Abstract
Pain is a complex multidimensional experience that integrates sensory and emotional components, presenting significant challenges for accurate assessment in clinical practice. Traditional methods of pain evaluation rely on subjective self-reporting and each individual’s ability to communicate their pain experience. In light of the effect of pain on the Autonomic Nervous System, researchers are interested in developing objective assessment techniques using physiological signals. This paper outlines the latest advances in pain biomarkers and machine learning methods for assessing pain using physiological signals, highlighting the growing interest and unmet demand in this area. A comprehensive literature review was conducted, covering studies between 2014 and 2024. The discussion is organised into two areas: first, an analysis of the variations in signal feature behaviour across different pain types, and second, a review of the current state-of-the-art models for pain assessment developed using classical machine learning and deep learning techniques.
Similar content being viewed by others
Introduction
The International Association for the Study of Pain (IASP) defines pain as “an unpleasant sensory and emotional experience associated with, or resembling that associated with actual or potential tissue damage”1. Pain perception involves complex interactions between the nervous system, the brain, and emotional factors, making it both a physical and psychological phenomenon. The sensory component encompasses nociception, the neural pathways transmitting pain sensation, and the physiological encoding and processing of those signals. The emotional component involves the individual’s anticipation of potential harm and the unique perception of pain1,2,3.
Due to this complexity, objective pain evaluation remains one of the biggest challenges that healthcare providers face in achieving optimal pain management4,5. The current standard of clinical practice is the use of self-assessment unidimensional tools like Visual Analog Scales (VAS), Numerical Rating Scales (NRS), and Verbal Rating Scales (VRS)2. Additionally, multidimensional questionnaires are often employed to capture broader aspects of the pain experience, including its quality, emotional impact, and interference with daily activities5.
Despite being simple and easy to use to gather insightful information from the patient, these methods have some limitations. They rely on the user’s ability to understand and communicate their pain perception, and the response provided may be influenced not only by the sensory information but by different social and psychological factors like anxiety, pain catastrophising and sensitivity3,6.
Objective pain assessment using physiological signals is an increasingly promising field. It is grounded in the premise that pain triggers changes in the Autonomic Nervous System (ANS). The ANS plays a fundamental role in regulating basal autonomic functions and biological processes such as blood flow, digestion, and respiration, leading to changes that can be measured using physiological signals7,8,9,10.
Despite extensive efforts to develop an objective tool for assessing pain, no objective method for pain assessment has been widely accepted or integrated into standard clinical practice. Previous reviews have explored these efforts, often focusing on specific pain types or technologies individually. For instance7, focused on the identification of relevant sensor technologies and analytical techniques specifically for acute pain, while11 discussed key signatures for chronic pain assessment, concentrating solely on Functional Magnetic Resonance Imaging (fMRI) and electroencephalography (EEG). Taking a technology-specific approach12,13,14, [13] aimed for the identification of photoplethysmography (PPG) derived indexes exclusively for the assessment of postoperative pain, and [14] provided an overview of signal processing methods and machine learning (ML) techniques used with electrodermal activity (EDA) in pain assessment.
However, to the best of our knowledge, no comprehensive review has examined how physiological signals and ML techniques are applied across different pain types, nor has one compared the behavioural and physiological signatures that distinguish these conditions. This review aims to address that by analysing the use of physiological signals and ML models in pain assessment, with a particular emphasis on identifying patterns and differences in behavioural features across acute, chronic, and perioperative/postoperative pain contexts.
Results
Search results
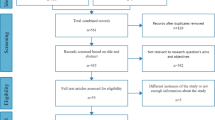
Figure 1 outlines the identification and selection process for the research papers retrieved. After duplicate removal, the search strategy yielded 518 scientific publications from the databases searched. Additionally, by screening the reference list of all literature reviews and relevant publications found, 23 papers were manually included, for a total of 541 papers screened against the inclusion criteria. Finally, after screening the metadata and abstracts, 89 papers were identified as meeting the criteria for inclusion in the review.
The figure presents the flow of information through the different stages of the systematic review. It summarizes the number of records identified by the search strategy, the screening steps applied, and the reasons for exclusion, leading to the final set of studies included.
A preliminary analysis of publication trends through the years reveals a growing interest among researchers in assessing pain through physiological signals. This increased interest is illustrated in Fig. 2, which shows that the number of publications retrieved went from 3 in 2014 to 17 in 2024.
The figure illustrates the publication trends on the use of physiological signals in pain assessment over the past decade. Individual categories represent specific physiological signals, while the “Others” category includes all signals reported in the literature that were not assigned to a predefined group. The polynomial line labelled “Poly. (Total)” depicts the overall trajectory of publications addressing pain assessment with physiological signals during this period.
In total, the included studies reported a wide variety of physiological signals for pain assessment. The most investigated signals were electrocardiography (ECG) (n = 36 studies), where HRV features were the most widely discussed, followed by EDA (n = 33 studies), PPG (n = 26 studies), and EEG (n = 25 studies). Less frequently reported were electromyography (EMG) (n = 14 studies) and other signals, such as blood pressure (BP) in four studies, respiration rate (Resp) in four studies, Functional Near-Infrared Spectroscopy (fNIRS) in three studies, and pupillometry in two studies, which implemented those signals. The lower frequency of reports containing EMG and other signals could have been due to the nature of the search strategy, where the keywords did not include those signals strictly. Notably, several studies simultaneously recorded and analysed multiple physiological signals, either in combination or separately. Therefore, the reported frequencies do not sum to 100% of the included studies but rather reflect the number of times each signal appeared across the literature.
Among the 89 studies included, 36 papers reported the use of machine learning approaches for pain assessment. The remaining 53 papers focused solely on traditional statistical methods to validate and analyse the behaviour of various physiological signals and specific markers related to pain assessment.
Currently available commercial devices
From the reviewed studies, two commercial devices were identified as currently used for pain monitoring: the Medasense PMD-200 system15,16, and the Newborn Infant Parasympathetic Evaluation (NIPE) Monitor17.
The PMD-200 system is a physiological monitor developed by Medasense to assist clinicians with analgesic dosage for patients under general anaesthesia. It provides a numeric scale of nociceptive response levels, known as the Nociception Level (NOL) Index. This numeric non-linear value is generated by a machine learning model trained using the random forest algorithm, which takes PPG, EDA, peripheral temperature and accelerometry as the input to calculate the NOL Index18.
Recent studies have analysed the usefulness of this technology for pain assessment in awake individuals, where other factors besides nociception might activate autonomic nervous system responses. The NOL Index has shown a significant, mild positive correlation with nociception and the potential to discriminate between nociceptive vs non-nociceptive stimulation in postoperative patients15. It has also shown a mild correlation with acute pain in chronic pain patients16. However, this might not be suitable for assessing moderate acute pain levels or discriminating chronic pain15,16.
Similarly, a NIPE Monitor has been commercialised by MDoloris Medical Systems. This technology is specifically designed to evaluate intraoperative and postoperative pain in infants. It analyses continuous cardiac signals derived from ECG or PPG waveform and provides an index between 0 and 10019. This monitor was recently used to perform a comparison and characterisation of intraoperative and postoperative pain after open vs laparoscopic inguinal hernia repair in children under two years17.
Open access physiological databases for pain study
Based on the outcomes of the reviewed studies, summarised in Table 1, three publicly accessible databases containing physiological recordings for pain analysis were identified. The BioVid Heat Pain database was the most frequently used, appearing in nine of the included papers. The X-ITE Pain database was reported in three studies, while the PainMonit database was used in one.
The BioVid Heat pain database is the first publicly accessible database that combines physiological parameters, video signals, and stimulus intensity for studying acute pain in healthy subjects. The database was first introduced in 2013, and it was publicly released for use in non-commercial research in 2018. It contains data from 90 subjects divided into three age groups of 30 subjects each: 18–35, 36–50, and 51–65. All groups were equally divided between male and female participants. The data were acquired from an experimental study where pain was induced using thermal stimulation from a thermode block placed on the right arm, which produces heat on the skin. The physiological signals recorded include electrocardiography (ECG), electromyography (EMG), electroencephalography (EEG), and electrodermal activity (EDA). The video signals were recorded to capture participants’ facial expressions during the experimental pain induction. The database is available for non-commercial research upon request20.
On the other hand, the X-ITE Pain Database is a publicly available multimodal dataset designed to support research in automatic pain recognition. It was collected from 134 participants through controlled experimental pain induction (heat and electrical stimuli) at multiple intensities, including both phasic and tonic protocols. The dataset comprises approximately 24,000 phasic and 800 tonic stimuli, along with their associated responses. A wide range of modalities were recorded, including facial video (frontal, side-view, and thermal), body video (RGB and depth), audio, as well as physiological signals such as electrodermal activity (EDA), electrocardiogram (ECG), and surface electromyography (sEMG) from several muscle sites. This breadth makes the X-ITE database particularly valuable for multimodal approaches that combine behavioural and physiological data in the context of pain assessment. The database is available for research upon request21.
Recently, a new database called PainMonit was also released. This database has two datasets that include not only ECG, EMG, and EDA signals but also photoplethysmography (PPG), temperature (Temp) and respiration (Resp) signals for the study of pain. The first dataset, the PainMonit Experimental Dataset (PMED), represents experimental data collected from 55 healthy subjects, comprising 22 males and 33 females, with an average age of 27.47 ± 6.90 years. These subjects underwent thermal (heat) pain elicitation from a thermode block. In addition to information about stimulus intensity, like the BioVid database, this dataset provides self-reported pain intensity VAS score from the subjects acquired over the duration of the experiment22.
The second dataset includes a clinical dataset, PainMonit Clinical Dataset (PMCD), acquired from 49 subjects (21 males and 28 females) with an average age of 27.47 ± 6.90 years, diagnosed with chronic arm and neck pain. Signals were collected before, during and after a facial physiotherapy session that was prescribed for their pain management. Besides the signals, self-reporting NRS scores are included in the dataset information22.
Study heterogeneity and reporting indicators
To provide an overview of study characteristics and facilitate the interpretation of results, key features of the included studies were summarised in a heterogeneity table (Table 2). The table presents information such as the type of pain assessed, the study population, the sample size, sex distribution, methodological approach, measure used in comparative analysis, and whether data were derived from public databases. This summary allows readers to appreciate the diversity across studies in terms of design, population, and measured outcomes, highlighting potential sources of variability in the literature.
The studies demonstrated substantial heterogeneity across key characteristics. Most addressed acute pain (64%), with fewer focusing on chronic or intraoperative/postoperative pain. Study populations were nearly balanced between volunteers and patients, with some using mixed groups. Sample sizes varied widely, though most ranged between 20 and 50 participants, followed by studies with more than 50 participants and only a few reporting fewer than 20 subjects. Sex distribution was inconsistent, with balanced samples in only 21.3% of studies, while most showed either male or female predominance, and nearly a quarter did not report sex at all. Methodological approaches were diverse, dominated by pain induction (66.3%), followed by pain modulation, observational, and surgical studies. Public database use was limited, with only 14.6% of studies employing shared datasets.
Physiological responses to pain
This section explores the physiological signals linked to variations in pain perception, as reported in the reviewed literature. Table 1 summarises the extracted data, organised by signal type and further categorised by the type of pain studied to support a clear presentation of evidence. Each physiological signal identified as a potential pain marker is discussed in turn, highlighting features that have been previously investigated as possible biomarkers of autonomic nervous system activity and pain sensation. The section also explores how these features behave across different pain contexts, including acute, chronic, and perioperative/postoperative pain.
Electromyography (EMG)
Electromyography is defined as the measurement of muscle electrical impulses generated when the muscle fibres contract. EMG signals can be measured using an intramuscular needle or electrodes placed on the skin’s surface23. The signal’s potential for pain evaluation relies on the idea that electrical muscle activity indicates general psychophysiological stimulation. Higher muscle tone is linked to increased sympathetic nervous system activity, while a decrease in electrical muscle activity is associated with parasympathetic activity24.
Several studies have been conducted using the BioVid Heat Pain Database. Amplitude and similarity-derived parameters measured from the zygomaticus and corrugator muscles have demonstrated a significant correlation with thermal stimulation levels (4 levels) in healthy subjects24,25. Additionally, these parameters exhibit high accuracy, sensitivity and specificity to differentiate between no stimulus vs painful stimulation when used to train SVM models25.
In contrast, statistical features extracted from the database’s EMG signals, such as the ratio of mobility of the first derivative of the signal (FF), root mean square value (RMS), and zero crossing, or the number of times the pattern crosses the zero-potential line (ZCD), showed no significant correlation with different acute pain states in healthy subjects23.
Jurth et al. (2014) investigated the reliability of the Nociceptive Flexion Reflex (NFR) and self-assessment for quantifying pain through test-retest measures using a Conditioned Pain Modulation (CPM) approach26. NFR refers to a leg withdrawal reflex measured in the biceps femoris muscle after stimulation of the sural nerve at the foot. The study found no statistically significant correlation between this parameter and pain stimulation/reduction.
Electrocardiography (ECG)
Electrocardiography is a technique used to measure the electrical activity of the heart7. The most commonly used feature in pain research is Heart Rate Variability (HRV). HRV is an indicator of the variability in time between heartbeats that has been linked to cardiac autonomic regulation27. This measure contains time and frequency domain parameters with different information regarding the ANS.
Time-domain parameters such as Heart Rate (HR) and R-R Interval (RRI) are commonly used indicators of autonomic nervous system activity. HR represents the number of heartbeats per minute, reflecting the frequency of cardiac cycles. The RRI refers to the time elapsed between two consecutive heartbeats, measured as the interval between R-waves on the ECG28. These parameters reflect the dynamic balance between the two branches of the ANS: the parasympathetic and sympathetic nervous systems. Parasympathetic influence on HR is primarily mediated by the vagus nerve through the release of acetylcholine, which acts to slow the heart rate29.
With greater parasympathetic (vagal) activity, HR tends to decrease and RRI increases, indicating a relaxed state. Conversely, a shorter R-R Interval and elevated heart rate are often associated with increased sympathetic activity, which prepares the body for ‘fight or flight’ responses29. Studies have reported a significant positive correlation between HR and chronic pain intensity29,30.
On the other hand, regarding the RRI marker, research has demonstrated a significant negative correlation with chronic pain intensity29,30 and acute pain detection in healthy volunteers31. Similarly, the R-R slope indicates a strong correlation with acute pain levels in healthy volunteers24 while R-R standard deviation (RRSD) shows a significant positive correlation with pain intensity in cancer patients experiencing chronic pain32.
Other time domain parameters studied as potential pain biomarkers include the Standard Deviation of the Normal-to-Normal Beat Interval (SDNN) and the Square Root of the Mean Squared Differences of Successive NN intervals (RMSSD). Research has reported that SDNN has a negative correlation with chronic pain intensity30 but a significant positive correlation with acute pain in healthy volunteers31. Similarly, RMSSD has reported a negative correlation with chronic pain intensity30.
Also, HRV frequency domain parameters, which measure the distribution of power across different frequency bands in the heart rate signal, have been shown to be impacted by pain. High Frequency (HF), between 0.15 and 0.4 Hz, is affected by vagal parasympathetic activity via the release of acetylcholine by the vagus nerve33 and has shown a significant negative correlation with chronic pain intensity29,30 and acute pain in healthy volunteers31. In contrast, Lee et al. (2019) found that HF has described a negative correlation with postoperative pain. They also suggested that this marker itself might not be sufficient to distinguish moderate pain states34.
Low Frequency (LF), ranging from 0.04 to 0.15 Hz, reflects both sympathetic and vagal activity. Research shows a significant negative correlation between this parameter and chronic pain intensity29 and acute pain in healthy volunteers31. Similarly, Very Low Frequency (VLF), between 0.003 and 0.04 Hz, also reports a negative correlation with chronic pain intensity29.
Furthermore, the Analgesia Nociception Index (ANI) has been introduced to evaluate acute nociception. This index is derived from the RR and a frequency domain analysis of the HF component. Initially suggested for monitoring pain nociception during surgery, studies have demonstrated that it also negatively correlates with self-reported pain in postoperative patients35.
To improve the pain recognition capabilities of physiological signals, Jiang et al. (2024) proposed two methods for representing individual pain sensitivity using the HRV features SDNN, RMSSD and LF at resting state. These methods are named SensPredict and SensHRV and calculate a pain sensitivity score using linear regression and neural networks, respectively36.
Despite the evidence previously discussed, the reliability of HRV features for assessing pain remains uncertain. While some research suggests a link between self-reported pain and HRV features, other studies have reported a lack of correlation between these parameters in chronic pain scenarios, indicating that they can’t be used as surrogate biomarkers of pain28,37.
For example, the Low-to-high Frequency Ratio (LF/HF) parameter, hypothesised to represent the sympathetic and parasympathetic balance index33, has shown a strong positive correlation with chronic pain intensity30. Despite this, its accuracy in terms of sympathetic activity has been debated38. Particularly, Umeda & Okifuji (2022) discovered that, although there is a correlation with severe self-reported acute pain scores, the log-transformed LF and HF features do not correlate with moderate acute pain levels39.
Furthermore, Tracy et al. (2018) found that resting LF and HF features positively correlate with pain threshold in healthy subjects. However, they suggested that there are sex differences that need to be considered in HRV marker responses to pain40. Ling et al. (2014) particularly addressed the limitations of using HRV parameters in individuals taking antihypertensive agents, such as β-blockers. These medications can suppress the expected increase in heart rate and blood pressure in response to pain stimuli. In their study, they found no correlation between HRV markers and the reported pain intensity in postoperative patients41.
Photoplethysmography (PPG)
Photoplethysmography is a non-invasive optical technique that measures changes in light absorption or transmission caused by the volumetric changes in pulsating blood associated with the cardiac cycle. As a result, the PPG waveform is generated from light that is not absorbed by the irradiated tissue but rather scattered. This reflected or transmitted light is detected by an optical sensor, which processes it to indicate light absorbance. The intensity of light recorded is inversely proportional to the amount of light absorbed by the tissue42.
PPG’s potential use as a pain biomarker comes from its ability to monitor changes in the peripheral vascular system caused by pain. Noxious stimuli generating pain affect the peripheral vascular system by activating the sympathetic nervous system (SNS), a key regulator of the cardiovascular system that controls cardiac output and peripheral vascular resistance43,44. This activation induces vasoconstriction, reducing arterial compliance, a measure of arterial elasticity, and narrowing vessel diameter45. Those vascular changes associated with the pain sensation can be monitored through the analysis of the photoplethysmography signal46.
PPG signal can be analysed using a morphological approach. As illustrated in Fig. 3, the PPG signal can be decomposed into two major components associated with the absorbance of the pulsatile vs non-pulsatile tissue components: the AC and DC waves. The non-pulsatile wave (DC), or Baseline (BL), comes from the relatively constant light absorbed by the non-pulsatile tissue components. It is inversely proportional to the blood volume in the tissue41,42. It’s been reported to have a strong positive correlation with chronic pain30, acute pain in healthy subjects31 and postoperative pain47.
The figure shows a schematic of the characteristic morphology of a photoplethysmography waveform.
The pulsatile wave (AC) represents the arterial blood component that is synchronised with the heartbeat41,42. One of the most straightforward morphological features that can be extracted from the AC component is the PPG Amplitude (PPGA) – also referred to as Systolic Peak Amplitude (SPA), AC Amplitude from previous diastole (ACAdia), and pulse height. This amplitude has been shown to have an inverse correlation with sympathetic and vasoconstrictor nerve tone30. In addition, the ratio of the PPGA to the DC signal’s amplitude is defined as the AC/DC ratio or Perfusion Index (PI), which reflects changes in tissue microcirculation13.
Another parameter that can be extracted is the Pulse-Pulse Interval (PPI), also called Heart-Beat Interval (HBI), representing the time elapsed between successive pulses. The PPI can be systolic (PPIsys) if the interval is measured between systolic peaks or diastolic (PPIdia) if it’s calculated between diastolic peaks47. A negative correlation between this parameter and sympathetic tone has previously been reported30, as well as a significant negative correlation with chronic pain intensity29,30. However, no significant changes have been found between the diastolic and systolic components regarding postoperative pain47.
Paloheimo et al. (2010)48 introduced a numerical descriptor of the autonomic nervous system that uses the PPI and PPGA features to calculate the index, called Autonomic Nervous System State (ANSS). Equation 1 shows the formula to calculate this indicator, which has shown a negative correlation with sympathetic tone30.
Equation 1The equation to calculate the ANSS. The analysis is made pulse-by-pulse.
Overall, researchers have reported a significant negative correlation between acute pain intensity in healthy volunteers and PPGA31,45,49,50, and ANNS31,45. This indicates that as pain intensity increases, those variables tend to decrease. This negative correlation is also reported between postoperative pain intensity and PPGA41,47, ANNS, and AC/DC ratio41.
This behaviour may be linked to the excitability of the sympathetic nerves, which leads to a decrease in peripheral perfusion. This way, increased pain intensity could reduce the AC wave amplitude and the AC/DC value. In contrast to those outcomes, Kwon et al. (2019) found no significant correlation between postoperative pain and perfusion index or AC/DC ratio51.
On the other hand, Ye et al. (2017) found that, in acute pain scenarios, the negative correlation among the exposed features switches after a few minutes of applying the noxious stimuli, showing a positive correlation between pain intensity and PPGA/ANNS features associated with prolonged pain stimulation52. This trend is similar to the one reported in studies conducted on chronic pain patients undergoing radiofrequency therapy29, electrotherapy and thermotherapy30 for pain relief, where they found a significant decrease in PPGA and ANSS associated with chronic pain intensity reduction.
Another index developed from the PPGA and PPI features is the Surgical Pleth Index (SPI), formerly known as the Surgical Stress Index (SSI). It is a tool used to evaluate the effectiveness of intraoperative analgesia during general anaesthesia. It is particularly useful for detecting nociceptive events when administering total intravenous anaesthesia. The SPI values range from 0 to 100, reflecting the body’s level of surgical stress13,29. Equation 2 shows the equation used to calculate the SPI level, where \({{\rm{PPI}}}_{{\rm{norm}}}\) and \({{\rm{PPGA}}}_{{\rm{norm}}}\) denote the normalised values of the features.
Equation 2The equation to calculate the SPI.
Generally, studies have found statistically significant differences between pain and no pain states in studies conducted on chronic pain30, and postoperative pain34 patients. Nevertheless, the effectiveness of this parameter has been discussed since it has displayed notable interindividual variability compared to others, like ANI. Additionally, SPI alone has proven insufficient to distinguish moderate pain states34.
Yang et al. (2018) analysed morphological parameters that can be extracted from the PPG signal waveform. This study identified specific correlation patterns among some of the parameter groups and postoperative pain including: negative correlation with area-based parameters - PA, Area of a Systolic Phase (Asys), Area of a Diastolic Phase (Adia), Triangular Area of a Pulse (TriA), Triangular Area of a Systolic Phase (TriAsys), and Triangular Area of a Systolic Phase (TriAdia)-; a negative correlation with amplitude-based parameters -AC Amplitude from baseline (ACAbl)-; a negative correlation with slope-based parameters – Rising Slope (RS), Rising Slope Length (Lrs), Maximum Rising Slope (RSmax), Failing Slope (FS), and Failing Slope Length (Lfs)-47.
Researchers have also analysed combinations of morphological parameters in binary classification scenarios, finding that systolic peak variation (SYSvar, ACVsys/ACAdia), diastolic peak variation (DIAvar, ACVdia/ACAdia), PPI variation (HBIvar), ACAdia, ACVsys/ACAbl, ACVdia/ACAbl showed the best individual performance among all the combinations analysed47,53. In contrast, when trying to distinguish between three different pain levels, the best performance has been shown by median Atotal/ACAbl, max Asys/ACAbl, median Asys/ACAbl, median Adia/ACAbl, median (Atotal/ACAbl)/Lsys, median (Atotal/ACAbl)/Ldia, median (Atotal/ACAbl)/PPIonset, median (Asys/ACAbl)/PPIonset, median (Asys/ACAbl)/Ldia, median (Adia/ACAbl)/Lsys, median (Adia/ACAbl)/Ldia54.
Furthermore, Cho et al. (2018) introduced the Nasal Photopletismography Index (NPI), built based on the DIAvar and the PPIvar parameters. It uses a PPG sensor placed between the columella and the nasal septum. A value close to 100 represents severe pain, and 0 means no pain. They used this novel index in a study trying to assess postoperative pain intensity, showing a higher accuracy (75.3%) when compared to the SPI index (64.8%), whose PPG signals are acquired from a PPG finger probe53.
Equation 3Mathematical expression of the Nasal Photoplethysmography Index (NPI).
Pulse Rate Variability (PRV), a measurement of the changes in pulse rate (PR) over time, has been widely used in the last few years as a surrogate measure of Heart Rate Variability (HRV). Researchers often use both terms interchangeably, particularly when discussing measurements derived from the PPG waveform. However, a recent study has shown that PRV and HRV do not always provide the same information about autonomic activity, for example, under extreme weather conditions55. Therefore, this paper distinguishes the two measures based on the specific signals used for their calculations, regardless of the terminology employed in the original studies reviewed.
The features derived from HRV – See 4.2. Electrocardiography (ECG) - can also be calculated from PRV; however, PRV features are based on pulse rate signals rather than direct heart rate signals. Studies utilising time-domain parameters have revealed several key findings. Firstly, studies analysing Pulse Rate (PR) have found a strong positive correlation between this variable and chronic pain intensity30, but no significant correlation with acute pain in healthy volunteers23. Standard Deviation of the Normal-to-Normal Beat Interval (SDNN) and Square Root of the Mean Squared Differences of Successive NN intervals (RMSSD) have been associated with a positive statistical correlation to chronic pain intensity30. However, it’s been reported that RMSSD does not hold a significant correlation with postoperative pain56. Furthermore, the Average of the Peak-to-Peak intervals (AVNN) has shown a strong correlation with postoperative pain56. Finally, in terms of the PRV frequency domain parameters, High Frequency (HF), Low Frequency (LF) and Very Low Frequency (VLF) all reveal a significant negative correlation with chronic pain intensity29.
Ultimately, other features previously studied in pain assessment research include time domain features such as mean (M), variance (V), skewness (S), kurtosis (K), crest factor (CF), shape factor (SF), impulse factor (IF), margin factor (MF), Shannon energy (SE), log energy (LE), mobility (Mob), and complexity (Comp); and frequency domain features, like spectral flux (SpF), spectral crest (SpC), spectral flatness (SpFt), spectral centroid (SpCent), spectral kurtosis (SpK), spectral spread (SpSp), spectral roll-off (SpR), spectral slope (SpS), spectral decrease (SpD), spectral entropy (SpE), and mean frequency (SpM)23,57. Other time domain features with a potentially strong correlation include the pulse’s Rise Time (the average of the time between a valley and the next peak) and Fall Time (the average of the time between the peak and the next valley)56.
Electrodermal Activity (EDA)
Previously known as Galvanic Skin Response (GSR), EDA is a measurement of the electrical activity on the skin in response to sweat secretion. The reasoning behind this approach, as a potential signal for pain assessment, is that sweat glands are controlled by the sympathetic nervous system, with no contribution from the parasympathetic nervous system. Consequently, when a person experiences acute pain that activates the sympathetic nerve, it can affect the electrogalvanic properties of the skin due to changes in the eccrine sweat glands24,41.
Two methods are used for EDA measurements, based on measuring the electrical difference between two electrodes placed on the skin. In the exosomatic technique, a small electrical current from one of the electrodes is applied to the skin, and the resistance is then measured58. This is the most commonly used method, retrieving the Skin Conductance (SC), the reciprocal of skin resistance59. The signal obtained using this method has previously been studied for pain monitoring and has shown a significant correlation with acute pain intensity in healthy subjects23.
The second method, the endosomatic method, measures electrical skin potentials generated internally without applying an external current. This method is mainly used to measure Skin Potential (SP)59. This parameter, however, has shown no statistical correlation with acute pain in healthy volunteers60.
The EDA signal can be separated into its tonic (basal) and phasic components. The tonic component, called level (L), reflects the signal’s slow, almost DC variation (<0.05 Hz). The phasic component, named response (R), corresponds to the rapid variations of the signal (0.05-5 Hz). The Skin Conductance Response (SCR) is then the phasic component, and the Skin Conductance Level (SCL) is the tonic component of the EDA signal acquired using the exosomatic technique59,61. This last component has been attributed to a positive correlation with acute pain states62.
It is important to note that the decomposition techniques employed to extract the phasic and tonic components of the SC signal play a crucial role in the feasibility of those features as markers of pain. In this context, Posada-Quintero et al. (2021) discovered that features obtained through the sparse deconvolution approach (sparsEDA) exhibit greater classification power in distinguishing between pain and no pain states than those derived from the convex optimisation approach (cvxEDA)61. Furthermore, dynamic causal modelling (DCM) has been found to provide an SCR measure that is slightly more sensitive to acute pain levels when compared to sparsEDA, cvxEDA, and other methods like continuous decomposition (CDA) analysis and discrete decomposition analysis (DDA); it is also computationally demanding, making it less suitable for real-time applications63. In contrast to those findings, most articles reported the use of cvxEDA for SCR and/or SCL extraction36,61,64,65,66.
Most of the morphological features of the EDA signal are extracted from the SCR. One of them is the Amplitude of the SCR (SCR_Amp), which exhibits a positive statistical correlation with acute pain intensity in healthy volunteers60. The time interval between the onset of a new electrical variation in the SCR signal and the point at which it reaches its peak is known as the SCR rise time (SCR_Trise). Significant differences in this parameter have been identified between different acute pain intensities in healthy subjects60.
The derivative of the phasic component (dPhEDA) has also been presented as a potential marker for pain assessment, showing a positive correlation with pain sensation and stimulus61. Conversely, SCR latency, sum, average, and integrated ISCR -defined as the area of the detected phasic activity- have shown no statistically significant correlation with pain in chronic cancer patients32.
Researchers have also investigated time-series features. For example, Pouromran et al. (2021) analysed 22 features related to value distribution and outliers, simple temporal statistics, linear autocorrelation, successive differences, and fluctuation scaling properties. They identified three key features to train models designed to assess pain: the time interval between successive extreme events above the mean and below the mean, and the exponential fit to successive distances in 2-dimensional embedding space67.
Moreover, Posada-Quintero et al. (2021)68 introduced a new index of sympathetic activity based on time-frequency spectral analysis of the EDA signal, named the Time-Varying EDA Index of Sympathetic Control (TVSymp). A modified version, MTVSymp, is defined as the difference between TVSymp at a time (t) and the mean value of TVSymp over the previous five seconds. Both versions of the index have displayed a positive correlation with pain sensation and stimulus in healthy volunteers61. In contrast, TVSymp has shown a moderate positive correlation with acute tooth pain during cold stimulation but a weak correlation when electric pulp testing (EPT) was performed69.
Furthermore, the time-series spectrum of the SCR signal (TFS-phEDA) and its power coefficients have been reported to improve the performance of deep-learning pain detection models in comparison to models only using the phasic component (SCR) of the signal66.
Finally, it has been suggested that different information about pain perception may be extracted from both endosomatic and exosomatic EDA. Bari et al. (2018) proposed that the Skin Potential Relative Early Turn (SPRET) could serve as a score to predict pain sensation. This score is calculated using measures from both endosomatic and exosomatic EDA60. Equation 4 shows the formula to calculate this parameter, where SPR refers to the Skin Potential Response obtained using the endosomatic signal.
Equation 4Mathematical representation of the Skin Potential Relative Early Turn (SPRET).
Electroencephalography (EEG)
Electroencephalography is a technique that measures electrical activity in the brain. This is done by using a set of electrodes attached to the scalp, which record the electrical currents generated by cortical neurons. The electrodes are placed on the scalp in specific locations according to the standardised international 10-20 system to ensure consistent results across different EEG subjects. Each electrode placement point is labelled with a letter indicating the underlying brain region: ‘F’ for Frontal, ‘T’ for Temporal, ‘P’ for Parietal, and ‘O’ for Occipital. The numbers associated with each label indicate the brain hemisphere, with odd numbers representing the left and even numbers representing the right hemisphere70.
Changes in neural activity within the brain lead to fluctuations in the EEG signal amplitude over time. A power spectral analysis is typically performed to monitor these changes across signal frequency ranges. This method decomposes the EEG time signals into their constituent frequencies71. The EEG signals contain five principal frequency bands, each related to a specific brain function: Delta (~1–4 Hz), Theta (~4–8 Hz), Alpha (~8–13 Hz), Beta (~13-30 Hz), low Gamma (~30–50 Hz), and high Gamma (~52–100 Hz)23,72.
In comparison to other physiological signals, EEG directly shows neuronal activation associated with pain sensation, providing an advanced method for the processing of complex pain information71,73. Lin et al. (2022) identified a significant increase in the EEG power density around the parietal area over all bands and a significant decrease around the central parietal area in alpha, beta and gamma bands associated with pain increase in healthy volunteers23. Okolo & Omurtag (2018) reported a statistically significant decrease throughout the cortex for all bands associated with increasing pain intensity in healthy volunteers71.
Zhou et al. (2020) studied how frontal function preservation influences pain tolerance in healthy individuals as they age. They found that ageing and cognitive performance impact theta and low gamma brain wave activity, suggesting that these characteristics can affect pain tolerance72. In contrast, Wang et al. (2020) found that there are minimal significant differences in the frontal and temporal regions in healthy subjects undergoing acute pain sensation74.
Furthermore, Tripanpitak et al. (2020) introduced a method for signal feature extraction using a non-linear analysis to obtain information from the complex dynamic characteristics of brain waves. They explored various Fractal Dimension (FD) computational techniques and identified that the feature combinations derived from the correlation dimension, using a moving variance approach, yielded the best results75.
Later, Feng et al. (2021) studied the alpha-band oscillation changes between closed and open eyes conditions as a potential pain biomarker, observing a linear relationship between the parameter and perceived chronic pain intensity76.
Some indicators derived from EEG signals have been proposed for pain assessment. For example, Naro et al. (2017) examined the potential of the interpeak interval (IPI) variability between the N2 and P2 components of laser-evoked potentials (LEP) as an acute pain biomarker in patients with disorders of consciousness. Their findings indicated a statistically significant negative correlation between this marker and self-reported pain levels as measured by the Nociception Coma Scale-Revised (NCS-R)77.
Similarly, An et al. (2017) introduced the pain index (Pi) for pain recognition. The index gives a value between 0 and 100, calculated by taking the frequency band of the brain waves using a wavelength algorithm. The study found a statistically positive correlation between the Pi and the self-assessment pain score in chronic pain patients73.
In contrast, the Bispectral Index (BIS), commonly used in clinical settings to evaluate the level of consciousness in the cerebral cortex, has demonstrated no significant correlation with postoperative pain, indicating its unsuitability as a pain marker51.
Blood Pressure (BP)
Researchers have found a statistically significant increase in blood pressure associated with acute pain increase in healthy volunteers undergoing electrocutaneous49,50 and cold23 stimulation. Yet, Ling et al.41 discussed the unreliability of using this marker in individuals taking antihypertensive agents such as β-blockers. These agents can suppress the increase of heart and blood pressure relative to pain stimuli. They found no correlation between those markers and the reported pain intensity in postoperative patients.
Functional Near-Infrared Spectroscopy (fNIRS)
Researchers have shown interest in Functional Near-Infrared Spectroscopy to measure the variability of the nervous system associated with pain states. This optical technique measures changes in oxyhemoglobin (HbO) and deoxyhemoglobin concentrations78.
Pollonini et al. (2020) used this neuroimaging technique to explore the efficacy of non-pharmacological pain treatment for osteoarthritis patients that combines transcranial direct current stimulation (tDCS) and mindfulness-based meditation (MBM)79. Similarly, Du et al. (2023) employed this technique to study the hemodynamic effects of Transcutaneous Electrical Nerve Stimulation (TENS) for pain management in chronic pain patients, revealing that variation in pain sensation is associated with HbO levels in the prefrontal cortex78. Most recently, Khan et al. (2024) used the same TENS method, in this case, for pain stimulation in healthy volunteers, integrating ΔHbO2 and ΔHHB features80.
Signal combination features
Matsubara et al. (2018) proposed a log-linearised peripheral arterial viscoelastic model, named the Arterial Stiffness Index (β), that considers the nonlinear relationship between arterial wall impedance and blood pressure. The model uses the signals for PPG, ECG, and BP49. This model could accurately detect pain vs no-pain states49 showing a positive correlation with acute pain in healthy volunteers and with brain activity evoked by painful stimuli50.
Pulse Transit Time (PTT) refers to the time it takes for a pulse wave to travel from the heart to the peripheral arteries45,81. This parameter combines the ECG and PPG signals to calculate the pulse wave propagation time through the blood vessels. Research by Van Velzen et al. (2015) found that PTT decreased in response to painful stimuli and remains reduced even after those stimuli are reduced; while the marker’s immediate variation is slight, it becomes more significant over time45.
Other physiological techniques
Respiration Rate (Resp) is a physiological signal obtained through a sensor placed around the chest or abdomen. The strap experiences deformation due to the expansion of the rib cage, leading to alterations in the recorded voltage. This measurement has been researched for pain assessment; however, no statistical correlation has been found across different pain states in healthy volunteers23. Despite this, Luebke et al. (2023) proposed that this measure could be better suited for chronic pain patients, as the fluctuations in respiration caused by acute pain stimulation may be more significant in those with chronic pain than in healthy controls82.
Pupillometry refers to the measurement of variations in pupil size, pupillary reflexes, and Pupillary Unrest in Ambient Light (PUAL). This measurement is conducted using a portable infrared pupilometer. Pupil diameter has shown significant differences between different acute pain states in healthy volunteer23. In contrast, research using the amplitude variations of pupil oscillations, referred to as PUAL, has shown that postoperative pain reduction in chronic pain patients does not affect this parameter83.
Algorithms for pain assessment models
This section will discuss the latest advancements in the implementation of Machine Learning (ML) algorithms for pain recognition and assessment. Table 3 summarizes the findings related to algorithms implemented for pain detection and pain intensity evaluation. It also includes results from the best-performing models in each study, providing an illustrative comparison. From the 36 papers collected, 26 implemented ML for the assessment of acute pain, using traditional ML techniques (n = 14), deep learning approaches (n = 11), and a combination of both (n = 1). Additionally, seven studies addressed perioperative or postoperative pain, with the majority (71.4%, n = 5) relying primarily on photoplethysmography (PPG) signals. Only two studies explored the use of machine learning for chronic pain assessment, both utilising shallow learning methods. One study conducted a comparative analysis across multiple pain types, targeting both acute and chronic pain.
Shallow Learning Algorithms
Logistic regression is the most straightforward machine learning approach. It is a linear model used for binary classification, represented as a continuous function (See Eq. 5) that retrieves a value between 0 and 1. Seok et al. (2019) used the regression approach to build a model that uses PPG waveform features to assess postoperative pain84.
The algorithm can also be used for classification approaches by focusing the training objective on obtaining a binary outcome. Yang et al. (2018) implemented this algorithm as a classification model to train a model that takes PPG waveform features to differentiate between pain and no-pain states in patients undergoing surgery47. Similarly, Jhang et al. (2021) implemented the model to calculate the probability of pain in healthy volunteers using HRV and PPG features.
Equation 5Mathematical representation of the Logistic Classification Function.47
Linear Discriminant Analysis (LDA) is a linear binary classifier that uses a linear discriminant function to classify new data points. A hyperplane is constructed by finding a linear combination of features to separate or categorise two classes of objects or events85. Research has shown the use of this algorithm to develop binary classification models that differentiate between pain and no-pain states. Some aimed to analyse the predictive power of individual parameters extracted from PPG signals47. Chu et al. (2017) implemented this algorithm using a one-vs-rest strategy to assess different levels of pain using PPG, ECG and EDA signals as input85.
Random Forest (RF) is an algorithm that uses a collection of decision tree classifiers to categorise the input data into multiple classes that correspond to the branches of the trees. Van der Vaart et al. (2019) found that RF models to detect noxious stimulation in newborns showed better results using a multimodal approach instead of single-signal models86. A study conducted using the BioVid pain dataset showed that models trained using RF had better accuracy when compared to SVM, LR, DT, NB, KNNs87. Patterson et al. (2023) implemented this algorithm to train a model capable of assessing four different pain intensity states in chronic pain patients with SCS systems88.
Extreme Gradient Boosting Regression (XGBoost) is an ensemble technique based on the sequential addition of new models, or trees, to correct errors made by previous learning trees. It uses the gradient descent algorithm to optimise the loss function67,89. Light Gradient Boosting Machine (LightGBM) is a tree-based ensemble designed to overcome the efficiency and scalability difficulties of the XGBoost algorithm by implementing Gradient-based One-Side Sampling and Exclusive Feature Bundling90. Chu et al. (2024) implemented this algorithm to train a model that takes ECG signals and classifies pain and no pain states in women during labour, demonstrating better discriminant power compared to the models obtained using XGBoost, SnapDecision-Tree and SnapLogisticRegression algorithms91.
According to the literature review’s findings, one of the most commonly used shallow learning algorithms is the Support Vector Machine (SVM). It is a supervised machine learning algorithm, especially useful in binary Classification and Regression (SVR) applications. Some authors use this model to compare its performance with their proposed ML approaches. Others have adopted it as the primary algorithm for the development of models that incorporate PPG85, EEG71, and EDA57 signals alone or using a multimodal approach57,67,85.
Chu et al. (2017) found that this algorithm outperforms models trained with Linear Discriminant Analysis (LDA) and K-Nearest Neighbours (KNN) algorithms using ECG, PPG and EDA signals for acute pain intensity classification across multiple subjects and days85. Susam et al. (2022) proposed an architecture that combines a Linear SVM model with a Kernel Density Estimation (KDE) that processes the model’s output and weighted Bayesian fusion of class conditional distributions for maximum likelihood classification using EDA and Video as input92. Lin et al. (2022)
The SVM classifier algorithm has also been used as a cascade-based classification tree to differentiate among various pain states (non-binary approaches), employing unimodal inputs (solely fNIRS signals)80 and multimodal inputs23. This approach has shown better results when compared with KNN and Disc algorithms80.
Deep learning algorithms
Artificial neural networks (ANNs) are designed based on biological neural structures to learn and identify complex patterns within data. A typical feedforward neural network is structured with input, hidden, and output layers. The input layer receives the feature data and transmits it to the hidden layers, where the input weights are adjusted for optimisation. Ultimately, the processed information is sent to the output layer for final results56,93.
Extreme Learning Machine (ELM) is an algorithm designed for single-layer feedforward neural networks (SLFNs). This approach involves the random initialisation of the weights and biases of the hidden layer, followed by analytically determining the output weights through least-squares optimisation. Yu et al. (2020) studied the implementation of an ELM classifier and a kernel ELM classifier to classify four different acute pain states. Using EEG frequency bands. Their findings suggest that the kernel ELM classifier could be better for the task, even compared to the SVM model’s performance. Additionally, they identified that the Alpha and Beta bands were the most effective for pain prediction using these algorithms94.
Recurrent Neural Networks (RNNs) are designed to capture temporal relationships in sequential data. They are particularly beneficial for natural language processing and time series modelling. RNNs retain model outputs in internal memory and use them as additional input variables for predicting subsequent samples. Long Short-Term Memory (LSTM) algorithms are a specialised type of RNN that introduces a data-forgetting mechanism to overcome vanishing gradient problems, making it suitable for analysing temporal patterns over long sequences65.
Othman et al. (2022) analysed the performance of the LSTM algorithm, including a variation that incorporates sample weighting (LSTM-SW) across uni-, bi-, and multimodal approaches. Their findings suggest that the LSTM-SW algorithm is better for models combining two different modalities, namely EDA and Face Expression, in continuous pain intensity evaluation95. Furthermore, a combination of individually trained classification LSTM-SW models with Decision Fusion (DF) has also been implemented to increase performance in continuous pain monitoring96
Bidirectional LSTM (BiLSTM) algorithms have also been implemented independently and combined with XGBoost (BiLSTM-XGB) for a multiclass classification architecture using EDA signals, finding that BiLSTM-XGB using both whole signals and extracted features as input outperformed the model trained using BiLSTM alone65.
Multilayer Perceptron Neural Networks (MLPNN) is an algorithm that models biological brain structures. One input layer, at least one hidden layer, and an output layer according to a hierarchy. Weights indicate the degree of connection between each layer56. Jean et al. (2022) compared models derived from MLPNN and LSTM algorithms using the Analgesia Nociception Index as input. They found that the outcome score of the MLPNN model exhibited the strongest correlation with the Expert Assessment Patient Score (EAPS) used as a reference97.
In contrast, Lim et al. (2019) proposed the implementation of a Deep Belief Network (DBN) to model the complex relationship between physiological signals (PPG) and self-reported pain, demonstrating that this approach performs better than MLPNN. DBN is a probabilistic generative model built by stacking Restricted Boltzmann Machines (RBMs) layers. The process starts with pre-training using unsupervised learning to initialise the weights and bias of the model and supervised fine-tuning using the back-propagation algorithm of the MLPNN to minimise the error56.
Convolutional Neural Networks (CNNs or ConvNets) are algorithms inspired by biological NNs. They are designed to automatically and adaptively learn spatial hierarchies of features, also known as patterns, from visual data. CNNs’ architecture consists of layers processing the input data, with different layer types serving specific functions. The core layers, called Convolutional Layers, contain a set of neurons that scan the input sequentially and extract a particular pattern of information to build a set of feature maps98.
Several variations of this model have been implemented. Yu et al. (2020) successfully implemented diverse frequency band CNNs (DFB-ConvNets) models that extract features from each one of the bands that compose the EEG signal to classify acute pain between 3 different states99.
Subramaniam & Dass (2021) proposed a hybrid approach using CNN in combination with the LSTM algorithm named CNN_LSTM. This approach aimed to leverage the spatial feature extraction capabilities of the CNNs, along with the temporal sequence modelling ability of the LSTM to train models for binary classification between different pain states using ECG and EDA signals100. These algorithms have also been used in a parallel architecture incorporating a Temporal Convolutional Network (TCN) alongside a Stacked Bidirectional and Unidirectional LSTM (SBU-LSTM). This combined model, named Parallel TCN-SBU-LSTM, was used to detect acute pain using EDA signals66.
Moreover, Jiang et al. (2024) proposed an architecture that implemented a hybrid attention approach for pain intensity assessment using ECG and EDA signals. The algorithm contains a deep feature extraction module, temporal and channel attention modules, and a base module that uses temporal-channel attention weights to calculate the output101.
Metaheuristic hybrid approach
Genetic Algorithms fall into this category. A stochastic algorithm based on an optimisation criterion that simulates biological evolution selects fitter sets of solutions (also called chromosomes or individuals) in a population at each generation. Feature selection has an exponential search space, making the genetic algorithm a potential fit for this data analysis stage. Chu et al. (2017) implemented this algorithm alongside the Principal Component Analysis (PCA) to perform feature selection and reduction among 12 statistical features, each from PPG, ECG and EDA signals85.
Discussion
This literature review analysed the state of the art in pain assessment using physiological signals. While many physiological signals are currently explored for pain assessment, EDA has gathered considerable interest among other physiological signals for acute pain assessment in healthy subjects. A study comparing 112 features extracted from EDA, PPG and Resp signals found that statistical features from EDA are the most suitable for binary classification and pain intensity assessment in healthy subjects57. Similarly, studies conducted using the BioVid Heat Pain database have revealed that EDA is the most information-rich signal, compared with EMG and ECG signals, for pain classification and continuous pain intensity prediction in healthy volunteers67,98. Still, there is not enough evidence regarding the usefulness of this signal for chronic pain assessment paradigms.
Similarly, PPG signals show a high potential for use in pain intensity assessment. Nevertheless, there is still no conclusive evidence for the PPG features’ behaviour, as they show contrasting correlation feature behaviour, suggesting variability associated with different pain types. Furthermore, contrary to EDA, EMG, and ECG signals, before October 2024, there was no open-access pain database that included PPG signals for pain study, shortening the possibility of further analysing the potential of PPG for pain assessment.
Several physiological signals show strong potential for clinical translation in pain assessment; however, increased research focus is needed in this area, particularly for chronic pain. Of the studies reviewed, only 18 addressed chronic pain scenarios, with seven employing brain activity-related signals (EEG and fNIRS). Among these, EEG emerged as the most used modality, reflecting its value in capturing cortical responses to pain. While EEG offers important insights into the brain regions involved in pain processing, its use in long-term or continuous monitoring remains limited due to hardware complexity, susceptibility to artefacts, and poor wearability.
In contrast, HRV derived from ECG offers a more practical solution for wearable applications, reflecting autonomic responses to pain. However, HRV remains an indirect and non-specific marker, sensitive to confounding factors such as stress, respiration, physical activity, and emotional state. Its reliability in chronic pain contexts also requires further investigation.
Similarly, PPG signals—currently the most widely used in perioperative and postoperative monitoring—hold strong potential for broader application beyond surgical or ICU settings. PRV, extracted from PPG, presents a promising alternative to HRV, showing comparable trends under resting and controlled conditions, with the added advantage of easier integration into low-power wearable devices. However, like HRV, PRV is sensitive to multiple physiological and environmental confounders, and its correlation with HRV under dynamic or uncontrolled conditions has yet to be fully validated. This limits its current effectiveness in conscious and active states, highlighting the need for more targeted research before it can be reliably used in diverse clinical scenarios.
Likewise, the non-invasive nature and ease of integration into wearable devices of EDA sensors make it a practical choice for real-time monitoring in controlled settings, such as during experimental pain induction or perioperative care. However, the use of EDA in chronic pain scenarios remains limited, with relatively few studies exploring its reliability or clinical relevance in long-term conditions.
Overall, the combination of multiple modalities, such as ECG and PPG, or EDA and EMG, often yields improved accuracy compared to single-signal approaches, suggesting that multimodal strategies may be more promising for real-world applications. Despite these promising findings, several barriers hinder the translation of these approaches into routine clinical practice. These include high interindividual variability in physiological responses to pain and a lack of validation across diverse patient populations and clinical conditions, which limit generalizability. Further research is necessary to gain a comprehensive understanding of how different types of pain affect signal measurements, considering autonomic nervous system dysregulation102 and structural and functional brain alterations that can appear among different types of chronic pain37.
Generally, models and statistical analyses for various painful conditions rely on the self-assessment pain scale, i.e., the Numerical Rating Scale (NRS) or Visual Analogue Scale (VAS), as a measure of the actual pain reported by individuals. However, there is a subjective psychological component that affects the self-reported pain intensity, influenced by different social and psychological factors like anxiety, pain catastrophising and pain sensitivity3,6,103.
As a result, this approach could potentially introduce bias to the models when self-reported tools are used as the standard assessment methods for treatment and research purposes. Additionally, given that self-reporting scales and physiological signals may reflect different dimensions of pain, they are less likely to show a strong correlation when other measures that also reflect the subjective aspect are not incorporated into the analysis.
Moreover, psychophysical studies have demonstrated that there are gender differences in pain perception among adults, and it is necessary to carefully consider the interactions between gender and age in pain research104. Despite this evidence, only a limited number of studies have explored the role of gender, age and psychological phenomena like pain catastrophising and pain sensitivity in physiological signal variability and bias induction in pain assessment through self-reporting3,26,36,40,79,100,103. It is essential to address this issue during the model training process. One potential approach is to incorporate subject profiling as part of the data input, which could help mitigate the accuracy bias associated with interindividual variability.
Few measurements effectively predict pain intensity as a level indicator67. Most methodologies for pain assessment are based on binary classification, which categorises pain and no-pain states or between two different pain levels, like no-pain and low-pain or low-pain and high-pain. Unfortunately, this methodology often exhibits low performance in assessing mild and even moderate pain states. Additionally, this model is unsuitable for most clinical and research applications, where treatment relies on a fairly accurate objective analysis of pain. Thus, the main challenge remains in the objective quantification of pain intensity.
Given that pain is a multidimensional experience, multiple authors have explored combining different physiological signals and information sources, aiming to overcome the limitations of each signal. This approach aims to improve the sensitivity and specificity of the designed models for pain assessment. For instance, studies comparing unimodal and multimodal approaches generally show that multimodal methods enhance model outcomes, with some improvements being minimal while others are significant57,67,98. However, while taking this approach, it is essential to maintain a balance between incorporating more than one signal to enhance a model’s predictive capabilities and keeping the complexity of the technology manageable for practical, real-world applications.
In brief, this paper has reviewed the current state-of-the-art methodologies and techniques for the assessment of pain using physiological signals. Two main areas were addressed. A comprehensive analysis of the behaviour of different signal features related to pain states highlighted the differences among different pain types. It examined the current advances in machine learning approaches for the identification and measurement of pain using physiological signals. The review intended to highlight the currently available objective methods of acute and chronic pain assessment and the need to substantiate a method with better sensitivity and validity for clinical and research purposes.
It is important to note that no attempts were made to identify or translate non-English-language publications, which may have limited the inclusion of some relevant studies in this review. Also, publication bias may have occurred since only journal articles were included. Moreover, including only studies published since 2014 considerably restricted the number of studies analysed in this review. While this restriction reduced the total number of studies, it allowed us to focus on literature aligned with technological advancements from the past decade. Finally, despite our comprehensive search, we acknowledge that some relevant publications may not have been captured due to limitations in indexing and keywording. This is an inherent limitation of all literature reviews, including scoping reviews.
Methods
Review framework
The review was developed in accordance with the PRISMA extension for Scoping Reviews (PRISMA-ScR) guidelines105. The primary research question guiding this review was: “What are the current state-of-the-art methods for pain assessment using physiological signals?” In line with this question, the review aimed to analyse the behaviour of physiological signals across different pain types, highlighting key differences in signal patterns and features reported in the literature, and to summarise the machine learning models implemented in studies using physiological signals for pain assessment, including the modelling approaches, features used, and reported outcomes.
Literature search strategy
A literature search was conducted using four online databases: PubMed, Embase, Scopus, and Web of Science. Since this review focuses on analysing the latest technological advancements in pain assessment using these signals, only studies published within the last ten years (2014-2024) were included. The search was done in April 2024 and updated in October 2024. The strategy implemented was structured as shown in Fig. 4 where the keywords respond to the research question and the previously introduced aims.
The figure illustrates the search strategy used for the systematic review. Three categories of terms were combined: pain, assessment, and physiological signals. Terms for pain and assessment were restricted to article titles, while physiological signals were searched in titles, abstracts, and keywords. The dollar sign in “Signal$” indicates truncation, allowing the inclusion of variations such as “signal” and “signals”.
Eligibility criteria
The criteria for inclusion were as follows: (1) Studies that either (a) report statistically significant relationships or correlations between physiological signal variations and pain intensity or pain states, or (b) describe the development of machine learning models using physiological signals to assess pain. (3) Articles with full text accessible, (4) articles at the final stage of publication, (5) papers with available abstracts. All types of pain were included to create a comparative review that examines differences in signal patterns, feature behaviour, and trends in pain assessment methods across various pain types.
The criteria for exclusion were as follows: (1) conference abstracts, conference papers, letters, editorials, case-control studies, summaries, expert opinions, and comments; (2) studies conducted in animals; (3) studies not using physiological signals; (4) articles not available in English; and (5) full text not available/retrievable. Conference papers were excluded from this review because they often lack detailed methodology and complete experimental data. To ensure the inclusion of robust, fully developed studies that provide a complete picture of the research conducted, only peer-reviewed journal articles and full-length studies were considered.
Data charting and study selection
All search results were imported into Microsoft Excel for deduplication and screening. Reasons for exclusion at the full-text stage were recorded in an Excel spreadsheet. No specialised systematic review software was used. First, titles and abstracts were reviewed to exclude studies that were unrelated based on the predefined eligibility criteria. Second, the full texts of all potentially relevant articles were retrieved and assessed for final inclusion. No significant disagreements arose during the screening process. Minor uncertainties regarding eligibility were discussed and resolved by consensus among all authors.
Data extraction and synthesis
From each included study, the following information was extracted where reported: type of pain assessed, study population (volunteers or patients), sample size, participants’ age and sex distribution, methodological approach (pain modulation method), self-report instruments used as the reference for pain, whether the data comes from a publicly available database, physiological signals recorded, extracted physiological markers, and reported behaviour of signals or features.
For studies implementing machine learning models, we additionally extracted: the type of machine learning model applied, the modelling approach (e.g., pain detection, pain level classification including the number of levels, or pain quantification), features or markers used for training, testing and validation methodology, and reported model outcomes.
Finally, the extracted data were synthesised descriptively and grouped into two main thematic domains: 1) physiological signals used for pain assessment or modulation, and 2) machine learning models implemented for analysis or prediction. Findings are presented in summary tables and narrative form. Given the scoping nature of the review, no meta-analysis or formal critical appraisal was conducted; however, basic indicators of study context are reported to provide context for interpreting the evidence.
It is important to note that in this review, a feature or effect is described as statistically significant only if the original study explicitly reported a p-value less than or equal to 0.05. This ensures that statements about significance directly reflect the findings reported by the authors of the included studies.
Data availability
The datasets generated and/or analysed during the current study are either published in this article or available on the Zenodo research repository, 10.5281/zenodo.17135805.
References
Raja, S. N. et al. The Revised IASP definition of pain: concepts, challenges, and compromises HHS Public Access. Pain 161, 1976–1982 (2020).
Mccormick, T. & Frampton, C. Assessment of acute and chronic pain. Anaesth. Intensive Care Med. 20, 405–409 (2019).
Kakon, G. et al. Elevated Heart Rate and Pain During a Cold Pressor Test Correlates to Pain Catastrophizing. Appl Psychophysiol. Biofeedback 46, 359–366 (2021).
Fink, R. Pain assessment: the cornerstone to optimal pain management. Proc. (Bayl. Univ. Med. Cent.) 13, 236 (2000).
Wang, J. & Doan, L. V. Clinical pain management: Current practice and recent innovations in research. Cell Rep. Med 5, 101786 (2024).
Cimpean, A. & David, D. The mechanisms of pain tolerance and pain-related anxiety in acute pain. Health Psychol. Open 6, 1–13 (2019).
Fernandez Rojas, R., Brown, N., Waddington, G. & Goecke, R. A systematic review of neurophysiological sensing for the assessment of acute pain. NPJ Digit. Med. 6, 76 (2023).
Henderson, L. et al. Linking pain sensation to the autonomic nervous system: the role of the anterior cingulate and periaqueductal gray resting-state networks. Front Neurosci. 14, 147 (2020).
Hamunen, K. et al. Effect of pain on autonomic nervous system indices derived from photoplethysmography in healthy volunteers. Br. J. Anaesth. 108, 838–844 (2012).
Werner, P. et al. Automatic Recognition Methods Supporting Pain Assessment: A Survey. IEEE Trans. Affect Comput. 13, 530–552 (2022).
Xu, X. & Huang, Y. Objective Pain Assessment: a Key for the Management of Chronic Pain. F1000 Res. 9, 35 (2020).
Forte, G., Troisi, G., Pazzaglia, M., De Pascalis, V. & Casagrande, M. Heart Rate Variability and Pain: A Systematic Review. Brain Sci. 12, 153 (2022).
Argüello-Prada, E. J. & Molano Valencia, R. D. On the use of indexes derived from photoplethysmographic (PPG) signals for postoperative pain assessment: A narrative review. Biomed. Signal Process Control 80, 1746–8094 (2023).
Kong, Y. & Chon, K. H. Electrodermal activity in pain assessment and its clinical applications. Appl. Phys. Rev. 11, 031316 (2024).
Gélinas, C. et al. Exploration of a Multi-Parameter Technology for Pain Assessment in Postoperative Patients After Cardiac Surgery in the Intensive Care Unit: The Nociception Level Index (NOL). J. Pain. Res. 14, 3723–3731 (2021).
Santella, T. M., Richebé, P., Godin, N. & Brulotte, V. Nociception level index variations in patients with complex regional pain syndrome: a pilot study. J. Clin. Monit. Comput 36, 1851–1858 (2022).
Sakthivel, M. et al. An Objective Evaluation of Intraoperative and Postoperative Pain in Infants Undergoing Open Inguinal Herniotomy and Laparoscopic Inguinal Hernia Repair Using the Newborn Infant Parasympathetic Evaluation (NIPETM) Monitor. J. Pediatr. Surg. 161651 https://doi.org/10.1016/J.JPEDSURG.2024.07.036 (2024).
Medasense Biometrics Ltd. Introducing the NOL ® (Nociception Level) Index Algorithm. (2022).
Butruille, L. et al. Development of a pain monitoring device focused on newborn infant applications: The NeoDoloris project. IRBM 36, 80–85 (2015).
Walter, S. et al. The biovid heat pain database. in 2013 IEEE International Conference on Cybernetics, CYBCONF 2013 128–131 (IEEE, Laussane, 2013). https://doi.org/10.1109/CYBCONF.2013.6617456.
Gruss, S. et al. Multi-modal signals for analyzing pain responses to thermal and electrical stimuli. J. Vis. Exp. 2019, (2019).
Gouverneur, P. et al. An experimental and clinical physiological signal dataset for automated pain recognition. Sci. Data 2024 11, 1–13 (2024).
Lin, Y. et al. Experimental exploration of objective human pain assessment using multimodal sensing signals. Front. Neurosci. 16, 831627 (2022).
Walter, S. et al. Automatic pain quantification using autonomic parameters. Psychol. Neurosci. 7, 363–380 (2014).
Gruss, S. et al. Pain intensity recognition rates via biopotential feature patterns with support vector machines. PLoS One 10, e0140330 (2015).
Jurth, C., Rehberg, B. & Von Dincklage, F. Reliability of subjective pain ratings and nociceptive flexion reflex responses as measures of conditioned pain modulation. Pain. Res. Manag.: J. Can. Pain. Soc. 19, 93 (2014).
Mejia-Mejia, E., May, J. M., Torres, R. & Kyriacou, P. A. Pulse rate variability in cardiovascular health: a review on its applications and relationship with heart rate variability. Physiol. Meas. 41, 07TR01 (2020).
Cascella, M. et al. Advancing the integration of biosignal-based automated pain assessment methods into a comprehensive model for addressing cancer pain. BMC Palliat. Care 23, 1–14 (2024).
Ye, J. J., Chuang, C. C., Tai, Y. T., Lee, K. T. & Hung, K. S. Use of heart rate variability and photoplethysmograph-derived parameters as assessment signals of radiofrequency therapy efficacy for chronic pain. Pain. Pract. 17, 879–885 (2017).
Ye, J. J. et al. Assessing pain intensity using photoplethysmography signals in chronic myofascial pain syndrome. Pain. Pract. 18, 296–304 (2018).
Jhang, D. F., Chu, Y. S., Cai, J. H., Tai, Y. Y. & Chuang, C. C. Pain Monitoring Using Heart Rate Variability and Photoplethysmograph-Derived Parameters by Binary Logistic Regression. J. Med Biol. Eng. 41, 669–677 (2021).
Cascella, M. et al. Exploring biosignals for quantitative pain assessment in cancer patients: a proof of concept. Electronics (Switzerland) 12, (2023).
Camm, A. et al. Heart rate variability: standards of measurement, physiological interpretation, and clinical use. Circulation 93, 1043–1065 (1996).
Lee, J.-H. et al. Evaluation of Surgical Pleth Index and Analgesia Nociception Index as surrogate pain measures in conscious postoperative patients: an observational study. J. Clin. Monit. Comput. 34, 1087–1093 (2020).
Abdullayev, R., Uludag, O. & Celik, B. Analgesia Nociception Index: assessment of acute postoperative pain. Rev. Bras. Anestesiol. 69, 396–402 (2019).
Jiang, M. et al. Personalized and adaptive neural networks for pain detection from multi-modal physiological features. Expert Syst. Appl. 235, 121082 (2024).
Moens, M. et al. Heart rate variability is not suitable as a surrogate marker for pain intensity in patients with chronic pain. Pain 164, 1741–1749 (2023).
Billman, G. E. The LF/HF ratio does not accurately measure cardiac sympatho-vagal balance. Front Physiol. 4, 26 (2013).
Umeda, M. & Okifuji, A. Prediction of pain responses to subsequent cold pressor test via baseline heart rate variability in healthy adults. Eur. J. Pain. (U. Kingd.) 26, 1811–1820 (2022).
Tracy, L. M., Koenig, J., Georgiou-Karistianis, N., Gibson, S. J. & Giummarra, M. J. Heart rate variability is associated with thermal heat pain threshold in males, but not females. Int. J. Psychophysiol. 131, 37–43 (2018).
Ling, P., Siyuan, Y., Wei, W., Quan, G. & Bo, G. Assessment of postoperative pain intensity by using photoplethysmography. J. Anesth. 28, 846–853 (2014).
Kyriacou, P. A. & Chatterjee, S. The origin of photoplethysmography. in Photoplethysmography: Technology, Signal Analysis and Applications (eds. Kyriacou, P. & Allen, J.) 17–42 (Academic Press). https://doi.org/10.1016/B978-0-12-823374-0.00004-9 (2022).
Schlereth, T. & Birklein, F. The sympathetic nervous system and pain. Neuromolecular Med 10, 141–147 (2008).
Jaenig, W. & Baron, R. Sympathetic Nervous System and Pain. in Encyclopedia of Pain (eds. Gebhart, G. & Schmidt, R.) 3763–3779 (Springer, Berlin, 2013). https://doi.org/10.1007/978-3-642-28753-4_4327.
van Velzen, M. H. et al. Effect of heat-induced pain stimuli on pulse transit time and pulse wave amplitude in healthy volunteers. Physiol. Meas. 37, 52 (2015).
Liu, B. et al. The Assessment of Autonomic Nervous System Activity Based on Photoplethysmography in Healthy Young Men. Front Physiol. 12, 733264 (2021).
Yang, Y. et al. Postoperative Pain Assessment Indices Based on Photoplethysmography Waveform Analysis. Front. Physiol. 9, 1199 (2018).
Paloheimo, M. P. J., Sahanne, S. & Uutela, K. H. Autonomic nervous system state: The effect of general anaesthesia and bilateral tonsillectomy after unilateral infiltration of lidocaine. Br. J. Anaesth. 104, 587–595 (2010).
Matsubara, H. et al. Quantitative evaluation of pain during electrocutaneous stimulation using a log-linearized peripheral arterial viscoelastic model. Sci. Rep. 8, 3091 (2018).
Tsuji, T. et al. Peripheral arterial stiffness during electrocutaneous stimulation is positively correlated with pain-related brain activity and subjective pain intensity: an fMRI study. Sci. Rep. 11, 4425 (2021).
Kwon, J. H. et al. Evaluation of the Intraoperative Perfusion Index for Correlation with Acute Postoperative Pain in Patients Undergoing Laparoscopic Colorectal Cancer Surgery. J. Clin. Med. 8, 1299 (2019).
Ye, J. J., Lee, K. T., Lin, J. S. & Chuang, C. C. Observing continuous change in heart rate variability and photoplethysmography-derived parameters during the process of pain production/relief with thermal stimuli. J. Pain. Res 10, 527–533 (2017).
Choi, B. M. et al. Development of a new analgesic index using nasal photoplethysmography. Anaesthesia 73, 1123–1130 (2018).
Ryu, G. et al. Analysis of Photoplethysmography-Based Surgical Pain Severity Assessment Markers. J. Electr. Eng. Technol. 19, 3665–3674 (2024).
Mejia-Mejia, E., Budidha, K., Abay, T. Y., May, J. M. & Kyriacou, P. A. Heart Rate Variability (HRV) and Pulse Rate Variability (PRV) for the Assessment of Autonomic Responses. Front. Physiol. 11, 779 (2020).
Lim, H., Kim, B., Noh, G.-J. & Yoo, S. K. A Deep Neural Network-Based Pain Classifier Using a Photoplethysmography Signal. Sensors 19, 384 (2019).
Fernandez Rojas, R. et al. Multimodal physiological sensing for the assessment of acute pain. Front. Pain Res. 4, 1150264 (2023).
Dawson, M. E., Schell, A. M. & Filion, D. L. The Electrodermal System. In Handbook of Psychophysiology (eds. Cacioppo, J., Tassinary, L. & Berntson, G.) 217–243 (Cambridge University Press). https://doi.org/10.1017/9781107415782.010 (2017).
Stern, R. M., Ray, W. J. & Quigley, K. S. Skin: Electrodermal Activity. in Psychophysiological Recording (eds. Stern, R., Ray, W. & Quigley, K.) 206–220 (Oxford University Press, New York). https://doi.org/10.1093/ACPROF:OSO/9780195113594.003.0013 (2000).
Bari, D. S. et al. Electrodermal activity responses for quantitative assessment of felt pain. J. Electr. Bioimp 9, 52–58 (2018).
Posada-Quintero, H. F., Kong, Y. & Chon, K. H. Objective pain stimulation intensity and pain sensation assessment using machine learning classification and regression based on electrodermal activity. Am. J. Physiol. 321, R186–R196 (2021).
Bhatkar, V., Picard, R. & Staahl, C. Combining electrodermal activity with the peak-pain time to quantify three temporal regions of pain experience. Front. Pain Res. 3, 764128 (2022).
Kong, Y., Posada-Quintero, H. F. & Chon, K. H. Sensitive Physiological Indices of Pain based on differential characteristics of electrodermal activity. IEEE Trans. Biomed. Eng. 68, 3122–3130 (2021).
Aqajari, S. A. H. et al. Pain Assessment Tool With Electrodermal Activity for Postoperative Patients: Method Validation Study. JMIR Mhealth Uhealth 9, 25258 (2021).
Pouromran, F., Lin, Y. & Kamarthi, S. Personalized Deep Bi-LSTM RNN based model for pain intensity classification using EDA signal. Sensors 22, 8087 (2022).
Pinzon-Arenas, J. O., Kong, Y., Chon, K. H. & Posada-Quintero, H. F. Design and evaluation of deep learning models for continuous acute pain detection based on phasic electrodermal activity. IEEE J. Biomed. Health Inf. 27, 4250–4260 (2023).
Pouromran, F., Radhakrishnan, S. & Kamarthi, S. Exploration of physiological sensors, features, and machine learning models for pain intensity estimation. PLoS One 16, e0254108 (2021).
Posada-Quintero, H. F., Florian, J. P., Orjuela-Cañón, ÁD. & Chon, K. H. Highly sensitive index of sympathetic activity based on time-frequency spectral analysis of electrodermal activity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 311, R582–R591 (2016).
Tran, H. T. et al. The use of electrodermal activity in pulpal diagnosis and dental pain assessment. Int. Endod. J. 56, 356 (2023).
Sazgar, M. & Young, M. G. Overview of EEG, Electrode Placement, and Montages. Absolute Epilepsy and EEG Rotation Review 117–125. https://doi.org/10.1007/978-3-030-03511-2_5 (2019).
Okolo, C. & Omurtag, A. Use of Dry Electroencephalogram and Support Vector for Objective Pain Assessment. Biomed. Instrum. Technol. 52, 372–378 (2018).
Zhou, S. et al. Involvement of frontal functions in pain tolerance in aging: evidence from neuropsychological assessments and Gamma-band oscillations. Front Aging Neurosci. 12, 131 (2020).
An, J. X., Wang, Y., Cope, D. K. & Williams, J. P. Quantitative evaluation of pain with Pain Index Extracted from Electroencephalogram. Chin. Med J. (Engl.) 130, 1926 (2017).
Wang, L., Xiao, Y., Urman, R. D. & Lin, Y. Cold pressor pain assessment based on EEG power spectrum. SN Appl Sci. 2, 1–8 (2020).
Tripanpitak, K., Viriyavit, W., Huang, S. Y. & Yu, W. Classification of Pain Event Related Potential for Evaluation of Pain Perception Induced by Electrical Stimulation. Sensors 20, 1491 (2020).
Feng, L. et al. Low back pain assessment based on alpha oscillation changes in spontaneous Electroencephalogram (EEG). Neural Plast. 2021, 8537437 (2021).
Naro, A., Bramanti, P., Bramanti, A. & Calabrò, R. S. Assessing pain in patients with chronic disorders of consciousness: Are we heading in the right direction?. Conscious Cogn. 55, 148–155 (2017).
Du, J., Shi, P., Fang, F. & Yu, H. Cerebral cortical hemodynamic metrics to aid in assessing pain levels? A pilot study of functional near-infrared spectroscopy. Front Neurosci. 17, 1136820 (2023).
Pollonini, L. et al. Functional near-infrared spectroscopy to assess central pain responses in a non-pharmacologic treatment trial of osteoarthritis. J. Neuroimaging 30, 808 (2020).
Khan, M. U. et al. Multilevel Pain Assessment with Functional Near-Infrared Spectroscopy: Evaluating ΔHBO2 and ΔHHB Measures for Comprehensive Analysis. Sensors 24, 458 (2024).
Mejía-Mejía, E. et al. Photoplethysmography signal processing and synthesis. in Photoplethysmography: Technology, Signal Analysis and Applications (eds. Kyriacou, P. & Allen, J.) 69–146 (Academic Press). https://doi.org/10.1016/B978-0-12-823374-0.00015-3 (2022).
Luebke, L. et al. Objective measurement of subjective pain perception with autonomic body reactions in healthy subjects and chronic back pain patients: an experimental heat pain study. Sensors 23, 8231 (2023).
Behrends, M. & Larson, M. D. Measurements of pupillary unrest using infrared pupillometry fail to detect changes in pain intensity in patients after surgery: a prospective observational study. Can. J. Anaesth. 71, 611 (2024).
Seok, H. S., Choi, B. M., Noh, G. J. & Shin, H. Postoperative pain assessment model based on pulse contour characteristics analysis. IEEE J. Biomed. Health Inf. 23, 2317–2324 (2019).
Chu, Y., Zhao, X., Han, J. & Su, Y. Physiological signal-based method for measurement of pain intensity. Front. Neurosci. 11, 279 (2017).
van der Vaart, M. et al. Multimodal pain assessment improves discrimination between noxious and non-noxious stimuli in infants. Paediatr. Neonatal Pain. 1, 21–30 (2019).
Albahdal, D., Aljebreen, W. & Ibrahim, D. M. PainMeter: automatic assessment of pain intensity levels from multiple physiological signals using machine learning. IEEE Access 12, 48349–48365 (2024).
Patterson, D. G. et al. Objective wearable measures correlate with self-reported chronic pain levels in people with spinal cord stimulation systems. NPJ Digit. Med. 6, 146 (2023).
Shi, H., Chikhaoui, B. & Wang, S. Tree-based models for pain detection from biomedical signals. Lect. Notes Computer Sci. (including Subser. Lect. Notes Artif. Intell. Lect. Notes Bioinforma.) 13287, 183–195 (2022).
Ke, G. et al. LightGBM: A Highly Efficient Gradient Boosting Decision Tree. in 31st Conference on Neural Information Processing Systems (Long Beach, 2017).
Chu, Y. C. et al. Enhanced labor pain monitoring using machine learning and ECG waveform analysis for uterine contraction-induced pain. BioData Min. 17, 32 (2024).
Susam, B. et al. Automated pain assessment in children using electrodermal activity and video data fusion via machine learning. IEEE Trans. Biomed. Eng. 69, 422–431 (2022).
Khan, M. U. et al. Experimental exploration of multilevel human pain assessment using Blood Volume Pulse (BVP) Signals. Sensors 23, 3980 (2023).
Yu, M. et al. EEG-based tonic cold pain assessment using extreme learning machine. Intell. Data Anal. 24, 163–182 (2020).
Othman, E., Werner, P., Saxen, F., Fiedler, M. A. & Al-Hamadi, A. An automatic system for continuous pain intensity monitoring based on analyzing data from uni-, bi-, and multi-modality. Sensors 22, 4492 (2022).
Othman, E. et al. Automated electrodermal activity and facial expression analysis for continuous pain intensity monitoring on the X-ITE Pain Database. Life 13, 1828 (2023).
Jean, W. H., Sutikno, P., Fan, S. Z., Abbod, M. F. & Shieh, J. S. Comparison of deep learning algorithms in predicting expert assessments of pain scores during surgical operations using Analgesia Nociception Index. Sensors 22, 5496 (2022).
Thiam, P., Bellmann, P., Kestler, H. A. & Schwenker, F. Exploring deep physiological models for nociceptive pain recognition. Sensors 2019 19, 4503 (2019).
Yu, M. et al. Diverse frequency band-based convolutional neural networks for tonic cold pain assessment using EEG. Neurocomputing 378, 270–282 (2020).
Subramaniam, S. D. & Dass, B. Automated nociceptive pain assessment using physiological signals and a hybrid deep learning network. IEEE Sens J. 21, 3335–3343 (2021).
Jiang, M. et al. Physiological time-series fusion with hybrid attention for adaptive recognition of pain. IEEE J. Biomed. Health Inform. (2024). https://doi.org/10.1109/JBHI.2024.3456441 (2024).
Yeater, T. D. et al. Chronic pain is associated with reduced sympathetic nervous system reactivity during simple and complex walking tasks: potential cerebral mechanisms. Chronic Stress 5, 1–8 (2021).
Etherton, J., Lawson, M. & Graham, R. Individual and gender differences in subjective and objective indices of pain: Gender, fear of pain, pain catastrophizing and cardiovascular reactivity. Appl. Psychophysiol. Biofeedback 39, 89–97 (2014).
Failla, M. D. et al. Gender Differences in Pain Threshold, Unpleasantness, and Descending Pain Modulatory Activation Across the Adult Life Span: A Cross Sectional Study. J. Pain. 25, 1059–1069 (2024).
Tricco, A. C. et al. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation. Ann. Intern Med 169, 467–473 (2018).
Deepika, C. L. & Brindha, D. Design of Non-invasive Multi-parameter Pain Monitoring System. J. Sci. Ind. Res 74, 330–333 (2015).
Choi, B. M., Yim, J. Y., Shin, H. & Noh, G. Novel Analgesic Index for Postoperative Pain Assessment Based on a Photoplethysmographic Spectrogram and Convolutional Neural Network: Observational Study. J. Med. Internet Res. 23, 23920 (2021).
Hamed, M. A. et al. Correlation of different time measurements of the surgical pleth index with postoperative pain: a prospective observational study. Open Anesth. J. 18, 25896458318051 (2024).
Badura, A., Bienkowska, M., Mysliwiec, A. & Pietka, E. Continuous short-term pain assessment in temporomandibular joint therapy using LSTM models supported by heat-induced pain data patterns. IEEE Trans. Neural Syst. Rehabil. Eng. https://doi.org/10.1109/TNSRE.2024.3461589. (2024).
Wu, F. et al. Learning Spatial-Spectral-Temporal EEG representations with deep attentive-recurrent-convolutional neural networks for pain intensity assessment. Neuroscience 481, 144–155 (2022).
Valentini, E. et al. Assessing the specificity of the relationship between brain alpha oscillations and tonic pain. Neuroimage 255, 119143 (2022).
Tan, B. et al. Differential analgesic effects of high-frequency or accelerated intermittent theta burst stimulation of M1 on experimental tonic pain: Correlations with cortical activity changes assessed by TMS-EEG. Neurotherapeutics 21, 00451 (2024).
Chen, L. et al. PainVision-based evaluation of brain potentials: a novel approach for quantitative pain assessment. Front Bioeng Biotechnol. 11, 1197070 (2023).
Mujib, M. D. et al. Comparative neurological and behavioral assessment of central and peripheral stimulation technologies for induced pain and cognitive tasks. Biomedicines 2024 12, 1269 (2024).
Fu, Z. et al. A Spatiotemporal Deep Learning Framework for Scalp EEG-based Automated Pain Assessment in Children. IEEE Trans. Biomed. Eng. 71, 1889–1900 (2024).
Coviello, C. et al. Evaluation of the relationship between pain exposure and somatosensory evoked potentials in preterm infants: a prospective cohort study. Children 11, 676 (2024).
May, E. S. et al. Dynamics of brain function in patients with chronic pain assessed by microstate analysis of resting-state electroencephalography. Pain 162, 2894 (2021).
Jessen, J. et al. Posttraumatic headache: pain related evoked potentials (PREP) and conditioned pain modulation (CPM) to assess the pain modulatory function. Sci. Rep. 14, 16306 (2024).
Hunt, A. M. G. et al. Neuronal effects of listening to entrainment music versus preferred music in patients with chronic cancer pain as measured via EEG and LORETA Imaging. Front Psychol. 12, 588788 (2021).
Pu, L., Lion, K. M., Todorovic, M. & Moyle, W. Portable EEG monitoring for older adults with dementia and chronic pain - A feasibility study. Geriatr. Nurs. 42, 124–128 (2021).
Gkikas, S. et al. Multimodal automatic assessment of acute pain through facial videos and heart rate signals utilizing transformer-based architectures. Front. Pain. Res. 5, 1372814 (2024).
Rokicki, J. P., Ivanauskas, A. & Januzis, G. The role of facial expression analysis and electrodermal activity as an objective evaluation of persistent idiopathic facial pain. J. Craniofac. Surg. 33, e14 (2022).
MacNeill, A. L. & Mayich, D. J. A physiological assessment of patient pain during surgery with wide-awake local anesthesia. J. Orthop. 19, 158–161 (2020).
Rousseaux, F. et al. Virtual reality hypnosis in the management of pain: Self-reported and neurophysiological measures in healthy subjects. Eur. J. Pain 27, 148–162 (2023).
Sen, K. & Pal, S. Alternative method for pain assessment using EMG and GSR. J. Mech. Med. Biol. 21, 2150039 (2021).
Acknowledgements
This research is part of a project co-funded by City St George’s, University of London and Barts Health NHS Trust. The authors declare that they have no known competing financial or non-financial interests that could influence the work reported in this paper.
Author information
Authors and Affiliations
Contributions
C.C-N. Conceptualisation, study design, literature search, data extraction, data curation and data interpretation. Writing - original draft, review & editing. L.L. Data interpretation. Writing - review & editing. K.P. & V.M. Writing - review & editing. Funding acquisition. P.A.K. Conceptualisation and data curation. Writing - review & editing. Project administration and funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Camacho-Navas, C., Li, L., Poply, K. et al. Pain assessment using physiological responses/markers in different types of pain: a scoping review. npj Digit. Med. 9, 64 (2026). https://doi.org/10.1038/s41746-025-02241-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41746-025-02241-6