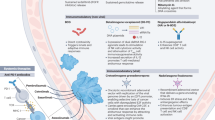
Abstract
Neoadjuvant cisplatin-based combination chemotherapy or perioperative durvalumab with neoadjuvant gemcitabine–cisplatin has been the primary treatment for localized muscle-invasive bladder cancer (MIBC). Nevertheless, many patients are either cisplatin-ineligible or relapse after standard therapy. This multicenter, retrospective real-world study evaluated disitamab vedotin (RC48) plus PD-1 inhibitors as neoadjuvant therapy for localized MIBC. Twenty-five patients (cT2-4aN0-2M0) received at least four cycles of RC48 (2.0 mg/kg, Q2W or Q3W) with toripalimab, tislelizumab, or pembrolizumab, followed by radical cystectomy. The pathological complete response rate was 48%, and the pathological downstaging rate was 88%. After a median follow-up of 17.0 months, 12-month disease-free and overall survival rates were 91.5% and 100%, respectively. HER2 overexpression (IHC 3+) was significantly associated with higher response (odds ratio [OR] = 6.75, 95% confidence interval [CI]: 1.16–39.20, p = 0.033), whereas advanced stage (>T2N0M0) predicted poorer outcomes (OR = 0.15, 95% CI: 0.03–0.86, p = 0.033). Treatment-related adverse events were manageable. These findings suggest that RC48 combined with PD-1 inhibitors is a promising neoadjuvant strategy for localized MIBC and warrants further validation in biomarker-selected populations.
Similar content being viewed by others
Introduction
Currently, neoadjuvant cisplatin-based combination chemotherapy or perioperative durvalumab with neoadjuvant gemcitabine–cisplatin followed by radical cystectomy has been the primary treatment for localized muscle-invasive bladder cancer (MIBC). However, more than 40% of patients with MIBC experience disease recurrence or death within three years after standard therapy1,2,3,4,5. Furthermore, a substantial proportion of patients are deemed ineligible for cisplatin-based neoadjuvant chemotherapy (NAC) due to comorbidities or renal impairment6. Thus, novel neoadjuvant strategies for patients with MIBC are urgently needed.
Immunotherapy has been actively investigated in this paradigm. Several phase II trials have demonstrated the efficacy and safety of immune checkpoint inhibitors (ICI) monotherapy in the neoadjuvant setting, such as PURE-01 (pembrolizumab) and ABACUS (atezolizumab), which reported pathologic complete response (pCR) rates of approximately 30–40% with acceptable safety profiles7,8. More recently, combination regimens of ICIs with platinum-based chemotherapy have yielded numerically higher pCR rates of 40–50%9,10,11. However, the incremental benefit over NAC remains modest, and durable survival advantages have yet to be conclusively demonstrated. Moreover, both ICI monotherapy and chemo-immunotherapy exhibit heterogeneous efficacy, with a substantial subset of patients failing to achieve meaningful tumor regression.
Given these limitations, novel strategies that can potentiate antitumor immunity while providing direct cytotoxic effects are needed. Disitamab vedotin (RC48) is an antibody–drug conjugate (ADC) targeting human epidermal growth factor receptor 2 (HER2), comprising a HER2-targeted monoclonal antibody linked to the cytotoxic agent monomethyl auristatin E (MMAE). The combination of RC48 and PD-1 inhibitors leverages synergistic mechanisms to enhance antitumor efficacy: RC48-mediated cytotoxicity releases tumor antigens, thereby priming and potentiating the immune response initiated by PD-1 blockade12. Preclinical studies have shown that RC48 induces immunogenic cell death and dendritic-cell maturation and antigen presentation, which enhances PD-1 blockade efficacy, especially in HER2-positive tumors, providing a biological rationale for combining RC48 with PD-1 inhibitors in HER-expressing MIBC13,14. Clinical trials (RC48-C005&C009, C014, C016) have demonstrated the promising efficacy and manageable toxicity of RC48 alone or in combination with ICIs in advanced urothelial carcinoma (UC) regardless of HER2 expression levels15,16,17. These results support further investigation of combining RC48 with ICIs in earlier disease settings such as neoadjuvant therapy for localized MIBC.
Recently, the single-arm phase II trial RC48-C017 reported encouraging efficacy and manageable safety of RC48 combined with toripalimab in the neoadjuvant treatment of MIBC18,19. Besides, the ongoing RC48-C022 study is evaluating perioperative RC48 plus toripalimab and XELOX in patients with resectable locally advanced gastric or gastroesophageal junction adenocarcinoma, highlighting the clinical applicability and rationale for RC48-based immunocombination regimens in the neoadjuvant setting. Nevertheless, clinical trials often have strict eligibility criteria and controlled conditions that may not fully reflect routine clinical practice. Here, we conducted a multicenter, real-world study to evaluate the efficacy and safety of RC48 combined with PD-1 inhibitors as neoadjuvant therapy for localized MIBC. Our study aims to explore pathological response rates and survival outcomes in a real-world context, offering insights into the feasibility of incorporating this novel combination into clinical practice.
Results
Patient and treatment characteristics
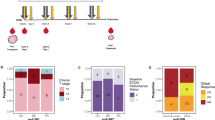
Figure 1 shows the patient selection process and study design. A total of 25 patients with localized MIBC receiving RC48 with PD-1 inhibitor combination therapy were included in the final analysis. The median age was 69 years (range, 46–79 years), and 21 (84.0%) were male. All patients had an ECOG performance status of 0 (92.0%, n = 23) or 1 (8.0%, n = 2). A history of smoking was reported in 24.0% of cases.
Patient Selection Flow Diagram.
The majority of patients had clinical stage T2 disease (72.0%, n = 18), and 9 patients (36.0%) presented with divergent differentiation or histologic subtypes20. HER2 expression by IHC on pre-treatment specimens was distributed as follows: IHC 0 (4.0%, 1/25), IHC 1+ (4.0%, 1/25), IHC 2+ (40.0%, 10/25), and IHC 3+ (52.0%, 13/25). Regarding treatment regimens, 13 patients (52.0%) received RC48 combined with toripalimab, while 9 patients (36.0%) receiving RC48 combined with tislelizumab and the other 3 patients (12.0%) receiving RC48 combined with pembrolizumab. The median number of neoadjuvant RC48 with PD-1 inhibitor administered cycles was 5 (range, 4–7), and radical cystectomy was completed in 25 of 25 patients (100%). Detailed patient characteristics were shown in Table 1.
Efficacy outcomes
All 25 patients underwent radical cystectomy. Pathologic assessment after neoadjuvant therapy revealed a pCR in 12 of 25 patients (48.0%) as shown in Table 2. Pathological downstaging was observed in 22 patients with a pDS rate of 88.0%. Persistent muscle-invasive disease (≥ypT2) remained in five patients (20.0%), and only one patient (4.0%) experienced disease progression. After a median follow-up of 17.0 months, the 12-month DFS rate was 91.5% (95%CI: 80.8–100%), and the 12-month OS rate was 100%. Kaplan-Meier survival curves are shown in Fig. 2.
A Disease-free survival (DFS). B Overall survival (OS).
The results of univariate analysis for all available pathological response-related factors between pathological complete responders (pCR, n = 12) and others (pPR+pSD+pPD, n = 13) are shown in Fig. 3. In the univariate analysis, most baseline characteristics, including age (≤65 vs. >65 years), ECOG performance status (0 vs. ≥1), treatment frequency (Q2W vs. Q3W), and treatment regimens (RC48 + ICIs) were not significantly associated with treatment response. And the pCR rates were generally consistent between patients with conventional UC and UC patients with other differentiation or subtypes (OR = 0.80, 95% CI: 0.16–4.12, p = 0.790). Notably, patients with clinical stage >T2N0M0 had a significantly lower likelihood of achieving pCR compared with those with T2N0M0 disease (OR = 0.15, 95% CI: 0.03-0.86, p = 0.033). Similarly, HER2 overexpression (IHC 3+) was associated with improved treatment outcomes (OR = 6.75, 95% CI: 1.16–39.20, p = 0.033).
Results of univariate logistic regression of pathological response-related factors with pCR rate.
We also conducted a Sankey plot (Fig. 4) illustrating each patient’s tumor stage from baseline (cTNM) to post-treatment (ypTNM) following neoadjuvant therapy, as well as HER2 expression dynamics. Analysis of individual patient trajectories demonstrated substantial downstaging from cTNM to ypTNM following neoadjuvant therapy, although the extent of regression varied across cases. Notably, a decline in HER2 expression between pre-treatment and post-treatment specimens was observed in patients who achieved pathological downstaging. Among the 10 patients with residual tumors after radical cystectomy, 6 (60%) showed a decrease in HER2 expression from positive (IHC 2+/3+) to negative (IHC 0/1+). In contrast, as for the three patients without pathological downstaging, HER2 expression remained stable in two and increased in one.
Each line represents an individual patient (n = 25), illustrating the change in Clinical stage from baseline (cTN) to post-treatment (ypTN). Line color indicates HER2 expression status, while symbols denote assessment timepoints: triangles represent pretreatment HER2, and squares represent posttreatment HER2. Twelve patients achieved pathological complete response (pCR), for whom HER2 evaluation was not feasible due to the absence of residual tumor tissue.
Safety profiles
The safety profile of RC48 with PD-1 inhibitors combination therapy is summarized in Table 3. Treatment-related adverse events (TRAEs) occurred in 92.0% (23/25) of patients (any grade), with grade ≥3 TRAEs in 16.0% (4/25). The most common TRAEs were peripheral neuropathy (32.0%, 2 patients grade ≥3), rash (24.0%, 2 patients grade ≥3), and elevated transaminases (20.0%, all grade 1-2). Most AEs were manageable with supportive care or dose adjustment, and no new safety signals were identified compared to previous RC48 trials15,16. No treatment-related deaths were reported.
Discussion
In the evolving landscape of neoadjuvant therapy for MIBC, several approaches have been evaluated beyond standard cisplatin-based chemotherapy, which yield pCR rates around 25-35%. ICI monotherapy has demonstrated clinically meaningful activity in phase II trials, with PURE-01 (pembrolizumab) and ABACUS (atezolizumab) reporting pCR rates of 42% and 31%, respectively, along with favorable safety profiles7,8. More recently, chemo-immunotherapy combinations such as gemcitabine–cisplatin plus atezolizumab (BLASST-1) and gemcitabine ± cisplatin plus nivolumab (HCRN GU14-188) achieved the pCR rates of approximately 41%10,21. The phase III NIAGARA trial evaluated perioperative durvalumab combined with neoadjuvant gemcitabine–cisplatin followed by cystectomy in cisplatin-eligible patients with MIBC. Compared to chemotherapy alone, this approach significantly improved event-free survival (EFS), as well as OS4. Despite these promising findings, the incremental benefit of adding immunotherapy to chemotherapy remains uncertain. More importantly, these studies also highlight a key limitation: a significant proportion of patients derive little or no benefit, underscoring the need for novel strategies capable of improving response rates while maintaining tolerability.
This multicenter, real-world study of neoadjuvant RC48 combined with immunotherapy in localized MIBC, demonstrated a pCR rate of 48%, with the pathological downstaging occurring in 88% of patients. Moreover, the 12-month DFS rate was 91.5% and 100% OS rate at a median follow-up of 17.0 months. Compared with the phase II trial RC48-C017, which reported a pCR rate of 63.6% with RC48 plus toripalimab, our study showed a slightly lower pCR rate, likely attributing to the heterogeneity of patients and treatment regimens in the real-world setting19. These findings suggest that the RC48 + PD-1 inhibitors regimen may represent a highly efficacious neoadjuvant option in MIBC patients. Mechanistically, the synergy between RC48 and PD-1 inhibitors offers a compelling rationale for these promising results. As a HER2-targeted ADC delivering MMAE, RC48 induces direct tumor cytotoxicity and immunogenic cell death, which may enhance antigen presentation and T-cell infiltration, thereby amplifying antitumor immunity13. Recently, another ADC, enfortumab vedotin (EV), has been explored as monotherapy or in combination with PD-1 inhibitors in the neoadjuvant setting for MIBC. Early results from the EV-103 Cohort H study demonstrated that neoadjuvant EV monotherapy achieved a pCR rate of 36% in cisplatin-ineligible patients, with an overall pathological downstaging rate of 50%22. Notably, the EV-303 (KEYNOTE-905) study evaluating perioperative EV in combination with pembrolizumab reported significant improvements in EFS (HR 0.40, 95% CI 0.28–0.57; one-sided p < 0.0001) and OS (HR 0.50, 95% CI 0.33–0.74; one-sided p = 0.0002) compared with surgery alone, yielding a pCR rate of 57.1%23,24. These data collectively suggest that ADC-based strategies, particularly when combined with PD-1 inhibitors, may hold promising potential in the neoadjuvant treatment of MIBC.
In our study, we found that the pCR rate for the HER2 IHC 3+ subgroup was higher than those for IHC 0/1+/2+ subgroups (OR 6.75, 95% CI 1.16–39.20, p = 0.033), which is consistent with data obtained from the RC48-C017 clinical trial. This observation may suggest that patients with HER2 overexpression could derive greater benefit from RC48-based neoadjuvant regimens, supporting that HER2 might serve not only as a therapeutic target but also as a predictive biomarker for treatment selection25. Some studies had also found that Her2 expression might provide additional prognostic information for MIBC patients, while others reported divergent results25,26,27. Moreover, previous studies have confirmed significant intratumor heterogeneity, and differences between primary tumors and metastases in the expression of ADC targets such as HER2, which further complicate its role as a biomarker28. Our study also observed that some patients experienced dynamic changes in HER2 expression following neoadjuvant RC48 + PD-1 inhibitor treatment. Therefore, the prognostic and predictive value of HER2 requires validation in larger, prospective cohorts.
Another potential factor associated with treatment response was clinical stage. Patients with >T2N0M0 disease tended to have lower pCR rates compared with those with T2N0M0 disease (OR = 0.15, 95% CI: 0.03-0.86, p = 0.033), consistent with previous evidence in the RC48-C017 trial indicating that the pCR rate for T2N0 patients was evidently higher than those for the other subgroups (T3N0, T4N0 and TanyN1)18. However, for patients who achieve pCR, whether certain selected MIBC patients might be suitable for bladder-sparing approaches remains an intriguing question29. This concept has been explored in recent trials, such as the phase II study of gemcitabine-cisplatin plus nivolumab as organ-sparing treatment for MIBC, where patients achieving clinical CR (cCR) after neoadjuvant therapy were considered for bladder preservation without compromising short-term outcomes30. Future prospective, biomarker-driven studies incorporating molecular markers, immune signatures, or circulating tumor DNA (ctDNA) will be essential to validate these observations and refine patient selection for bladder-sparing strategies.
Compared with clinical trials, this study was conducted in a real clinical practice setting to supplement and refine the efficacy and safety data of RC48 plus PD-1 inhibitors in real-world patient populations. Several limitations should be acknowledged. First, the retrospective nature inherently introduces potential selection bias, and the small, heterogeneous cohort limits the generalizability of our findings and precludes robust multivariate analysis. Second, variations in PD-1 inhibitor type, dosage, and treatment cycles may introduce confounding effects. Moreover, the short median follow-up restricts evaluation of long-term treatment outcomes and late-onset adverse events, which are needed to confirm the durability of responses. Given these limitations, future multicenter, prospective study with a standard treatment control group should be conducted to address the limitations and provide stronger evidence.
In summary, our findings suggest that neoadjuvant RC48 plus PD-1 inhibitors may represent a feasible and potentially effective approach for localized MIBC, warranting validation in larger, prospective biomarker-driven studies.
Methods
Study design and patients
This was a multicenter, retrospective study conducted at Peking University First Hospital and Peking University First Hospital Daxing District, Beijing, China, between July 2022 and December 2024. Patient data were collected from clinical records. Eligible patients were those diagnosed with localized MIBC (cT2-4aN0-1M0) and received at least four cycles of neoadjuvant therapy with RC48 (2.0 mg/kg, IV, Q2W or Q3W) plus a PD-1 inhibitor (e.g., toripalimab, tislelizumab, or pembrolizumab) as neoadjuvant therapy followed by planned radical cystectomy. Patients with prior systemic therapy, distant metastases, or incomplete clinical data were excluded. HER2 expression status was determined by immunohistochemistry (IHC) on pre-treatment transurethral resection of bladder tumor (TURBT) specimen, and HER2 positivity was not mandated for inclusion in this real-world study, reflecting clinical practice.
The study protocol was approved by the Ethics Committee of Peking University First Hospital (Approval No.: 2025R0556) and conducted in accordance with the Declaration of Helsinki. The requirement for informed consent was waived by the ethics committee given the participants could not be contacted due to the retrospective nature of the research.
HER2 testing
In our study, HER2 expression was evaluated by IHC staining method based on the criteria from previous studies evaluating anti-HER2 therapies27,31,32. The primary antibody used was the Ventana anti-HER2/Neu (4B5) rabbit monoclonal antibody (mAb) (Roche), and staining was performed with the BenchMark GX (Roche). IHC scores were reviewed by an independent pathologist at the laboratory. The staining scores were assessed according to the HER2 test guidelines for breast cancer and were classified into IHC 0, IHC 1+, IHC 2 + , and IHC 3+33,34. HER2 3+ and 2+ were defined as HER2 positive35,36. A detailed description of the IHC scoring protocol is provided in the Supplementary Material.
Data collection and effectiveness evaluation
Baseline demographics, tumor characteristics, treatment regimens, treatment-related adverse events (TRAEs) and follow-up data were retrospectively collected. For all patients, clinical staging was conducted using computed tomography (CT) of the chest, abdomen and pelvis with intravenous contrast, and contrast-enhanced pelvic magnetic resonance imaging (MRI). After the completion of neoadjuvant therapy followed by RC, pathological restaging was performed, including ypTN classification, resection margin status, and tumor regression grade (TRG) according to the Mandard scoring system37.
The primary effectiveness endpoint was pathological complete response rate (pCR; ypT0N0, defined as the absence of tumor cells in the surgical specimen, both at the primary tumor site and regional lymph nodes).
Secondary endpoints included: pathological downstaging rate (pDS; defined as lower pathologic stage compared with the pre-treatment clinical stage), disease-free survival (DFS), overall survival (OS), and safety, assessed by the incidence and severity of adverse events (AEs) according to Common Terminology Criteria for Adverse Events (CTCAE) version 5.0.
Statistical analysis
Descriptive statistics were used to summarize baseline characteristics, treatment details and safety outcomes. Categorical variables were reported as frequencies and percentages; continuous variables were reported as medians with ranges or interquartile ranges (IQR). pCR rate was calculated with 95% confidence intervals (CIs). Univariate logistic regression analyses were explicated to model the pCR rate using pathological response-related factors relevant variables. DFS was calculated from the day of treatment initiation to the observation of progression or recurrence, and OS was measured from the day of treatment initiation to death from any cause. Patients without recurrence or death were censored with the date of last follow-up, with no documented recurrence or death as the date of censoring. Both DFS and OS curves were estimated using the Kaplan-Meier method.
A two-sided P value < 0.05 was considered statistically significant. Statistical analyses were performed using R software (version 4.3.2) or SPSS version 26.0 (IBM Corp., Armonk, NY, USA). Data cut-off date was May 31st, 2025.
Data availability
The datasets generated and analyzed during the current study are not publicly available due to patient privacy concerns but are available from the corresponding author on reasonable request.
Code availability
No new algorithms were developed for this article. All code generated for analysis is available from the corresponding author upon request.
References
Lopez-Beltran, A., Cookson, M. S., Guercio, B. J. & Cheng, L. Advances in diagnosis and treatment of bladder cancer. BMJ 384, e076743 (2024).
Grossman, H. B. et al. Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N. Engl. J. Med. 349, 859–866 (2003).
Griffiths, G., Hall, R., Sylvester, R., Raghavan, D. & Parmar, M. K. International phase III trial assessing neoadjuvant cisplatin, methotrexate, and vinblastine chemotherapy for muscle-invasive bladder cancer: long-term results of the BA06 30894 trial. J. Clin. Oncol. 29, 2171–2177 (2011).
Powles, T. et al. Perioperative durvalumab with neoadjuvant chemotherapy in operable bladder cancer. N. Engl. J. Med. 391, 1773–1786 (2024).
Patel, V. G., Oh, W. K. & Galsky, M. D. Treatment of muscle-invasive and advanced bladder cancer in 2020. CA Cancer J. Clin. 70, 404–423 (2020).
Jiang, D. M. et al. Defining cisplatin eligibility in patients with muscle-invasive bladder cancer. Nat. Rev. Urol. 18, 104–114 (2021).
Necchi, A. et al. Pembrolizumab as neoadjuvant therapy before radical cystectomy in Patients With Muscle-Invasive Urothelial Bladder Carcinoma (PURE-01): an open-label, single-arm, phase II study. J. Clin. Oncol. 36, 3353–3360 (2018).
Powles, T. et al. Clinical efficacy and biomarker analysis of neoadjuvant atezolizumab in operable urothelial carcinoma in the ABACUS trial. Nat. Med. 25, 1706–1714 (2019).
Gupta, S. et al. Results from BLASST-1 (Bladder Cancer Signal Seeking Trial) of nivolumab, gemcitabine, and cisplatin in muscle-invasive bladder cancer (MIBC) undergoing cystectomy. J. Clin. Oncol. 38, 439–439 (2020).
Brown, J. et al. HCRN GU14-188: Phase Ib/II study of neoadjuvant pembrolizumab and chemotherapy for T2-4aN0M0 urothelial cancer. J. Clin. Oncol. 41, 448–448 (2023).
Li, K. et al. Neoadjuvant gemcitabine–cisplatin plus tislelizumab in persons with resectable muscle-invasive bladder cancer: a multicenter, single-arm, phase 2 trial. Nat. Cancer 5, 1465–1478 (2024).
Wei, Y. et al. Disitamab vedotin in combination with immune checkpoint inhibitors for locally and locally advanced bladder urothelial carcinoma: a two-center’s real-world study. Front. Pharmacol. 14, https://doi.org/10.3389/fphar.2023.1230395 (2023).
Xiao, J. et al. Overcoming immunotherapy resistance in bladder cancer with a novel antibody-drug conjugate RC48. J. Immunother. Cancer 13, e011881 (2025).
Wu, X. et al. A HER2-targeting antibody-MMAE conjugate RC48 sensitizes immunotherapy in HER2-positive colon cancer by triggering the cGAS-STING pathway. Cell Death Dis. 14, 550 (2023).
Sheng, X. et al. Efficacy and safety of disitamab vedotin in patients with human epidermal growth factor receptor 2-positive locally advanced or metastatic urothelial carcinoma: a combined analysis of two phase II clinical trials. J. Clin. Oncol. 42, 1391–1402 (2024).
Zhou, L. et al. Disitamab vedotin plus toripalimab in patients with locally advanced or metastatic urothelial carcinoma (RC48-C014): a phase Ib/II dose-escalation and dose-expansion study. Ann. Oncol. 36, 331–339 (2025).
Sheng, X. et al. Disitamab vedotin plus toripalimab in HER2-expressing advanced urothelial cancer. N. Engl. J. Med. https://doi.org/10.1056/NEJMoa2511648 (2025).
Sheng, X. et al. Preliminary efficacy and safety results from RC48-C017: a phase II study of neoadjuvant treatment with disitamab vedotin plus toripalimab in patients with muscle-invasive bladder cancer (MIBC) with HER2 expression. J. Clin. Oncol. 42, 4568–4568 (2024).
Sheng, X. et al. Neoadjuvant treatment with disitamab vedotin plus perioperative toripalimab in patients with muscle-invasive bladder cancer (MIBC) with HER2 expression: Updated efficacy and safety results from the phase II RC48-C017 trial. J. Clin. Oncol. 43, 665–665 (2025).
Netto, G. J. et al. The 2022 World Health Organization classification of tumors of the urinary system and male genital organs—Part B: prostate and urinary tract tumors. Eur. Urol. 82, 469–482 (2022).
Funt, S. A. et al. Neoadjuvant atezolizumab with gemcitabine and cisplatin in patients with muscle-invasive bladder cancer: a multicenter, single-arm, phase II trial. J. Clin. Oncol. 40, 1312–1322 (2022).
Flaig, T. W. et al. Study EV-103: neoadjuvant treatment with enfortumab vedotin monotherapy in cisplatin-ineligible patients (pts) with muscle invasive bladder cancer (MIBC): Updated results for Cohort H. J. Clin. Oncol. 41, 4595–4595 (2023).
Necchi, A. et al. Phase 3 KEYNOTE-905/EV-303: Perioperative pembrolizumab (pembro) or pembro + enfortumab vedotin (EV) for muscle-invasive bladder cancer (MIBC). J. Clin. Oncol. 41, TPS585–TPS585 (2023).
Vulsteke, C. et al. in Annals of Oncology 36 S1-S60. https://doi.org/10.1016/annonc/annonc1965 (2025).
Kim, S. W. et al. HER2 overexpression predicts pathological T2 stage and improved survival in de novo muscle-invasive bladder cancer after immediate radical cystectomy: a retrospective cohort study. Int. J. Surg. 110, 847–858 (2024).
Kolla, S. B. et al. Prognostic significance of Her2/neu overexpression in patients with muscle invasive urinary bladder cancer treated with radical cystectomy. Int. Urol. Nephrol. 40, 321–327 (2008).
Jimenez, R. E. et al. Her-2/neu overexpression in muscle-invasive urothelial carcinoma of the bladder: prognostic significance and comparative analysis in primary and metastatic tumors. Clin. Cancer Res. 7, 2440–2447 (2001).
Raggi, D. et al. HER2 and urothelial carcinoma: current understanding and future directions. Nat. Rev. Urol. https://doi.org/10.1038/s41585-025-01075-x (2025).
Grobet-Jeandin, E. et al. A systematic review of oncological outcomes associated with bladder-sparing strategies in patients achieving complete clinical response to initial systemic treatment for localized muscle-invasive bladder cancer. Eur. Urol. Oncol. 6, 251–262 (2023).
Galsky, M. D. et al. Gemcitabine and cisplatin plus nivolumab as organ-sparing treatment for muscle-invasive bladder cancer: a phase 2 trial. Nat. Med. 29, 2825–2834 (2023).
Hussain, M. H. et al. Trastuzumab, paclitaxel, carboplatin, and gemcitabine in advanced human epidermal growth factor receptor-2/neu-positive urothelial carcinoma: results of a multicenter phase II National Cancer Institute trial. J. Clin. Oncol. 25, 2218–2224 (2007).
Powles, T. et al. Phase III, double-blind, randomized trial that compared maintenance lapatinib versus placebo after first-line chemotherapy in patients with human epidermal growth factor receptor 1/2-positive metastatic bladder cancer. J. Clin. Oncol. 35, 48–55 (2017).
Wolff, A. C. et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J. Clin. Oncol. 31, 3997–4013 (2013).
Zhang, H., Moisini, I., Ajabnoor, R. M., Turner, B. M. & Hicks, D. G. Applying the new guidelines of HER2 testing in breast cancer. Curr. Oncol. Rep. 22, 51 (2020).
Gårdmark, T., Wester, K., De La Torre, M., Carlsson, J. & Malmström, P.-U. Analysis of HER2 expression in primary urinary bladder carcinoma and corresponding metastases. BJU Int. 95, 982–986 (2005).
Sheng, X. et al. Open-label, multicenter, phase II study of RC48-ADC, a HER2-targeting antibody–drug conjugate, in patients with locally advanced or metastatic urothelial carcinoma. Clin. Cancer Res. 27, 43–51 (2021).
Mandard, A. M. et al. Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer 73, 2680–2686 (1994).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (Grant No. 82273347) and the Clinical Research Foundation of Central High-level Hospital (Peking University First Hospital Research Fund Program) (Grant No. 2023HQ6).
Author information
Authors and Affiliations
Contributions
C.Z. had full access to all the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: X.G., Y.G., Z.H., C.Z. Acquisition of data: X.G., S.W., Y.M., Y.D., Y.C., Q.W. Analysis and interpretation of data: X.G., Y.D., Y.C., K.W., Q.T., Y.F., H.H. Drafting of the manuscript: X.G., Y.G.Critical revision of the manuscript for important intellectual content: X.G., Z.H., Y.G., C.Z. Statistical analysis: X.G., S.W. Administrative, technical, or material support: None. Supervision: Z.H. and C.Z.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, X., Wang, S., Ma, Y. et al. Disitamab vedotin (RC48-ADC) combined with immunotherapy as neoadjuvant therapy for localized muscle-invasive bladder cancer: a multicenter real-world study. npj Precis. Onc. 10, 11 (2026). https://doi.org/10.1038/s41698-025-01211-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41698-025-01211-1