional two-sample MR analysis.
Flowchart of our MR analysis. Include three MR assumptions and enrolled autoimmune diseases.
Autoimmune disease samples
Summary statistics for each of the eight autoimmune disease samples were sourced from multiple large, independent European GWAS meta-analyses, with none being part of the FinnGen or UK Biobank datasets. Summary statistics for AS were obtained from International Genetics of Ankylosing Spondylitis Consortium (IGAS), including 22,647 samples13. Summary statistics for CeD were extracted from Spanish Consortium on the Genetics of Coeliac Disease (CEGEC), encompassing 23,649 samples14. Summary statistics for IBD, UC, and CD were sourced from The International Inflammatory Bowel Disease Genetics Consortium (IIBDGC), with a total of 34,652 27,432 20,883 samples15. Summary statistics of MN were derived from the a latest GWAS meta-analysis, comprising 7,979 sample16. Summary statistics of RA came from a global GWAS meta-analysis, including 58,284 samples17. Finally, Summary statistics for SLE were obtained from a publicly available GWAS meta-analyses with 14,267 samples18. All participants in these studies were of European ancestry, detailed information on the datasets is provided in Table 1.
Iron deficiency anemia samples
To avoid participant overlap, we sourced genomic data on IDA traits from the FinnGen (R10). During the discovery stage, we extracted GWAS summary statistics for IDA from FinnGen Biobank’s latest release (R10, December 2023), which comprised 15,153 cases and 393,684 controls of European ancestry19. Further information about FinnGen is availabe at https://www.finngen.fi/en/access_results. Details of the included GWAS studies are summarized in Table 1.
Instrumental variable selection and validation
We conducted a series of stringent quality control measures to select IVs that adhere to the three fundamental assumptions of MR analysis, thereby ensuring its robustness and reliability. Initially, genetic variants linked to the exposure were pinpointed at the genome-wide significance level (p < 5 × 10− 8). Secondly, any SNP significantly associated with the outcome was excluded. Thirdly, SNPs were clumped with a threshold of r2 = 0.001 and a window size of 10mb assessed in the European 1,000 genomes reference panel. Fourth, SNPs potentially demonstrating pleiotropic effects were eliminated. Specifically, we utilized the Radial regression method to assess outliers for pleiotropy20. Fifth, we calculated the F statistic \((\varvec{F}={\varvec{b}\varvec{e}\varvec{t}\varvec{a}}^{2}/{\varvec{s}\varvec{e}}^{2})\) to address potential bias from weak IVs; an F statistic less than 10 suggested an invalid IV. Ambiguous and palindromic SNPs were harmonized to ensure accuracy, and Steiger filtering was employed to further refine the selection of SNPs21.
Statistical analyses
We utilized a two-sample MR approach to investigate the causal relationship between ADs and IDA. The foundation of our study was inverse variance weighted (IVW) analysis, which acted as the principal method of analysis. Our analysis employed a random-effects inverse-variance model. We also adopted complementary methods, such as MR-Egger and weighted median. Then, A multistep process was conducted for sensitivity analysis and to assess the second and third assumptions. Initially, Cochran’s Q test was used to evaluate heterogeneity among the IVs. Subsequently, the MR-Egger regression and MR-PRESSO global were employed to assess the potential horizontal pleiotropy of the IVs. A leave-one-out analysis was also performed to determine if a single SNP with a significant horizontal pleiotropic effect could substantially influence the MR estimates. The MR analysis was conducted within the R software environment (version 4.3.1), employing the TwoSampleMR (version 0.5.7), RadialMR (version 1.0) and MRPRESSO (version 1.0) packages.
In this study, we performed eight independent MR analyses for the same outcome variable (IDA), corresponding to eight different autoimmune disease exposures. Given the multiple testing involved, we applied Bonferroni correction to adjust the significance threshold, the corrected significance level was set at 0.00625 (0.05/8). With associations exhibiting p-values < 0.00625 considered statistically significant, while those with p-values between 0.00625 and 0.05 were regarded as suggestive evidence warranting further investigation.
Results
Selection of instrumental variables
For the two-sample MR analysis of the relationships between IDA and 8 Autogenous immune diseases, the number of SNPs used as genetic instruments ranged from 3 (MN) to 115 (IBD).
Causal effects of autoimmune diseases on iron deficiency anemia
To systematically evaluate the causal effects of eight ADs on IDA, we first performed a preliminary analysis including all instrumental variables. IVW analysis revealed significantly increased risks of IDA associated with genetically predicted RA, IBD, UC, CeD, MN, and AS after Bonferroni correction (P < 0.00625), with corresponding ORs ranging from 1.04 to 1.19. Additionally, SLE and CD exhibited suggestive associations (0.00625 ≤ P < 0.05), meriting further investigation (Fig. 2).
Given the strong association of the MHC region with multiple autoimmune diseases, and considering its complex linkage disequilibrium structure—which increases the risk of horizontal pleiotropy and potential violations of MR assumptions—we excluded genetic variants located within the MHC region (chr6: 28,477,797–33,448,354, GRCh37). After excluding MHC variants, IVW analysis demonstrated that RA, IBD, and UC remained significantly associated with an increased risk of IDA (P < 0.00625), with ORs consistently ranging from 1.04 to 1.05, in agreement with pre-exclusion results. In contrast, the associations for CeD and AS were attenuated, shifting from statistically significant to suggestive levels (P = 0.021 and 0.036, respectively), while the association for MN lost statistical significance entirely (P = 0.117). The associations for SLE and CD remained suggestive, with minimal changes in effect estimates (Fig. 3). These findings suggest that the observed associations between CeD, MN, AS, and IDA may have been partly driven by genetic variants within the MHC region, indicating potential pleiotropic bias in the instrumental variables for these diseases.
Forest plot of our MR analysis. Include IVW, MR Egger and Weighted median methods.
Forest plot of MR analysis after excluded the MHC region. Include IVW, MR Egger and Weighted median methods.
Sensitivity analyses
All P values from the Cochran Q tests exceeded 0.05, signifying a lack of observable heterogeneity. Insufficient evidence was found for horizontal pleiotropy, as all P values from subsequent Egger intercept and MR-PRESSO global test exceeded 0.05. Sensitivity analysis results are presented in Table 2 and Supplementary Fig. 2.
The MR analysis demonstrated that for all exposures (e.g., AS, CeD, CD, etc.), neither heterogeneity tests (Q tests for both MR-Egger and IVW with p-values > 0.05) nor horizontal pleiotropy assessments (Egger intercept p-values > 0.05) reached statistical significance. The MR-PRESSO global tests (p > 0.05) further confirmed the absence of influential outlier SNPs, indicating no substantial heterogeneity or pleiotropy among the instrumental variables. The high consistency between IVW and MR-Egger results supports the robustness of the study conclusions. While MN showed a relatively lower Q-test p-value (0.111), suggesting potential minor heterogeneity, this did not significantly impact the overall findings. In summary, the strong concordance across all analytical methods indicates minimal likelihood of pleiotropic bias affecting the study outcomes.
Discussion
In this study, in order to explore the causal association between common autoimmune disorders and IDA, we performed a two-sample MR approach with massive GWAS datasets. Primarily, our investigation demonstrated substantial evidence underpinning a causal connection between eight autoimmune disorders (SLE, RA, IBD, UC, CD, CeD, MN, and AS) and IDA based on IVW’s estimate.
Our MR investigation not only validated and expanded the previous observational studies, but also enhanced the evidence that ADs have an adverse impact on hematopoietic well-being and increases a broad range of risk in IDA. A great number of traditional observational studies demonstrated that anemia was common among the autoimmune diseases mentioned in our research, including SLE, AS, IBD, CeD, and RA8,22,23,24,25. As for SLE, a study indicated that anemia in SLE usually took the form of anemia of chronic disease (ACD) (37.1%) and IDA (35.6%)7. In 2012, a retrospective study conducted by KIM et al. revealed that anemia in patients with AS consists of ACD (33.3%) and IDA (66.7%) of cases9. In 2017, Bulut et al. also observed an increased incidence of IDA (27%) in patients with AS when compared to the normal population26. Digestive tract disease was identified as a well- established risk factor for IDA1. The occurrence rate of iron deficiency in anemia associated with IBD was approximated to range between 36% and 90%27. Similarly, according to the report of Woźniak et al., iron deficiency among newly diagnosed IBD patients represents a prevalence of 77.53%28. A systematic review and individual patient data meta-analysis by Filmann et al. found that the general occurrence of anemia in patients with CD and UC was 27% and 21%, respectively. Concomitantly, it was observed that 57% of the patients with anemia exhibited iron deficiency29. CeD was another gastrointestinal disorder that played a crucial role in IDA30. In a multicenter study involving 1026 patients with CeD, IDA was the predominant extraintestinal manifestation, occurring in approximately 39% of cases25. Ertekin et al. and Karaman et al. reported that positive serology for CeD was observed in approximately 21.3% and 8.4% of the IDA cases, respectively31,32. In addition, in a cross-sectional transverse prospective study including 184 children, refractory IDA patients had the higher frequency of positive serologic test for CeD compared to those responsive to iron supplement33. Similarly, in the adult population, Dube et al. documented an incidence of CeD ranged from 3% to 6% in patients IDA and upregulated to 10%-15% in IDA individuals presenting with digestive symptoms10. IDA was also a prevailing hematological abnormality observed in RA34. Agrawal et al. reported that among RA patients with anemia, ACD accounted for 51.6%, while IDA accounted for 48.4%8. Likewise, Imtiaz et al. found that within the RA patient cohort, anemia was prevalent in 60% of cases, with iron deficiency accounting for anemia in 23% of RA patients35. However, the association between MN and IDA has received limited attention in research. In recent, a cohort study assessed for the first time that the prevalence of anemia in MN was 23.81%, and the cumulative anemia prevalence was 50.72% during the follow-up period36. Although studies examining the occurrence of IDA in MN were limited, numerous studies have demonstrated an association between chronic kidney disease (CKD), possibly attributed to MN, and IDA37. Building upon current research and the causal relationship we discovered between MN and IDA in our study, there are promising directions for further investigation in the future.
Iron deficiency (ID) is distinguished as two primary forms: absolute or functional. Absolute iron deficiency occurs when there is a depletion or exhaustion of the body’s total iron stores. Blood loss due to menstrual periods and gastrointestinal bleeding, malabsorption, and impaired iron absorption play a vital role in absolute ID38. Wincup et al. reported that menorrhagia exhibited a higher incidence in women with SLE and was more prevalent than in the general population39. Additionally, the administration of non-steroidal, anti-inflammatory drugs, and aspirin contribute to menorrhagia and elevate gastrointestinal blood loss40. It is well-established that intestinal disorder are often associated with chronic hemorrhage, reduced iron absorption, or malnutrition41. For example, the proximal duodenum of patients with CeD is dysfunctional. However, this segment serves as the principal locus for the process of iron absorption. This may contribute to a diminished absorption of iron and subsequent development of IDA42. Interestingly, Bulut et al. observed a substantial proportion of AS patients with IDA exhibited mucosal inflammation26. Gökşenoğluet al. reported the upper gastrointestinal system disorder (42.1%), lower gastrointestinal system disorder (18%), and inflammatory bowel disease (4%) was found in AS patients43. Thorough these studies, we hypothesize that the activity of AS may induce mucosal inflammation, potentially diminishing the absorption of iron in the intestines, consequently giving rise to IDA.
However, the total body iron stores are normal or elevated in functional iron deficiency, but the utilization of iron stores is inefficient38. Several studies demonstrated that functional iron deficiency occurred in numerous acute and chronic inflammatory conditions, with hepcidin contributing to the pathogenesis38. Hepcidin, a 25 amino acid peptide primarily synthesized in the liver, serves as a significant iron-regulatory hormone that modulates the absorption of iron from the gastrointestinal tract and the release of iron from the iron stores44. It is known that pro-inflammatory cytokines are increased in ADs45. Inflammatory cytokines like IL-1β and IL-6 elicit the upregulation of hepcidin expression via the JAK/STAT3 pathways44,46. High levels of hepcidin induced by an increase in the production of cytokines can impede the absorption of iron in the duodenum and suppress iron export by Ferroportin (FPN) from enterocytes, hepatocytes and macrophages into the circulation, resulting in functional iron deficiency anemia44.Our study justifies researches to clarify how autoimmune and inflammation diseases trigger this condition.
This investigation examined the effects of various ADs on IDA. IDA is a prevalent condition worldwide and cause a significant public health burden1. Based on our findings, advocating public awareness of the detrimental effects of ADs on hematopoietic health and implementing public health interventions targeting both the ADs and the associated IDA is important, thereby alleviating the burden of IDA. Additionally, our results may help facilitate the development of guidelines for IDA prevention and the management of patients at high risk of developing IDA as a consequence of autoimmune diseases.
Our research possesses several noteworthy merits. First, in comparison to randomized controlled trials, the MR design offers reduced susceptibility to bias originating from reverse causation and unmeasured confounding, which are pervasive issues in traditional observational studies. Second, our study performed MR analyses to systematically analyze the impact of ADs on IDA, which had never been reported in previous studies. By conducting comprehensive analysis of eight common AD diseases, an opportunity was given to evaluate a wide range of ADs that are causally related to IDA. Third, not only the large sample size in this study guaranteed enough statistical power, but also strict selection of IVs ensured they met the randomization assumption, reducing the risk of endogeneity and strengthening causal inference. Fourth, to mitigate population stratification bias, this study utilized GWAS summary statistics derived from individuals of European ancestry. Furthermore, to reduce bias due to genetic pleiotropy, we excluded variants located in the MHC region. The MHC region, particularly the HLA gene cluster, plays a central role in the pathogenesis of various autoimmune diseases and may indirectly influence iron metabolism and anemia risk by modulating inflammatory factors such as IL-6 and hepcidin, thereby potentially violating the independence assumption of instrumental variables in MR analysis. After excluding the MHC region, the associations between RA, IBD, UC, and IDA remained robust, suggesting that the potential causal effects of these diseases on IDA are relatively independent of MHC-related pathways. In contrast, the associations of CeD and AS with IDA were attenuated to suggestive levels, while the association with MN was no longer significant. This pattern indicates that the initial associations observed for CeD, MN, and AS with IDA may have been partly driven by pleiotropic variants within the MHC region. Therefore, caution is warranted in causal inference for these diseases, and further validation using more refined genetic instruments or multivariable MR approaches is needed in future studies. While the effect sizes for some results were small (OR ~ 1.04–1.05), we note that although the risk increase for an individual is minimal, autoimmune diseases and IDA are both common in the general population. From a population perspective, even such a slight risk increase could lead to a substantial number of additional IDA cases, underscoring its significant public health importance. Furthermore, we also noted the non-significant results among the findings. We pointed out that after excluding the MHC region, the IVW analysis showed reduced significance for CeD and AS (P = 0.021 and 0.036, respectively), while MN lost its statistical significance (P = 0.117). We therefore indicated that the associations involving CeD, MN, and AS may be partially driven by variants within the MHC region. This provides a more balanced interpretation of all our results, rather than focusing solely on the significant positive associations.
Of course, our study also had several limitations. First, all the participants included in this study were of European descent, but SLE exhibits a higher prevalence among African Americans, Hispanics, and Asians compared to whites47. This could constrain the external validity of our results to other populations. Additional investigations are needed to corroborate our results in populations with non-European ancestry. Second, certain ADs, such as SLE and RA, are more prevalent in women than in men48, while AS is predominantly observed in males49. However, our study did not conduct a separate analysis based on gender, potentially impacting our findings. Performing a gender-specific MR analysis in future research would offer valuable insights. Third, the ADs data we used were mostly from adult individuals, whereas the most patients develop CeD before age 10 years50.It would be helpful to perform an age stratification MR analysis to reduce the impact of confounding factors, and enhances the internal and external validity of the study. Additional, while certain exposures (e.g., RA, CeD) showed statistically significant associations with IDA, their relatively small effect sizes (ORs) and wide confidence intervals suggest limited clinical relevance. These findings highlight that statistical significance does not necessarily translate to substantial biological or clinical impact, warranting cautious interpretation of these marginal associations.
Conclusions
In summary, this study suggests that RA, IBD, and UC may have a causal impact on IDA, and these associations remained robust after excluding the MHC region. In contrast, the associations of SLE, CD, CeD, MN, and AS with IDA may be influenced by pleiotropic bias and thus require further validation in future research. Healthcare providers are advised to heighten vigilance regarding the potential development of IDA in individuals with autoimmune disorders. The underlying biological mechanisms of these associations and further gender-specific and ethnic-stratified analyses need to be explored in future investigations.
Data availability
All GWAS summary data of ADs (SLE, RA, IBD, UC, CD, CeD, MN, and AS) were obtained from the GWAS Catalog database (https://www.ebi.ac.uk/gwas/). And genomic data of IDA can be accessible through FinnGen study (https://www.finngen.fi/en/access_results).
References
Camaschella, C. Iron-deficiency anemia. N Engl. J. Med. 372, 1832–1843. https://doi.org/10.1056/NEJMra1401038 (2015).
Benson, C. S. et al. The effect of iron deficiency and anaemia on women’s health. Anaesthesia 76 (Suppl 4), 84–95. https://doi.org/10.1111/anae.15405 (2021).
Snook, J. et al. British Society of Gastroenterology guidelines for the management of iron deficiency anaemia in adults. Gut 70, 2030–2051. https://doi.org/10.1136/gutjnl-2021-325210 (2021).
Ansari, T. et al. Nutritional iron deficiency in women of child bearing age–what to do? J. Ayub Med. Coll. Abbottabad. 21, 17–20 (2009).
Gheibi, S., Farrokh-Eslamlou, H. R., Noroozi, M. & Pakniyat, A. Refractory iron deficiency anemia and Helicobacter Pylori Infection in pediatrics: A review. Iran. J. Ped Hematol. Oncol. 5, 50–64 (2015).
Camaschella, C. Iron deficiency. Blood 133, 30–39. https://doi.org/10.1182/blood-2018-05-815944 (2019).
Voulgarelis, M. et al. Anaemia in systemic lupus erythematosus: aetiological profile and the role of erythropoietin. Ann. Rheum. Dis. 59, 217–222. https://doi.org/10.1136/ard.59.3.217 (2000).
Agrawal, S., Misra, R. & Aggarwal, A. Anemia in rheumatoid arthritis: high prevalence of iron-deficiency anemia in Indian patients. Rheumatol. Int. 26, 1091–1095. https://doi.org/10.1007/s00296-006-0133-4 (2006).
Ki-Jo, K. & Chul-Soo, C. Anemia of chronic disease in ankylosing spondylitis: improvement following anti-TNF therapy. Archives Rheumatol. 27, 090–097 (2012).
Dubé, C. et al. The prevalence of celiac disease in average-risk and at-risk Western European populations: a systematic review. Gastroenterology 128, S57–67. https://doi.org/10.1053/j.gastro.2005.02.014 (2005).
Wang, T. et al. Causal relationship between obesity and iron deficiency anemia: a two-sample Mendelian randomization study. Front. Public. Health. 11, 1188246. https://doi.org/10.3389/fpubh.2023.1188246 (2023).
Greenland, S. An introduction to instrumental variables for epidemiologists. Int. J. Epidemiol. 29, 722–729. https://doi.org/10.1093/ije/29.4.722 (2000).
Cortes, A. et al. Identification of multiple risk variants for ankylosing spondylitis through high-density genotyping of immune-related loci. Nat. Genet. 45, 730–738. https://doi.org/10.1038/ng.2667 (2013).
Trynka, G. et al. Dense genotyping identifies and localizes multiple common and rare variant association signals in celiac disease. Nat. Genet. 43, 1193–1201. https://doi.org/10.1038/ng.998 (2011).
Liu, J. Z. et al. Association analyses identify 38 susceptibility loci for inflammatory bowel disease and highlight shared genetic risk across populations. Nat. Genet. 47, 979–986. https://doi.org/10.1038/ng.3359 (2015).
Xie, J. et al. The genetic architecture of membranous nephropathy and its potential to improve non-invasive diagnosis. Nat. Commun. 11, 1600. https://doi.org/10.1038/s41467-020-15383-w (2020).
Okada, Y. et al. Genetics of rheumatoid arthritis contributes to biology and drug discovery. Nature 506, 376–381. https://doi.org/10.1038/nature12873 (2014).
Bentham, J. et al. Genetic association analyses implicate aberrant regulation of innate and adaptive immunity genes in the pathogenesis of systemic lupus erythematosus. Nat. Genet. 47, 1457–1464. https://doi.org/10.1038/ng.3434 (2015).
Kurki, M. I. et al. FinnGen provides genetic insights from a well-phenotyped isolated population. Nature 613, 508–518. https://doi.org/10.1038/s41586-022-05473-8 (2023).
Bowden, J. et al. Improving the visualization, interpretation and analysis of two-sample summary data Mendelian randomization via the Radial plot and Radial regression. Int. J. Epidemiol. 47, 2100. https://doi.org/10.1093/ije/dyy265 (2018).
Hemani, G., Tilling, K. & Davey Smith, G. Orienting the causal relationship between imprecisely measured traits using GWAS summary data. PLoS Genet. 13, e1007081. https://doi.org/10.1371/journal.pgen.1007081 (2017).
Velo-García, A., Castro, S. G. & Isenberg, D. A. The diagnosis and management of the haematologic manifestations of lupus. J. Autoimmun. 74, 139–160. https://doi.org/10.1016/j.jaut.2016.07.001 (2016).
Zviahina, O. V., Shevchuk, S. V., Kuvikova, I. P. & Segeda, I. S. Anemia in patients with ankylosing spondylitis, association with the activity of the inflammatory process and the severity of the disease. Wiad Lek. 73, 715–721 (2020).
Akhuemonkhan, E., Parian, A., Miller, K., Hanauer, S. & Hutfless, S. Prevalence and screening for anaemia in mild to moderate Crohn’s disease and ulcerative colitis in the United States, 2010–2014. BMJ Open. Gastroenterol. 4, e000155. https://doi.org/10.1136/bmjgast-2017-000155 (2017).
Bottaro, G., Cataldo, F., Rotolo, N., Spina, M. & Corazza, G. R. The clinical pattern of subclinical/silent celiac disease: an analysis on 1026 consecutive cases. Am. J. Gastroenterol. 94, 691–696. https://doi.org/10.1111/j.1572-0241.1999.00938.x (1999).
Bulut, Y., Tas, D. A., Ozturk, O. G. & Karaogullarindan, U. SAT0396 Investigation of iron deficiency anemia in ankylosing spondylitis patients. Ann. Rheum. Dis. 76, 921 (2017).
Aksan, A. et al. Iron Formulations for the Treatment of Iron Deficiency Anemia in Patients with Inflammatory Bowel Disease: A Cost-Effectiveness Analysis in Switzerland. Adv. Ther. 38, 660–677. https://doi.org/10.1007/s12325-020-01553-1 (2021).
Woźniak, M., Barańska, M., Małecka-Panas, E. & Talar-Wojnarowska, R. The prevalence, characteristics, and determinants of anaemia in newly diagnosed patients with inflammatory bowel disease. Prz Gastroenterol. 14, 39–47. https://doi.org/10.5114/pg.2019.83424 (2019).
Filmann, N. et al. Prevalence of anemia in inflammatory bowel diseases in european countries: a systematic review and individual patient data meta-analysis. Inflamm. Bowel Dis. 20, 936–945. https://doi.org/10.1097/01.MIB.0000442728.74340.fd (2014).
Bergamaschi, G. et al. Pathophysiology and therapeutic management of anemia in gastrointestinal disorders. Expert Rev. Gastroenterol. Hepatol. 16, 625–637. https://doi.org/10.1080/17474124.2022.2089114 (2022).
Ertekin, V., Tozun, M. S. & Küçük, N. The prevalence of celiac disease in children with iron-deficiency anemia. Turk. J. Gastroenterol. 24, 334–338. https://doi.org/10.4318/tjg.2013.0529 (2013).
Karaman, K., Akbayram, S., Kar, S. & Demirören, K. Prevalence of Celiac Disease in Children With Iron Deficiency Anemia in Van Lake Region of Turkey. J. Pediatr. Hematol. Oncol. 38, 143–146. https://doi.org/10.1097/mph.0000000000000495 (2016).
Shahriari, M., Honar, N., Yousefi, A. & Javaherizadeh, H. Association Of Potential Celiac Disease And Refractory Iron Deficiency Anemia In Children And Adolescents. Arq. Gastroenterol. 55, 78–81. https://doi.org/10.1590/s0004-2803.201800000-15 (2018).
Masson, C. Rheumatoid anemia. Joint Bone Spine. 78, 131–137. https://doi.org/10.1016/j.jbspin.2010.05.017 (2011).
Imtiaz, T., Saleem, M. W., Zahid, M. & Abbasi, S. H. Rheumatoid Arthritis Related Presentation of Different Types of Anemia and Their Evaluation. Asian J. Multidisciplinary Stud. 7, 6 (2019).
Li, Z. et al. Clinical features of anemia in membranous nephropathy patients: a Chinese cohort study. Ren. Fail. 45, 2152692. https://doi.org/10.1080/0886022x.2022.2152692 (2023).
Batchelor, E. K., Kapitsinou, P., Pergola, P. E., Kovesdy, C. P. & Jalal, D. I. Iron Deficiency in Chronic Kidney Disease: Updates on Pathophysiology, Diagnosis, and Treatment. J. Am. Soc. Nephrol. 31, 456–468. https://doi.org/10.1681/asn.2019020213 (2020).
Lopez, A., Cacoub, P., Macdougall, I. C. & Peyrin-Biroulet, L. Iron deficiency anaemia. Lancet 387, 907–916. https://doi.org/10.1016/s0140-6736(15)60865-0 (2016).
Wincup, C., McDonnell, T. C. R. & Rahman, A. Menorrhagia: an underappreciated problem in pre-menopausal women with systemic lupus erythematosus. Lupus 28, 916–917. https://doi.org/10.1177/0961203319851868 (2019).
Tai, F. W. D. & McAlindon, M. E. Non-steroidal anti-inflammatory drugs and the gastrointestinal tract. Clin. Med. (Lond). 21, 131–134. https://doi.org/10.7861/clinmed.2021-0039 (2021).
Tulewicz-Marti, E., Moniuszko, A. & Rydzewska, G. Management of anemia in inflammatory bowel disease: a challenge in everyday clinical practice. Prz Gastroenterol. 12, 239–243. https://doi.org/10.5114/pg.2017.72096 (2017).
Talarico, V., Giancotti, L., Mazza, G. A., Miniero, R. & Bertini, M. Iron deficiency anemia in celiac disease. Nutrients 13 https://doi.org/10.3390/nu13051695 (2021).
Gökşenoğlu, G., Buğdaycı, D., Paker, N., Yıldırım, M. A. & Etli, Ö. The prevalence of comorbidity and predictors in ankylosing spondylitis. Turk. J. Phys. Med. Rehabil. 65, 132–138. https://doi.org/10.5606/tftrd.2019.2822 (2019).
Ganz, T. Systemic iron homeostasis. Physiol. Rev. 93, 1721–1741. https://doi.org/10.1152/physrev.00008.2013 (2013).
Wahren-Herlenius, M. & Dörner, T. Immunopathogenic mechanisms of systemic autoimmune disease. Lancet 382, 819–831. https://doi.org/10.1016/s0140-6736(13)60954-x (2013).
Hentze, M. W., Muckenthaler, M. U., Galy, B. & Camaschella, C. Two to tango: regulation of Mammalian iron metabolism. Cell 142, 24–38. https://doi.org/10.1016/j.cell.2010.06.028 (2010).
Kiriakidou, M. & Ching, C. L. Systemic Lupus Erythematosus. Ann. Intern. Med. 172 (96), Itc81–itc. https://doi.org/10.7326/aitc202006020 (2020).
Durcan, L., O’Dwyer, T. & Petri, M. Management strategies and future directions for systemic lupus erythematosus in adults. Lancet 393, 2332–2343. https://doi.org/10.1016/s0140-6736(19)30237-5 (2019).
Zhu, W. et al. Ankylosing spondylitis: etiology, pathogenesis, and treatments. Bone Res. 7, 22. https://doi.org/10.1038/s41413-019-0057-8 (2019).
Lebwohl, B., Rubio-Tapia, A. & Epidemiology Presentation, and Diagnosis of Celiac Disease. Gastroenterology 160, 63–75. https://doi.org/10.1053/j.gastro.2020.06.098 (2021).
Acknowledgements
None.
Funding
Key Clinical Specialty Discipline Construction Program of Fuzhou, 20220301 Wen Chen. National Natural Science Foundation of China, 82160114 Daoming Liang. Yunnan Fundamental Research Kunming Medical University Joint Projects, 202101AY070001-165 Li Zhai, 202301AY070001-025 Daoming Liang, 202501AY070001-086 Qiyu Lu. Yunnan Fundamental Research Projects, 202501AT070276 Yixiong Shu. Yunnan Revitalization Talent Support Program, XDYC-MY-2022-0100 Daoming Liang. Graduate Student Innovation Fund of Kunming Medical University, 2025S259 Yinuo Wang.
Author information
Authors and Affiliations
Contributions
Yixiong Shu, Daoming Liang, and Li Zhai designed the study. Wen Chen, Yinuo Wang, and Haojun Long an-alyzed the datasets and interpreted the results. Yongyu Yang, Yingliang Liu, Qiyu Lu and Jie Xiao downloaded the data and provided software support. Wen Chen, Yinuo Wang, and Haojun Long wrote and edited the man-uscript. Daoming Liang, Li Zhai, Qiyu Lu, Yixiong Shu, Wen Chen and Yinuo Wang provided financial support. All the authors have read and agreed to the pub-lished version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
NA. The data originated from a publicly accessible database, we did not collect any new data, nor was further ethical approval required.
Consent for publication
NA. No identifying human images were used in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.


Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, W., Wang, Y., Long, H. et al. Causal relationship between autoimmune diseases and iron deficiency anemia: a two-sample mendelian randomization study. Sci Rep 16, 12935 (2026). https://doi.org/10.1038/s41598-026-42356-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-42356-8