Abstract
The goal of post-cesarean analgesia includes minimizing pain and enabling early ambulation. Quadratus lumborum block has emerged as a part of multimodal analgesia for this procedure. This randomized controlled trial compared the analgesic effects of anterior quadratus lumborum block (QLB3) and intramuscular quadratus lumborum block (QLB4). A total of 128 parturients undergoing cesarean delivery (64 per group) were enrolled in this study. Parameters including butorphanol consumption, PCA utilization, pain scores, block duration, adverse events and maternal satisfaction were assessed. QLB4 was associated with significantly lower 24/48-hour butorphanol use, less PCA pressing at 6–24 h, lower 6/12-hour NRS pain scores and a much lower block resolution rate at 12–24 h than QLB3 (all p < 0.05). QLB4 had a lower incidence of lower extremity hypoesthesia (0% vs. 46.88%), shorter procedural time, and higher maternal satisfaction scores (all p < 0.05). No significant between-group differences in late-stage opioid use, pain scores or other adverse events were found. QLB4 provides more effective, longer-lasting post-cesarean analgesia with superior safety and patient satisfaction, and greater procedural efficiency, making it a valuable multimodal analgesia option for cesarean delivery.
Clinical trial registration: This trial was registered on the Chinses Clinical Trial Registry on 27/08/2025 (ChiCTR2500108272).
Similar content being viewed by others
Introduction
The quadratus lumborum block (QLB) is an effective regional anesthetic technique for post-cesarean delivery1. The QLB is categorized into four distinct approaches based on needle tip placement: the lateral approach (QLB1), posterior approach (QLB2), anterior approach (QLB3), and intramuscular approach (QLB4)2,3. The anatomical basis of the QLB centers on the thoracolumbar fascia (TLF). The TLF, constituting the deep fascia of the thoracodorsal and lumbar regions, comprises three layers in the lumbar region: the anterior, middle, and posterior layers. The quadratus lumborum muscle is enveloped anteriorly by the anterior layer and posteriorly by the middle layer of the TLF4. The TLF serves not only as a conduit for the diffusion of local anesthetic towards the paravertebral space3, but is also innervated by somatic nerves and sympathetic fibers5. Consequently, the QLB provides simultaneous blockade of both somatic and visceral nociceptive transmission.
Previous studies have reported the analgesic efficacy of QLB performed via different approaches (QLB1, QLB2, and QLB3), with results indicating that the QLB3 provides superior analgesia6,7,8. However, clinical observation has revealed a relatively high incidence of lower extremity hypoesthesia in patients receiving QLB3 for post-operative analgesia following abdominal surgery9. Additionally, performing the QLB3 requires the patient to be positioned laterally. Although some case reports reported that QLB3 was performed successfully in the supine position, there are still challenges such as technical difficulties in ultrasound operation and probe fixation, anatomical interferences during the procedure, and elevated safety risks of puncture10,11. This presents technical challenges when administering the block to patients who have received neuraxial anesthesia for cesarean delivery.
The QLB4 involves the direct injection of local anesthetic into the substance of the quadratus lumborum muscle. This technique offers the advantages of not requiring identification of the thoracolumbar fascial plane and can be performed with the patient in the supine position, making it relatively straightforward to perform. In addition, the incidence of lower extremity hypoesthesia caused by QLB4 is much lower9. Some studies have reported its controversial role in postoperative pain management12,13,14. Herein, we performed a randomized controlled trial to compare the effects of QLB3 and QLB4 on post-cesarean pain.
Results
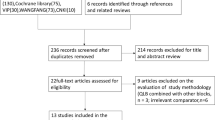
From August 28, 2025 to November 20, 2025, 364 puerperas were assessed for eligibility, with 128 of them enrolled and analyzed (Fig. 1). As shown in Table 1, the age, body weight, and BMI were comparable between the groups (29.19 ± 3.59 vs. 30.20 ± 2.77 years, p = 0.076; 66.52 ± 6.93 vs. 68.13 ± 7.56 kg, p = 0.210; 26.48 ± 2.50 vs. 26.51 ± 2.47 kg/m2, p = 0.949, respectively), though the height of QLB4 group was higher than that of QLB3 group (158.47 ± 3.55 vs. 160.25 ± 4.78 cm, p = 0.018).
Flowchart of this study. QLB3, anterior quadratus lumborum block. QLB4, intramuscular quadratus lumborum block.
The consumption of butorphanol 24 h after QLB3 was 67.22 ± 16.44 mL (100 µg/mL), which is significantly higher than 60.42 ± 14.31 mL in the QLB4 group (p = 0.014, Table 2). Similarly, the consumption of butorphanol 48 h after QLB3 was significantly higher than after QLB4 (115.36 ± 16.35 vs. 108.70 ± 14.36 mL, p = 0.016, Table 2). However, the consumption from 24 to 48 h had no significant difference between the groups (48.14 ± 0.64 vs. 48.28 ± 1.58 mL, p = 0.510, Table 2).
In the QLB3 group, 56 patients pressed the PCA button at least once within 24–48 h, similar to 54 in the QLB4 group (p = 0.611, Table 2). The first time of pressing PCA button was 4.53 (3.17, 7.13) hours in the QLB3 group, and it was 5.02 (3.60, 12.43) in the QLB4 group, with no significant difference (p = 0.209, Table 2). The times of PCA within 6 post-block hours in the QLB3 group was comparable with that in the QLB4 group (2 (1, 3) vs. 1.5 (1, 2), p = 0.057, Table 2), while the times of PCA within 12, 24, and 48 post-block hours in the QLB3 group was significantly more than that in the QLB4 group (2 (1, 5) vs. 1 (0, 3), p = 0.008; 5 (2.25, 9) vs. 3 (1, 5), p = 0.007; 5 (2.25, 9) vs. 3 (1, 5), p = 0.008; respectively, Table 2). Further analyses indicated that patients in the QLB3 group pressed more times of PCA button from 6 to 12 and 12 to 24 post-block hours than those in the QLB4 group (1 (0, 3) vs. 0.5 (0, 2), p = 0.023; 3 (1, 4) vs. 1 (0, 3.75), p = 0.022; respectively, Table 2), whereas the difference was not significant from 24 to 48 post-block hours (0 (0, 0) vs. 0 (0, 0), p = 0.984, Table 2).
The NRS scores of both resting state and exercising state in the QLB3 group were significantly higher than that in the QLB4 group at 6 and 12 post-block hours, but the differences at 24–48 h were not significant (Table 3). At 6 post-block hours, all patients had blocking planes (Table 3). At 12 post-block hours, 76.56% patients in the QLB3 group had blocking planes, significantly less than 98.44% in the QLB4 group (p < 0.001, Table 3). At 24 post-block hours, the difference of blocking rate was not significant (29.69% vs. 37.50%, p = 0.349, Table 3). When it came to 48 post-block hours, the blocking effects disappeared in all patients (Table 3).
During the study period, adverse events we observed included nausea and vomiting, dizziness, drowsiness, and lower extremity hypoesthesia. The incidence of lower extremity hypoesthesia in the QLB3 group was significantly higher than that in the QLB4 group (46.88% vs. 0, p < 0.001, Table 4). However, the differences of the other adverse events between the groups were not significant (Table 4). In addition, the operation time (from position preparation to injection completion) was significantly longer in the QLB3 group (13.25 ± 3.14 vs. 10.87 ± 2.98, P < 0.001).
The patients in the QLB4 group felt significantly more satisfied with the pain management than those in the QLB3 group (8 (8,9) vs. 9 (8, 10), p = 0.025, Table 4).
Discussion
The present study compared the analgesic effects of QLB3 and QLB4 on post-cesarean pain. The results of this randomized controlled trial demonstrates that the QLB4 technique provides superior analgesic efficacy compared to QLB3 in the first 24 h following cesarean delivery.
Our primary outcome, 24-hour or 48-hour butorphanol consumption, was significantly lower in the QLB4 group. However, the consumption from 24 to 48 h was comparable between the groups, which indicates the effect duration of a single injection of ropivacaine. We further compared the effect duration of QLB3 and QLB4 in an indirect way. The block duration of ropivacaine for neuraxial anesthesia is within 6 h15. The block rate of QLB at 6 h were 100% in both groups. However, a significantly higher rate of sustained sensory blockade was observed in the QLB4 group at 12 h (98.44% vs. 76.56%). These findings suggest QLB4 provides more prolonged dermatomal coverage. At 24 post-block hours, only a small part of patients sustained sensory blockade (37.50% vs. 29.69%), indicating the median action time of ropivacaine via QLB3 or QLB4 is between 12 and 24 h. Zhang et al.2 found that QLB4 could provide a sensory loss of 5 to 24 h, which is generally consistent with our results.
The temporal pattern of analgesia further elucidates QLB4’s advantages. While both groups showed comparable PCA usage and pain scores within the first 6 h, significant differences emerged between 6 and 24 h after block. QLB4 patients pressed the PCA button significantly less frequently during the 6–12 h and 12–24 h intervals and reported lower NRS scores at rest and during exercise at 6 h and 12 h. The convergence of NRS scores and PCA usage between groups by 24–48 h indicates a comparable analgesic profile once the primary block effects subside.
A critical finding was the significantly higher incidence of transient lower extremity hypoesthesia in the QLB3 group (46.88% vs. 0%), which is in line with a previous study performed in pediatric patients9. This suggests QLB3 may involve a greater degree of lateral spread affecting lumbar plexus branches, potentially limiting early mobilization, a key goal in enhanced recovery after cesarean protocols. While no permanent neurological deficits or other serious complications (organ damage, significant bleeding, local anesthetic toxicity) were reported, the hypoesthesia difference of lower limbs underscores a tolerability advantage for QLB4. The shorter operation time for QLB4 (10.87 ± 2.98 min vs. 13.25 ± 3.14 min), together with no position change, also supports its practical efficiency in clinical settings.
The reduction in opioid requirement did not lead to statistically significant differences in nausea, vomiting, drowsiness or dizziness. This may attribute to the difference of butorphanol (within 10 mL or 1 mg).
The significantly higher maternal satisfaction scores in the QLB4 group (9 (8, 10) vs. 8 (8, 9)) strongly supports its clinical relevance. This enhanced satisfaction likely integrates the combined benefits of lower pain intensity during mobilization in the first 12 h, fewer PCA interventions, and avoidance of lower extremity sensory changes.
We hypothesized that the observed differences likely stem from anatomical spread patterns. QLB3 deposits local anesthetic between the quadratus lumborum (QL) and psoas major muscles (TLF plane). While this facilitates diffusion to the paravertebral space, the spread is relatively uncontrolled and biased laterally/caudally to the L1–L3 lumbar plexus (the primary cause of lower extremity hypoesthesia). This excessive lateral/caudal diffusion diverts local anesthetic from cephalad spread to the T6–T10 thoracic dermatomes—the critical level for cesarean analgesia16,17. QLB4 deposits anesthetic within the quadratus lumborum, potentially facilitating more consistent spread within the thoracolumbar fascial plane and medial diffusion towards the paravertebral space. Since post-cesarean pain involves somatosensory innervation (abdominal wall trauma) and sympathetic innervation (pelvic peritoneal and uterine surgical manipulation), ropivacaine via the intramuscular pathway may preferentially block the sympathetic neural network within the thoracolumbar fascia, thereby augmenting relief of visceral nociceptive components of post-cesarean pain. Even though these hypotheses need to be validated by imaging studies, this may explain both the prolonged analgesia and reduced motor/sensory side effects seen with QLB4. A previous study reported that the block range was T7-T12, making it suitable for lower abdominal surgery18. Yoshida et al.13 performed a study to investigate the effects of QLB4 vs. saline on post-cesarean pain. They found that the first time of additional analgesic in the QLB4 group was longer (230 ± 103 vs. 194 ± 89 min), but without statistical significance. Their result may be caused by small sample size (18 in each group), low ropivacaine concentration (0.25%), and absence of sensory testing or motor function assessment to verify block success.
Despite the meaningful findings, we have to acknowledge some limitations. The first is the lack of formal assessment of dermatomal level of blockade, which precludes comparative analysis of the block range between the groups and further hinders the explanation of our results. It takes much time to confirm the dermatomal level and repeated test can compromise the judgement of patients, which impacts the resting time and satisfaction of patients, as well as the credibility of measured findings. Alternatively, we tested whether there were bloke planes to help us interpret the results. Secondly, we did not test the exact time of block duration. However, we determined the longer duration of QLB4 by comparing the block rate at different time points. In addition, long-term outcomes were not assessed, as the study only evaluated endpoints within 48 h postoperatively, precluding determination of potential long-term tissue injury from the intramuscular approach. A further notable limitation is that the differences observed in partial key outcomes, such as the dose of butorphanol, do not inevitably indicate clinically meaningful differences, and these modest numerical gaps should be interpreted with caution in the context of clinical practice.
In conclusion, QLB4 demonstrates clinically significant advantages over QLB3 for post-cesarean analgesia, providing more effective and sustained pain relief in the first 24 h, a superior safety profile regarding lower extremity sensation, higher patient satisfaction, and greater procedural efficiency. These findings support the adoption of QLB4 as a valuable component of multimodal analgesia regimens for cesarean delivery.
Methods
This randomized controlled study was reviewed and approved by the Medical Ethics Committee of Sichuan Jinxin Xinan Women & Children Hospital. All patients signed the written informed consent. The study followed international standards: the Consolidated Standards of Reporting Trials (CONSORT)19 guidelines for reporting and the Declaration of Helsinki for ethics.
Patients
Pregnant women scheduled to undergo a cesarean were assessed for eligibility.
Inclusion criteria
(1) Nulliparous women scheduled for elective cesarean delivery via lower uterine segment transverse incision under neuraxial anesthesia; (2) Singleton pregnancy at term; (3) Age between 23 and 35 years; (4) American Society of Anesthesiologists (ASA) physical status classification II or III; (5) Body Mass Index (BMI) < 30 kg/m2; (6) Demonstrated sensory block level within T6-S5 upon completion of surgery; (7) Provision of written informed consent.
Exclusion criteria
(1) Patient refusal to receive a regional nerve block technique; (2) Pre-existing neuropathy involved in studied area; (3) Infection at the proposed block injection sites; (4) Chronic analgesic use or history of chronic pain; (5) History of psychiatric illness; (6) History of opioid abuse or tolerance; (7) Known allergy to any study-related medications; (8) Any other condition potentially affecting patient enrollment or outcome assessment.
Withdrawal criteria
Study withdrawal is defined as the discontinuation of a participant’s involvement in the clinical study for any reason. Participants must be withdrawn from the study if any of the following occur: Intraoperative change in the planned surgical approach or anesthetic technique, intraoperative placement of an intrauterine balloon tamponade, requirement for postoperative angiographic embolization for hemorrhage control, necessity for a secondary surgical procedure, failure of the administered nerve block, surgical duration exceeding 3 h, and participant withdrawal of informed consent for the clinical trial.
Randomization and blinding
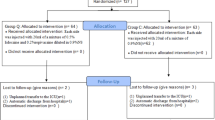
Participants were randomly assigned to either the QLB3 group or the QLB4 group using SPSS-generated randomization sequences (1:1 allocation ratio). The allocation sequence was concealed using sequentially numbered, opaque, sealed envelopes. Before the nerve block was carried out, the anesthesiologist cut open the envelope and performed the QLB as instructed in the letter. All the nerve blocks for the parturients were performed by a highly skilled anesthesiologist who did not participate in post-block management. Patients, surgeons, and outcome assessors were blinded to group assignment. Unblinding occurred only for data analysis or in emergency scenarios per predefined protocols.
Interventions
Following surgery completion, participants in both the QLB3 group and the QLB4 group received a bilateral quadratus lumborum block under ultrasound guidance. Each side was administered 20 mL of 0.375% ropivacaine. For the QLB3 group, the injectate was deposited between the quadratus lumborum and psoas major muscles. For the QLB4 group, the injectate was deposited intramuscularly within the substance of the quadratus lumborum muscle.
Anesthetic procedure
Upon arrival in the operating room, all participants received standard monitoring, including electrocardiography (ECG), pulse oximetry (SpO₂), respiratory rate, and non-invasive blood pressure (NIBP). All patients underwent cesarean delivery under combined spinal-epidural anesthesia (CSEA). Epidural puncture was performed at the L3-L4 intervertebral space. Ropivacaine was diluted to a concentration of 0.5% using cerebrospinal fluid (after confirming free flow of CSF). A volume of 3.0–3.2.0.2 mL of the 0.5% ropivacaine solution was injected intrathecally at a constant rate over 10–15 s. An epidural catheter was inserted cephalad and secured with its tip residing 3–5 cm within the epidural space. Immediately following anesthetic administration, participants were placed in the supine position, and the operating table was tilted 30 degrees to the left to prevent aortocaval compression. As for postoperative analgesia, both groups received patient-controlled intravenous analgesia (PCIA) postoperatively. Upon completion of surgery, an electronic PCIA pump was connected to the peripheral intravenous access. The PCIA solution is butorphanol 100 µg/mL. The PCIA Parameters were as follows: Background infusion: 2 mL/h; Bolus dose: 3 mL; Lockout interval: 20 min; Maximum hourly dose: 10 mL/h. Participants and their families received standardized education on the use of the PCIA device. If the Numerical Rating Scale (NRS) pain score remained ≥ 4 despite PCIA, rescue analgesia consisted of an intravenous bolus of butorphanol 1 mg were administrated.
Implementation of QLB
Immediately following transfer to the Post-Anesthesia Care Unit (PACU) after surgery, bilateral quadratus lumborum blocks were performed. Utilizing an ultrasound system (S-II; FUJIFILM SonoSite Inc., Bothell, WA, USA) equipped with a low-frequency convex transducer and a 19G × 100 mm needle, two approaches were employed. For the anterior (transmuscular) approach, the patient was positioned laterally with the target side uppermost. The operator, standing posterior to the patient, prepared the skin with povidone-iodine and applied a sterile fenestrated drape; the transducer was coated with gel and covered with a sterile transparent sheath. Placed vertically between the costal margin and iliac crest, the transducer was slid anteroposteriorly to identify the origins of the external oblique, internal oblique, and transversus abdominis muscles. It was then gradually moved dorsally to locate the “shamrock sign”: continuous hyperechoic structures representing the transverse process (stem) and vertebral body, with the quadratus lumborum muscle superior to the transverse process apex (central leaf), the psoas major muscle overlying the vertebral body, and the erector spinae dorsally (lateral leaves). The anterior thoracolumbar fascia appeared as a hyperechoic layer between the psoas major and quadratus lumborum muscles. Using an in-plane technique, the needle was inserted approximately 1 cm from the transducer edge. Upon reaching the target fascial plane between quadratus lumborum and psoas major, aspiration confirmed no blood return. Hydrodissection with 1–2 mL saline verified the correct plane before injecting 20 mL of 0.375% ropivacaine, often observable as quadratus lumborum muscle lifting. The transducer was then rotated 90 degrees longitudinally to confirm craniocaudal spread of local anesthetic between the quadratus lumborum and psoas major muscles from the iliac crest to the 12th rib. Alternatively, for the intramuscular approach, the patient was placed supine. The operator, standing on the target side, performed skin preparation and transducer sheathing following standard aseptic technique. The transducer was positioned slightly cephalad to the iliac crest and moved anteroposteriorly to identify the quadratus lumborum muscle. Using an in-plane technique, the needle was advanced to penetrate the fascia and enter the quadratus lumborum muscle substance. After confirming no blood aspiration, hydrodissection with 1–2 mL saline ensured intramuscular spread prior to injecting 20 mL of 0.375% ropivacaine intramuscularly. The images of QLB3 and QLB4 were shown in the supplementary material.
Approximately 6 h following the injection of local anesthetic, sensory assessment was conducted over the thoracic, abdominal, and dorsal regions innervated by the nerves targeted by the specific QLB approach. This assessment involved inquiring whether the patient experienced spontaneous numbness in the skin over the chest wall, abdominal wall, and lumbar back; the presence of numbness suggests successful blockade. Additionally, a blunt needle was used to lightly prick the skin within the anticipated block distribution area to test for the presence of pain sensation (pinprick test). A partial sensory block is defined by the loss of pinprick sensation while tactile sensation remains intact, whereas a complete sensory block is characterized by the loss of both pinprick sensation and tactile sensation. Concurrently, sensory abnormalities (such as paresthesia or numbness) or motor deficits (including decreased muscle strength) in the lower limbs were also monitored.
Outcomes
The primary outcome was the consumption of butorphanol 24 h after QLB. The secondary outcomes included the consumption of butorphanol 48 h after QLB, time of the first use of PCA, block duration, operation time, number of PCA use, NRS scores at rest and during exercise, and maternal satisfaction score. Maternal satisfaction scores were assessed at 48 h after surgery via a 0–10 rating scale, according to their subjective feelings of postoperative pain and adverse events. The occurrence of complications such as infection at the puncture site, nausea and vomiting, dizziness, organ damage, bleeding, and local anesthetic poisoning were employed as the safety indicators during the study period.
Statistics
Based on our preliminary data, the 24-hour butorphanol consumption was 68.5 ± 9.8 mg in the QLB3 group versus 63.2 ± 8.9 mg in the QLB4 group. Using the G*Power 3.1 software with a two-sided α level of 0.05 and statistical power of 0.8, a minimum of 58 participants per group was calculated for independent samples t-test analysis. Accounting for an anticipated 10% dropout rate, the final enrollment target was set at 64 patients per group, yielding a total enrollment of 128 participants.
All analyses were conducted using SPSS 22.0. Continuous variables were expressed as mean ± standard deviation or median [interquartile range] based on distributional assumptions verified by Shapiro-Wilk and Levene tests. Parametric comparisons between the groups employed t-test; non-parametric equivalents used Mann-Whitney U and Friedman tests. Categorical variables were analyzed by Chi-Square test or Fisher’s exact test. Statistical significance was defined as two-tailed P < 0.05.
Data availability
The data will be available upon reasonable request from the corresponding author.
References
Xu, M. et al. Quadratus lumborum block for postoperative analgesia after cesarean delivery: A systematic review and meta-analysis. Int. J. Obstet. Anesth. 42, 87–98 (2020).
Zhang, S. et al. Sensory assessment of intramuscular quadratus lumborum block at the L2 level in open inguinal hernia repair patients. BMC Anesthesiol. 24(1), 377 (2024).
Ueshima, H., Otake, H. & Lin, J. A. Ultrasound-guided quadratus lumborum block: An updated review of anatomy and techniques. Biomed Res Int 2017, 2752876 (2017).
Willard, F. H. et al. The thoracolumbar fascia: anatomy, function and clinical considerations. J. Anat. 221(6), 507–536 (2012).
Schilder, A. et al. Sensory findings after stimulation of the thoracolumbar fascia with hypertonic saline suggest its contribution to low back pain. Pain 155(2), 222–231 (2014).
Koksal, E. et al. Comparison of the analgesic effects of two quadratus lumborum blocks (QLBs), QLB type II vs QLB type III, in caesarean delivery: A randomised study. Int. J. Clin. Pract. 75(10), e14513 (2021).
Elsayed Elashry, H. et al. Analgesic effect of quadratus lumborum block type III and type II versus lateral transversus abdominis plane block in cesarean section: A randomized controlled multicenter trial. Anesth. Pain Med. 14(1), e140464 (2024).
Singh, N. P. et al. Efficacy of different approaches of quadratus lumborum block for postoperative analgesia after cesarean delivery: A Bayesian network meta-analysis of randomized controlled trials. Clin. J. Pain 39(11), 634–642 (2023).
Hussein, M. M. Ultrasound-guided quadratus lumborum block in pediatrics: trans-muscular versus intra-muscular approach. J. Anesth. 32(6), 850–855 (2018).
D’Souza, N., Himashweta, G. & Diwan, S. Transmuscular quadratus lumborum block (QLB) in supine position for abdominal surgeries: Pros and cons. Saudi J. Anaesth. 17(1), 138–140 (2023).
Diwan, S. et al. [The supine coronal midaxillary approach to anterior quadratus lumborum block: case report]. Braz J. Anesthesiol. 70(4), 443–447 (2020).
Yoshida, K. et al. Intramuscular quadratus lumborum block can be a good analgesic option for lumbar spine surgery. JA Clin. Rep. 10(1), 76 (2024).
Yoshida, K. et al. The effectiveness of the intramuscular quadratus lumborum block in postoperative analgesia after cesarean section with vertical incision: a randomized, double-blinded placebo-controlled study. J. Anesth. 34(6), 849–856 (2020).
Aboelfadl, G. M. et al. Comparison of two approaches to quadratus lumborum block for postoperative analgesia in radical cystectomy: A randomized clinical trial. Minerva Anestesiol. 91(1–2), 36–44 (2025).
Hansen, T. G. Ropivacaine: a pharmacological review. Expert Rev. Neurother. 4(5), 781–791 (2004).
Carline, L., McLeod, G. A. & Lamb, C. A cadaver study comparing spread of dye and nerve involvement after three different quadratus lumborum blocks. Br. J. Anaesth. 117(3), 387–394 (2016).
Balocco, A. L. et al. Quadratus lumborum block: an imaging study of three approaches. Reg. Anesth. Pain Med. 46(1), 35–40 (2021).
Murouchi, T., Iwasaki, S. & Yamakage, M. Quadratus lumborum block: Analgesic effects and chronological ropivacaine concentrations after laparoscopic surgery. Reg. Anesth. Pain Med. 41(2), 146–150 (2016).
Hopewell, S. et al. CONSORT 2025 statement: Updated guideline for reporting randomised trials. BMJ 389, e081123 (2025).
Funding
This study is supported by Health Commission of Chengdu Municipality (No. 2022075).
Author information
Authors and Affiliations
Contributions
Study design: Yang Zhong and Dan Li; Data collection: Yang Zhong, Yong Tang, Jianping Zhang, Yajing Huang, and Dan Li; Project management: Yong Tang and Dequan Li; Statistics: Xuelin Pan; Funding: Yang Zhong; Drafting manuscript: Yang Zhong, Yong Tang, Jianping Zhang, Xuelin Pan, Dequan Li, Yajing Huang, and Dan Li. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhong, Y., Tang, Y., Zhang, J. et al. Effects of intramuscular quadratus lumborum block on post-cesarean pain: a randomized controlled trial. Sci Rep 16, 10734 (2026). https://doi.org/10.1038/s41598-026-46025-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-46025-8