Abstract
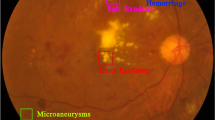
Diabetic Retinopathy (DR) is a major cause of vision loss and blindness in diabetic individuals. DR is conventionally diagnosed by assessing retinal lesion findings from fundus photographs taken during exams and applying a scale like International Classification of Diabetic Retinopathy (ICDR). The expected rise in future DR cases highlights the need for deep learning models capable of identifying relevant lesions and delivering explainable results. To this end we present BigEye, a novel framework that uses extracted lesion features to predict ICDR stage. A dataset of fundus images from a local hospital and a public dataset, annotated with segmentation masks and DR stages, is assembled to train a DeepLabV3 + model on six retinal lesions. Lesion quantities and pixel area features are integrated by a classifier model evaluated through 10-fold nested cross validation (0.77 ± 0.07 precision, 0.71 ± 0.06 recall, 0.72 ± 0.07 F1 score, 0.95 ± 0.02 ROC-AUC, 0.83 ± 0.03 accuracy). A Shapely Additive Explanations (SHAP) value analysis notably shows close alignment between discriminative lesions for each DR stage and corresponding ICDR stage criteria. These results demonstrate that BigEye is well suited for providing explainable ICDR stage predictions grounded in clinical knowledge.
Similar content being viewed by others
Data availability
e-ophtha original data: https://www.adcis.net/en/third-party/e-ophtha/.IU Health data: Some fundus images used in this study contain protected health information from the Indiana University Health system and cannot be made publicly available in accordance with patient privacy regulations. Data access requests may be directed to the Indiana University Data Management Council (iudata@iu.edu), subject to ethical approval and execution of appropriate data use agreements.BigEye source code and model weights: https://github.com/Janga-Lab/BigEye/.
References
Chong, D. D., Das, N. & Singh, R. P. Diabetic retinopathy: Screening, prevention, and treatment. Cleve. Clin. J. Med. 91, 503–510 (2024).
Kaur, A., Kumar, R. & Sharma, A. Diabetic retinopathy leading to blindness- A review. Curr. Diabetes Rev. 20, e240124225997 (2024).
Henricsson, M. et al. The incidence of retinopathy 10 years after diagnosis in young adult people with diabetes: results from the nationwide population-based Diabetes Incidence Study in Sweden (DISS). Diabetes Care. 26, 349–354 (2003).
Berrabeh, S. et al. Prevalence and risk factors of retinopathy in Type 1 Diabetes: A cross-sectional study. Cureus 15, e47993 (2023).
Romero-Aroca, P. et al. Ten-year incidence of diabetic retinopathy and macular edema. Risk factors in a sample of people with Type 1 Diabetes. Diabetes Res. Clin. Pract. 94, 126–132 (2011).
Romero-Aroca, P. et al. Differences in incidence of diabetic retinopathy between Type 1 and 2 Diabetes mellitus: A nine-year follow-up study. Br. J. Ophthalmol. 101, 1346–1351 (2017).
Voigt, M. et al. Prevalence and progression rate of diabetic retinopathy in Type 2 Diabetes patients in correlation with the duration of diabetes. Exp. Clin. Endocrinol. Diabetes 126, 570–576 (2018).
Yin, L., Zhang, D., Ren, Q., Su, X. & Sun, Z. Prevalence and risk factors of diabetic retinopathy in diabetic patients: A community based cross-sectional study. Medicine (Baltimore) 99, e19236 (2020).
Aljehani, E. A., Alhawiti, A. E. & Mohamad, R. M. Prevalence and determinants of diabetic retinopathy among type 2 diabetic patients in Saudi Arabia: A systematic review. Cureus 15, e42771 (2023).
Song, P., Yu, J., Chan, K. Y., Theodoratou, E. & Rudan, I. Prevalence, risk factors and burden of diabetic retinopathy in China: A systematic review and meta-analysis. J. Glob. Health. 8, 010803 (2018).
Lundeen, E. A. et al. Prevalence of diabetic retinopathy in the US in 2021. JAMA Ophthalmol. 141, 747–754 (2023).
Perais, J. et al. Prognostic factors for the development and progression of proliferative diabetic retinopathy in people with diabetic retinopathy. Cochrane Database Syst. Rev. 2, CD013775 (2023).
Grossniklaus, H. E., Geisert, E. E. & Nickerson, J. M. Introduction to the retina. Prog. Mol. Biol. Transl. Sci. 134, 383–396 (2015).
Singh, C. Metabolism and vascular retinopathies: Current perspectives and future directions. Diagnostics https://doi.org/10.3390/diagnostics12040903 (2022).
Country, M. W. Retinal metabolism: A comparative look at energetics in the retina. Brain Res. 1672, 50–57 (2017).
Hudson, N. & Campbell, M. Tight junctions of the neurovascular unit. Front. Mol. Neurosci. 14, 752781 (2021).
Rudraraju, M., Narayanan, S. P. & Somanath, P. R. Regulation of blood-retinal barrier cell-junctions in diabetic retinopathy. Pharmacol. Res. 161, 105115 (2020).
Eshaq, R. S., Aldalati, A. M. Z., Alexander, J. S. & Harris, N. R. Diabetic retinopathy: Breaking the barrier. Pathophysiology 24, 229–241 (2017).
Park, D. Y. et al. Plastic roles of pericytes in the blood-retinal barrier. Nat. Commun. 8, 15296 (2017).
Willis, J. R. et al. Vision-related functional burden of diabetic retinopathy across severity levels in the United States. JAMA Ophthalmol. 135, 926–932 (2017).
Curtis, T. M., Gardiner, T. A. & Stitt, A. W. Microvascular lesions of diabetic retinopathy: Clues towards understanding pathogenesis?. Eye 23, 1496–1508 (2009).
Horie, S. & Ohno-Matsui, K. Progress of imaging in diabetic retinopathy-from the past to the present. Diagnostics https://doi.org/10.3390/diagnostics12071684 (2022).
Takamura, Y., Yamada, Y. & Inatani, M. Role of microaneurysms in the pathogenesis and therapy of diabetic macular edema: A descriptive review. Medicina (Kaunas) https://doi.org/10.3390/medicina59030435 (2023).
An, D., Tan, B., Yu, D.-Y. & Balaratnasingam, C. Differentiating microaneurysm pathophysiology in diabetic retinopathy through objective analysis of capillary nonperfusion, inflammation, and pericytes. Diabetes 71, 733–746 (2022).
Santos, A. R. et al. Microaneurysm turnover in mild non-proliferative diabetic retinopathy is associated with progression and development of vision-threatening complications: A 5-year longitudinal study. J. Clin. Med. https://doi.org/10.3390/jcm10102142 (2021).
Shukla, U. V., Gurnani, B. & Kaufman, E. J. Intraocular Hemorrhage. in StatPearls (StatPearls Publishing, 2025).
Kanukollu, V. M. & Ahmad, S. S. Retinal Hemorrhage. in StatPearls (StatPearls Publishing, (2025).
Chew, E. Y. Association of elevated serum lipid levels with retinal hard exudate in diabetic retinopathy. Early Treatment Diabetic Retinopathy Study (ETDRS) Report 22. Arch. Ophthalmol. 114, 1079–1084 (1996).
Raman, R., Nittala, M. G., Gella, L., Pal, S. S. & Sharma, T. Retinal sensitivity over hard exudates in diabetic retinopathy. J. Ophthalmic Vis. Res. 10, 160–164 (2015).
Chui, T. Y. P., Thibos, L. N., Bradley, A. & Burns, S. A. The mechanisms of vision loss associated with a cotton wool spot. Vision Res. 49, 2826–2834 (2009).
Kim, J. S. The microperimetry of resolved cotton-wool spots in eyes of patients with hypertension and diabetes mellitus. Arch. Ophthalmol. 129, 879–884 (2011).
Chaudhary, S., Zaveri, J. & Becker, N. Proliferative diabetic retinopathy (PDR). Dis. Mon. 67, 101140 (2021).
Lange, J., Hadziahmetovic, M., Zhang, J. & Li, W. Region-specific ischemia, neovascularization and macular oedema in treatment-naïve proliferative diabetic retinopathy. Clin. Exp. Ophthalmol. 46, 757–766 (2018).
Agrawal, M. et al. Pattern and distribution of neovascularization in proliferative diabetic retinopathy on fundus fluorescein angiography: A growing paradigm. Med. J. Armed Forces India 79, 207–212 (2023).
Iyer, S. S. R., Regan, K. A., Burnham, J. M. & Chen, C. J. Surgical management of diabetic tractional retinal detachments. Surv. Ophthalmol. 64, 780–809 (2019).
Stewart, M. W., Browning, D. J. & Landers, M. B. Current management of diabetic tractional retinal detachments. Indian J. Ophthalmol. 66, 1751–1762 (2018).
Muqit, M. M. K. et al. Pascal panretinal laser ablation and regression analysis in proliferative diabetic retinopathy: Manchester Pascal Study Report 4. Eye 25, 1447–1456 (2011).
Pande, G. S. & Tidake, P. Laser treatment modalities for diabetic retinopathy. Cureus 14, e30024 (2022).
Stefánsson, E. The therapeutic effects of retinal laser treatment and vitrectomy. A theory based on oxygen and vascular physiology. Acta Ophthalmol. Scand. 79, 435–440 (2001).
Wang, X. et al. Panretinal photocoagulation plus intravitreal conbercept for diabetic retinopathy in real world: a retrospective study. BMC Ophthalmol. 23, 400 (2023).
Zhan, H. Q., Zhou, J. L., Zhang, J., Wu, D. & Gu, C. Y. Conbercept combined with laser photocoagulation in the treatment of diabetic macular edema and its influence on intraocular cytokines. World J. Diabetes. 14, 1271–1279 (2023).
Lin, Z., Deng, A., Hou, N., Gao, L. & Zhi, X. Advances in targeted retinal photocoagulation in the treatment of diabetic retinopathy. Front. Endocrinol. (Lausanne). 14, 1108394 (2023).
Gawęcki, M., Kiciński, K., Bianco, L. & Battaglia Parodi, M. Regression of neovascularization after panretinal photocoagulation combined with Anti-VEGF injection for proliferative diabetic retinopathy-a review. Diagnostics https://doi.org/10.3390/diagnostics14010031 (2023).
Cleland, C. Comparing the International Clinical Diabetic Retinopathy (ICDR) severity scale. Community Eye Health. 36, 10 (2023).
Romero-Aroca, P. et al. A clinical decision support system for diabetic retinopathy screening: creating a clinical support application. Telemed J. E Health. 25, 31–40 (2019).
Nakayama, L. F. et al. Artificial intelligence for telemedicine diabetic retinopathy screening: a review. Ann. Med. 55, 2258149 (2023).
Mohanty, C. et al. Using deep learning architectures for detection and classification of diabetic retinopathy. Sensors 23, (2023).
Jabbar, M. K., Yan, J., Xu, H., Ur Rehman, Z. & Jabbar, A. Transfer learning-based model for diabetic retinopathy diagnosis using retinal images. Brain Sci. https://doi.org/10.3390/brainsci12050535 (2022).
Narayanan, B. N., Hardie, R. C., De Silva, M. S. & Kueterman, N. K. Hybrid machine learning architecture for automated detection and grading of retinal images for diabetic retinopathy. J. Med. Imaging (Bellingham). 7, 034501 (2020).
Chilukoti, S. V., Shan, L., Tida, V. S., Maida, A. S. & Hei, X. A reliable diabetic retinopathy grading via transfer learning and ensemble learning with quadratic weighted kappa metric. BMC Med. Inf. Decis. Mak. 24, 37 (2024).
Larsen, T. J. et al. The use of artificial intelligence to assess diabetic eye disease among the Greenlandic population. Int. J. Circumpolar Health 83, 2314802 (2024).
Gu, Z. et al. Classification of diabetic retinopathy severity in fundus images using the vision transformer and residual attention. Comput. Intell. Neurosci. 1305583 (2023). (2023).
Yao, Z. et al. FunSwin: A deep learning method to analysis diabetic retinopathy grade and macular edema risk based on fundus images. Front. Physiol. 13, 961386 (2022).
Manan, M. A. et al. Semantic segmentation of retinal exudates using a residual encoder-decoder architecture in diabetic retinopathy. Microsc. Res. Tech. 86, 1443–1460 (2023).
Jiang, Y. et al. Segmentation of Laser Marks of Diabetic Retinopathy in the Fundus Photographs Using Lightweight U-Net. J. Diabetes Res. 8766517 (2021). (2021).
Gao, W., Fan, B., Fang, Y. & Song, N. Lightweight and multi-lesion segmentation model for diabetic retinopathy based on the fusion of mixed attention and ghost feature mapping. Comput. Biol. Med. 169, 107854 (2024).
Xu, Y. et al. FFU-Net: Feature fusion U-Net for lesion segmentation of diabetic retinopathy. Biomed. Res. Int. 2021, 6644071 (2021).
Alam, M., Zhao, E. J., Lam, C. K. & Rubin, D. L. Segmentation-assisted fully convolutional neural network enhances deep learning performance to identify proliferative diabetic retinopathy. J. Clin. Med. https://doi.org/10.3390/jcm12010385 (2023).
Andersen, J. K. H., Hubel, M. S., Rasmussen, M. L., Grauslund, J. & Savarimuthu, T. R. Automatic detection of abnormalities and grading of diabetic retinopathy in 6-Field retinal images: Integration of segmentation into classification. Transl. Vis. Sci. Technol. 11, 19 (2022).
Hassan, D. et al. Combining transfer learning with retinal lesion features for accurate detection of diabetic retinopathy. Front. Med. 9, 1050436 (2022).
Dai, L. et al. A deep learning system for detecting diabetic retinopathy across the disease spectrum. Nat. Commun. 12, 3242 (2021).
Decencière, E. et al. TeleOphta: Machine learning and image processing methods for teleophthalmology. IRBM 34, 196–203 (2013).
Dutta, A. & Zisserman, A. ACM Press,. The VIA annotation software for images, audio and video. in Proceedings of the 27th ACM International Conference on Multimedia - MM ’19 2276–2279 (2019). https://doi.org/10.1145/3343031.3350535
Riotto, E. et al. Accuracy of Autonomous Artificial Intelligence-Based Diabetic Retinopathy Screening in Real-Life Clinical Practice. J Clin. Med 13, (2024).
Rajalakshmi, R., Subashini, R., Anjana, R. M. & Mohan, V. Automated diabetic retinopathy detection in smartphone-based fundus photography using artificial intelligence. Eye 32, 1138–1144 (2018).
Karabeg, M. et al. A pilot cost-analysis study comparing AI-based EyeArt® and ophthalmologist assessment of diabetic retinopathy in minority women in Oslo, Norway. Int. J. Retina Vitreous 10, 40 (2024).
Raumviboonsuk, P. et al. Deep learning versus human graders for classifying diabetic retinopathy severity in a nationwide screening program. npj Digit. Med. 2, 25 (2019).
Lee, A. Y. et al. Head-to-Head, Real-World Validation Study of Seven Automated Artificial Intelligence Diabetic Retinopathy Screening Systems. Diabetes Care. 44, 1168–1175 (2021). Multicenter.
Kalavar, M. et al. Applications of artificial intelligence for the detection, management, and treatment of diabetic retinopathy. Int. Ophthalmol. Clin. 60, 127–145 (2020).
Kong, M. & Song, S. J. Artificial intelligence applications in diabetic retinopathy: What we have now and what to expect in the future. Endocrinol. Metab. 39, 416–424 (2024).
Bora, A. et al. Predicting the risk of developing diabetic retinopathy using deep learning. Lancet Digit. Health 3, e10–e19 (2021).
Vujosevic, S., Limoli, C., Luzi, L. & Nucci, P. Digital innovations for retinal care in diabetic retinopathy. Acta Diabetol. 59, 1521–1530 (2022).
Bienefeld, N. et al. Solving the explainable AI conundrum by bridging clinicians’ needs and developers’ goals. npj Digit. Med. 6, 94 (2023).
Amann, J. et al. Explainability for artificial intelligence in healthcare: A multidisciplinary perspective. BMC Med. Inform. Decis. Mak. 20, 310 (2020).
Singh, A., Sengupta, S. & Lakshminarayanan, V. Explainable deep learning models in medical image analysis. J. Imaging https://doi.org/10.3390/jimaging6060052 (2020).
Esmaeilkhanian, H. et al. The relationship of diabetic retinopathy severity scales with frequency and surface area of diabetic retinopathy lesions. Graefes Arch. Clin. Exp. Ophthalmol. 261, 3165–3176 (2023).
Rahimi, M. et al. Inner retinal oxygen delivery and metabolism in progressive stages of diabetic retinopathy. Sci. Rep. 14, 4414 (2024).
Dai, L. et al. A deep learning system for predicting time to progression of diabetic retinopathy. Nat. Med. 30, 584–594 (2024).
Chen, L.-C., Zhu, Y., Papandreou, G., Schroff, F. & Adam, H. Encoder-decoder with atrous separable convolution for semantic image segmentation. arXiv https://doi.org/10.48550/arxiv.1802.02611 (2018).
Zuiderveld, K. Contrast Limited Adaptive Histogram Equalization. https://www.cse.unr.edu/~bebis/CS474/StudentPaperPresentations/1994%20-%20CLAHE.pdf
Lundberg, S. A unified approach to interpreting model predictions. arXiv https://doi.org/10.48550/arxiv.1705.07874 (2017).
Acknowledgements
The authors thank Indiana University Information Technology Services Research Technologies (UITS RT) for maintaining the Big Red 200 cluster used in this study. We also thank Indiana University Health and the French Research Agency’s ANR-TECSAN-TELEOPHTA and OPHDIAT© projects for the public data used in this study. Figures were created in part with BioRender (https://www.biorender.com).
Funding
This study was supported by NIH/NEI grant 5T35EY031282-05, the Indiana University Indianapolis Institute of Integrative Artificial Intelligence (iAI), and the Lilly Endowment, Inc., through its support for the Indiana University Pervasive Technology Institute. The funding providers had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
H.M.G., D.H.S., A.R.H., and S.C.J contributed to study conceptualization and design. M.H. and A.R.H. performed data curation. M.H. performed initial initial fundus image annotation, and A.R.H. reviewed and approved all annotations. H.M.G, D.H.S, and O.B.O. contributed to result organization and visualization. H.M.G and D.H.S. wrote the BigEye codebase. M.H., F.D.B., J.X.L., and A.R.H provided clinical insight. H.M.G., D.H.S., F.D.B., and S.C.J performed manuscript preparation. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gill, H.M., Salem, D.H., Omoru, O.B. et al. BigEye: a clinically interpretable deep learning framework for diabetic retinopathy detection and stage prediction. Sci Rep (2026). https://doi.org/10.1038/s41598-026-43573-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-026-43573-x