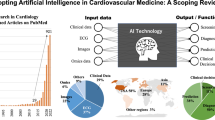
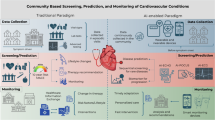
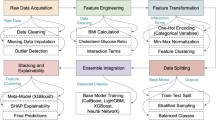
Abstract
Cardiovascular disease remains the leading cause of death and disability worldwide. The convergence of big data and artificial intelligence (AI) is reshaping precision cardiovascular medicine through multimodal integration of electronic health records (EHRs), imaging, omics, and wearable data across the care continuum, enabling predictive, diagnostic, therapeutic, and system-level optimization. However, translation into durable clinical benefit remains constrained by evidentiary gaps, implementation complexity, and fragmented governance architectures.
Similar content being viewed by others
Data availability
There are no research data in this paper.
References
Mensah, G. A., Roth, G. A. & Fuster, V. The global burden of cardiovascular diseases and risk factors: 2020 and beyond. J. Am. Coll. Cardiol. 74, 2529–2532 (2019).
Partridge, L., Deelen, J. & Slagboom, P. E. Facing up to the global challenges of ageing. Nature 561, 45–56 (2018).
Li, Y., Cao, G. Y., Jing, W. Z., Liu, J. & Liu, M. Global trends and regional differences in incidence and mortality of cardiovascular disease, 1990-2019: findings from 2019 global burden of disease study. Eur. J. Prev. Cardiol. 30, 276–286 (2023).
Chong, B. et al. Global burden of cardiovascular diseases: projections from 2025 to 2050. Eur. J. Prev. Cardiol. 32, 1001–1015 (2025).
Wang, R. S., Maron, B. A. & Loscalzo, J. Multiomics network medicine approaches to precision medicine and therapeutics in cardiovascular diseases. Arterioscler. Thromb. Vasc. Biol. 43, 493–503 (2023).
Doenst, T., Kirov, H., Bagiella, E., Scherag, A. & Omerovic, E. Challenges of conventional and novel approaches to clinical trial designs in cardiovascular medicine. Eur. J. Cardiothorac. Surg. 67, ezaf056 (2025).
Fountzilas, E., Tsimberidou, A. M., Vo, H. H. & Kurzrock, R. Clinical trial design in the era of precision medicine. Genome Med. 14, 101 (2022).
Duan, X. P. et al. New clinical trial design in precision medicine: discovery, development and direction. Signal Transduct. Target Ther. 9, 57 (2024).
Maiorino, E. & Loscalzo, J. Phenomics and robust multiomics data for cardiovascular disease subtyping. Arterioscler. Thromb. Vasc. Biol. 43, 1111–1123 (2023).
Nagarajan, V. D. et al. Artificial intelligence in the diagnosis and management of arrhythmias. Eur. Heart J. 42, 3904–3916 (2021).
Hosny, A., Parmar, C., Quackenbush, J., Schwartz, L. H. & Aerts, H. Artificial intelligence in radiology. Nat. Rev. Cancer 18, 500–510 (2018).
Topol, E. J. High-performance medicine: the convergence of human and artificial intelligence. Nat. Med. 25, 44–56 (2019).
Deng, J. et al. So you’ve got a high AUC, now what? an overview of important considerations when bringing machine-learning models from computer to bedside. Med. Decis. Mak. 45, 640–653 (2025).
Borges do Nascimento, I. J. et al. Barriers and facilitators to utilizing digital health technologies by healthcare professionals. NPJ Digit. Med. 6, 161 (2023).
Rosenthal, J. T., Beecy, A. & Sabuncu, M. R. Rethinking clinical trials for medical AI with dynamic deployments of adaptive systems. NPJ Digit. Med. 8, 252 (2025).
Dainis, A. M. & Ashley, E. A. Cardiovascular precision medicine in the genomics era. JACC Basic Transl. Sci. 3, 313–326 (2018).
Shameer, K., Johnson, K. W., Glicksberg, B. S., Dudley, J. T. & Sengupta, P. P. Machine learning in cardiovascular medicine: are we there yet? Heart 104, 1156–1164 (2018).
Sarker, I. H. Machine learning: algorithms, real-world applications and research directions. SN Comput. Sci. 2, 160 (2021).
Johnson, K. W. et al. Artificial intelligence in cardiology. J. Am. Coll. Cardiol. 71, 2668–2679 (2018).
LeCun, Y., Bengio, Y. & Hinton, G. Deep learning. Nature 521, 436–444 (2015).
Botvinick, M. et al. Reinforcement learning, fast and slow. Trends Cogn. Sci. 23, 408–422 (2019).
Leopold, J. A. & Loscalzo, J. Emerging role of precision medicine in cardiovascular disease. Circ. Res. 122, 1302–1315 (2018).
Hemingway, H. et al. Big data from electronic health records for early and late translational cardiovascular research: challenges and potential. Eur. Heart J. 39, 1481–1495 (2018).
Caufield, J. H. et al. Cardiovascular informatics: building a bridge to data harmony. Cardiovasc. Res. 118, 732–745 (2022).
Cowie, M. R. et al. Electronic health records to facilitate clinical research. Clin. Res. Cardiol. 106, 1–9 (2017).
Silverio, A., Cavallo, P., De Rosa, R. & Galasso, G. Big health data and cardiovascular diseases: a challenge for research, an opportunity for clinical care. Front. Med. 6, 36 (2019).
Denaxas, S. C. & Morley, K. I. Big biomedical data and cardiovascular disease research: opportunities and challenges. Eur. Heart J. Qual. Care Clin. Outcomes 1, 9–16 (2015).
Tsao, C. W. & Vasan, R. S. Cohort profile: the Framingham Heart Study (FHS): overview of milestones in cardiovascular epidemiology. Int. J. Epidemiol. 44, 1800–1813 (2015).
Sudlow, C. et al. UK Biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12, e1001779 (2015).
Brendel, J. M. et al. Coronary artery disease detection using deep learning and ultrahigh-resolution photon-counting coronary CT angiography. Diagn. Inter. Imaging 106, 68–75 (2025).
Attia, Z. I. et al. Screening for cardiac contractile dysfunction using an artificial intelligence-enabled electrocardiogram. Nat. Med. 25, 70–74 (2019).
Guazzo, A. et al. Deep-learning-based natural-language-processing models to identify cardiovascular disease hospitalisations of patients with diabetes from routine visits’ text. Sci. Rep. 13, 19132 (2023).
Wu, J. et al. Multi-rater prism: learning self-calibrated medical image segmentation from multiple raters. Sci. Bull. 69, 2906–2919 (2024).
Houssein, E. H., Mohamed, R. E. & Ali, A. A. Heart disease risk factors detection from electronic health records using advanced NLP and deep learning techniques. Sci. Rep. 13, 7173 (2023).
Hongn, A., Bosch, F., Prado, L. E., Ferrandez, J. M. & Bonomini, M. P. Wearable physiological signals under acute stress and exercise conditions. Sci. Data 12, 520 (2025).
Kuppe, C. et al. Spatial multi-omic map of human myocardial infarction. Nature 608, 766–777 (2022).
Joshi, A., Rienks, M., Theofilatos, K. & Mayr, M. Systems biology in cardiovascular disease: a multiomics approach. Nat. Rev. Cardiol. 18, 313–330 (2021).
Dong, Y. et al. Single-cell profile reveals the landscape of cardiac immunity and identifies a cardio-protective Ym-1(hi) neutrophil in myocardial ischemia-reperfusion injury. Sci. Bull. 69, 949–967 (2024).
Wang, W. et al. Spatiotemporal transcriptomics unveil gelsolin as a critical regulator in ventricular remodeling after myocardial infarction. Sci. China Life Sci. 68, 1835–1838 (2025).
Jin, K. et al. Single-cell RNA sequencing reveals the temporal diversity and dynamics of cardiac immunity after myocardial infarction. Small Methods 6, e2100752 (2022).
Li, Y. et al. Oral-gut microbial transmission promotes diabetic coronary heart disease. Cardiovasc. Diabetol. 23, 123 (2024).
Wang, Z. et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 472, 57–63 (2011).
Ji, W. et al. Air pollution, lifestyle, and cardiovascular disease risk in northwestern China: a cohort study of over 5.8 million participants. Environ. Int. 199, 109459 (2025).
Lehne, M., Sass, J., Essenwanger, A., Schepers, J. & Thun, S. Why digital medicine depends on interoperability. NPJ Digit Med 2, 79 (2019).
Bender, D. & Sartipi, K. in Proc. 26th IEEE International Symposium on Computer-Based Medical Systems 326-331 (IEEE, 2013).
Ahmadi, N. et al. Methods used in the development of common data models for health data: scoping review. JMIR Med. Inf. 11, e45116 (2023).
Ward, R., Hallinan, C. M., Ormiston-Smith, D., Chidgey, C. & Boyle, D. The OMOP common data model in Australian primary care data: building a quality research ready harmonised dataset. PLoS ONE 19, e0301557 (2024).
Li, Y. et al. A review of deep learning-based information fusion techniques for multimodal medical image classification. Comput. Biol. Med. 177, 108635 (2024).
Shaik, T., Tao, X., Li, L., Xie, H. & Velásquez, J. D. A survey of multimodal information fusion for smart healthcare: mapping the journey from data to wisdom. Inf. Fusion 102, 102040 (2024).
Zhou, Y. et al. Applying multimodal AI to physiological waveforms improves genetic prediction of cardiovascular traits. Am. J. Hum. Genet. 112, 1562–1579 (2025).
DeGroat, W. et al. Multimodal AI/ML for discovering novel biomarkers and predicting disease using multi-omics profiles of patients with cardiovascular diseases. Sci. Rep. 14, 26503 (2024).
Kline, A. et al. Multimodal machine learning in precision health: a scoping review. NPJ Digit. Med. 5, 171 (2022).
Stahlschmidt, S. R., Ulfenborg, B. & Synnergren, J. Multimodal deep learning for biomedical data fusion: a review. Brief Bioinform 23, bbab569 (2022).
Guarrasi, V. et al. A systematic review of intermediate fusion in multimodal deep learning for biomedical applications. Image Vis. Comput. 158, 32 (2025).
Nahai, F. General data protection regulation (GDPR) and data breaches: what you should know. Aesthet. Surg. J. 39, 238–240 (2019).
Voigt, P. & Bussche, A. V. D. The EU General Data Protection Regulation (GDPR): A Practical Guide (Springer, 2017).
Marks, M. & Haupt, C. E. AI chatbots, health privacy, and challenges to HIPAA compliance. JAMA 330, 309–310 (2023).
Health informatics - pseudonymization (ISO 25237:2017). (2017).
Atreya, R. V., Smith, J. C., McCoy, A. B., Malin, B. & Miller, R. A. Reducing patient re-identification risk for laboratory results within research datasets. J. Am. Med. Inf. Assoc. 20, 95–101 (2013).
El Emam, K. & Dankar, F. K. Protecting privacy using k-anonymity. J. Am. Med. Inf. Assoc. 15, 627–637 (2008).
Machanavajjhala, A., Kifer, D., Gehrke, J. & Venkitasubramaniam, M. L-diversity: privacy beyond k-anonymity. ACM Trans. Knowl. Discov. Data 1, 3–es (2007).
Dyda, A. et al. Differential privacy for public health data: an innovative tool to optimize information sharing while protecting data confidentiality. Patterns 2, 100366 (2021).
Shen, A., Francisco, L., Sen, S. & Tewari, A. Exploring the relationship between privacy and utility in mobile health: algorithm development and validation via simulations of federated learning, differential privacy, and external attacks. J. Med. Internet Res. 25, e43664 (2023).
Bai, Y., Zhao, H., Shi, X. & Chen, L. Towards practical and privacy-preserving CNN inference service for cloud-based medical imaging analysis: a homomorphic encryption-based approach. Comput. Methods Prog. Biomed. 261, 108599 (2025).
Cepova, L. et al. Improving privacy-preserving multi-faceted long short-term memory for accurate evaluation of encrypted time-series MRI images in heart disease. Sci. Rep. 14, 20218 (2024).
Froelicher, D. et al. Truly privacy-preserving federated analytics for precision medicine with multiparty homomorphic encryption. Nat. Commun. 12, 5910 (2021).
Joly, Y., Dyke, S. O. M., Knoppers, B. M. & Pastinen, T. Are data sharing and privacy protection mutually exclusive? Cell. 167, 1150–1154 (2016).
Psarra, E., Apostolou, D., Verginadis, Y., Patiniotakis, I. & Mentzas, G. Permissioned blockchain network for proactive access control to electronic health records. BMC Med. Inf. Decis. Mak. 24, 303 (2024).
Fairbairn, T. A. et al. Real-world clinical utility and impact on clinical decision-making of coronary computed tomography angiography-derived fractional flow reserve: lessons from the ADVANCE Registry. Eur. Heart J. 39, 3701–3711 (2018).
Benton, S. M. Jr. et al. Noninvasive derivation of fractional flow reserve from coronary computed tomographic angiography: a review. J. Thorac. Imaging. 33, 88–96 (2018).
Bastiaansen, J. A. M., van Heeswijk, R. B., Stuber, M. & Piccini, D. Noncontrast free-breathing respiratory self-navigated coronary artery cardiovascular magnetic resonance angiography at 3 T using lipid insensitive binomial off-resonant excitation (LIBRE). J. Cardiovasc. Magn. Reson. 21, 38 (2019).
Gulati, M. et al. 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR guideline for the evaluation and diagnosis of chest pain: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 144, e368–e454 (2021).
Eng, D. et al. Automated coronary calcium scoring using deep learning with multicenter external validation. NPJ Digit. Med. 4, 88 (2021).
Li, Y. et al. Automatic coronary artery segmentation and diagnosis of stenosis by deep learning based on computed tomographic coronary angiography. Eur. Radiol. 32, 6037–6045 (2022).
Zou, L. M. et al. Ultra-low-dose coronary CT angiography via super-resolution deep learning reconstruction: impact on image quality, coronary plaque, and stenosis analysis. Eur. Radiol. 35, 4674–4684 (2025).
Williams, M. C. et al. Low-attenuation noncalcified plaque on coronary computed tomography angiography predicts myocardial infarction: results from the multicenter SCOT-HEART trial (Scottish computed tomography of the HEART). Circulation 141, 1452–1462 (2020).
Alven, J. et al. PlaqueViT: a vision transformer model for fully automatic vessel and plaque segmentation in coronary computed tomography angiography. Eur. Radiol. 35, 4461–4471 (2025).
Lee, J. et al. Automated analysis of fibrous cap in intravascular optical coherence tomography images of coronary arteries. Sci. Rep. 12, 21454 (2022).
Niioka, H. et al. Automated diagnosis of optical coherence tomography imaging on plaque vulnerability and its relation to clinical outcomes in coronary artery disease. Sci. Rep. 12, 14067 (2022).
Hong, H. et al. Risk stratification in acute coronary syndrome by comprehensive morphofunctional assessment with optical coherence tomography. JACC Asia 2, 460–472 (2022).
Park, S. et al. A novel deep learning model for a computed tomography diagnosis of coronary plaque erosion. Sci. Rep. 13, 22992 (2023).
Wang, Z. et al. Semiautomatic segmentation and quantification of calcified plaques in intracoronary optical coherence tomography images. J. Biomed. Opt. 15, 061711 (2010).
Hwang, Y. N., Lee, J. H., Kim, G. Y., Shin, E. S. & Kim, S. M. Characterization of coronary plaque regions in intravascular ultrasound images using a hybrid ensemble classifier. Comput. Methods Prog. Biomed. 153, 83–92 (2018).
Gessert, N. et al. Automatic plaque detection in IVOCT pullbacks using convolutional neural networks. IEEE Trans. Med. Imaging 38, 426–434 (2019).
Chu, M. et al. Artificial intelligence and optical coherence tomography for the automatic characterisation of human atherosclerotic plaques. EuroIntervention 17, 41–50 (2021).
Koch, V. et al. Deep learning model DeepNeo predicts neointimal tissue characterization using optical coherence tomography. Commun. Med. 5, 124 (2025).
Khera, A. V. et al. Genetic risk, adherence to a healthy lifestyle, and coronary disease. N. Engl. J. Med. 375, 2349–2358 (2016).
Khera, A. V. et al. Genome-wide polygenic scores for common diseases identify individuals with risk equivalent to monogenic mutations. Nat. Genet. 50, 1219–1224 (2018).
Inouye, M. et al. Genomic risk prediction of coronary artery disease in 480,000 adults: implications for primary prevention. J. Am. Coll. Cardiol. 72, 1883–1893 (2018).
Lu, X. et al. A polygenic risk score improves risk stratification of coronary artery disease: a large-scale prospective Chinese cohort study. Eur. Heart J. 43, 1702–1711 (2022).
Patel, A. P. et al. A multi-ancestry polygenic risk score improves risk prediction for coronary artery disease. Nat. Med. 29, 1793–1803 (2023).
Lee, M., Park, T., Shin, J. Y. & Park, M. A comprehensive multi-task deep learning approach for predicting metabolic syndrome with genetic, nutritional, and clinical data. Sci. Rep. 14, 17851 (2024).
Oh, H. S. et al. Organ aging signatures in the plasma proteome track health and disease. Nature 624, 164–172 (2023).
Nurmohamed, N. S. et al. Polygenic risk is associated with long-term coronary plaque progression and high-risk plaque. JACC Cardiovasc. Imaging 17, 1445–1459 (2024).
Chan, K. et al. Inflammatory risk and cardiovascular events in patients without obstructive coronary artery disease: the ORFAN multicentre, longitudinal cohort study. Lancet 403, 2606–2618 (2024).
Pezel, T. et al. A machine learning model using cardiac CT and MRI data predicts cardiovascular events in obstructive coronary artery disease. Radiology 314, e233030 (2025).
Chen, S. F. et al. Meta-prediction of coronary artery disease risk. Nat. Med. 31, 2277–2288 (2025).
Zhang, J. et al. A non-invasive prediction model for coronary artery stenosis severity based on multimodal data. Front. Physiol. 16, 1592593 (2025).
Sardar, P. et al. Impact of artificial intelligence on interventional cardiology: from decision-making aid to advanced interventional procedure assistance. JACC Cardiovasc. Interv. 12, 1293–1303 (2019).
Spitaleri, G. et al. Quantitative flow ratio identifies nonculprit coronary lesions requiring revascularization in patients with ST-segment-elevation myocardial infarction and multivessel disease. Circ. Cardiovasc. Interv. 11, e006023 (2018).
Jie, L. et al. Using machine learning to aid treatment decision and risk assessment for severe three-vessel coronary artery disease. J. Geriatr. Cardiol. 19, 367–376 (2022).
Ghasemi, P. et al. Personalized decision making for coronary artery disease treatment using offline reinforcement learning. NPJ Digit. Med. 8, 99 (2025).
Migliaro, S. et al. Comparing AI-driven and heart team decision-making in multivessel coronary artery disease. J. Clin. Med. 14, 4452 (2025).
Li, F. et al. Dynamic prognosis prediction for patients on dapt after drug-eluting stent implantation: model development and validation. J. Am. Heart Assoc. 13, e029900 (2024).
van den Broek, W. W. A. et al. Cost-effectiveness of implementing a genotype-guided de-escalation strategy in patients with acute coronary syndrome. Eur. Heart J. Cardiovasc. Pharmacother. 11, 230–240 (2025).
Savarese, G. et al. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc. Res. 118, 3272–3287 (2023).
Campbell, P., Rutten, F. H., Lee, M. M., Hawkins, N. M. & Petrie, M. C. Heart failure with preserved ejection fraction: everything the clinician needs to know. Lancet 403, 1083–1092 (2024).
Roh, J., Hill, J. A., Singh, A., Valero-Munoz, M. & Sam, F. Heart failure with preserved ejection fraction: heterogeneous syndrome, diverse preclinical models. Circ. Res. 130, 1906–1925 (2022).
Choi, E., Schuetz, A., Stewart, W. F. & Sun, J. Using recurrent neural network models for early detection of heart failure onset. J. Am. Med. Inf. Assoc. 24, 361–370 (2017).
Blecker, S. et al. Early identification of patients with acute decompensated heart failure. J. Card. Fail. 24, 357–362 (2018).
Rao, S. et al. An explainable transformer-based deep learning model for the prediction of incident heart failure. IEEE J. Biomed. Health Inf. 26, 3362–3372 (2022).
Li, Y. et al. Hi-BEHRT: hierarchical transformer-based model for accurate prediction of clinical events using multimodal longitudinal electronic health records. IEEE J. Biomed. Health Inf. 27, 1106–1117 (2023).
Stehlik, J. et al. Continuous wearable monitoring analytics predict heart failure hospitalization: the LINK-HF multicenter study. Circ. Heart Fail 13, e006513 (2020).
Pan, X. et al. Deep cross-modal feature learning applied to predict acutely decompensated heart failure using in-home collected electrocardiography and transthoracic bioimpedance. Artif. Intell. Med. 140, 102548 (2023).
Fox, H. et al. Early identification of heart failure deterioration through respiratory monitoring with adaptive servo-ventilation. J. Sleep. Res. 32, e13749 (2023).
Dhingra, L. S. et al. Artificial intelligence-enabled prediction of heart failure risk from single-lead electrocardiograms. JAMA Cardiol. 10, 574–584 (2025).
Poterucha, T. J. et al. Detecting structural heart disease from electrocardiograms using AI. Nature 644, 221–230 (2025).
Oexner, R. R. et al. Serum metabolomics improves risk stratification for incident heart failure. Eur. J. Heart Fail. 26, 829–840 (2024).
Emilsson, V. et al. Proteomic prediction of incident heart failure and its main subtypes. Eur. J. Heart Fail. 26, 87–102 (2024).
Ouyang, D. et al. Video-based AI for beat-to-beat assessment of cardiac function. Nature 580, 252–256 (2020).
Akerman, A. P. et al. Automated echocardiographic detection of heart failure with preserved ejection fraction using artificial intelligence. JACC Adv. 2, 100452 (2023).
Akerman, A. P. et al. External validation of artificial intelligence for detection of heart failure with preserved ejection fraction. Nat. Commun. 16, 2915 (2025).
Lau, E. S. et al. Deep learning-enabled assessment of left heart structure and function predicts cardiovascular outcomes. J. Am. Coll. Cardiol. 82, 1936–1948 (2023).
Downie, C. G. et al. Molecular phenogroups in heart failure: large-scale proteomics in a population-based cohort. Circ. Genom. Precis. Med. 18, e004953 (2025).
Kobayashi, M. et al. Machine learning-derived echocardiographic phenotypes predict heart failure incidence in asymptomatic individuals. JACC Cardiovasc. Imaging 15, 193–208 (2022).
Shah, S. J. et al. Phenomapping for novel classification of heart failure with preserved ejection fraction. Circulation 131, 269–279 (2015).
Banerjee, A. et al. Identifying subtypes of heart failure from three electronic health record sources with machine learning: an external, prognostic, and genetic validation study. Lancet Digit. Health 5, e370–e379 (2023).
Fan, Z., Mamouei, M., Li, Y., Rao, S. & Rahimi, K. Identification of heart failure subtypes using transformer-based deep learning modelling: a population-based study of 379,108 individuals. EBioMedicine 114, 105657 (2025).
Li, R. et al. Machine learning-based phenotyping and assessment of treatment responses in heart failure with preserved ejection fraction. eClinicalMedicine 88, 103462 (2025).
Bertrand, A. et al. Sex-specific cardiometabolic multimorbidity, metabolic syndrome and left ventricular function in heart failure with preserved ejection fraction in the UK Biobank. Cardiovasc. Diabetol. 24, 238 (2025).
Gevaert, A. B. et al. Clinical phenogroups are more effective than left ventricular ejection fraction categories in stratifying heart failure outcomes. ESC Heart Fail. 8, 2741–2754 (2021).
Sotomi, Y. et al. Medications for specific phenotypes of heart failure with preserved ejection fraction classified by a machine learning-based clustering model. Heart 109, 1231–1240 (2023).
Karwath, A. et al. Redefining beta-blocker response in heart failure patients with sinus rhythm and atrial fibrillation: a machine learning cluster analysis. Lancet 398, 1427–1435 (2021).
Choy, M. et al. Phenotypes of heart failure with preserved ejection fraction and effect of spironolactone treatment. ESC Heart Fail. 9, 2567–2575 (2022).
Chowdhury, S. et al. Stratifying heart failure patients with graph neural network and transformer using electronic health records to optimize drug response prediction. J. Am. Med. Inf. Assoc. 31, 1671–1681 (2024).
Solomon, S. D. et al. Angiotensin-neprilysin inhibition in heart failure with preserved ejection fraction. N. Engl. J. Med. 381, 1609–1620 (2019).
Jhund, P. S. et al. Dapagliflozin across the range of ejection fraction in patients with heart failure: a patient-level, pooled meta-analysis of DAPA-HF and DELIVER. Nat. Med. 28, 1956–1964 (2022).
Poh, M. Z. et al. Diagnostic assessment of a deep learning system for detecting atrial fibrillation in pulse waveforms. Heart 104, 1921–1928 (2018).
Guo, Y. et al. Mobile photoplethysmographic technology to detect atrial fibrillation. J. Am. Coll. Cardiol. 74, 2365–2375 (2019).
van Vliet, M. et al. Ambulatory atrial fibrillation detection and quantification by wristworn AI device compared to standard holter monitoring. NPJ Digit. Med. 8, 177 (2025).
Gavidia, M. et al. Early warning of atrial fibrillation using deep learning. Patterns 5, 100970 (2024).
Attia, Z. I. et al. An artificial intelligence-enabled ECG algorithm for the identification of patients with atrial fibrillation during sinus rhythm: a retrospective analysis of outcome prediction. Lancet 394, 861–867 (2019).
Khurshid, S. et al. ECG-based deep learning and clinical risk factors to predict atrial fibrillation. Circulation 145, 122–133 (2022).
Krasteva, V., Stoyanov, T., Naydenov, S., Schmid, R. & Jekova, I. Detection of atrial fibrillation in Holter ECG recordings by ECHOView images: a deep transfer learning study. Diagnostics 15, 865 (2025).
Lim, J., Han, D. & Chon, K. H. QRS-centric beat-wise atrial fibrillation detection in ECG signals using deep neural networks. Comput. Biol. Med. 192, 110282 (2025).
Lin, C., Lu, H., Sang, P. & Pan, C. A knowledge embedded multimodal pseudo-siamese model for atrial fibrillation detection. Sci. Rep. 15, 3133 (2025).
Veltkamp, R. et al. Direct oral anticoagulants versus no anticoagulation for the prevention of stroke in survivors of intracerebral haemorrhage with atrial fibrillation (PRESTIGE-AF): a multicentre, open-label, randomised, phase 3 trial. Lancet 405, 927–936 (2025).
Pirruccello, J. P. et al. Deep learning of left atrial structure and function provides link to atrial fibrillation risk. Nat. Commun. 15, 4304 (2024).
Yuan, N. et al. Deep learning evaluation of echocardiograms to identify occult atrial fibrillation. NPJ Digit. Med. 7, 96 (2024).
Schoels, M. et al. Artificial intelligence for prediction of atrial fibrillation in the stroke unit: a retrospective derivation validation cohort study. EBioMedicine 118, 105869 (2025).
Zhang, Z. et al. Development of an MRI based artificial intelligence model for the identification of underlying atrial fibrillation after ischemic stroke: a multicenter proof-of-concept analysis. EClinicalMedicine 81, 103118 (2025).
Pieszko, K. et al. Artificial intelligence in detecting left atrial appendage thrombus by transthoracic echocardiography and clinical features: the left atrial thrombus on transoesophageal echocardiography (LATTEE) registry. Eur. Heart J. 45, 32–41 (2024).
Zhao, Y. et al. Harnessing risk assessment for thrombosis and bleeding to optimize anticoagulation strategy in nonvalvular atrial fibrillation. Thromb. Haemost. 125, 492–504 (2025).
Somani, S. et al. Understanding reasons for oral anticoagulation nonprescription in atrial fibrillation using large language models. J. Am. Heart Assoc. 14, e040419 (2025).
Liu, W. T. et al. Artificial intelligence-enabled ECGs for atrial fibrillation identification and enhanced oral anticoagulant adoption: a pragmatic randomized clinical trial. J. Am. Heart Assoc. 14, e042106 (2025).
Santamaria, A. et al. LOLATAO-an artificial-intelligence-based virtual assistant for clinical follow-up of patients with non-valvular atrial fibrillation (AF) undergoing oral anticoagulant therapy (OAT): a feasibility study. J. Clin. Med. 14, 3023 (2025).
Deisenhofer, I. et al. Artificial intelligence for individualized treatment of persistent atrial fibrillation: a randomized controlled trial. Nat. Med. 31, 1286–1293 (2025).
Gutierrez-Fernandez-Calvillo, M. et al. Non-invasive estimation of atrial fibrillation driver position using long-short term memory neural networks and body surface potentials. Comput Methods Prog. Biomed. 246, 108052 (2024).
Narita, M. et al. Comparison of the characteristics between machine learning and deep learning algorithms for ablation site classification in a novel cloud-based system. Heart Rhythm 22, e572–e577 (2025).
Zhou, X. et al. Deep learning-based recurrence prediction of atrial fibrillation after catheter ablation. Circ. J. 86, 299–308 (2022).
Tang, S. et al. Machine learning-enabled multimodal fusion of intra-atrial and body surface signals in prediction of atrial fibrillation ablation outcomes. Circ. Arrhythm. Electrophysiol. 15, e010850 (2022).
Zhou, Z. et al. Multimodal prediction of catheter ablation outcomes in patients with persistent atrial fibrillation. Neural Netw. 191, 107835 (2025).
Poplin, R. et al. Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nat. Biomed. Eng. 2, 158–164 (2018).
Zekavat, S. M. et al. Deep learning of the retina enables phenome- and genome-wide analyses of the microvasculature. Circulation 145, 134–150 (2022).
Yoshihara, H., Tsugawa, Y., Fukuda, M., Okiyama, S. & Nakayama, T. Detection of hypertension from pharyngeal images using deep learning algorithm in primary care settings in Japan.BMJ Health Care Inform. 31, e100824 (2024).
Ding, Y. et al. Using deep learning to screen OCTA images for hypertension to reduce the risk of serious complications. Front. Cell Dev. Biol. 13, 1581785 (2025).
Reel, P. S. et al. Machine learning for classification of hypertension subtypes using multi-omics: a multi-centre, retrospective, data-driven study. EBioMedicine 84, 104276 (2022).
Wu, L. et al. Differential diagnosis of secondary hypertension based on deep learning. Artif. Intell. Med. 141, 102554 (2023).
Ye, C. et al. Prediction of incident hypertension within the next year: prospective study using statewide electronic health records and machine learning. J. Med. Internet Res. 20, e22 (2018).
Datta, S. et al. Predicting hypertension onset from longitudinal electronic health records with deep learning. JAMIA Open 5, ooac097 (2022).
Hrytsenko, Y. et al. Machine learning models for predicting blood pressure phenotypes by combining multiple polygenic risk scores. Sci. Rep. 14, 12436 (2024).
Hall, J. M. et al. Unsupervised learning using EHR and census data to identify distinct subphenotypes of newly diagnosed hypertension patients. PLoS ONE 20, e0326776 (2025).
Writing Committee, M. AHA/ACC/AANP/AAPA/ABC/ACCP/ACPM/AGS/AMA/ASPC/NMA/PCNA/SGIM guideline for the prevention, detection, evaluation and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines.Circulation 152, e114–e218 (2025).
Thomas, S. S. et al. BioWatch: a noninvasive wrist-based blood pressure monitor that incorporates training techniques for posture and subject variability. IEEE J. Biomed. Health Inf. 20, 1291–1300 (2016).
Wang, L., Tian, S. & Zhu, R. A new method of continuous blood pressure monitoring using multichannel sensing signals on the wrist. Microsyst. Nanoeng. 9, 117 (2023).
Li, J. et al. Thin, soft, wearable system for continuous wireless monitoring of artery blood pressure. Nat. Commun. 14, 5009 (2023).
Li, S. et al. Monitoring blood pressure and cardiac function without positioning via a deep learning-assisted strain sensor array. Sci. Adv. 9, eadh0615 (2023).
Cavero-Redondo, I. et al. Antihypertensive drug recommendations for reducing arterial stiffness in patients with hypertension: machine learning-based multicohort (RIGIPREV) study. J. Med. Internet Res. 26, e54357 (2024).
Chunyu, L. et al. Characterizing the critical features when personalizing antihypertensive drugs using spectrum analysis and machine learning methods. Artif. Intell. Med. 104, 101841 (2020).
Yi, J. et al. Development of a machine learning-based model for predicting individual responses to antihypertensive treatments. Nutr. Metab. Cardiovasc. Dis. 34, 1660–1669 (2024).
Song, J. et al. Learning implementation of a guideline based decision support system to improve hypertension treatment in primary care in China: pragmatic cluster randomised controlled trial. BMJ 386, e079143 (2024).
van Vliet, M. et al. Assessment of photoplethysmography-based blood pressure determinations during long-term and short-term remote cardiac monitoring: the RECAMO study. Eur. Heart J. Digit. Health 6, 763–771 (2025).
Teng, T. Q. et al. Efficiency of remote monitoring and guidance in blood pressure management: a randomized controlled trial : the role of remote monitoring in improving hypertension management. BMC Med. 23, 459 (2025).
Mikulski, B. S., Bellei, E. A., Biduski, D. & De Marchi, A. C. B. Mobile health applications and medication adherence of patients with hypertension: a systematic review and meta-analysis. Am. J. Prev. Med. 62, 626–634 (2022).
Eze, C. E. et al. eHealth literacy and participation in remote blood pressure monitoring among patients with hypertension: cross-sectional study. J. Med. Internet Res. 27, e71926 (2025).
Wang, Y. et al. Hyper-DREAM, a multimodal digital transformation hypertension management platform integrating large language model and digital phenotyping: multicenter development and initial validation study. J. Med. Syst. 49, 42 (2025).
Baharoon, M. et al. HyMNet: a multimodal deep learning system for hypertension prediction using fundus images and cardiometabolic risk factors. Bioengineering 11, 1080 (2024).
Wang, J. et al. Multimodal data-driven, vertical visualization prediction model for early prediction of atherosclerotic cardiovascular disease in patients with new-onset hypertension. J. Hypertens. 42, 1757–1768 (2024).
Wang, Y. et al. Efficacy of a WeChat-based multimodal digital transformation management model in new-onset mild to moderate hypertension: randomized clinical trial. J. Med. Internet Res. 25, e52464 (2023).
Mathkor, D. M. et al. Multirole of the internet of medical things (IoMT) in biomedical systems for managing smart healthcare systems: an overview of current and future innovative trends. J. Infect. Public Health 17, 559–572 (2024).
Xie, X. et al. Efforts to improve survival outcomes of out-of-hospital cardiac arrest in China: BASIC-OHCA. Circ. Cardiovasc. Qual. Outcomes 16, e008856 (2023).
Kass-Hout, T. A., Stevens, L. M. & Hall, J. L. American Heart Association precision medicine platform. Circulation 137, 647–649 (2018).
Ahalt, S. et al. Building a collaborative cloud platform to accelerate heart, lung, blood, and sleep research. J. Am. Med. Inf. Assoc. 30, 1293–1300 (2023).
Semler, S. C. et al. [The medical informatics initiative at a glance-establishing a health research data infrastructure in Germany]. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 67, 616–628 (2024).
Seth, M. et al. Technologies for interoperable internet of medical things platforms to manage medical emergencies in home and prehospital care: scoping review. J. Med. Internet Res. 27, e54470 (2025).
Facile, R., Chronaki, C., van Reusel, P. & Kush, R. Standards in sync: five principles to achieve semantic interoperability for TRUE research for healthcare. Front. Digit. Health 7, 1567624 (2025).
Lin, H. et al. Using big data to improve cardiovascular care and outcomes in China: a protocol for the CHinese Electronic Health Records Research in Yinzhou (CHERRY) Study. BMJ Open 8, e019698 (2018).
All of Us Research Program, I. The “All of Us” research program. N. Engl. J. Med. 381, 668–676 (2019).
Klein, D. et al. Building a digital health research platform to enable recruitment, enrollment, data collection, and follow-up for a highly diverse longitudinal US Cohort of 1 million people in the All of Us Research Program: design and implementation study. J. Med. Internet Res. 27, e60189 (2025).
Sun, T. et al. Development of a health behavioral digital intervention for patients with hypertension based on an intelligent health promotion system and WeChat: randomized controlled trial. JMIR Mhealth Uhealth 12, e53006 (2024).
Al Khatib, H. S. et al. Patient-centric knowledge graphs: a survey of current methods, challenges, and applications. Front. Artif. Intell. 7, 1388479 (2024).
Kuan, P. X. et al. Efficacy of telemedicine for the management of cardiovascular disease: a systematic review and meta-analysis. Lancet Digit. Health 4, e676–e691 (2022).
Ong, M. K. et al. Effectiveness of remote patient monitoring after discharge of hospitalized patients with heart failure: the better effectiveness after transition—heart failure (BEAT-HF) randomized clinical trial. JAMA Intern. Med. 176, 310–318 (2016).
WHO Guideline: Recommendations on Digital Interventions for Health System Strengthening (WHO, 2019).
Wah, J. N. K. Revolutionizing e-health: the transformative role of AI-powered hybrid chatbots in healthcare solutions. Front. Public Health 13, 1530799 (2025).
Yamashita, S. et al. Electronic patient-reported outcome system implementation in outpatient cardiovascular care: a randomized clinical trial. JAMA Netw. Open 8, e2454084 (2025).
Fonseca, M. et al. The use of online consultation systems or remote consulting in England characterized through the primary care health records of 53 million people in the OpenSAFELY platform: retrospective cohort study. JMIR Public Health Surveill. 10, e46485 (2024).
Small, W. R. et al. Large language model-based responses to patients’ in-basket messages. JAMA Netw. Open 7, e2422399 (2024).
Lam, K., Lu, A. D., Shi, Y. & Covinsky, K. E. Assessing telemedicine unreadiness among older adults in the United States during the COVID-19 pandemic. JAMA Intern. Med. 180, 1389–1391 (2020).
Zhao, B. Y. et al. Digital health literacy and associated factors among internet users from China: a cross-sectional study. BMC Public Health 24, 908 (2024).
Richardson, S., Lawrence, K., Schoenthaler, A. M. & Mann, D. A framework for digital health equity. NPJ Digit. Med. 5, 119 (2022).
Gani, I. et al. Understanding “alert fatigue” in primary care: qualitative systematic review of general practitioners attitudes and experiences of clinical alerts, prompts, and reminders. J. Med. Internet Res. 27, e62763 (2025).
Paccoud, I., Leist, A. K., Schwaninger, I., van Kessel, R. & Klucken, J. Socio-ethical challenges and opportunities for advancing diversity, equity, and inclusion in digital medicine. Digit Health 10, 20552076241277705 (2024).
Shi, Z. et al. Factors influencing digital health literacy among older adults: a scoping review. Front. Public Health 12, 1447747 (2024).
Qiu, C. S. et al. Determinants of digital health literacy: international cross-sectional study. J. Med. Internet Res. 27, e66631 (2025).
Fomo, M. et al. Empowering capabilities of people with chronic conditions supported by digital health technologies: scoping review. J. Med. Internet Res. 27, e68458 (2025).
Virani, S. S. et al. 2023 AHA/ACC/ACCP/ASPC/NLA/PCNA guideline for the management of patients with chronic coronary disease: a report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation 148, e9–e119 (2023).
Duggan, J. P., Peters, A. S., Trachiotis, G. D. & Antevil, J. L. Epidemiology of coronary artery disease. Surg. Clin. North Am. 102, 499–516 (2022).
Pamporis, K. et al. Prevalence of asymptomatic atrial fibrillation and risk factors associated with asymptomatic status: a systematic review and meta-analysis. Eur. J. Prev. Cardiol. zwaf138 (2025).
Inoue, T. Unawareness and untreated hypertension: a public health problem needs to be solved. Hypertens. Res. 48, 1639–1642 (2025).
van de Veerdonk, M. C. et al. Multimorbidity in heart failure: leveraging cluster analysis to guide tailored treatment strategies. Curr. Heart Fail. Rep. 20, 461–470 (2023).
Meyer, M. et al. Association between beta-blockers and outcomes in heart failure with preserved ejection fraction: current insights from the SwedeHF Registry. J. Card. Fail. 27, 1165–1174 (2021).
Iding, A. F. J., Ten Cate, V., Ten Cate, H., Wild, P. S. & Ten Cate-Hoek, A. J. Untangling profiles of postthrombotic syndrome using unsupervised machine learning. Blood Adv. 9, 3631–3641 (2025).
Wu, E. et al. How medical AI devices are evaluated: limitations and recommendations from an analysis of FDA approvals. Nat. Med. 27, 582–584 (2021).
Jalepalli, S. K., Gupta, P., Dekker, A., Bermejo, I. & Kar, S. Development and validation of multicentre study on novel Artificial Intelligence-based Cardiovascular Risk Score (AICVD).Fam. Med. Community Health 12, e002340 (2024).
Persell, S. D. et al. Effect of home blood pressure monitoring via a smartphone hypertension coaching application or tracking application on adults with uncontrolled hypertension: a randomized clinical trial. JAMA Netw. Open 3, e200255 (2020).
Rakai, E. et al. Systematic, randomized atrial fibrillation screening using detailed phenotyping with a risk prediction model combined with patch electrocardiogram in a Swedish population aged 65 years or older: the CONSIDERING-AF trial. Europace 27, euaf190 (2025).
Yang, J. et al. On-site computed tomography-derived fractional flow reserve to guide management of patients with stable coronary artery disease: the TARGET randomized trial. Circulation 147, 1369–1381 (2023).
Trivedi, R. et al. Conversational AI phone calls to support patients with atrial fibrillation: randomized controlled trial. JMIR Cardio 9, e64326 (2025).
Lee, J. S. et al. Antihypertensive medication adherence and medical costs, health care use, and labor productivity among people with hypertension. J. Am. Heart Assoc. 13, e037357 (2024).
Ali, D. H. et al. Therapeutic inertia in the management of hypertension in primary care. J. Hypertens. 39, 1238–1245 (2021).
Kimmoun, A. et al. Temporal trends in mortality and readmission after acute heart failure: a systematic review and meta-regression in the past four decades. Eur. J. Heart Fail. 23, 420–431 (2021).
Clark, H. et al. Hospitalisation costs associated with heart failure with preserved ejection fraction (HFpEF): a systematic review. Heart Fail. Rev. 27, 559–572 (2022).
Bundgaard, J. S. et al. Healthcare cost variation in patients with heart failure: a nationwide study. Public Health 207, 88–93 (2022).
Salih, A. et al. Explainable artificial intelligence and cardiac imaging: toward more interpretable models. Circ. Cardiovasc. Imaging 16, e014519 (2023).
Haupt, M., Maurer, M. H. & Thomas, R. P. Explainable artificial intelligence in radiological cardiovascular imaging-a systematic review. Diagnostics 15, 1399 (2025).
Leschka, S. et al. Noninvasive coronary angiography with 64-section CT: effect of average heart rate and heart rate variability on image quality. Radiology 241, 378–385 (2006).
Kini, A. S. et al. Fibrous cap thickness by optical coherence tomography in vivo. J. Am. Coll. Cardiol. 69, 644–657 (2017).
Young, M. N. et al. Multidisciplinary heart team approach for complex coronary artery disease: single center clinical presentation. J. Am. Heart Assoc. 9, e014738 (2020).
Koshy, A. N. et al. Outcomes following percutaneous coronary intervention in patients with multivessel disease who were recommended for but declined coronary artery bypass graft surgery. J. Am. Heart Assoc. 13, e033931 (2024).
Predel, C. & Steger, F. Ethical challenges with smartwatch-based screening for atrial fibrillation: putting users at risk for marketing purposes? Front. Cardiovasc. Med. 7, 615927 (2020).
McIntyre, W. F. et al. Direct oral anticoagulants for stroke prevention in patients with device-detected atrial fibrillation: a study-level meta-analysis of the NOAH-AFNET 6 and ARTESiA trials. Circulation 149, 981–988 (2024).
Biswas, D. et al. Artificial intelligence for cardiovascular care in action: from learning to implementation in health systems. JACC Adv. 4, 102307 (2025).
Leung, T. I., Coristine, A. J. & Benis, A. AI scribes in health care: balancing transformative potential with responsible integration. JMIR Med. Inf. 13, e80898 (2025).
Wu, W. T., Chao, Y. W., Lin, T. K., Huang, C. K. & Hsieh, P. H. Economic evaluation of AI-assisted technologies in healthcare: a systematic review. J. Food Drug Anal. 33, 487–500 (2025).
El Arab, R. A. & Al Moosa, O. A. Systematic review of cost effectiveness and budget impact of artificial intelligence in healthcare. NPJ Digit. Med. 8, 548 (2025).
Odendaal, W. A. et al. Health workers’ perceptions and experiences of using mHealth technologies to deliver primary healthcare services: a qualitative evidence synthesis. Cochrane Database Syst. Rev. 3, CD011942 (2020).
Gao, C. et al. Pitfalls of defacing whole-head MRI: re-identification risk with diffusion models and compromised research potential. Comput. Biol. Med. 197, 111112 (2025).
Panch, T., Mattie, H. & Celi, L. A. The “inconvenient truth” about AI in healthcare. NPJ Digit. Med. 2, 77 (2019).
Warraich, H. J., Tazbaz, T. & Califf, R. M. FDA Perspective on the regulation of artificial intelligence in health care and biomedicine. JAMA 333, 241–247 (2025).
Tang, D., Xi, X., Li, Y. & Hu, M. Regulatory approaches towards AI Medical Devices: a comparative study of the United States, the European Union and China. Health Policy 153, 105260 (2025).
Inan, O. T. et al. Digitizing clinical trials. NPJ Digit. Med. 3, 101 (2020).
ElZarrad, M. K., Lee, A. Y., Purcell, R. & Steele, S. J. Advancing an agile regulatory ecosystem to respond to the rapid development of innovative technologies. Clin. Transl. Sci. 15, 1332–1339 (2022).
Carvalho, E. et al. Predetermined change control plans: guiding principles for advancing safe, effective, and high-quality AI-ML technologies. JMIR AI 4, e76854 (2025).
Vardas, E. P., Marketou, M. & Vardas, P. E. Medicine, healthcare and the AI act: gaps, challenges and future implications. Eur. Heart J. Digit. Health 6, 833–839 (2025).
Cunningham, J. W. et al. Artificial Intelligence in cardiovascular clinical trials. J. Am. Coll. Cardiol. 84, 2051–2062 (2024).
Cubukcu, H. C. et al. Regulating the future of laboratory medicine: European regulatory landscape of AI-driven medical device software in laboratory medicine. Clin. Chem. Lab Med. 63, 1891–1914 (2025).
Babu, M., Lautman, Z., Lin, X., Sobota, M. H. B. & Snyder, M. P. Wearable devices: implications for precision medicine and the future of health care. Annu. Rev. Med. 75, 401–415 (2024).
Stark, C., Rytkin, E., Mircea, A. A. & Efimov, I. R. Advances in cardiac devices and bioelectronics augmented with artificial intelligence. J. Physiol. https://doi.org/10.1113/JP287135 (2025).
Matsuoka, Y. et al. Phenotypic trajectories from acute to stable phase in heart failure with preserved ejection fraction: insights from the PURSUIT-HFpEF study. J. Am. Heart Assoc. 14, e037567 (2025).
Soltani, F. et al. Phenogrouping heart failure with preserved or mildly reduced ejection fraction using electronic health record data. BMC Cardiovasc. Disord. 24, 343 (2024).
Kyodo, A. et al. Heart failure with preserved ejection fraction phenogroup classification using machine learning. ESC Heart Fail. 10, 2019–2030 (2023).
Woolley, R. J. et al. Machine learning based on biomarker profiles identifies distinct subgroups of heart failure with preserved ejection fraction. Eur. J. Heart Fail. 23, 983–991 (2021).
Acknowledgements
This work was supported by Major Science and Technology Special Projects for Cancer, Cardiovascular, Respiratory and Metabolic Diseases (2025ZD0547200), and Excellent Young Science and Technology Talent Cultivation Special Project of CACMS (CI2023D006).
Author information
Authors and Affiliations
Contributions
Y.L. and YF.L. conceived this topic and arranged the outlines, revised the review. Q.X., YW.L., and M.Z. researched data for the article. Q.X., M.Z., Y.L., Y.C., X.C., W.W., Y.L., J.J., Y.X., and Y.L. substantially contributed to the discussion of content. Q.X., YW.L., and M.Z. wrote the article. All authors reviewed/edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, Q., Li, Y., Zhu, M. et al. Precision cardiovascular medicine with big data and AI. npj Digit. Med. (2026). https://doi.org/10.1038/s41746-026-02538-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-026-02538-0