Abstract
Previous studies of the relationship between lipid levels and depression have yielded inconsistent findings. This inconsistency may relate to inter-lipid interactions. The current study employs a novel analytical approach to evaluate interactions among lipid parameters in relation to the odds of depression. This study utilized datasets obtained from the U.S. National Health and Nutrition Examination Survey spanning 2005 to 2018. Depressive symptoms were assessed using the Patient Health Questionnaire-9, with a score ≥ 10 used to define depression. Survey-weighted generalized linear models first tested interaction effects between continuous triglyceride (TG) and cholesterol subtypes (low-density lipoprotein cholesterol [LDL-C], high-density lipoprotein cholesterol, total cholesterol). For statistically significant interactions, participants were dichotomized at each of 39 quantile points (2.5–97.5% at 2.5% intervals) of lipid 2. Sequential analyses determined whether these binary subgroups significantly modified the association between lipid 1 and depression. The optimal lipid 2 cutoff value was selected based on the multiple-comparison-adjusted P value for interaction across all quantiles. Sensitivity analyses evaluated cutoff value stability. A total of 8962 participants were included. The interaction analysis revealed a synergistic effect between TG and LDL-C on depression (interaction term β = 0.158, P = 0.03). Sequential interaction threshold analysis identified LDL-C = 3.6 mmol/L as the most significant effect modifier. In the subgroup with LDL-C levels ≥ 3.6 mmol/L, TG were significantly positively associated with depression (odds ratio 1.78; 95% confidence interval 1.30–2.42; P < 0.001), with a pronounced interaction effect (Pinteraction = 0.002). Sensitivity analyses incorporating eight distinct analytical conditions consistently confirmed LDL-C = 3.6 mmol/L as a significant effect modifier of the TG-depression association. TG showed a significant positive association with depression in the context of high LDL-C, with 3.6 mmol/L emerging as an optimal threshold for identifying this interaction effect. This finding underscores the importance of considering lipid–lipid interactions when evaluating the link between lipid profiles and depression.
Similar content being viewed by others
Introduction
Depressive disorder is a prevalent mental health condition characterized by prolonged periods of low mood or diminished interest in activities. In severe cases, it can lead to suicide, with an estimated 727,000 people worldwide losing their lives to suicide in 20211. In 2019, depression accounted for 37.3% of disability-adjusted life-years attributable to mental disorders and ranked second among the top causes of years lived with disability globally2. Despite the widespread use of antidepressant medications, nearly one-third of patients remain treatment-resistant3,4, underscoring the need to identify novel and modifiable factors beyond conventional neurotransmitter-based mechanisms.
Emerging evidence suggests that dyslipidemia may be involved in the pathophysiology of depression. Elevated triglyceride (TG) levels may contribute to depression through inflammatory pathways5,6. Similarly, hypercholesterolemia has been associated with increased inflammation and impaired serotonin neurotransmission, potentially contributing to depressive symptoms7. However, evidence of the link between lipids and depression remains inconclusive. One study reported an association between low total cholesterol (TC) levels and a higher risk of depression8, whereas others found no significant association9,10. Similarly, studies based on the National Health and Nutrition Examination Survey (NHANES) also yielded conflicting results regarding the association between TG levels and depression11,12,13.
Most prior studies have focused on individual lipid parameters rather than their potential interactive effects. Cardiovascular studies have demonstrated that the combination of high TG and low high-density lipoprotein cholesterol (HDL-C) exerts synergistic effects on disease risk14. Building on this evidence, we hypothesized that interactions between TG and cholesterol subtypes would be associated with depression. Therefore, this study aimed to evaluate the interactive effects of specific lipid pairs (TG/TC, TG/low-density lipoprotein cholesterol [LDL-C], and TG/HDL-C) on the odds of depression.
Methods
Study design and participants
The NHANES assesses the health and nutritional status of the non-institutionalized U.S. civilian population through household interviews, standardized physical examinations, laboratory testing, and detailed demographic and dietary data. The study protocol was approved by the National Center for Health Statistics Research Ethics Review Board, and all participants provided written informed consent. For this study, we pooled data from seven consecutive survey cycles (2005–2006 to 2017–2018) to increase the sample size and generate nationally representative estimates.
Study variables
Serum lipids, including TG, TC, HDL-C, and LDL-C, served as exposure variables, and detailed analytical protocols for their quantification are comprehensively documented in the NHANES Laboratory Procedure Manual.
Depression was measured using the Patient Health Questionnaire-9 (PHQ-9), a validated 9-point instrument quantifying symptom frequency (0 = never to 3 = nearly daily), during the preceding fortnight. Higher total scores correspond to greater depression severity15. In this study, participants were stratified into non-depression (score range, 0–9) and depression (score range, 10–27) groups based on a validated clinical cutoff10.
Covariates
Covariates included age stratification, sex, race/ethnicity, educational attainment, marital status, family poverty-to-income ratio (PIR) categories, body mass index (BMI) categories, smoking status, alcohol consumption status16, physical activity (PA), estimated glomerular filtration rate (eGFR), diabetes, hypertension, and use of antidepressants and antihyperlipidemic drugs. The classification criteria for the indicators are listed in Table 1.
PA was quantified as weekly metabolic equivalent of task (MET)-minutes. MET values were computed using established methods17. Activity levels were categorized according to American guidelines: inactive (0 MET-min/week), insufficiently active (1–599 MET-min/week), and sufficiently active (≥ 600 MET-min/week)18. eGFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration 2021 equation (CKD-EPI 2021) and categorized according to established criteria19,20. Diabetes was defined by clinical diagnosis (self-reported physician diagnosis or current use of antidiabetic medications) or laboratory confirmation (fasting plasma glucose ≥ 7.0 mmol/L or HbA1c ≥ 6.5%)21. Hypertension was defined as physician-diagnosed hypertension, active antihypertensive medication use, or blood pressure ≥ 130/80 mmHg22. Antidepressant and antihyperlipidemic drug use was identified using a prescription medication questionnaire from NHANES.
Statistical analysis
The NHANES data analysis incorporated sampling weights to ensure national representativeness23. In this study, the fasting subsample 2-year mobile examination center weight (WTSAF2YR) was divided by 7 and used as the final weight. Differences in variables were assessed using weighted linear regression or Rao-Scott adjusted chi-squared tests.
Three survey-weighted generalized linear models (GLMs) were used to evaluate lipid-depression associations: Model 1 (unadjusted); Model 2 (adjusted for sociodemographic variables); and Model 3 (Model 2 additionally adjusted for smoking status, alcohol consumption, hypertension, diabetes, physical activity, and eGFR). The potential nonlinear relationship between lipid levels and depression were evaluated in Model 3 using survey-weighted restricted cubic spline (RCS) regression with 3 knots and pre-smoothing was applied to minimize overfitting prior to formal analysis. Variance inflation factors (VIFs) were calculated to assess multicollinearity, excluding variables with VIFs ≥ 524. Final VIFs are provided in Tables S1–S4.
This study employed survey-weighted GLMs (using the survey R package) to examine the interaction effects between continuous TG and various cholesterol levels, adjusting for the covariates in Model 3. For significant interaction terms, participants were dichotomized using lipid 2 thresholds corresponding to percentiles ranging from 2.5 to 97.5% in 2.5% increments. Within each subgroup, survey-weighted GLMs assessed the association between lipid 1 and depression odds, interaction testing was performed using likelihood ratio tests, with resulting P values adjusted for multiple comparisons via Bonferroni, Holm, and Benjamini–Hochberg methods. The optimal lipid 2 cutoff value was determined by selecting the points where the P values in the three multiple comparison adjustments are the smallest, with at least one being less than 0.05. In addition, to visualize the interaction effect between lipid parameters, the marginal effect of lipid 1 on depression across the observed range of lipid 2 was estimated as: βlipid 1 + βinteraction × lipid 2. Odds ratios (ORs) were calculated at varying levels of LDL-C, and corresponding 95% confidence intervals (CIs) were derived using the delta method under the complex survey design.
Sensitivity analyses were conducted under eight distinct analytical conditions to evaluate the robustness of the primary outcomes (Table 5). For conditions requiring handling of missing data, multiple imputations were performed using the mice R package. Detailed specifications of the multiple imputation procedure, including imputation methods, variables incorporated, and diagnostic checks, are provided in Table S5. All statistical procedures were performed using R version 4.3.3 (R Foundation) and Free Statistics platform v2.3, with P < 0.05 determining significance.
Results
Baseline characteristics
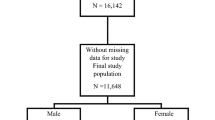
Following the screening protocol in Fig. 1, a total of 8962 eligible participants were included in the analysis. Significant differences were observed between depression and non-depression groups. Females predominated in the depression cohort (62.0%, P < 0.001). Although age showed no significant difference (P = 0.571), notable disparities were found in marital status, race, education, PIR, smoking, diabetes, BMI, PA, eGFR, TG, and HDL-C (all P < 0.05) (Table 1), collectively underscoring the multifactorial interplay between depression and sociodemographic, behavioral, and cardiometabolic determinants.
Participant inclusion flowchart.
Baseline characteristics of included and excluded participants are shown in Table S6. Except for age group and education level, all standardized mean differences were < 0.20, indicating generally comparable distributions between groups. The observed differences in age and education are consistent with the NHANES study design, as the PHQ-9 questionnaire was administered only to participants aged ≥ 18 years.
Association between individual lipids and depression
In the crude analyses, TG showed a positive association with depression, whereas HDL-C demonstrated a negative association (TG: P = 0.036; HDL-C: P = 0.028). These associations were attenuated to marginal significance in Model 2 (TG, P = 0.057; HDL-C, P = 0.079) and became non-significant in Model 3 (Table 2). Subsequent survey-weighted RCS analyses of lipid parameters based on Model 3 revealed no evidence of nonlinear associations with depression (Fig. 2; all P values for nonlinearity > 0.05).
Association between lipids and odds of depression. Panels: (a) Triglycerides; (b) Total Cholesterol; (c) High Density Lipoprotein Cholesterol; (d) Low Density Lipoprotein Cholesterol.
Interaction analysis of TG and cholesterol subtypes on the odds of depression
Survey-weighted GLMs demonstrated a significant multiplicative interaction between continuous triglyceride (TG) and LDL-cholesterol (LDL-C) levels in their association with depression (interaction β = 0.158; P = 0.03) (Table 3). The marginal effect analysis showed that the association between TG and depression progressively strengthened as LDL-C increased (Fig. 3). This interaction indicated that the relationship between TG and depression depended on LDL-C, prompting a search for relevant LDL-C thresholds. The optimal stratification point was identified at LDL-C = 3.6 mmol/L (Bonferroni, Holm, and Benjamini–Hochberg adjusted P were 0.061, 0.061, and 0.023, respectively, see Table S7). In contrast, no statistically valid TG cutoff value emerged to modify LDL-C–depression associations (all adjusted P > 0.05) (Table S8).
Marginal effect of TG across LDL-C levels.
The participants were stratified into subgroups based on predefined LDL-C cutoff: LDL-C levels ≥ 3.6 mmol/L, TG was found to have a positive association with depression (OR 1.78, 95% CI 1.30–2.42; P < 0.001), with a significant interaction effect (Pinteraction = 0.002) (Table 4). These findings underscore the lipid-level-dependent synergism in the odds of depression.
Sensitivity analyses
Across all tested conditions, a consistently positive association between triglycerides (TG) and depression was observed in the LDL-C ≥ 3.6 mmol/L subgroup (adjusted ORs 1.41–1.79; all P ≤ 0.005), whereas no significant association was detected in the LDL-C < 3.6 mmol/L subgroup (adjusted ORs 0.90–1.07; all P > 0.05). Interaction tests were significant in four of eight analytic conditions (Pinteraction ≤ 0.03) (Table 5).
To further minimize potential optimism bias, bootstrap resampling under the complex survey design was conducted using 1,000 replicate weights. The replicate-weighted GLM, based on the fully adjusted Model 3 and incorporating the TG × LDL-C group interaction term, demonstrated a significant interaction (OR 1.73, 95% CI 1.25–2.41, P = 0.002). These findings further support the robustness of the identified LDL-C cutoff.
Discussion
Individual analyses revealed no significant association between lipid components and depression. In contrast, when exploring the interaction effects, a significant interaction between LDL-C and TG levels on depression was identified. Quantile-driven analysis revealed a significant positive association between TG levels and depression specifically when LDL-C exceeded 3.6 mmol/L. Conversely, no such association was observed below this LDL-C threshold. By identifying LDL-C ≥ 3.6 mmol/L as a key effect modifier amplifying the TG-depression link, our findings provide an exploratory cutoff value for advancing ‘precision psychiatry’ approaches. Although the magnitude of some associations was modest, statistical significance does not necessarily imply clinical importance at the individual level. However, given the high prevalence of dyslipidemia and depression in the general population, even relatively small increases in relative risk may have meaningful implications at the population level, potentially affecting a substantial number of individuals.
Our analysis revealed a positive interaction between LDL-C and TG levels and the odds of depression. Critically, neither 3.388 mmol/L nor 3.465 mmol/L—values bracketing the conventional 3.4 mmol/L clinical threshold25,26—demonstrated statistically significant adjusted interaction P values, confirming that 3.4 mmol/L does not represent the optimal modifying threshold for TG-depression associations. Instead, statistical significance emerged at LDL-C = 3.595 mmol/L (Padjusted < 0.05), establishing 3.6 mmol/L as a meaningful modifying threshold for TG-depression relationships in this population, with robustness confirmed through sensitivity analyses.
The interaction analysis revealed that LDL-C significantly modified the association between TG and depression which may operate through sequential neuroinflammatory mechanisms. Elevated LDL-C levels impair blood–brain barrier integrity27,28,29, facilitating the entry of TG-derived free fatty acids into the central nervous system. These fatty acids activate toll-like receptor 4 receptors on microglia30,31, triggering neuroinflammatory cascades that exacerbate depression pathogenesis, particularly during the concomitant elevation of both lipids. Furthermore, both LDL particles and TG-rich lipoproteins are apolipoprotein B-containing lipoproteins, and elevated LDL-C levels may reflect an increased burden of circulating atherogenic particles. This cumulative apolipoprotein B-containing lipoprotein load has been implicated in endothelial dysfunction and systemic low-grade inflammation, potentially predisposing individuals to neurovascular impairment.
Although the association between LDL-C and depression is theoretically expected to vary significantly across TG levels, our analysis failed to identify statistically significant cutpoints after multiple comparison adjustment. As shown in Table 3, the negative main effect of LDL-C combined with its positive interaction with TG suggests that the net association may manifest as protective, null, or detrimental effects depending on TG concentrations—a finding consistent with prior reports of context-dependent lipid-depression relationships32,33,34. The absence of significant TG thresholds could stem from reduced statistical power in extreme percentiles or undetected nonlinearities.
This study has several strengths. First, combining a nationally representative sample with rigorously implemented inclusion and exclusion criteria and comprehensive sensitivity analyses strengthened the validity of our findings. Second, to our knowledge, this is the first study to demonstrate the synergistic interaction between TG and LDL-C levels on the odds of depression in a cross-sectional cohort. Third, our results further underscore the necessity of incorporating interactive effects into the evaluation of associations between lipid profiles and disease risk.
Nonetheless, several limitations should be acknowledged. First, depression may influence lipid metabolism through behavioral and biological mechanisms. Depressive symptoms are associated with altered dietary intake, reduced physical activity, and dysregulation of the hypothalamic–pituitary–adrenal axis, all of which may contribute to adverse lipid profiles. Therefore, reverse causality cannot be excluded in this cross-sectional analysis. Second, as the study population was derived from U.S. adults, caution is warranted when extrapolating these findings to populations with different racial and ethnic compositions. Finally, LDL-C was calculated using the Friedewald equation in the included NHANES cycles. Because this method does not yield valid LDL-C values when TG are ≥ 400 mg/dL, individuals with very high TG concentrations were excluded. Consequently, the findings may not be generalizable to populations with TG ≥ 400 mg/d. Future research should address these limitations to strengthen the clinical and mechanistic interpretations.
Conclusion
This study demonstrates that TG exhibit a significant positive association with depression specifically at LDL-C levels ≥ 3.6 mmol/L. Clinicians should prioritize comprehensive lipid profiling over single-metric evaluations when assessing the susceptibility to depression. Future large-scale prospective studies are critical to validate the interaction cutoffs and establish causality, which could reshape preventive strategies for depression.
Data availability
The analytical data for this investigation were obtained from the publicly accessible data repository of NHANES (https://wwwn.cdc.gov/Nchs/Nhanes/).
Abbreviations
- BMI:
-
Body Mass Index
- CI:
-
Confidence Interval
- CKD-EPI:
-
Chronic Kidney Disease Epidemiology Collaboration
- eGFR:
-
Estimated Glomerular Filtration Rate; GLM: Generalized Linear Models
- HDL-C:
-
High Density Lipoprotein Cholesterol
- LDL-C:
-
Low Density Lipoprotein Cholesterol
- NHANES:
-
National Health and Nutrition Examination Survey
- MET:
-
Metabolic Equivalent of Task
- OR:
-
Odds Ratio
- PA:
-
Physical Activity
- PHQ-9:
-
Patient Health Questionnaire 9
- PIR:
-
Poverty-to-Income Ratio
- RCS:
-
Restricted Cubic Spline
- TC:
-
Total Cholesterol
- TG:
-
Triglyceride
- VIF:
-
Variance inflation factors
References
Depressive disorder (depression) [https://www.who.int/news-room/fact-sheets/detail/depression]
Global, regional, and national burden of 12 mental disorders in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. The lancet Psychiatry 2022, 9(2):137–150.
Anand, A. et al. Ketamine versus ECT for nonpsychotic treatment-resistant major depression. N. Engl. J. Med. 388(25), 2315–2325 (2023).
Falchi-Carvalho, M. et al. Rapid and sustained antidepressant effects of vaporized N, N-dimethyltryptamine: A phase 2a clinical trial in treatment-resistant depression. Neuropsychopharmacology 50(6), 895–903 (2025).
Osimo, E. F., Baxter, L. J., Lewis, G., Jones, P. B. & Khandaker, G. M. Prevalence of low-grade inflammation in depression: A systematic review and meta-analysis of CRP levels. Psychol. Med. 49(12), 1958–1970 (2019).
Welty, F. K. How do elevated triglycerides and low HDL-cholesterol affect inflammation and atherothrombosis?. Curr. Cardiol. Rep. 15(9), 400 (2013).
Chen, S. J. et al. Pitavastatin attenuates hypercholesterolemia-induced decline in serotonin transporter availability. Lipids Health Dis. 23(1), 250 (2024).
Segoviano-Mendoza, M. et al. Hypocholesterolemia is an independent risk factor for depression disorder and suicide attempt in Northern Mexican population. BMC Psychiatry 18(1), 7 (2018).
Zhang, Q., Liu, Z., Wang, Q. & Li, X. Low cholesterol is not associated with depression: Data from the 2005-2018 National Health and Nutrition Examination Survey. Lipids Health Dis. 21(1), 35 (2022).
Zhong, X., Ming, J. & Li, C. Association between dyslipidemia and depression: A cross-sectional analysis of NHANES data from 2007 to 2018. BMC Psychiatry 24(1), 893 (2024).
Lin, Z., Chan, Y. H. & Cheung, B. M. Y. Dissecting relations between depression severity, antidepressant use, and metabolic syndrome components in the NHANES 2005–2020. J. Clin. Med. 12(12), 3891 (2023).
Shokeen, D. & Sokal-Gutierrez, K. Association between cardio-metabolic risks and depressive symptoms among US adults (NHANES 2013–2018). J. Affect. Disord. 299, 166–173 (2022).
Cepeda, M. S., Kern, D. M., Blacketer, C. & Drevets, W. C. Low levels of cholesterol and the cholesterol type are not associated with depression: Results of a cross-sectional NHANES study. J. Clin. Lipidol. 14(4), 515–521 (2020).
Carey, V. J. et al. Contribution of high plasma triglycerides and low high-density lipoprotein cholesterol to residual risk of coronary heart disease after establishment of low-density lipoprotein cholesterol control. Am. J. Cardiol. 106(6), 757–763 (2010).
Zimmerman, M. Using the 9-Item Patient Health Questionnaire to screen for and monitor depression. JAMA 322(21), 2125–2126 (2019).
Shen, Y. et al. Association between insulin resistance indices and kidney stones: Results from the 2015-2018 National Health and Nutrition Examination Survey. Front. Nutr. 11, 1444049 (2024).
Wei, X. et al. Joint association of physical activity and dietary quality with survival among US cancer survivors: A population-based cohort study. Int. J. Surg. (London, England) 110(9), 5585–5594 (2024).
Piercy, K. L. et al. The physical activity guidelines for Americans. JAMA 320(19), 2020–2028 (2018).
Grams, M. E. et al. The kidney failure risk equation: Evaluation of novel input variables including eGFR estimated using the CKD-EPI 2021 equation in 59 cohorts. J. Am. Soc. Nephrol. 34(3), 482–494 (2023).
Qin, Z. et al. Systemic immune-inflammation index is associated with increased urinary albumin excretion: A population-based study. Front. Immunol. 13, 863640 (2022).
Kianersi, S., Liu, Y. & Guasch-Ferré, M. Chronotype, unhealthy lifestyle, and diabetes risk in middle-aged U.S. women: A prospective cohort study. Ann. Intern. Med. 176(10), 1330–1339 (2023).
Huang, Z. Association between blood lead level with high blood pressure in US (NHANES 1999–2018). Front. Public Health. 10, 836357 (2022).
Johnson, C. L. et al. National health and nutrition examination survey: analytic guidelines, 1999–2010: Vital and health statistics Series 2. Data Eval. Methods Res. 161, 1–24 (2013).
Li, F. et al. Association between the cumulative average triglyceride glucose-body mass index and cardiovascular disease incidence among the middle-aged and older population: A prospective nationwide cohort study in China. Cardiovasc. Diabetol. 23(1), 16 (2024).
Wiegman, A. et al. Efficacy and safety of Inclisiran in adolescents with genetically confirmed homozygous familial hypercholesterolemia: Results from the double-blind, placebo-controlled part of the ORION-13 randomized trial. Circulation 151(25), 1758–1766 (2025).
Errigo, A., Dore, M. P., Portoghese, M. & Pes, G. M. The cholesterol paradox in long-livers from a Sardinia longevity hot spot (blue zone). Nutrients 17(5), 765 (2025).
Griffiths, H., Irundika, D., Lip, G., Spickett, C. & Polidori, C. Oxidised LDL lipids, statins and a blood-brain barrier. Free Radic. Biol. Med. 75(Suppl 1), S15-16 (2014).
de Oliveira, J. et al. High cholesterol diet exacerbates blood-brain barrier disruption in LDLr-/- mice: Impact on cognitive function. J. Alzheimers. Dis. 78(1), 97–115 (2020).
Zhou, L. Q. et al. Ldl-stimulated microglial activation exacerbates ischemic white matter damage. Brain Behav. Immun. 119, 416–430 (2024).
Dasu, M. R. & Jialal, I. Free fatty acids in the presence of high glucose amplify monocyte inflammation via Toll-like receptors. Am. J. Physiol. Endocrinol. Metab. 300(1), E145-154 (2011).
Pišlar, A. et al. Cysteine peptidase Cathepsin X as a therapeutic target for simultaneous TLR3/4-mediated microglia activation. Mol. Neurobiol. 59(4), 2258–2276 (2022).
So, H. C., Chau, C. K., Cheng, Y. Y. & Sham, P. C. Causal relationships between blood lipids and depression phenotypes: A Mendelian randomisation analysis. Psychol. Med. 51(14), 2357–2369 (2021).
Wagner, C. J. et al. LDL cholesterol relates to depression, its severity, and the prospective course. Prog. Neuropsychopharmacol. Biol. Psychiatry 92, 405–411 (2019).
Enko, D. et al. Prospective plasma lipid profiling in individuals with and without depression. Lipids Health Dis. 17(1), 149 (2018).
Acknowledgements
The authors express profound gratitude to the National Center for Health Statistics research team for their rigorous execution of complex survey protocols and commitment to open data dissemination through the NHANES repository.
Funding
This work was supported by the Bureau of Science and Technology, Nanchong City [Grant Numbers 23YYJCYJ0158, and 25KJCXRC0013] and the Natural Science Foundation of Sichuan Province [Grant Number 2023JDRC0091].
Author information
Authors and Affiliations
Contributions
Shun Li: Writing—original draft and data curation; Jing Li: Formal Analysis & Writing—review, and editing; Fuqin Xiang: Data curation; Qi Huang: Data curation; Xiaohua Xian: Writing—review, editing, and funding acquisition; Jingjing Liu: Validation & Funding acquisition; Yin Xian: Conceptualization & Visualization & Data curation, Formal Analysis & Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The National center for health statistics Ethics Review Board approved the survey, and all participants provided written informed consent. This research was conducted in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, S., Li, J., Xiang, F. et al. LDL-C modifies the association between triglycerides and depression identified by threshold scanning in US adults. Sci Rep 16, 11710 (2026). https://doi.org/10.1038/s41598-026-44940-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-44940-4