Abstract
The early identification and risk stratification of metabolic-associated fatty liver disease (MAFLD) urgently require effective, non-invasive tools. This study aimed to comprehensively evaluate the association of the triglyceride-glucose-body mass index (TyG-BMI) with the risk of MAFLD prevalence and the degree of steatosis, and to further assess its potential as a biomarker for liver fibrosis. This cross-sectional study included 1,457 participants. Logistic regression were employed to assess the association between TyG-BMI and MAFLD risk, with diagnostic performance evaluated by ROC curve analysis. The relationships of TyG-BMI with MAFLD severity and liver stiffness measurement (LSM) were investigated. Individuals with MAFLD had a significantly higher TyG-BMI index than those without (242.02 ± 41.04 vs. 198.44 ± 33.99). After adjusting for confounders, each 1-unit increase in TyG-BMI was associated with a 3.4% increase in the risk of MAFLD (aOR = 1.034, 95% CI: 1.030–1.038). The AUC for TyG-BMI in diagnosing MAFLD was 0.806. This association was significantly modified by age, being stronger in younger individuals (≤ 60 years). Furthermore, trend tests indicated a significant increase in TyG-BMI levels with advancing degree of steatosis, and TyG-BMI was identified as an independent risk factor for the degree of steatosis (OR = 1.033). Additionally, the TyG-BMI index showed a significant positive correlation with LSM (Spearman’s r = 0.255). A linear regression confirmed that TyG-BMI was an independent contributing factor to LSM (β = 0.011) after adjusting for age and sex. The TyG-BMI index is independently associated with the prevalence, severity, and fibrosis risk of MAFLD. As a simple, non-invasive index integrating routine metabolic parameters, it holds potential for initial screening and may provide ancillary information for fibrosis risk assessment in clinical practice.
Similar content being viewed by others
Introduction
Metabolic-associated fatty liver disease (MAFLD), a chronic liver condition closely linked to metabolic dysfunction, affects more than one-third of the adult population worldwide, with its prevalence increasing proportionally1,2. The disease spectrum of MAFLD can progress from simple hepatic steatosis to steatohepatitis, liver fibrosis, and even cirrhosis, significantly elevating the risk of hepatocellular carcinoma and extrahepatic complications3,4,5. Although liver biopsy remains the gold standard for diagnosis and staging, its invasive nature, high cost, and other limitations severely restrict its application in population-wide screening and long-term follow-up. Consequently, there is an urgent clinical need for accurate, non-invasive, and easily accessible biomarkers for the early identification and risk stratification of MAFLD.
Insulin resistance (IR) is a key mechanism in the pathogenesis and progression of MAFLD6,7. IR not only promotes hepatic steatosis but also exacerbates systemic metabolic dysfunction, ultimately leading to increased mortality8. However, the hyperinsulinemic-euglycemic clamp, the gold standard for assessing IR, is not suitable for routine use due to its operational complexity9. The triglyceride-glucose index (TyG), calculated from two routine biochemical parameters—fasting triglycerides and fasting blood glucose—has been widely validated as a reliable surrogate marker of IR10,11. In recent years, to enhance predictive performance, researchers have combined the TyG index with body mass index (BMI) to create the triglyceride-glucose-body mass index (TyG-BMI)12,13. This composite index simultaneously integrates two core risk factors for MAFLD: dysregulated glucose and lipid metabolism and overall adiposity. A study focusing on non-diabetic populations confirmed that TyG-BMI predicts MAFLD with an AUC of up to 0.835, indicating good discriminatory ability14.TyG-BMI has been robustly associated with MAFLD prevalence in numerous studies, including large-scale cohorts and meta-analyses reporting area under the curve (AUC) values ranging from 0.83 to 0.8915,16,17,18.
However, the vast majority of current research has focused solely on the association between TyG-BMI and the prevalence of MAFLD. There is a lack of systematic evaluation regarding its ability to differentiate the degree of steatosis and its independent association with liver fibrosis—a critical prognostic indicator. Furthermore, there are relatively few studies comparing the TyG-BMI index with traditional liver fat scoring systems such as fatty liver index (FLI)19. Based on these gaps, this study aims to comprehensively assess the association of the TyG-BMI index with the risk of MAFLD prevalence and the degree of steatosis, and to further evaluate its potential as a biomarker for liver fibrosis.
Materials and methods
Study design and participants
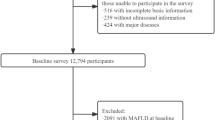
This cross-sectional study enrolled adults (age ≥ 18 years) who underwent health examinations at the Qingdao Sixth People’s Hospital between June 2025 and September 2025.The exclusion criteria were as follows: (1) presence of other primary liver diseases, such as viral hepatitis, autoimmune liver disease, or primary hepatocellular carcinoma; (2) history of liver surgery; (3) missing key data, including body mass index (BMI), laboratory parameters, controlled attenuation parameter (CAP), or liver stiffness measurement (LSM) values; (4) pregnancy; (5)Individuals reporting an average daily alcohol intake exceeding 30 g/day for males and 20 g/day for females. This study was conducted in accordance with the Declaration of Helsinki. The study protocol was approved by the Ethics Review Committee of the Qingdao Sixth People’s Hospital (No.2025030602). The patients/participants provided written informed consent to participate in this study.
General data and laboratory tests
Demographic data (age, sex) and lifestyle information (smoking history, alcohol consumption, etc.) were collected using standardized questionnaires. Trained healthcare staff measured height, weight, and waist circumference (WC). BMI was calculated as weight (kg) divided by height squared (m²). Fasting blood samples were collected from the antecubital vein in the morning after at least 8 h of fasting. Biochemical parameters, including fasting plasma glucose (FPG), glycated hemoglobin (HbA1c), triglycerides (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT), and platelet (PLT) were measured using an automatic biochemical analyzer.
Liver transient elastography examination and hepatic steatosis grading
All the subjects were assessed with the FibroScan device to obtain the LSM and CAP values. The operating procedure strictly adhered to guideline recommendations: (a) participants were placed in a supine position with the right arm fully abducted to maximize the intercostal space; (b) the operator placed the probe perpendicular to the skin surface on the participant’s right intercostal space with coupling gel applied; (c) measurements were taken while the participant held their breath for 3–5 s during quiet respiration; and (d) each valid measurement required at least 10 valid readings, and the final result was considered reliable only if the interquartile range (IQR) to median ratio was less than 30%. The examination was performed by an experienced sonographer.
The degree of hepatic steatosis was graded based on the CAP value (in dB/m) as follows20: S0 CAP < 248 dB/m; S1 248 ≤ CAP < 268 dB/m༛S2 268 ≤ CAP < 280 dB/m༛S3 CAP ≥ 280 dB/m。.
Calculation of the important index
TyG index was calculated using the formula: TyG index = Ln [fasting TG (mg/dL) × fasting FPG (mg/dL) / 2].
TyG-BMI index was subsequently calculated as: TyG-BMI index = TyG index × BMI (kg/m²).
FLI index was subsequently calculated as: (e0.953×ln(TG)+0.139×BMI+0.718×ln(GGT)+0.053×WC−15.745) ÷ (1 + e0.953×ln(TG)+0.139×BMI+0.718×ln(GGT)+0.053×WC−15.745)×100.
For the calculation of TyG-BMI, TG and FPG values were converted from mmol/L to mg/dL using the following equations: TG (mg/dL) = TG (mmol/L) × 88.5; FPG (mg/dL) = FPG (mmol/L) × 18.
Definition of MAFLD
MAFLD was diagnosed based on the presence of hepatic steatosis (determined by CAP ≥ 248 dB/m) plus at least one of the following three criteria21: (1) overweight/obesity (BMI ≥ 23 kg/m²); (2) type 2 diabetes mellitus (T2DM); or (3) evidence of metabolic dysregulation. Metabolic dysregulation was defined as the presence of at least two of the following components: central obesity (waist circumference ≥ 90 cm in men and ≥ 80 cm in women), elevated blood pressure (systolic ≥ 130 mmHg or diastolic ≥ 85 mmHg) or antihypertensive treatment, hypertriglyceridemia (serum triglycerides ≥ 150 mg/dL [1.7 mmol/L]) or lipid-lowering treatment, reduced high-density lipoprotein (HDL) cholesterol (< 40 mg/dL [1.0 mmol/L] in men or < 50 mg/dL [1.3 mmol/L] in women) or lipid-lowering treatment, prediabetes (fasting plasma glucose 5.6–6.9 mmol/L, 2-hour post-load glucose 7.8–11.0 mmol/L, or glycated hemoglobin [HbA1c] 5.7%-6.4%), an insulin resistance score ≥ 2.5, or elevated high-sensitivity C-reactive protein (hs-CRP) > 2 mg/L.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics (version 26.0). The software is available at: https://www.ibm.com/products/spss-statistics. Continuous variables with a normal distribution were presented as mean±standard deviation and compared between groups using the Student’s t-test. Non-normally distributed data were presented as median (interquartile range) and compared using the Mann-Whitney U test or the Kruskal-Wallis H test. Categorical variables were expressed as numbers (percentages) and compared using the chi-square test. The association between the TyG-BMI index and the risk of MAFLD was assessed using multivariable logistic regression, calculating odds ratios (ORs) and 95% confidence intervals (CIs). The diagnostic performance of the TyG-BMI index for MAFLD was evaluated by the area under the receiver operating characteristic (ROC) curve and compared with other indices. One-way analysis of variance was employed to compare TyG-BMI index levels across different MAFLD severity grades. A trend test was further conducted to assess whether the TyG-BMI index exhibited a significant linear trend across these ordinal severity categories. Spearman’s rank correlation analysis was used to assess the correlations between the TyG-BMI index and CAP values, as well as LSM. Multiple linear regression was employed to examine the independent association between the TyG-BMI index and LSM. All tests were two-sided, and a P-value < 0.05 was considered statistically significant.
Results
Baseline characteristics of the study population (N = 1457)
A total of 1,457 participants were included in this study, comprising 675 individuals in the MAFLD group and 782 in the non-MAFLD group. The comparative baseline characteristics of the two groups are detailed in Table 1. The mean age of participants in the MAFLD group was 52.20 ± 11.79 years, which was significantly higher than that of the non-MAFLD group (49.93 ± 12.00 years, P < 0.001). A higher proportion of males was observed in the MAFLD group, although the difference was of borderline significance (P = 0.05). The MAFLD group had a higher proportion of individuals with a history of smoking. Furthermore, the prevalence of hypertension and diabetes was significantly greater in the MAFLD group. Anthropometric measurements indicated that body weight and body mass index (BMI) were significantly higher in the MAFLD group compared to the non-MAFLD group (P < 0.01). However, no statistically significant difference was observed in height between the two groups (P = 0.620). In terms of liver function and lipid profiles, the MAFLD group exhibited significantly higher levels of ALT, AST, and GGT compared to the non-MAFLD group (all P < 0.001). Lipid metabolism was also more dysregulated in the MAFLD group, with elevated TG, TC, and LDL-C, alongside reduced HDL-C (all P < 0.01). In terms of liver steatosis and fibrosis assessment, the MAFLD group demonstrated significantly higher CAP values (296.51 ± 36.46 dB/m vs. 208.42 ± 25.50 dB/m, P < 0.001) and LSM values (5.28 ± 1.84 kPa vs. 4.61 ± 1.73 kPa, P < 0.001) compared to the non-MAFLD group. These findings are consistent with the characteristic metabolic disturbances associated with MAFLD. The TyG-BMI index and FLI was calculated according to the formula. The level of the TyG-BMI index was significantly higher in the MAFLD group (242.02 ± 41.04) compared to the non-MAFLD group (198.44 ± 33.99, P < 0.001).
Association between the TyG-BMI Index and MAFLD Status
Furthermore, a multivariable logistic regression analysis was performed to investigate the association between the TyG-BMI index and MAFLD status after adjusting for age and sex. The results, presented in Table 2, demonstrated that after additional adjustment for BMI, each 1-unit increase in the TyG-BMI index was independently associated with a 3.4% increase in the risk of MAFLD (aOR = 1.034, 95% CI: 1.030–1.038, P < 0.001).
To further illustrate the association between the TyG-BMI index and MAFLD risk, participants were divided into four groups (Q1-Q4) based on the quartiles of the TyG-BMI index. The TyG-BMI index quartiles were defined as follows: Q1 (< 188.07), Q2 (188.07–213.16), Q3 (213.16–244.12), and Q4 (> 244.12). Using Q1 as the reference, a multivariate logistic regression analysis was conducted, adjusting for age and sex. As shown in Table 3, the risk of MAFLD increased significantly with ascending TyG-BMI index quartiles (P < 0.001). Compared to the Q1 group, the adjusted ORs (95% CI) for MAFLD in the Q2, Q3, and Q4 groups were 3.04 (2.08–4.45), 9.56 (6.54–13.87), and 28.56 (18.99–42.95), respectively. This indicates that individuals in the highest quartile of TyG-BMI index had a 28.56-fold higher risk of MAFLD compared to those in the lowest quartile, revealing a pronounced dose-response relationship. This strong association is consistent with findings from other studies, reinforcing the value of the TyG-BMI index .
Diagnostic performance of the TyG-BMI Index for MAFLD
The diagnostic performance of the TyG-BMI index for diagnosing MAFLD was evaluated using the ROC curve(Fig. 1A). The results showed an AUC of 0.806 (95% CI: 0.784–0.828). The optimal cut-off value of TyG-BMI index, determined by the maximum Youden index, was 214.54, corresponding to a sensitivity of 73.7% and a specificity of 74.5%. Furthermore, we compared the diagnostic performance of the TyG-BMI index with the FLI for diagnosing MAFLD. The optimal cut-off value of FLI, determined by the maximum Youden index, was 26.54, corresponding to a sensitivity of 73.4% and a specificity of 73.9%. The results indicated that the TyG-BMI index and FLI had comparable diagnostic efficacy(Fig. 1B). Our findings suggest that the TyG-BMI index demonstrated good discrimination for MAFLD, which aligns with research highlighting its utility in risk stratification.
Comparison of Diagnostic Performance between TyG-BMI Index and FLI for MAFLD. (A) Diagnostic performance of TyG-BMI index for MAFLD diagnosis. (B) Comparison of diagnostic performance between TyG-BMI index and FLI for MAFLD diagnosis.
Subgroup Analysis and Interaction Tests
Subgroup analyses were conducted by age and gender to explore the stability of the TyG-BMI index’s diagnostic efficacy for MAFLD across different populations. The subgroups included 350 males and 1,107 females, 1,114 young individuals (18–60 years), and 343 elderly individuals (> 60 years). ROC curves were plotted for each subgroup. ROC curves were plotted for each subgroup, and the corresponding AUC values of TyG-BMI were as follows: 0.801 for males (Fig. 2A), 0.809 for females (Fig. 2B), 0.834 for young individuals (Fig. 2C), and 0.700 for elderly individuals (Fig. 2D). The results demonstrated that the association of the TyG-BMI index with MAFLD was consistent across genders(Fig. 2A and B). However, its diagnostic performance was generally lower in the elderly subgroup(Fig. 2C and D). Additionally, in comparison with the FLI, the TyG-BMI index showed more satisfactory diagnostic efficacy in the female population.
Diagnostic Performance of TyG-BMI Index for MAFLD across Different Age and Sex Subgroups. (A) diagnostic performance of TyG-BMI and FLI in males. (B) Diagnostic performance of TyG-BMI and FLI in females. (C) diagnostic performance of TyG-BMI and FLI in young population. (D) diagnostic performance of TyG-BMI and FLI in elderly population.
Interaction analysis using multivariate logistic regression was performed to test the potential effect modification by age and gender on the association between the TyG-BMI index and MAFLD. As shown in Table 4, the interaction term between the TyG-BMI index and age was statistically significant (OR = 0.984, 95% CI: 0.976–0.993). This indicates that for each 1-unit increase in the TyG-BMI index, the increase in MAFLD risk was 1.6% smaller in the elderly (> 60 years) compared to the young population (18–60 years), meaning the diagnostic effect of the TyG-BMI index for MAFLD was stronger in the young population than in the elderly. In contrast, the interaction term between the TyG-BMI index and gender was not significant (OR = 0.999, 95% CI: 0.989–1.008), suggesting that the strength of its association with MAFLD was consistent between males and females. In summary, a significant age modification effect was observed in the association between the TyG-BMI index and MAFLD risk (P < 0.001), but no significant gender modification effect was found (P = 0.794).
Correlation between the TyG-BMI Index and the degree of hepatic steatosis
To investigate the correlation between the TyG-BMI index and hepatic steatosis, participants were categorized based on the degree of hepatic steatosis into S0(n = 782), S1 (n = 178), S2 (n = 97), and S3 (n = 403) groups for subsequent analysis. One-way analysis of variance (ANOVA)revealed a significant difference in TyG-BMI index levels across the severity groups(Fig. 3). A trend testfurther confirmed that TyG-BMI index levels increased significantly with advancing hepatic steatosis (P for trend < 0.001). Post-hoc pairwise comparisons showed that the differences between all groups were statistically significant (all P < 0.001).
Distribution of TyG-BMI Index across different degree of hepatic steatosis.
To quantify the impact of the TyG-BMI index on the degree of hepatic steatosis, an ordinal logistic regression analysis was performed. The results, shown in Table 5, indicated that after adjusting for age and sex, each 1-unit increase in the TyG-BMI index was associated with a 3.3% increase in the risk of progressing to a higher severity grade of hepatic steatosis (aOR = 1.033, 95% CI: 1.029–1.036, P < 0.001).
To further explore the correlation between the TyG-BMI index and MAFLD progression, a Spearman correlation analysis was conducted based on CAP values and the TyG-BMI index. The results demonstrated a significant positive correlation between the TyG-BMI index and CAP values (r = 0.601, P < 0.001), as shown in Fig. 4. This strong correlation with a quantitative measure of liver fat content underscores the index’s relevance to disease severity.
Correlation between TyG-BMI Index and CAP Values.
Correlation between TyG-BMI Index and LSM Values
A Spearman correlation analysis was performed to examine the relationship between the TyG-BMI index and liver fibrosis indicators. The results showed a significant positive correlation between the TyG-BMI index and LSM (Spearman’s rho = 0.255, P < 0.001), as presented in Fig. 5.
Correlation between TyG-BMI Index and LSM Values.
To further assess the independent effect of the TyG-BMI index on LSM, a multiple linear regression model was established, adjusting for age and sex. The results, shown in Table 6, indicated that the TyG-BMI index was significantly and positively associated with LSM (β = 0.011, 95% CI: 0.008–0.013, P < 0.001). Age was also an independent factor influencing LSM (β = 0.013, P = 0.001), while the association between gender and LSM did not reach statistical significance (β= -0.135, P = 0.216). This regression analysis suggests that the TyG-BMI index is significantly associated with liver fibrosis severity, with higher values indicating a greater risk of fibrosis.
Discussion
This cross-sectional study systematically investigated the association between the TyG-BMI and the risk, severity, and liver fibrosis of MAFLD. The main findings indicate that the TyG-BMI index is not only an independent risk factor for MAFLD prevalence but also significantly correlates with the degree of hepatic steatosis and liver fibrosis. These results highlight the substantial potential of the TyG-BMI index for early screening and risk stratification of MAFLD in clinical practice.
In this study, MAFLD was diagnosed using CAP ≥ 248 dB/m, a well-validated threshold for hepatic steatosis20. The application of CAP and LSM provided quantitative assessment of both steatosis severity and fibrosis. Our study found a significant positive correlation between the TyG-BMI index and the risk of MAFLD, with each 1-unit increase associated with a 3.4% elevation in disease risk (aOR = 1.034). Compared to the Q1 group, the adjusted ORs (95% CI) for MAFLD in the Q2, Q3, and Q4 groups were 3.04 (2.08–4.45), 9.56 (6.54–13.87), and 28.56 (18.99–42.95), respectively. The high OR of 28.56 for the highest versus lowest TyG-BMI quartile must be interpreted with caution, as it may be influenced by selection bias from the single-center health check-up population which could inflate the observed association. The AUC for diagnosing MAFLD reached 0.806. This finding is highly consistent with several recent large-scale studies. or instance, research based on the National Health and Nutrition Examination Survey (NHANES) data also reported a high AUC of 0.886 for TyG-BMI in predicting MAFLD22. Another cohort study in a Chinese population confirmed the good diagnostic performance of the TyG-BMI index for MAFLD, with an AUC of 0.8523. The excellent predictive efficacy likely stems from its pathophysiological basis: the TyG index core reflects IR, while BMI quantifies overall adiposity, a major risk factor for MAFLD24,25. Therefore, the TyG-BMI index simultaneously captures two core pathways—metabolic dysregulation and obesity burden—providing a more comprehensive risk assessment than any single component. It is notable that the AUC value of TyG-BMI in this study (0.806) is lower than that in most previous studies. We consider this to be due to the heterogeneity of the study population and the complexity of the real clinical scenario. Nevertheless, the performance of TyG-BMI in this study was comparable to that of the established FLI (AUC 0.801), supporting the robustness of its clinical application.
Our results demonstrated that the diagnostic ability of the TyG-BMI index for MAFLD was significantly stronger in the younger population (≤ 60 years) compared to the elderly (> 60 years) (AUC 0.834 vs. 0.700). This discrepancy may be attributed to the more heterogeneous etiology of MAFLD in the elderly, which involves mechanisms beyond metabolic factors, such as aging-related inflammation, sarcopenia, and gut microbiota changes, potentially diminishing the explanatory power of the TyG-BMI index centered on IR26,27,28. This finding underscores that future development of non-invasive diagnostic models for MAFLD should consider introducing age-specific cut-offs or algorithms to optimize performance across all age groups. The stronger association observed in younger individuals aligns with findings by Hu and Li who reported more pronounced associations in younger or non-obese individuals with relatively normal metabolic dysregulation, suggesting a unique value of TyG-BMI in identifying early-stage, non-typical MAFLD populations15,23,29. Nevertheless, the association between TyG-BMI and MAFLD in our study showed no difference between genders, enhancing its potential as a universal screening tool.
Furthermore, our results indicated a significant positive correlation between the TyG-BMI index and the degree of hepatic steatosis in MAFLD (OR = 1.033). This suggests that TyG-BMI can not only identify the presence of the disease but also reflect the severity of fat accumulation within hepatocytes. This finding shifts the application of the index from mere diagnosis to severity stratification, holding significant clinical importance. The mechanism may involve enhanced hepatic de novo lipogenesis and impaired fatty acid oxidation due to worsening IR and obesity, leading to greater triglyceride deposition in liver cells30,31. A study involving 9,627 MAFLD patients similarly confirmed that the BMI-TyG index was an independent risk factor for the severity of steatosis32. In comparison, our study extended these findings by performing subgroup analyses to define the predictive performance of TyG‑BMI across different populations and by examining its correlation with LSM, thereby providing a more comprehensive assessment of its clinical utility.
Additionally, this study established an independent correlation between the TyG-BMI index and the degree of liver fibrosis (β = 0.011). Liver fibrosis is the most critical determinant of long-term prognosis in MAFLD patients. This study provides robust evidence that TyG-BMI has the potential to predict the prognosis of MAFLD patients33,34. The mechanisms linking TyG-BMI to fibrosis may involve chronic inflammation and oxidative stress triggered by persistent IR and obesity, which activate hepatic stellate cells, promote extracellular matrix deposition, and ultimately lead to fibrosis6,35。Using multiple linear regression models adjusted for age and sex, this study quantified the independent effect of TyG-BMI on LSM, providing epidemiological evidence from a large population supporting the core Metabolism-Inflammation-Fibrosis pathway in MAFLD progression. Future research should directly quantify inflammatory and oxidative stress markers to determine whether these factors mediate the observed association between TyG-BMI and liver fibrosis.
The strengths of this study include a relatively large sample size (N = 1457). The association between TyG-BMI and the severity of MAFLD and liver fibrosis was systematically evaluated, providing a more comprehensive understanding of the disease spectrum. Through detailed subgroup and trend analysis, we found that the diagnostic ability of TyG-BMI is significantly stronger in the young population. This finding is of great significance for formulating targeted screening strategies. Additionally, the comparative analysis demonstrating comparable diagnostic performance between TyG-BMI and the established FLI index further supports the clinical utility of this index. However, this study also has several limitations. First, the cross-sectional design cannot infer a causal relationship between TyG-BMI and MAFLD; future prospective cohort studies are needed for verification. Second, although the multivariate models adjusted for age and sex, they did not account for other potential confounders such as smoking status, alcohol intake, medication use, dietary patterns, and physical activity, which may have resulted in residual confounding. Third, the diagnosis of MAFLD and its steatosis severity relied on non-invasive imaging rather than the gold standard of liver biopsy. Although the former is widely validated and more ethical, some misclassification bias might remain. The TyG-BMI index shares conceptual overlap with the MAFLD diagnostic criteria, which may introduce inherent collinearity, and future studies could explore alternative modeling strategies to account for this. Finally, as a single-center study, the samples were mainly from the Chinese population. The results may be affected by population characteristics and geographical restrictions. Caution should be exercised when extending it to other ethnic groups, community screening populations, or different medical resource environments. This study provides evidence to support for the application of TyG-BMI in MAFLD screening. However, before wide promotion, ethical issues such as the risk of overdiagnosis, resource accessibility and patient preferences need to be considered. Future research should focus on multi-center prospective studies to validate our findings in diverse populations, establish age-specific TyG-BMI cut-off values, and assess the cost-effectiveness of risk-based screening strategies.
In conclusion, this study confirms that the TyG-BMI index is independently associated with the prevalence, severity, and fibrosis risk of MAFLD, demonstrating significant clinical application value for early screening and risk stratification. Future research should focus on large-scale prospective cohorts to validate its predictive ability and explore its applicability across diverse populations and its potential as a dynamic monitoring indicator for intervention effects.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Lim, G. E. H. et al. An Observational Data Meta-analysis on the Differences in Prevalence and Risk Factors Between MAFLD vs NAFLD [J]. Clinical gastroenterology and hepatology: the official clinical practice. J. Am. Gastroenterological Association. 21 (3), 619–629e617 (2023).
Miao, L. et al. Current status and future trends of the global burden of MASLD [J]. Trends Endocrinol. Metab. 35 (8), 697–707 (2024).
YOUNOSSI Z M. Non-alcoholic fatty liver disease - A global public health perspective [J]. J. Hepatol. 70 (3), 531–544 (2019).
Huang, D. Q., El-serag, H. B. & Loomba, R. Global epidemiology of NAFLD-related HCC: trends, predictions, risk factors and prevention [J]. Nat. reviews Gastroenterol. Hepatol. 18 (4), 223–238 (2021).
Powell, E. E., Wong, V. W. & Rinella, M. Non-alcoholic fatty liver disease [J]. Lancet (London England). 397 (10290), 2212–2224 (2021).
Khan, R. S. et al. Modulation of Insulin Resistance in Nonalcoholic Fatty Liver Disease [J]. Hepatol. (Baltimore Md). 70 (2), 711–724 (2019).
Zhang, C. H. et al. Molecular mechanisms of hepatic insulin resistance in nonalcoholic fatty liver disease and potential treatment strategies [J]. Pharmacol. Res. 159, 104984 (2020).
Jeppesen, J. et al. Insulin resistance, the metabolic syndrome, and risk of incident cardiovascular disease: a population-based study [J]. J. Am. Coll. Cardiol. 49 (21), 2112–2119 (2007).
Defronzo, R. A., Tobin, J. D. & Andres, R. Glucose clamp technique: a method for quantifying insulin secretion and resistance [J]. Am. J. Physiol. 237 (3), E214–223 (1979).
sánchez-garcía, A. et al. Diagnostic Accuracy of the Triglyceride and Glucose Index for Insulin Resistance: A Systematic Review [J]. International journal of endocrinology, 2020: 4678526. (2020).
Tahapary, D. L. et al. Challenges in the diagnosis of insulin resistance: Focusing on the role of HOMA-IR and Tryglyceride/glucose index [J]. Diabetes metabolic syndrome. 16 (8), 102581 (2022).
Er, L. K. et al. Triglyceride Glucose-Body Mass Index Is a Simple and Clinically Useful Surrogate Marker for Insulin Resistance in Nondiabetic Individuals [J]. PloS one. 11 (3), e0149731 (2016).
Li, H. et al. Relationship Between Six Insulin Resistance Surrogates and Nonalcoholic Fatty Liver Disease Among Older Adults: A Cross-Sectional Study [J]. Diabetes metabolic syndrome obesity: targets therapy. 16, 1685–1696 (2023).
Zhang, S. et al. Triglyceride glucose-body mass index is effective in identifying nonalcoholic fatty liver disease in nonobese subjects [J]. Medicine 96 (22), e7041 (2017).
Hu, H. et al. The triglyceride glucose-body mass index: a non-invasive index that identifies non-alcoholic fatty liver disease in the general Japanese population [J]. J. translational Med. 20 (1), 398 (2022).
Sheng, G. et al. The usefulness of obesity and lipid-related indices to predict the presence of Non-alcoholic fatty liver disease [J]. Lipids Health Dis. 20 (1), 134 (2021).
Kim, A. H. Son d h, Lee y j. Modified triglyceride-glucose index indices are reliable markers for predicting risk of metabolic dysfunction-associated fatty liver disease: a cross-sectional study [j]. Front. Endocrinol. 14, 1308265 (2023).
Boushehri, Y. G. et al. Association of triglyceride glucose-body mass index (tyg-bmi) with metabolic dysfunction-associated steatotic liver disease: a systematic review and meta-analysis [j]. Plos one. 20 (8), e0324483 (2025).
Bedogni, G. et al. The fatty liver index: a simple and accurate predictor of hepatic steatosis in the general population [j]. BMC Gastroenterol. 6, 33 (2006).
Karlas, T. et al. Individual patient data meta-analysis of controlled attenuation parameter (cap) technology for assessing steatosis [j]. J. Hepatol. 66 (5), 1022–1030 (2017).
Eslam, M. et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement [j]. J. Hepatol. 73 (1), 202–209 (2020).
Wang, R. et al. Usefulness of the triglyceride glucose-body mass index in evaluating nonalcoholic fatty liver disease: insights from a general population [j]. Lipids Health Dis. 20 (1), 77 (2021).
Li et al. Association between triglyceride glucose-body mass index and non-alcoholic fatty liver disease in the non-obese chinese population with normal blood lipid levels: a secondary analysis based on a prospective cohort study [j]. Lipids Health Dis. 19 (1), 229 (2020).
Zeng, Z. Y. et al. Association of triglyceride glucose index and its combination of obesity indices with prehypertension in lean individuals: a cross-sectional study of chinese adults [j]. Journal of clinical hypertension (greenwich). 22(6), 1025–1032 (2020).
Sinn, D. H. & park Gwak, G. Y. Ultrasonographically detected non-alcoholic fatty liver disease is an independent predictor for identifying patients with insulin resistance in non-obese, non-diabetic middle-aged asian adults [j]. Am. J. Gastroenterol. 107 (4), 561–567 (2012).
Torres, D. M., Williams, C. D. & Harrison, S. A. Features, diagnosis, and treatment of nonalcoholic fatty liver disease [j]. Clin. Gastroenterol. hepatology: official Clin. Pract. J. Am. gastroenterological association. 10 (8), 837–858 (2012).
Jiang, W. et al. Dysbiosis gut microbiota associated with inflammation and impaired mucosal immune function in intestine of humans with non-alcoholic fatty liver disease [j]. Sci. Rep. 5, 8096 (2015).
Liu, C. H. et al. Sarcopenia and masld: novel insights and the future [j] (Nature reviews endocrinology, 2025).
Han & Zhang, K. Triglycerides mediate the influence of body mass index on non-alcoholic fatty liver disease in a non-obese chinese population with normal low-density lipoprotein cholesterol levels [j]. Obes. Facts. 17 (2), 191–200 (2024).
Li, M. et al. Trends in insulin resistance: insights into mechanisms and therapeutic strategy [j]. Signal. Transduct. Target. therapy. 7 (1), 216 (2022).
Ameer, F. et al. De novo lipogenesis in health and disease [j]. Metab. Clin. Exp. 63 (7), 895–902 (2014).
Huang, X. et al. Tyg-bmi as a superior predictor of mafld and pre-mafld in chinese adults: a cross-sectional study [j]. BMC Gastroenterol. 25 (1), 495 (2025).
Ochoa-allemant, P. et al. Adverse liver outcomes, cardiovascular events, and mortality in steatotic liver disease [j]. Jama Intern. Med. 185 (8), 986–995 (2025).
Ekstedt, M., Hagström, H. & nasr p, et al. Fibrosis stage is the strongest predictor for disease-specific mortality in nafld after up to 33 years of follow-up [j]. Hepatology. 61(5), 1547–1554 (2015).
Kisseleva, T. & Brenner, D. Molecular and cellular mechanisms of liver fibrosis and its regression [J]. Nat. reviews Gastroenterol. Hepatol. 18 (3), 151–166 (2021).
Funding
Supported by Qingdao Key Medical and Health Discipline Project.
Author information
Authors and Affiliations
Contributions
Qi designed this theme and statistical methodology, as well as the analysis. Wang drafted and interpreted this study. Han and Liu critically reviewed the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study protocol was approved by the Ethics Review Committee of the Qingdao Sixth People’s Hospital (No.2025030602). The patients/participants provided written informed consent to participate in this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, X., Han, X., Liu, J. et al. The association of TyG-BMI with MAFLD and liver fibrosis: a cross-sectional study. Sci Rep 16, 10590 (2026). https://doi.org/10.1038/s41598-026-46378-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-46378-0