Abstract
De novo metastatic nasopharyngeal carcinoma (dmNPC) is a heterogeneous disease that exhibits variable failure patterns after first-line immunochemotherapy, complicating the decision-making for subsequent locoregional radiotherapy (LRRT). This retrospective real-world analysis enrolled 398 dmNPC patients treated with first-line immunochemotherapy with or without subsequent LRRT. We developed and validated a clinically applicable two-step risk stratification model that categorizes patients into three phenotypic subgroups based on different failure patterns. Durable responders were defined as patients likely to achieve long-term remission with immunotherapy maintenance alone. Partial responders were prone to experience isolated locoregional progression, while resistant patients had a high risk of developing distant progression, regardless of locoregional control. After inverse probability of treatment weighting adjustment, LRRT significantly improved 2-year progression-free survival (PFS) in partial responders (69.8% vs. 45.1%, HR = 0.45, P < 0.001), but conferred no benefit in durable responders (81.4% vs. 73.4%, P = 0.28) or resistant patients (12.4% vs. 8.6%, P = 0.33). Our findings suggest that only patients prone to isolated locoregional progression may derive survival benefit from LRRT, while others could safely omit LRRT without compromising prognosis. A failure-pattern–based strategy could personalize LRRT decisions and guide future clinical trial design in the immunotherapy era.
Similar content being viewed by others
Introduction
Nasopharyngeal carcinoma (NPC) is a head and neck cancer highly endemic in Southeast Asia, with 120,416 new cases and 73,476 cancer-related deaths globally, according to GLOBOCAN 20221. Despite the generally favorable prognosis for patients with early or locoregionally advanced NPC in the intensity modulated radiotherapy (IMRT) era, approximately 4%–10% of patients are diagnosed with de novo metastatic NPC (dmNPC), which has limited therapeutic options and inferior survival outcomes2,3. In recent years, several clinical trials have collectively demonstrated that chemotherapy combined with anti-programmed cell death protein 1 (PD-1) immunotherapy significantly improves the prognosis of recurrent or metastatic NPC (RM-NPC)4,5,6. Therefore, immunochemotherapy has been recommended as a standard therapeutic paradigm by the European Society for Medical Oncology (ESMO), Chinese Society of Clinical Oncology (CSCO), and National Comprehensive Cancer Network (NCCN) guidelines7,8,9, and widely adopted as first-line treatment for dmNPC. However, the need for locoregional radiotherapy (LRRT) to the primary tumor following immunochemotherapy, and the identification of optimal candidates, remains unresolved.
The heterogeneous nature of dmNPC makes it impractical to establish a universal treatment strategy applicable to all10,11. Although robust evidence from large-scale phase III randomized controlled trials remains scarce, accumulating retrospective studies suggest that subsequent LRRT may confer a survival benefit for patients with dmNPC, particularly for a subgroup characterized by low-risk features such as lower Epstein-Barr virus (EBV) DNA levels, no liver involvement, fewer metastatic lesions, and satisfactory treatment response12,13,14,15,16. In the pre-immunotherapy era, a phase III clinical trial reported that subsequent LRRT improved the 2-year overall survival (OS) rate from 54.5% to 76.4% in chemotherapy responders17. The proportion of cases with locoregional recurrence was 15.9% (10/63) in the LRRT arm vs 69.8% (44/63) in the chemotherapy-alone arm. Presumably, the improvement in survival was primarily attributable to the prevention of death due to locoregional progression, as nearly 20% of deaths in patients with advanced NPC were ascribed to the complications of uncontrolled locoregional disease, such as embolism, hemorrhage, and aspiration18. However, 58.7% (37/63) of responders experienced treatment failure despite LRRT, with most cases (91.9%, 34/37) manifesting as distant progression. For patients who developed distant progression, the clinical benefit of prior LRRT was substantially compromised. Meanwhile, the integration of immunotherapy has improved overall treatment efficacy remarkably, rendering the necessity for LRRT and the identification of optimal candidates more complex and intriguing. Despite the immature data, some published small-sample retrospective studies suggest that low-risk patients are still more likely to benefit from LRRT in the immunotherapy era, particularly those with fewer metastatic lesions, rapid EBV DNA clearance, and favorable responses19,20,21,22. However, whether all low-risk patients should receive LRRT remains unexplored. According to the definitive survival analysis of the JUPITER-02 trial, immunotherapy maintenance alone was adequate to achieve a 2-year progression-free survival (PFS) rate of 44.8% and a 2-year OS rate of 78.0%23, which is comparable to the historical survival data of patients who received LRRT in the pre-immunotherapy era (2-year OS: 76.4%)17. Immediate LRRT following immunochemotherapy may not yield additional survival benefits for patients demonstrating a durable response to immunotherapy. Instead, it may impair quality of life due to radiation-related complications and restrict subsequent treatment options.
Therefore, we conducted this retrospective study to analyze the data of patients with dmNPC who received first-line immunochemotherapy, aiming to develop a novel scoring model to identify the failure patterns and optimal candidates for subsequent LRRT. We hypothesized that for patients who achieve long-term remission of both the primary and metastatic lesions following immunochemotherapy, or those who experience distant progression, LRRT may not confer a survival benefit and may instead increase treatment-related toxicity. Patients with well-controlled distant metastases and progression confined to locoregional sites may be the optimal candidates for LRRT.
Results
Study population
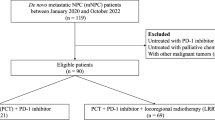
A total of 398 patients treated with first-line immunochemotherapy from Oct 2018 to Dec 2023, without (non-LRRT cohort, n = 178) or with (LRRT cohort, n = 220) subsequent LRRT, were included. The baseline characteristics are shown in Table 1. The median age of the included patients was 47 (IQR: 37-55) years, with males comprising 79.4% of the cohort. Most patients had T3-4 (91.0%) and/or N2-3 (89.4%) disease. Bone was the most common metastatic site (70.9%), followed by the liver (36.2%), lungs (23.9%), and distant lymph nodes (22.6%). Overall, compared with the patients in the non-LRRT cohort, patients that received subsequent LRRT had lower baseline plasma EBV DNA ( < 95000 copies/mL, 91.4% vs 74.2%, P < 0.001) and serum lactate dehydrogenase (LDH) levels (<500 U/L, 94.1% vs 82.0%, P < 0.001), fewer metastatic organs (only one, 66.8% vs 51.7%, P = 0.002) or lesions (three or fewer, 52.7% vs 41.6%, P = 0.027), better response evaluation results (CR/PR, 90.9% vs 82.6%, P = 0.012), and undetectable EBV DNA (85.5% vs 68.0%, P < 0.001) after immunochemotherapy. Most patients (356, 89.4%) completed the full six cycles of first-line immunochemotherapy, whereas the remaining patients received at least four cycles. The detail of concurrent chemotherapy during LRRT was summarized in Supplementary Table 6. The median follow-up time was 32.0 (IQR, 19.7–41.2) months. In the non-LRRT cohort, the 1-, 2-, and 3-year PFS rates were 61.8%, 38.2%, and 29.8%, respectively, and the corresponding OS rates were 91.6%, 80.9% and 74.7%. In the LRRT cohort, the 1-, 2-, and 3-year PFS rates were 83.2%, 64.1%, and 57.3%, respectively, whereas the corresponding OS rates were 99.1%, 89.5%, and 85.5%.
Failure patterns of dmNPC with or without LRRT
The failure patterns in the non-LRRT and LRRT cohorts are shown in Fig. 1. During the follow-up period, disease progression occurred in 133 patients (74.7%) in the non-LRRT cohort and 100 patients (45.5%) in the LRRT cohort. Of the 178 patients who did not receive LRRT or any treatment for distant metastases, 79 (44.4%) experienced distant metastasis progression, 54 (30.3%) had isolated locoregional progression, and 45 (23.2%) achieved durable disease control. A significantly higher proportion of patients in the non-LRRT cohort experienced failure at the primary site than did those in the LRRT cohort (30.3% vs. 4.1%, P < 0.001). However, the proportion of patients experiencing distant failure was similar between the two groups (44.4% vs. 41.4%, P = 0.55). Most failure events occurred within two years of completion of first-line treatment (non-LRRT cohort, 82.7%, 110/133; LRRT cohort, 79.0%, 79/100). Detailed characteristics of patients with three distinct failure patterns are shown in Supplementary Table 1. Overall, compared with patients that developed locoregional failure, those developed distant metastasis progression exhibited shorter PFS (median 9.7 months), unsatisfactory response to immunochemotherapy (SD/PD, 29.1%, P = 0.004), and poorer clinical characteristics, such as the baseline LDH level (≥500 U/L, 29.1%, P = 0.002) and metastatic lesions (>3, 78.5%, P < 0.001).
The distribution of failure patterns in patients (A) without and (B) with locoregional radiotherapy (LRRT) following first-line immunochemotherapy are shown as pie charts. Local failure was defined as relapse confined to the nasopharynx, regional failure referred to recurrence in cervical lymph nodes, and local and regional failure indicated simultaneous recurrence at both sites. The cumulative incidence of failure events over time in (C) non-LRRT and (D) LRRT cohorts. Kaplan–Meier survival curves for (E–G) progression-free survival and (H–J) overall survival after matching between patients with and without LRRT across the three risk groups. E, H Resistant patients, (F, I) partial responders, and (G, J) durable responders. P values were calculated through log-rank tests. Data are presented as weighted proportions rather than absolute numbers. dmNPC de novo metastatic nasopharyngeal carcinoma, LRRT locoregional radiotherapy.
Development of the risk stratification model
We developed a two-step scoring model in the non-LRRT cohort to identify patients predisposed to three distinct failure patterns. The detailed model construction procedure is presented in Supplementary A1. In the first step, the goal was to distinguish patients at high risk of distant progression. Four independent predictors were identified through LASSO and multivariate Cox regression (Table 2-A). These variables were integrated into Scoring Model I (Table 3), in which patients with a total score of two or higher were defined as resistant patients who were predisposed to distant progression, regardless of locoregional control. In the second step, we focused on patients without distant progression, aiming to differentiate those with isolated locoregional progression from those with durable disease control. Two independent predictors were identified (Table 2-B) and were used to construct Scoring Model II (Table 3), in which patients with a score of two were considered durable responders capable of achieving long-term disease control even without LRRT. Finally, the remaining patients were classified as partial responders prone to isolated locoregional progression. Based on the two-step scoring model, patients could be stratified into three clinically relevant risk groups with distinct failure patterns. Resistant patients were defined as those with a high risk of developing distant progression, regardless of locoregional control. Partial responders referred to patients who were prone to experience isolated locoregional progression. Durable responders were patients likely to achieve long-term remission with immunotherapy maintenance alone.
Validation of the risk stratification model
The two-step scoring model was applied to the entire patient cohort (n = 398) for validation. Detailed procedure of validation is presented in Supplementary A2. The application of Scoring Models I and II recognized 101 (25.4%) resistant patients predisposed to distant progression, 94 (23.6%) durable responders likely to achieve long-term disease control, and 203 (51.0%) partial responders vulnerable to isolated locoregional progression. To compare the survival benefit of LRRT within each subgroup, IPTW was applied to balance baseline covariates (Supplementary Table 5). The unadjusted survival analyses are provided in Supplementary Fig. 3. After IPTW adjustment, LRRT did not confer a survival advantage in resistant patients (2-year PFS, 12.4% vs 8.6%, HR = 0.78, 95% CI 0.47–1.29, P = 0.33) or durable responders (2-year PFS, 81.4% vs 73.4%, HR = 0.63, 95% CI 0.27–1.46, P = 0.28). However, in partial responders, LRRT significantly improved the survival outcome (2-year PFS: 69.8% vs 45.1%, HR = 0.45, 95% CI: 0.29–0.70, P < 0.001), highlighting this subgroup as the optimal candidates for LRRT following first-line immunochemotherapy (Fig. 1-E, F, G). A similar trend was observed in the OS analysis (Fig. 1-H, I, J). The analyses of LRFS and DMFS are presented in Supplementary Fig. 4.
Treatment adaption based on the two-step model in patients with dmNPC
The final clinical decision scoring model consisted of two steps, and the application process is illustrated in Fig. 2 and Table 3. For a newly diagnosed patient with dmNPC, guideline-recommended first-line immunochemotherapy is initially administered. Subsequently, evaluation based on the scoring model is conducted upon completion of first-line treatment. First, in Model I, patients with a score ≥ two are classified as resistant patients, indicating a need for proactive modification of the treatment strategy. Recommended options include switching to alternative chemotherapy regimens, adding metronomic chemotherapy, introducing anti-angiogenic or targeted agents, or considering investigational agents from clinical trials. Patients with a score of less than two in Model I are recommended to proceed to Model II for a second step of risk stratification. In Model II, patients with a score of two are considered durable responders, for whom immunotherapy maintenance alone is advised. Finally, the remaining patients are identified as partial responders vulnerable to isolated locoregional progression, and subsequent LRRT is strongly recommended.
After first-line immunochemotherapy, patients with a Model I score ≥ two are classified as resistant patients and modification of the treatment strategy is recommended. Those scoring < two proceed to Model II. In Model II, patients with a score of two are considered durable responders and advised to receive immunotherapy maintenance alone, while those with a score < two are identified as partial responders and LRRT is recommended. NPC nasopharyngeal carcinoma, LRRT locoregional radiotherapy, LDH lactate dehydrogenase, EBV Epstein-Barr virus, SD stable disease, PD progressive disease. Depth of response (DpR) was defined as the percentage of tumor shrinkage from the baseline tumor burden to the timepoint of assessment.
Discussion
To the best of our knowledge, this is the first study to explore failure patterns and identify optimal candidates for LRRT in patients with dmNPC in the immunotherapy era. We analyzed the largest cohort to date of patients with dmNPC who did not undergo LRRT following first-line immunochemotherapy, and comprehensively characterized their failure patterns during long-term immunotherapy maintenance. Based on the observed failure patterns in the non-LRRT cohort, we developed a clinically practical two-step scoring model to identify patients at high risk of isolated locoregional progression during immunotherapy maintenance. Finally, IPTW-adjusted survival analyses of the full cohort confirmed that this subgroup comprises the optimal candidates for LRRT following first-line immunochemotherapy, while others may omit LRRT without compromising prognosis.
For patients with dmNPC, systemic therapy, represented by chemotherapy, is the main treatment approach. However, chemotherapy alone results in poor long-term survival, with a 1-year PFS rate of only 20% and a median OS of just 29.1 months24. In the pre-immunotherapy era, a phase III trial demonstrated that combined-modality treatment, consisting of palliative chemotherapy followed by LRRT, provided durable locoregional control in dmNPC responding to initial chemotherapy17. However, dmNPC is a heterogeneous group varying by distribution, extent of metastatic burden, and response to PCT11,14. In the aforementioned trial, although the 2-year OS benefit exceeded 20% (76.4% vs 54.5%), most cases (58.7%) in the LRRT arm still relapsed. The locoregional progression rates in the chemotherapy-alone group and the LRRT group were 69.8% and 15.9%, respectively, while the rates of distant metastasis progression were 68.3% and 54.0%, respectively. Collectively, the therapeutic benefit of LRRT was primarily attributed to improved locoregional disease control without effective prevention of the progression of distant metastases. Moreover, one-quarter patients died of NPC despite LRRT within the first two years. These findings indicate that not all responding patients can benefit from LRRT. For these patients, LRRT not only failed to provide a survival benefit but also increased radiation-related toxicity.
Recently, three large multicenter phase III clinical trials demonstrated that chemotherapy combined with PD-1 inhibitors can significantly prolong survival in patients with metastatic NPC, with a median PFS up to 21.4 months and manageable overall toxicity4,5,6,23. Consequently, the latest version of the ESMO, NCCN, and CSCO guidelines collectively recommended chemotherapy plus PD-1 inhibitors as first-line treatment for metastatic NPC, with level I evidence7,8,9, marking the transition of metastatic NPC treatment into the immunotherapy era. The long-term follow-up results of the JUPITER-02 study showed that the 2-year OS for RM-NPC treated with immunochemotherapy (without LRRT) reached 78.0%, which is comparable to the historical survival data of patients who received LRRT in the pre-immunotherapy era (2-year OS: 76.4%)17,23. Therefore, the role of LRRT in patients with dmNPC remains to be further explored, especially in the immunotherapy era.
In clinical practice, we observed that some patients developed distant progression shortly after completing LRRT, while others achieved durable disease control with immunotherapy maintenance alone. For the former, the survival benefit from LRRT was minimal, whereas for the latter, delayed LRRT could preserve quality of life and alleviate financial burdens without jeopardizing disease control. These observations motivated us to explore the failure patterns and identify the optimal beneficiaries accordingly. Therefore, in this study we first explored the failure patterns in patients with dmNPC without LRRT under immunochemotherapy. We found that among the patients receiving long-term immunotherapy maintenance, 25.3% (45/178) achieved durable disease control, 44.4% (79/178) developed distant progression, and 30.3% (54/178) experienced isolated locoregional progression. Using our two-step scoring model, these three distinct failure patterns could be effectively identified. We propose that only patients prone to isolated locoregional progression represent the true beneficiaries of LRRT.
Several published studies have explored the clinical value of LRRT in the immunotherapy era. A recent retrospective study reported the survival outcomes of 364 patients with dmNPC who underwent LRRT after immunochemotherapy. The study stratified patients into three risk groups based on the number of metastatic lesions, liver metastasis status, and post-treatment EBV DNA levels. Compared with low- and intermediate-risk patients, high-risk patients showed a poor survival outcome (2-year PFS, 13.7%, versus 94.4% and 69.4%) despite receiving LRRT after immunochemotherapy25. Based on these findings, the authors further included 133 patients who did not undergo LRRT and developed a prognostic nomogram integrating serum LDH level, number of metastatic lesions, liver metastases status, and post-treatment EBV DNA levels. A tree-based risk stratification derived from the model classified all patients into two prognostic subgroups, and only low-risk patients could obtain survival benefit from LRRT19. Another study with a relatively short follow-up period (median:18 months) reported the survival outcomes of 95 newly diagnosed patients with dmNPC who received immunochemotherapy alone, and those of 178 patients who received immunochemotherapy combined with LRRT. The results showed that patients who received LRRT had improved OS and PFS generally, particularly those with oligo-metastases, EBV DNA below 20,200 copies/mL, and CR/PR after immunochemotherapy21. Taken together, these findings suggest that LRRT remains clinically meaningful in the immunotherapy era. Nevertheless, more refined risk stratification is necessary to avoid futile radiation exposure.
In this study, we applied a two-step risk stratification scoring model to stratify patients into three subgroups and explored the clinical value of LRRT within each group. We found that LRRT did not confer additional survival benefits in resistant patients at high risk of distant progression, nor in durable responders who were likely to achieve long-term remission with immunotherapy maintenance alone. Survival benefit from LRRT was observed only in partial responders, who were prone to isolated locoregional progression. However, while the median follow-up duration of 32 months is adequate for evaluating major survival outcomes, it may be insufficient to fully capture late-onset events. Longer follow-up may reveal evolving differences in outcomes between LRRT and non-LRRT groups. At the same time, the possibility of radiotherapy-related late toxicities may further undermine the clinical benefit of LRRT group, which strengthen the rationale for omitting LRRT in resistant patients and durable responders. Taken together, extended follow-up is needed to fully assess the long-term impact of LRRT on both survival outcomes and quality of life in different patient subgroups.
In the LRRT cohort of our study, 47.7% of patients received concurrent platinum-based chemotherapy during LRRT (Supplementary Table 7). The proportion of patients receiving concurrent chemotherapy was balanced across the three risk subgroups identified by our scoring model. Therefore, the use of concurrent chemotherapy is unlikely to confound our main findings, as no specific subgroup received disproportionate more concurrent chemotherapy that may bias survival outcomes. In this context, we can regard LRRT and concurrent chemotherapy as a combined intensification strategy. Our results showed that: when comparing LRRT and non-LRRT group, even with combined intensified treatment (LRRT plus chemotherapy), no additional survival benefit was observed in those resistant patients and durable responders. This finding reinforces the rationale of our risk-adapted intensification and de-intensification strategy. As a matter of fact, the benefit of concurrent chemotherapy during LRRT for patients with dmNPC remains controversial in the literature. Zheng et al.26 suggested that adding platinum-based chemotherapy during LRRT could further improve overall survival compared with LRRT alone after palliative chemotherapy in dmNPC. In contrast, Sun et al.27 reported no significant difference in survival between patients receiving LRRT alone versus concurrent chemoradiotherapy, and observed that administering more than four cycles of chemotherapy did not provide additional survival benefit. However, both studies were conducted in the pre-immunotherapy era. There is a lack of evidence regarding whether concurrent chemotherapy provides additional survival benefit in immunotherapy era.
Moreover, although LRRT group demonstrated a clear advantage in LRFS compared to non-LRRT group, no additional benefit of DMFS was observed, even some patients received concurrent chemotherapy during LRRT (Supplementary Fig. 4). It could be attributed to that all included patients had received at least four cycles of platinum-based chemotherapy prior to radiotherapy. Patients who were sensitive to chemotherapy may have derived maximal benefit of distant control, while the resistant patients were similarly unlikely to benefit from extra cycles. In addition, we performed a supplementary analysis to evaluate the impact of concurrent chemotherapy on DMFS in patients receiving LRRT (n = 220). The results showed that the addition of concurrent chemotherapy did not confer DMFS benefit, either in the overall cohort or within the three subgroups (Supplementary Fig. 5). Taken together, the marginal value of further chemotherapy intensification may be limited. This observation is consistent with findings from our previous study in the pre-immunotherapy era27.
Technically, our model was based on routinely available clinical parameters and employed a simple point-based scoring system, which was more user-friendly than conventional nomograms and may facilitate clinical decision-making in future settings, following further external and prospective validation. In addition to incorporating several classical prognostic indicators, the model also included the timepoint of EBV DNA clearance and the early assessment of locoregional response, highlighting the prognostic significance of rapid clearance of EBV DNA and the depth of response, align with previous findings20,28. Our study challenged the traditional paradigm, suggesting that low-risk patients may safely defer LRRT without compromising prognosis. The scoring model allows clinicians to recognize distinct patterns of treatment failure, thereby facilitating individualized adjustment of therapeutic strategies. For durable responders, LRRT may be safely omitted in favor of immunotherapy maintenance alone. For partial responders, immediate LRRT is recommended. For resistant patients, alternative treatment strategies, such as switching chemotherapy regimens, adopting metronomic chemotherapy, introducing anti-angiogenic or targeted agents, or participating in clinical trials are advised to avoid futile radiotherapy.
Despite the promising findings, several limitations of the study should be acknowledged. First, the retrospective nature of the study inherently carries the risk of selection bias. Unmeasured or unknown confounding factors cannot be fully balanced although we employed IPTW for adjustment, which could influence treatment selection and outcomes. Prospective studies are warranted to further validate our findings. Second, the proposed scoring model was developed and validated using data exclusively from single-center and external validation is lacking. Its generalizability to other institutions and larger populations remains uncertain. Accordingly, the clinical applicability of the model should be interpreted with caution. Third, the heterogeneity in treatment protocols such as variation in chemotherapy regimens and PD-1 inhibitors was not balanced due to the limited sample size, which may introduce additional variability in outcomes. Finally, all patients were from endemic regions of southern China, thus the applicability of our findings in non-endemic areas or Western populations should be cautious. Taken together, these limitations highlight the need for prospective, multicenter validation before the model can be adopted in real-world clinical decision-making.
In conclusion, this study established a two-step scoring model to stratify patients with de novo metastatic NPC receiving first-line immunochemotherapy into three subgroups with distinct failure patterns. These subgroups may warrant different treatment intensities to optimize disease control and minimize adverse effects. Our findings suggest that patients with durable remission or distant progression may not benefit from LRRT, while those with isolated locoregional progression could derive a survival benefit. While this study is retrospective in design, it provides the first pioneering effort that highlights the heterogeneity of treatment responses in dmNPC for guiding LRRT use. We propose that these response phenotypes be incorporated into prospective clinical trials investigating personalized strategies of treatment intensification and de-intensification in the immunotherapy era.
Methods
Patient selection
The study retrospectively enrolled consecutive patients newly diagnosed with dmNPC and treated at Sun Yat-sen University Cancer Center (SYSUCC) from October 2018 to December 2023. The inclusion criteria were as follows: (i) male or non-pregnant female aged 18 to 70 years, (ii) histologically confirmed treatment-naïve nasopharyngeal carcinoma with WHO II or III pathological subtypes, (iii) definite evidence of distant metastasis at the time of diagnosis, (iv) received at least four cycles of first-line platinum-based chemotherapy combined with anti-PD-1 immunotherapy, with or without subsequent LRRT, (v) had at least one measurable lesion according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. The exclusion criteria were as follows: (i) previous or synchronous secondary malignancies, (ii) any incomplete clinical data, such as missing imaging data or EBV DNA testing, (iii) a severe coexisting illness.
All the patients received routine pretreatment evaluations, including collection of demographic information, physical examinations, routine blood tests, serum biochemistry, and electronic nasopharyngoscopy. Imaging evaluations included magnetic resonance imaging (MRI) or enhanced computed tomography (CT) of the nasopharynx and neck (CT was indicated only in patients with a contraindication to MRI), enhanced CT scans of the chest and abdomen, and whole-body bone scan or positron emission tomography/computed tomography (PET/CT). All the patients were staged at baseline according to the eighth American Joint Committee on Cancer Union for International Cancer Control (UICC/AJCC) staging system29. Real-time quantitative polymerase chain reaction was used to measure the level of plasma EBV DNA before, during, and after first-line immunochemotherapy, as described previously30. All clinical data were collected from the electronic medical records system of SYSUCC.
The study flowchart is shown in Fig. 3. The study was approved by the Research Ethics Committee of Sun Yat-sen University Cancer Center (approval number: B2025-298-01), and performed according to the ethical principles of the Declaration of Helsinki. Written informed consent was waived due to the retrospective nature.
Matching between patients with and without LRRT across the three risk groups was performed using inverse probability of treatment weighting (IPTW). NPC nasopharyngeal carcinoma, LRRT locoregional radiotherapy.
Treatments
All the patients received standard first-line immunochemotherapy every three weeks for up to six cycles, at the discretion of clinicians. The standard palliative chemotherapy regimens included the following: GP: gemcitabine 1.0 g/m2 on days 1 and 8, and cisplatin 80 mg/m2 on day 1; TP: docetaxel or paclitaxel 260 mg/m2 on day 1, and cisplatin 80 mg/m2 on day 1; PF: cisplatin 80 mg/m2 on day 1, and 5-fluorouracil 500 mg/m2 on days 1-5; TPF: docetaxel or paclitaxel 200 mg/m2 on day 1, cisplatin 75 mg/m2 on day 1, and 5-fluorouracil 500 mg/m2 on days 1-5; TPC: docetaxel or paclitaxel 200 mg/m2 on day 1, cisplatin 75 mg/m2 on day 1, and capecitabine 1.0 g/m2, BID, on days 1-14. Anti-PD-1 immunotherapy options included 200 mg camrelizumab, tislelizumab, sintilimab, and pembrolizumab on day 1, or 240 mg toripalimab and nivolumab on day 1. All these agents were administered intravenously, except capecitabine, which was administered orally. 5-fluorouracil was administered by continuous intravenous infusion over 120 hours. Dose modification was permitted according to individual tolerance, as determined by the treating physicians.
After completing first-line treatment, patients in the non-LRRT cohort received immunotherapy maintenance every three weeks until disease progression or up to two years. All the patients in the non-LRRT cohort did not receive any form of local treatment for distant metastases to eliminate potential confounding effects on failure patterns. For patients in the LRRT cohort, IMRT-based LRRT to the nasopharynx and cervical lymph nodes was initiated within four to six weeks after the last immunochemotherapy cycle.
The design of the IMRT plan was as follows: The IMRT target volumes were delineated using imaging data before immunochemotherapy according to a previously described institutional treatment protocol31. All the patients were immobilized in the supine position using head, neck, and shoulder thermoplastic masks. Two sets of images with and without contrast were obtained from the CT simulator for treatment planning. All patients were scanned using serial 3-mm slices from the vertex through the clavicles. Inverse IMRT planning was performed using the Corvus system (version 3.0; Peacock, Nomos, Deer Park, IL, USA) and a MiMi multileaf collimator (Nomos, Sewickly, PA, USA) was used for planning and treatment. The gross tumor volume (GTV) included all gross diseases of the primary nasopharyngeal tumor volume (including retropharyngeal nodes; GTVnx) and corresponding cervical lymph node volumes (GTVnd) visualized on CT and/or MRI, as well as 18F-PET/CT before immunochemotherapy. The high-risk clinical target volume (CTV1) was defined by the pre-treatment GTVnx plus a 5–10 mm margin (2–3 mm posteriorly if adjacent to the brainstem or spinal cord) to encompass the high-risk sites of microscopic extension and the entire nasopharynx. The low-risk clinical target volume (CTV2) was defined as CTV1 plus a 5–10 mm margin (2–3 mm posteriorly if adjacent to the brainstem or spinal cord) to encompass the low-risk sites of microscopic extension, including the skull base, clivus, sphenoid sinus, foramen lacerum, ovale and spinosum, parapharyngeal space, pterygoid fossa, posterior parts of the nasal cavity, pterygopalatine fossa, retropharyngeal nodal regions, the involved and at-risk elective cervical nodal levels from level IB to level V. The planning target volume (PTV) PTV1 and PTV2 were created by adding an additional 5-mm margin to CTV1 and CTV2 to allow for setup variability and internal organ motion. Critical normal structures, including the brainstem, spinal cord, parotid glands, optic nerves, chiasm, lens, eyeballs, temporal lobes, temporomandibular joints, mandible, and hypophysis, were contoured and set as organ at risk (OAR) during optimization. Dose–volume histograms of the treatment targets and critical normal structures were evaluated. The prescribed dose was 70 Gy (range: 68–70 Gy), 66 Gy (range: 60–70 Gy), 60 Gy (range: 56–60 Gy), and 54 Gy (range: 50–54 Gy) for the PTVs derived from GTVnx, GTVnd, CTV1, and CTV2, respectively. The accumulated doses were divided into 33 fractions, five times per week.
Concurrent chemotherapy was either administered or omitted at the discretion of the clinician (Supplementary Table 7). Subsequently, immunotherapy maintenance was continued for patients in the LRRT cohort in the same manner.
Outcome and follow-up
The primary endpoint was PFS, which was defined as the period from the initiation of treatment to the first recorded disease progression at any site or death from any cause, whichever occurred first. Secondary endpoints were OS (defined as the period from the initiation of treatment to death from any cause), locoregional relapse-free survival (LRFS, defined as the period from the initiation of treatment to local or regional relapse or death from any cause), and distant metastasis-free survival (DMFS, defined as the period from the initiation of treatment to distant metastasis progression or death from any cause). Tumor response was assessed by an independent review committee and two experienced investigators according to RECIST version 1.132. The depth of response (DpR) was defined as the percentage of tumor shrinkage from the baseline tumor burden to the timepoint of assessment28,33.
Patients were followed up at regular intervals. The initial assessment of locoregional and distant tumor response was conducted after two cycles of first-line immunochemotherapy using CT and MRI scans. All the patients underwent comprehensive radiological and hematological examinations at the end of four to six cycles of first-line immunochemotherapy to assess the treatment response. After completion of first-line treatment, patients were followed up at least once every three months during the first three years and every six months thereafter until death. Nasopharyngoscopy, MRI of the head and neck, enhanced CT of the chest and abdomen, and plasma EBV DNA testing were performed routinely or when clinically indicated for tumor progression. Patients who were alive and without a recorded event were censored at the date of last follow-up.
Statistical analyses
Continuous variables were transformed into categorical variables according to the cut-off values, which were based on the previously published thresholds, or optimal cut-off values determined by X-tile software (version 3.6.1; Yale University, New Haven, CT, USA)34. Group comparisons were conducted using the Chi-square or Fisher’s exact test, as appropriate. The impact of variables was first assessed by calculating hazard ratios (HRs) and 95% confidence intervals (CIs) using univariate Cox proportional hazards regression. Variables with a P value < 0.2 were included in the least absolute shrinkage and selection operator (LASSO) logistic regression model for further selection. Thereafter, forward stepwise multivariate Cox regression was employed to test the independent significance of each variable selected from LASSO and develop the final scoring model. To facilitate clinical application, the weight of each variable in the scoring model was determined by rounding its β coefficient to the nearest integer. Using the scoring model, all patients (non-LRRT and LRRT cohorts) were classified into three risk stratification groups. To evaluate the discriminatory performance of the scoring model, the inverse probability of treatment weighting (IPTW) was first applied to balance potential confounding factors that could influence the treatment effects within each risk group. A standardized mean difference (SMD) < 0.1 was considered ideal and an SMD < 0.2 was deemed acceptable. Subsequently, Kaplan–Meier analyses were performed to evaluate the survival benefit of LRRT within each risk group by comparing PFS, OS, LRFS, and DMFS, with differences assessed using the log-rank test. All statistical analyses were performed using SPSS (version 26.0; SPSS, Chicago, IL, USA) and R version 4.3.1 (https://www.r-project.org) software. A two-tailed P value < 0.05 was considered statistically significant.
Data availability
The data that support the findings of this study are available on reasonable request from the corresponding author.
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Pan, J. J. et al. Proposal for the 8th Edition of the AJCC/UICC Staging System for Nasopharyngeal. Cancer Era Intensity-Modulated Radiother. Cancer 122, 546–558 (2016).
Chen, Y. P. et al. Nasopharyngeal carcinoma. Lancet Lond. Engl. 394, 64–80 (2019).
Mai, H. Q. et al. Toripalimab or placebo plus chemotherapy as first-line treatment in advanced nasopharyngeal carcinoma: a multicenter randomized phase 3 trial. Nat. Med. 27, 1536–1543 (2021).
Yang, Y. et al. Tislelizumab plus chemotherapy as first-line treatment for recurrent or metastatic nasopharyngeal cancer: A multicenter phase 3 trial (RATIONALE-309). Cancer Cell 41, 1061–1072.e4 (2023).
Yang, Y. et al. Camrelizumab versus placebo in combination with gemcitabine and cisplatin as first-line treatment for recurrent or metastatic nasopharyngeal carcinoma (CAPTAIN-1st): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 22, 1162–1174 (2021).
Bossi, P., Chan, A. T., Even, C. & Machiels, J. P. ESMO–EURACAN Clinical Practice Guideline update for nasopharyngeal carcinoma: adjuvant therapy and first-line treatment of recurrent/metastatic disease. Ann. Oncol. 34, 247–250 (2023).
Tang, L. L. et al. The Chinese Society of Clinical Oncology (CSCO) clinical guidelines for the diagnosis and treatment of nasopharyngeal carcinoma. Cancer Commun. Lond. Engl. 41, 1195–1227 (2021).
Network NCC. Head and Neck Cancers, Version 2. 2025. 2025. Accessed 30 March 2025. https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf.
Liu, Q., Li, J., Ng, W. T. & Lee, A. W. M. Treatment strategy for de novo metastatic nasopharyngeal carcinoma: a literature review. Chin. Clin. Oncol. 12, 43–43 (2023).
McDowell, L., Rischin, D. & Lee, A. W. M. Locoregional radiation therapy for de novo metastatic nasopharyngeal cancer: One size fits all?. Int J. Radiat. Oncol. 109, 131–133 (2020).
Li, W. Z. et al. Development of a prognostic model to identify the suitable definitive radiation therapy candidates in de novo metastatic nasopharyngeal carcinoma: a real-world study. Int J. Radiat. Oncol. Biol. Phys. 109, 120–130 (2022).
Yang, J. H. et al. Subdivision of de-novo metastatic nasopharyngeal carcinoma based on tumor burden and pretreatment EBV DNA for therapeutic guidance of locoregional radiotherapy. BMC Cancer 21, 534 (2021).
Sun, X. S. et al. Identifying optimal candidates for local treatment of the primary tumor among patients with de novo metastatic nasopharyngeal carcinoma: a retrospective cohort study based on Epstein–Barr virus DNA level and tumor response to palliative chemotherapy. BMC Cancer 19, 92 (2019).
Zou, X. et al. Establishment and validation of M1 stage subdivisions for de novo metastatic nasopharyngeal carcinoma to better predict prognosis and guide treatment. Eur. J. Cancer 77, 117–126 (2017).
Rusthoven, C. G. et al. Metastatic nasopharyngeal carcinoma: patterns of care and survival for patients receiving chemotherapy with and without local radiotherapy. Radiother. Oncol. 124, 139–146 (2017).
You, R. et al. Efficacy and safety of locoregional radiotherapy with chemotherapy vs chemotherapy alone in de novo metastatic nasopharyngeal carcinoma: a multicenter phase 3 randomized clinical trial. JAMA Oncol. 6, 1345–1352 (2020).
Li, P. et al. Analysis on clinical data of 677 death cases with nasopharyngeal carcinoma]. Lin. Chuang Er Bi Yan Hou Ke Za Zhi 16, 15–16 (2002).
Wen, D. et al. Locoregional radiotherapy candidates in de novo metastatic nasopharyngeal carcinoma: real-world insights in the immunotherapy era. J. Natl Compr. Canc Netw. 23, e247086 (2025).
He, S. Q. et al. Efficacy of local–regional radiotherapy in de novo metastatic nasopharyngeal carcinoma patients receiving chemo-immunotherapy: a multicenter, propensity score matching study. Radiother. Oncol. 203, 110687 (2025).
Liu, Z. Q. et al. Immunochemotherapy alone or immunochemotherapy plus subsequent locoregional radiotherapy in de novo metastatic nasopharyngeal carcinoma. Oral. Oncol. 147, 106583 (2023).
Hu, Y. J. et al. Locoregional radiotherapy improves survival outcomes in de novo metastatic nasopharyngeal carcinoma treated with chemoimmunotherapy. ESMO Open 8, 101629 (2023).
Mai, H. Q. et al. Toripalimab plus chemotherapy for recurrent or metastatic nasopharyngeal carcinoma: the JUPITER-02 randomized clinical trial. JAMA 330, 1961 (2023).
Zhang, L. et al. Gemcitabine plus cisplatin versus fluorouracil plus cisplatin in recurrent or metastatic nasopharyngeal carcinoma: a multicentre, randomised, open-label, phase 3 trial. Lancet 388, 1883–1892 (2016).
Wen, D. et al. Recursive partitioning analysis model for de novo metastatic nasopharyngeal carcinoma treated with locoregional radiotherapy following chemoimmunotherapy. ESMO Open 9, 103960 (2024).
Zheng, S. H. et al. Addition of chemoradiotherapy to palliative chemotherapy in de novo metastatic nasopharyngeal carcinoma: a real-world study. Cancer Cell Int. 22, 36 (2022).
Sun, X. S. et al. Optimizing the treatment pattern for de novo metastatic nasopharyngeal carcinoma patients: a large-scale retrospective cohort study. Front. Oncol. 10, 543646 (2020).
Xie, X., Li, X. & Yao, W. A narrative review: depth of response as a predictor of the long-term outcomes for solid tumors. Transl. Cancer Res. 10, 1119–1130 (2021).
Tang, L. L. et al. Validation of the 8th edition of the UICC/AJCC staging system for nasopharyngeal carcinoma from endemic areas in the intensity-modulated radiotherapy era. J. Natl. Compr. Cancer Netw. 15, 913–919 (2017).
Shao, J. et al. Comparison of plasma Epstein–Barr virus (EBV) DNA levels and serum EBV immunoglobulin A/virus capsid antigen antibody titers in patients with nasopharyngeal carcinoma. Cancer 100, 1162–1170 (2004).
Lai, S. Z. et al. How does intensity-modulated radiotherapy versus conventional two-dimensional radiotherapy influence the treatment results in nasopharyngeal carcinoma patients?. Int J. Radiat. Oncol. Biol. Phys. 80, 661–668 (2011).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
King, A. D., Leung, S. F., Wong, J. K. T. & Chan, A. T. C. Nasopharyngeal cancers: which method should be used to measure these irregularly shaped tumors on cross-sectional imaging? Int. J. Radiat. Oncol. Biol. Phys. 69, 148–154 (2007).
Camp, R. L., Dolled-Filhart, M. & Rimm, D. L. X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin. Cancer Res. 10, 7252–7259 (2004).
Acknowledgements
This study was funded by grants from the National Key Research and Development Program of China (2022YFC2505800, and 2022YFC2705005), National Natural Science Foundation of China (82173287, 82372980, 82203776, 82203125, 82222050, 82272739, 82272882, 82073003, 82003267, 82002852, 82373258, 82361168664, 8247101588, 82473038, 82303873, and 32200651), Guangdong Basic and Applied Basic Research Foundation (2021B1515230002, and 2023B1515120092), Science and Technology Program of Guangzhou (2025A04J3608, 2023A04J2127, and 2024B03J1248), Sun Yat-sen University Clinical Research 5010 Program (2024003, 2015021, 2019023, and 2017010), Postdoctoral Innovative Talent Support Program (BX20220361), Planned Science and Technology Project of Guangdong Province (2019B020230002), Key Youth Teacher Cultivating Program of Sun Yat-sen University (20ykzd24), and Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Contributions
Conceptualization: He, Liu, Mai. Data curation: He, Xiao, J. Chen, Li, Cheng. Formal analysis: He. Funding acquisition: Liu, Mai, Tang. Investigation: He, Liu, Mai, Q. Chen. Methodology: He, Xiao, Wu, Zhang, TSE, Sun. Project administration: Liu, Mai, Tang, Q. Chen. Supervision: Liu, Mai, Tang, Sun. Validation: Xiao, Wu. Visualization: He, Zhang, TSE. Writing—original draft: He, Xiao, Wu, Zhang, TSE. Writing—review & editing: He, Liu, Mai.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
He, JR., Xiao, YX., Wu, LF. et al. Who may omit locoregional radiotherapy in de novo metastatic nasopharyngeal carcinoma: a failure-pattern–based strategy in the immunotherapy era. npj Precis. Onc. 9, 389 (2025). https://doi.org/10.1038/s41698-025-01176-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41698-025-01176-1