Abstract
The long-term recording of neural activity could be used to understand complex behaviours and disorders. However, the development of technology capable of such measurements faces a variety of technical challenges, including the relative motion between recording electrodes and tissue and the excessive displaced volume from implanted electronics. Here we report a subnanolitre-volume tetherless optoelectronic microsystem for neural recording. The system relies on light for photovoltaic power and data transfer, through a light-emitting diode, eliminating the need for wires or other tethers. It uses a single AlGaAs diode as both photovoltaic and light-emitting diode. Complementary metal–oxide–semiconductor circuits provide low-noise amplification, pulse-position-modulated encoding and electro-optical transduction. Two-dimensional materials processing techniques, vacuum annealing and atomic layer deposition, in conjunction with a standard complementary metal–oxide–semiconductor fabrication process, provide compact encapsulation against the corrosive conditions of biological media. We show that the subnanolitre neural implant is capable of chronic (365 days) in vivo recordings in awake mice.
Similar content being viewed by others
Main
Direct, chronic recording of cellular electrical activity can provide an insight into how an organism develops, functions and declines over time, but remains technically challenging. Tethered (wired) methods inflict damage from the relative motion between the tether and the surrounding tissue1, rendering them problematic for chronic measurements. Tetherless techniques are diverse, and are largely defined by the modality they use to transfer power and data between the implant and the outside world. Radio-frequency (RF)2,3,4,5,6,7,8,9,10, ultrasound11,12,13 and optical methods14,15,16 have been explored, and each has benefits and drawbacks. The minimum size of RF- and ultrasound-based systems is typically limited by the wavelength of their power/communication modality. Such implants, even on a per-channel basis, typically displace volumes on the scale of microlitres (mm3) or greater, equivalent to a sizable fraction of the mouse brain, the most common in vivo model; of these, only a few have actually been fully embedded for in vivo demonstration7,11,13,17. A similar observation can be made in non-neural recording implants, such as temperature18/pressure19/oxygen20 sensors and electrical/optical stimulators10,17,21,22,23 (Extended Data Table 1).
Optical imaging techniques, such as multiphoton microscopy (MPM)24,25 and associated protocols have been developed to measure activity at cellular resolution over many months, without any need for an implant except for a fibre in select cases26. However, such techniques are only applicable to fluorescently labelled neurons, with fundamentally limited imaging depth and low temporal resolution (Supplementary Sections 1 and 2). Recently, some hybrid implementations3,4,5,6,7,8,17 have shown promise in which the measurement electrodes are tethered to a ‘base station’, which sits atop the dura or skull and communicates, and is powered electromagnetically (RF) or with a battery. However, a compact, embeddable system on the scale of these measurement electrodes, or smaller, has remained elusive.
In this Article, we report a microscale optoelectronic tetherless electrode (MOTE) that combines electrical and optical approaches through the heterogeneous integration of silicon circuitry with compound semiconductors to make. Building on our previous work15,27,28 on a scaled neural implant that can function chronically in vivo (which were limited by insufficient encapsulation28, bulky size, inefficient encoding schemes29 and light-induced leakage current in complementary metal–oxide–semiconductor (CMOS) circuits16), MOTE provides an untethered, subnanolitre neural implant that is capable of chronic (365 days) in vivo recordings in awake mice (Fig. 1a).
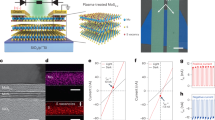
a, System overview in which a MOTE is implanted in an awake mouse brain to chronically record neural activity in vivo—incoming light powers the MOTE, and the MOTE, in turn, emits the PPM pulses communicating the recorded data. b, Optical microscopy image comparing a MOTE with a strand of human hair. c, MOTE is powered and is communicating optically; it is continuously powered at a shorter wavelength and communicates at a longer wavelength, making the powering system easier to implement and avoiding power–communication crosstalk. d, Scaling trends of tetherless autonomous microsystems for neural recording in which the MOTE is smaller than a single nanolitre. Credit: a, mouse illustration, SciDraw (E. Tyler and L. Kravitz).
In tetherless technology, where the system size is generally limited by the wavelength used to transfer power and data30, our optoelectronic approach is attractive for ultrasmall implants (Fig. 1b). MOTE relies on light for photovoltaic (PV) power and data transfer through a light-emitting diode (LED), eliminating the need for any wires or other tethers. It uses a single AlGaAs diode as both PV and LED (a photovoltaic light-emitting diode (PVLED))29,31 through time-division multiplexing (Fig. 1c), using it as a PV for 93.4% of the time and as an LED for 0.06% of the time15 (with the remainder of the time spent on transitions). An external 623-nm LED source provides power to the PVLED, and the MOTE emits 825-nm pulse-position-modulated (PPM) pulses15 that encode electrophysiological signals.
The underlying CMOS circuits provide low-noise amplification, stable biasing, PPM encoding and drive the PVLED (as an LED); the power consumption is dominated by low-noise amplification and the LED driver15. Red and infrared wavelengths (623 nm and 825 nm, respectively) are used for power and communication with minimal loss through the tissue32, improving the MOTE performance at depth; incident LED irradiance is limited to <70 mW mm−2, well below the threshold of 250 mW mm−2 for inflicting heat damage in the brain33. On-MOTE circuitry was optimized to minimize its size and power consumption, as well as allowing the external communications apparatus to be much larger and consume more power. Such heterogeneous integration and methodical system design allow MOTE to have a subnanolitre volume per channel (Fig. 1d).
MOTE fabrication
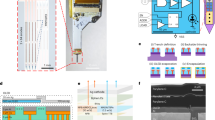
Various developments in nanofabrication, integration and system design have been used to create the MOTE. In fabrication, after PVLED placement on CMOS29, extremely low-pressure annealing (<10−6 torr at 300 °C; adopted from a two-dimensional materials cleaning procedure34) effectively removes transfer-related residues and promotes adhesion between heterogeneous elements without inflicting thermal damage28. Atomic layer deposition (ALD) of SiO2, Si3N4 and Al2O3 encapsulates the MOTE against corrosive biological media without substantially increasing its volume (total encapsulation thickness, <1.5 µm). High-pressure platinum (Pt) sputtering then provides not only favourable electrode impedance35 but also an effective and conformal36 light shield to prevent incident light from generating unwanted photocurrents in the electronics16. Critically, each fabrication step is done in parallel (Fig. 2a), simultaneously fabricating close to 100 MOTEs per chip (and scalable to thousands of MOTEs per square centimetre of silicon). Figure 2 illustrates how the CMOS and PVLED wafers are processed and integrated to yield an array of MOTEs in which the Pt light shield covers the surface, including the sides and bottom, of each MOTE, but without covering the input electrodes and the PVLED. The Pt light-shielded area is further encapsulated by 100 ± 10 nm of Al2O3 to electrically insulate the MOTE. The Methods provides further details on integration fabrication.
a, Bulk fabrication of MOTEs (left) integrating two disparate technologies—CMOS (silicon based) and PVLED (AlGaAs based)—and a cross-sectional view (right) of a fully fabricated MOTE illustrating how the ALD dielectrics and sputter Pt together constitute a shield against biological media and unwanted photocurrents16. VIN, input voltage. b, Key fabrication and integration steps: (i) PVLED transfer on CMOS. (ii) Vacuum annealing (300 ± 3 °C and <10−6 torr) for polymeric residue removal and high-quality ALD. (iii) Deposition of Pt contact on the PVLED and CMOS. (iv) Additional Pt deposition to connect the PVLED Pt contacts with the CMOS Pt contacts, followed by another ALD deposition to fill in the gaps and cracks. (v) Top etch to remove the CMOS intermetal dielectrics (IMDs) and bulk silicon between individual MOTEs—the etched silicon serves as an etch-stop mark during the backside etch (step (viii)). (vi) Selective Pt deposition (94 ± 1 nm) serves as a light shield to protect the inner CMOS circuits against light-induced excitons, followed by another ALD deposition for encapsulation. (vii) A pair of measurement Pt electrodes are exposed. (viii) The bulk CMOS Si is etched from the backside using a DRIE until the silicon trenches (step (v)) become visible. (ix) Additional Pt deposition on the backside (94 ± 1 nm) ensures that there are no light-induced carriers generated through the backside of the MOTEs either. PMMA, polymethyl methacrylate; PR, photoresist.
CMOS circuits
One of the critical innovations is the on-chip PPM (Fig. 3a) that optically communicates the measured electrophysiological signals. By concentrating the transmitted power into short, bright pulses and encoding information in the timing of those pulses, PPM is much more resistant to noise than amplitude modulation12,29. This advantage can be shown using Shannon’s channel theory37, and PPM is widely used in satellite communication, where high channel bandwidth (BW) (reflected in low-duty-cycle PPM pulses) compensates for low signal-to-noise ratio when data rates are significantly lower than the inverse pulse width38.
a, Systemic description of a MOTE and its external counterpart for communication—the MOTE’s output PPM pulses are detected by an external photodiode (PD) before being passed through a decoder (Methods). b. Schematics of the front-end amplifier based on pseudo-resistors27 (left) and the charge pump for optical pulse generation15,27 (right). VOUT, output voltage; VSS, system ground. c, Power and area distributions of a MOTE in which the amplifier and filter take the majority of the power for low-noise amplification (left), and the frame and integration overhead for protection against unwanted light and photocarriers take most of the area (right). Extended Data Table 2 and Supplementary Section 3 categorize 307 transistors and their power consumptions.
Figure 3a illustrates the block diagram of the underlying CMOS circuits in the MOTE, consisting mainly of the amplifier, PPM encoder and LED driver. The input-referred noise of the entire system is dominated by the amplifier (Fig. 3b, left), reflecting the fact that intrinsic transistor noise trades off against BW and power consumption39. We have designed the amplifier for a bandwidth from <10 Hz to >10 kHz and a noise floor of 14.8 µVr.m.s. to capture extracellular neural signals, including both local field potentials (LFPs) and action potentials and minimizing power consumption (500 nW)15—the MOTE electrodes (Pt) are 28.5 µm × 30.5 µm and 12.5 µm × 23 µm (294.25 µm apart, centre to centre), which sets its spatial resolution. The amplified signal then passes through the PPM encoder, which uses low-jitter timing circuitry to create the PPM voltage pulses. The LED driver (Fig. 3b, right) transduces voltage pulses into optical pulses by quickly discharging capacitors through the PVLED, and then slowly recharging those capacitors between pulses. To make this electro-optical transduction maximally efficient, the original voltage pulses are ‘stepped up’ by stacking three capacitors in series, thereby creating a higher voltage pulse to drive the PVLED; without this step-up, LED efficiency can degrade by orders of magnitude. The nominal power consumption is 1 µW, divided between the amplifier (50.0%), encoder (10.5%), LED driver (26.2%) and support circuits (13.3%). Figure 3c summarizes this power distribution (Fig. 3c, left) as well as notes that the physical requirements for light shielding (‘Frame’) and integration fabrication (‘Integration overhead’) together claim a substantial portion of the total area (Fig. 3c, right).
Validation in vitro
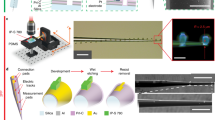
Once the heterogeneous elements of MOTEs are integrated and encapsulated, the MOTEs are suspended as individual chiplets in a solution of isopropanol and transferable using a pulled glass micropipette. Isopropanol, which dries soon after the MOTEs are loaded into the micropipette, also serves as a sterilizing agent without incurring any functional damage to the MOTEs. To verify that MOTEs function in biological media and faithfully record electrophysiological activity, we deployed MOTEs in a Petri dish and cultured induced pluripotent stem cell (iPSC)-derived cardiomyocytes40 (Extended Data Fig. 1) on them (Fig. 4a and Supplementary Video 1). Once the cardiomyocytes attached to the MOTEs, we stained their nuclei with a blue Hoechst dye41 (Fig. 4a, right) to confirm their presence on the MOTE electrodes.
a, MOTEs on a Petri dish next to a Singaporean dollar coin for scale (left and middle), and a MOTE surrounded by iPSC-derived cardiomyocytes (right) with blue fluorescent dye (Hoechst 33342) staining for DNA, labelling the cell nuclei; the inset has the same scale as the background image. b, A MOTE’s PPM encoding in action. A MOTE emits a series of pulses (top left; top right shows the zoomed-in view), which encodes cardiomyocyte voltage spikes; specifically, the interpulse intervals (green arrows) correspond to voltage (bottom left; bottom right shows the zoomed-in view). c, Chemical modulation of cardiac spike rates. (i) Modulation of cardiomyocytes spike frequency using isoproterenol (1 µM and 5 µM) and blebbistatin (5 µM) to confirm the cellular origin of the MOTE measurements. (ii) Isoproterenol increased the frequency of spontaneous cardiomyocyte contraction, whereas blebbistatin, an actomyosin inhibitor that also results in excitation–contraction uncoupling, resulted in a decrease in the contraction frequency, as expected. Different colours denote three different MOTEs. CTRL, control; ISO, isoproterenol; BLEB, blebbistatin.
Figure 4b depicts the encoded optical output of a MOTE (Fig. 4b, top) and the replicated cardiomyocytes voltage time series after decoding (Fig. 4b, bottom; Methods). Figure 4b (inset) illustrates the PPM optical encoding and decoding; red dots denote regular ‘reference’ pulses, whereas green dots denote ‘encoding’ pulses. The time between the reference and encoding pulses (green arrow) corresponds directly to the voltage measured across the two MOTE electrodes, enabling the voltage time series to be reconstructed (Fig. 4b, bottom). To confirm that the reconstructed cardiac spikes are associated with genuine cardiomyocyte contraction, we chemically modulated the beating rate of the cardiomyocytes using the beta adrenergic receptor agonist isoproterenol (1 µM and 5 µM) or the myosin inhibitor blebbistatin (5 µM), which are known to increase and decrease the cardiomyocyte contraction frequency, respectively42. The decoded MOTE output successfully tracked the changes in cardiomyocytes activity (Fig. 4c), matching the expected trends. Although MOTEs are probably measuring from multiple cardiomyocytes, the cells beat together in sync.
Validation in vivo
After confirming the functionality of MOTEs with cardiomyocytes in vitro, we conducted in vivo testing in the mouse barrel cortex, implanting MOTEs through transcranial surgery (Extended Data Fig. 2 and Methods) and delivering mechanical stimulation onto whiskers43 with a motorized rod (Supplementary Video 2). Although the link efficiency (in powering and communicating with a MOTE) generally decreases with the implant depth and tilt (with respect to the objective), interestingly, we noticed that the angular dependency weakens with an increase in the implant depth through Monte Carlo simulation33,44 as well as experiments (Extended Data Fig. 3 and Supplementary Section 4).
In two of the six implanted mice, we placed MOTEs on the brain surface, from which we were able to measure the electrocorticographic (ECoG) signals (Extended Data Figs. 4 and 5). In the other four mice, we inserted MOTEs into the barrel cortex (Fig. 5) using a pulled pipette-coupled nanoinjector, as previously reported28 (a separate group of mice were implanted with dummy MOTEs for a preliminary histological test (Fig. 6a), including mouse 0 in Fig. 5a(iv)). Figure 5a(i) depicts a typical measurement setup, in which the whiskers of an awake, head-fixed mouse are stimulated by a motorized rod (Extended Data Fig. 6). The implanted MOTEs can be repeatedly observed stereoscopically (Fig. 5a(ii),(iii)), localized in layers 1–3 (~100–400 µm in depth45) of the barrel cortex (Fig. 5a(iv)), and histologically examined in more detail post-mortem to confirm the health of the tissue surrounding the implantation sites (Fig. 6a).
a, (i) Measurement setup, in which a computer-controlled motor moves a rod to stimulate a whisker of an awake, head-fixed mouse during MOTE recording. (ii),(iii) Stereoscopic images of MOTEs implanted in the barrel cortex over 161 days. (iv) Neuron staining near a MOTE implant site 31 days after insertion. b, MOTE action potential recordings in mouse 2 after 13 days (top) and 102 days (bottom) of implantation. The overlay plots confirm the presence of action potentials, and the dot raster plots (indicating spike timing over 100 stimulation cycles) and peri-stimulus time histograms show a strong causal relationship between the whisker stimuli (grey zone) and spikes. The increase in stimulus-induced spikes on day 102 might be due to decreased inflammation over time and/or MOTE migration closer to an axon hillock. Grey windows denote the rod movements. c, LFP measurements from MOTEs in multiple mice and over multiple days. (i) Thirty sequential recordings of MOTE 1 in mouse 1 after 4 days (left), 161 days (middle) and 304 days (right) of implantation. Whisker contact timing may vary between measurements, probably causing the observed change in the time delay between the rod movement and onset of LFPs. (ii) Overlay plot and average of the 30 traces taken on day 4 (top), compared with the control case in which the stimulation rod is moving but does not touch the whisker, confirming that the LFP responses are not electrical artefacts. Control: no twitch. (iii) LFP recording, overlay and average, from the second MOTE in the same mouse (mouse 1) after 161 days of implantation; the LFP recordings became slightly weaker and noisier after 161 days, which may have been caused by electrode degradation, scar tissue formation and/or the movement of the MOTEs (~50–300 µm; measured using a three-photon microscope25 (Extended Data Fig. 7)) inside the brain as the initial inflammation subsided. (iv) Overlay and average plot of the LFP recording from the MOTE in mouse 2 after 102 days of implantation; this is the same MOTE and mouse as in the action potential measurement shown in b. Same mouse and MOTE as in Fig. 4b. 0 ms denotes the beginning of the rod movement. All the LFP data (c) and action potential data (b) shown were band-passed between 10–250 Hz and 300–4,000 Hz, respectively, using a fourth-order Butterworth filter. Panel a(i) adapted with permission from ref. 43, Springer Nature Limited.
a, Left: confocal imaging and immunohistochemical staining revealed distinct staining patterns of neurons (NeuN or GCAMP6), microglia (Iba1) and nuclei around MOTE implants (after 6 months) and optic fibres (after 3 months). Comparable Iba1 levels between the MOTE regions and the corresponding control areas (under the transcranial window but in areas in which no MOTE was inserted) suggest a minimal impact of MOTEs on surrounding brain tissues. On the contrary, optic fibre implants elicited higher microglial responses compared with MOTEs, indicating more severe tissue damage and foreign body reactions. The neurons beneath the optic fibre implants degenerated, contrasting with the healthy appearance of neurons surrounding MOTEs. It should be noted that the optical window above the MOTE implants induced a foreign body reaction, leading to elevated microglial levels in adjacent brain regions compared with control regions at similar depths. Scale bars, 20 µm. It should be noted that brain tissues can morph during clarity and index matching, and therefore, the MOTE dimensions in the above figures may not directly correspond to the physical MOTE dimensions. Right: quantitative analysis based on the images on the left. Data points represent the integrated density of Iba1 signals normalized to the image area, facilitating comparison between depths. DAPI, 4′,6-diamidino-2-phenylindole; OF, optical fibre. b, (i)–(iii) PPM output pulses from mouse 1’s MOTE on the first (i) and last (ii) day of in vivo measurement and after euthanasia (iii). (iv) Stereoscopic image of a MOTE within brain tissue extracted from mouse 1. (v)–(vii) PPM output pulses from mouse 2’s MOTE on the first (v) and last (vi) day of the in vivo measurement, and after euthanasia (vii). (viii) Stereoscopic image of a MOTE within brain tissue extracted from mouse 2. The ex vivo PPM outputs in (iii) and (vii) seem stronger than the earlier in vivo PPM outputs because the tissue thickness above the MOTEs is thinner in these brain tissue slices.
The bandwidth of implanted MOTEs suffices to capture both individual action potentials (Fig. 5b) and LFPs (Fig. 5c). Although MOTEs are still larger than individual neurons, they are capable of measuring local field potentials around them, in addition to action potentials from one or more neurons. Figure 5b shows a stimulus-correlated increase in the rate of spiking from recorded neurons over 100 days. Not only are MOTEs able to remain functional for well over 100 days but we have often observed that the LFP shapes remained stable over that time span, suggesting the stability of the MOTEs post-implantation. Minor differences in LFPs can be attributed to MOTE movement inside the brain (~50–300 µm; they can drift both laterally and vertically) as initial inflammation subsided46 and reached different cortical layers47, possible depositions that alter the electrode characteristics over time, as well as variability in rod positioning (and therefore response latency) between experiments. In control experiments, in which the motorized rod was moving but did not touch the whisker (Fig. 5c(ii), bottom), no discernible LFPs were recorded, ruling out purely electrical artefacts. As an additional verification, Fig. 5c depicts the MOTE LFP recording in the second MOTE of mouse 1 (Fig. 5c(iii)), as well as from a MOTE in a different mouse (Fig. 5c(iv), mouse 2). Of the six MOTEs tested, embedded in four different mice (mice 1–4), we observed LFPs from four MOTEs (in mice 1–3); the fifth MOTE in mouse 3 was embedded too deeply, and its PPM amplitudes, therefore, attenuated, for its signals to be reliably decoded, and the sixth in mouse 4 appeared to have been damaged during implant preparation. We were also able to measure LFP in mouse 1 (Fig. 5c(i)) on day 365, but the LFP has become noticeably weakened by then such that we had to average over traces (Extended Data Fig. 8 and Supplementary Section 5). All four mice with implanted MOTEs had remained healthy over 200 days after implantation and have not exhibited any signs of seizures or other maladies that can be induced by larger implants with substantially greater volumes of displaced brain. Unfortunately, their head-bar sites began to degrade near 300 days post-implantation, which may be attributable to imperfect transcraniotomy and expired dental cement. Following euthanasia, we have extracted MOTE-containing brain tissues from mice 1 and 2 to check their functionality. Figure 6b shows that both MOTEs were able to output PPM output pulses even after their extended stay inside the brain. Given the number of transistors needed to generate the PPM pulses (Extended Data Table 2), the weaker LFPs seen on day 365 were probably due to electrode degradation rather than encapsulation or circuit failures. A preliminary histological test (Fig. 6b), although not providing a rigorous conclusion, shows that the above-MOTE microglia reaction might be similar to that of the control area under the same cranial window (but without a MOTE), and much less compared with the optical fibre that had been implanted for just half as long (six months versus three months).
Conclusions
We have reported the development of a subnanolitre autonomous microsystem that is capable of chronic in vivo neural recording. Here 186 CMOS transistors provide low-noise amplification, PPM encoding and electro-optical transduction. PPM encoding enables more efficient data communication than other common approaches such as amplitude modulation29. Two-dimensional materials processing techniques, vacuum annealing and ALD, in conjunction with a standard CMOS fabrication process, provide compact encapsulation against the corrosive conditions of biological media. We have also developed methodologies, based on prior electrophysiological techniques, to implant such tiny microsystems into the mouse cortex and provide in vivo validation.
A MOTE not only works as a minimally invasive neural recording unit in the mouse brain but also provides a path towards chronic monitoring in models beyond mice, including organoids and invertebrates, where existing technologies struggle either due to their bulky size (measuring inside organoids), the lack of fluorescent gene lines or viral vectors (invertebrates and atypical animal models), or bandwidth and scattering (Supplementary Section 2). Furthermore, MOTEs are well suited for providing an electrophysiological side channel under the optical measurement of activity; by eliminating wires, they may be more compatible with functional magnetic resonance imaging. In addition, the small size of MOTEs, and the lack of tether, may enable recording from flexible non-brain tissue in moving animals, including small animals such as fruit flies; we have used a head-fixed stage for this demonstration, but a movement-tracking light source and detection apparatus for free-moving measurements are currently under development. Our technology provides the basis for accessing a wide variety of physiological signals48 with small and untethered instrumentation implanted on chronic timescales.
Methods
CMOS circuit design
Available power largely dictates the MOTE’s CMOS circuit design, which is, in turn, limited by the amount of optical power that can be used without excessive brain heating33. If one wants to access the entire brain of a mouse, an ideal implant should function up to 6 mm deep in the brain. Taking into account the scattering and absorption properties of the brain49,50, it could theoretically be possible for a MOTE to function at a depth approaching 6 mm; however, an improved AlGaAs PVLED and a photomultiplier tube (for detection) will be required. At a certain depth, although optical attenuation from brain tissue is generally undesirable, the highly scattering nature of the tissue may also provide the benefit of making MOTEs in the brain orientation insensitive (Extended Data Fig. 3 and Supplementary Section 4).
Given the 1 µW of electrical power, the AlGaAs PVLED defines the available supply voltage, VDD, of 1 V, the open-circuit voltage (VOC) of the PVLED, implying 1 µA of current to power the entire MOTE system. The front-end amplifier takes a large fraction of power (50.0%) to achieve the desired noise floor and bandwidth for neural measurements; the target input-referred noise floor was 10 μVr.m.s. and BW of 10 kHz, which are acceptable for many neural applications5, and post-measurement filtering can further reduce the noise at the expense of BW. Figure 5a shows the circuit topology of the front-end amplifier utilizing pseudo-resistors to set the input transistor biasing and the high-pass corner. It should be noted that because these pseudo-resistors form such high-impedance nodes of the order of hundreds of gigaohms, any current induced by light leakage into the CMOS substrate can incur significant voltage offsets. We prevent such offsets with a self-aligned Pt light shield.
Then, 26.2% of the power is spent on the charge pump (Fig. 5b) to maximize the PVLED emission efficiency, whereas the rest of the circuits such as start-up, biasing and encoding take up the other remaining 25% of the power budget (Extended Data Table 2 and Supplementary Section 3 provide the exact breakdown). Although we have aimed to minimize the physical size of the CMOS, maintaining sufficient size for the amplifier (to reduce its flicker noise) as well as the capacitances for input, decoupling and charge pump requires the presented scale of the CMOS.
Integration fabrication
The MOTE’s CMOS circuits are taped out through a foundry (TSMC 180-nm mixed-signal process), where 97 MOTE circuits are arrayed in a single die. The AlGaAs PVLEDs are separately fabricated, again arrayed like the CMOS circuits, on a sapphire substrate with epitaxially grown AlGaAs layers on top. Using a soft sacrificial layer called polymethyl methacrylate, a common practice in two-dimensional materials transfer51 (Fig. 2b(i)), the AlGaAs PVLED array is chemically delaminated from the sapphire substrate and transferred onto the CMOS die45. Following the polymethyl-methacrylate-based transfer, the elimination of polymeric residues and the promotion of adhesion between the PVLEDs and CMOS are imperative for the ensuing fabrication and encapsulation steps. High-vacuum annealing (<10−6 torr at 300 ± 3 °C) achieves this residue removal and adhesion (Fig. 2b(ii)) without damaging the underlying CMOS28 or the PVLED.
Throughout the fabrication processes, ALD is used liberally to fill any cracks and gaps originating from a fabrication step, thereby providing an atomically dense encapsulation layer (<1.5 µm in total) against biological media, in contrast to the common parylene-C-based encapsulation that are often several micrometres thick yet porous. It should be emphasized that without high-vacuum annealing or proper cleaning and rinsing between steps, an ALD may not properly ‘seed’ and the remaining residues can either turn the ALD into chemical vapour deposition or inhibit the deposition altogether.
For biocompatibility, we have only used Pt for metallization, and we have used high-pressure (20 mtorr) RF sputtering for conformal coating and electrode impedance optimization35. Figure 2b(iii) shows this Pt deposition after an opening etch to establish electrical contacts with the CMOS circuit and the PVLED anode and cathode, which are connected through Pt routing in Fig. 2b(iv). Individual MOTEs are then ‘outlined’ during top etch (Fig. 2b(v)), using inductively coupled plasma reactive ion etch for intermetal dielectric and deep reactive ion etch (DRIE) for CMOS bulk silicon, using a Cr hard mask to withstand prolonged etching. This silicon etch is intentionally made shallow (<10 µm) compared with the bulk thickness (330 µm for an as-delivered die) to set the total MOTE thickness to be <20 µm during the backside etch (Fig. 2b(viii)); a deeper depth incurs additional fabrication difficulties associated with photolithography on high-aspect-ratio features.
Although light is a medium through which MOTE is powered and communicates, any light ‘leakage’ into the silicon of the core CMOS circuits can generate excitons that can have a detrimental effect on the CMOS circuits16. This leakage is exacerbated at high-impedance nodes and has been a failure mode in previous attempts at MOTE-like implants28. To mitigate this leakage, Fig. 2b(vi) illustrates how we selectively deposit Pt (94 ± 1 nm) to cover the MOTEs. This Pt light shield is then encapsulated before we use a gentle reactive ion etch to expose the two measurement electrodes. High-energy inductively coupled plasma reactive ion etch or DRIE, when applied directly to the CMOS contacts, can damage the underlying CMOS circuits.
In the final step, the substrate is flipped upside down and placed on a four-inch Si carrier wafer with a photoresist (Microposit S1818) as a temporary adhesive. Figure 2b(viii) portrays the backside thinning in which the DRIE is used to etch the CMOS bulk Si until the predefined trenches (Fig. 2b(v)) become visible. Then, to prevent any light-induced carriers in the bulk silicon, an additional Pt (94 ± 1 nm) light shield is deposited on the etched backside in a self-aligned fashion (Fig. 2b(ix)). Although the deposited Pt initially covers and connects all of the MOTEs and the Si carrier, once the carrier substrate is immersed in photoresist-dissolving acetone, only the Pt deposited on MOTEs will remain with the Pt deposited on the photoresist lifted off. Though there are circuit techniques to mitigate the light leakage problem16, our Pt-shielding provides a robust physical means that can withstand more than an order of magnitude higher light (>70 mW mm−2) than the circuit technique.
Cardiomyocyte preparation and measurement
Extended Data Fig. 1 shows human iPSC-derived cardiomyocytes. Cells were differentiated according to the methods outlined in ref. 52 and seeded on fibronectin-coated 30-mm dishes. For seeding, we used a Roswell Park Memorial Institute medium supplemented with fetal bovine serum (10%) and the ROCK inhibitor (Y-27632) at a dilution of 1:1,000. A day after seeding, the media is changed to the Roswell Park Memorial Institute medium with a B27 supplement and penicillin–streptomycin. First, we measured the base heart rate (1.8 beats per second; Fig. 4c(i), left) and then applied isoproterenol (1 µM) to the iPSC-derived cardiomyocytes and let them stabilize for 30 min in an incubator before measurement (2.0 beats per second; Fig. 4c(i), middle). We increased the isoproterenol dose to 5 µM followed by another 30-min stabilization period, although we did not see noticeable changes in the beating rate compared with the 1-µM case. We then switched to fresh media and applied blebbistatin (5 µM) followed by the 30-min stabilization, after which we saw a reduction in the beating rate (0.8 beats per second; Fig. 4c(i), right). The iPSC-derived cardiomyocytes are BMWT1 from J.L.’s laboratory, which is originally from McNally’s laboratory—GM7924 (BMWT1): healthy, male, 17, white, QC (mycoplasma, bacteria, look undifferentiated) = PASS, and [CHIR] (µM) = 6–2 × 24’ from Coriell.
Transcranial surgery in mice
A mouse is placed inside a rodent-anaesthetizing box and anaesthetized through a gas mixture of oxygen (1 s.c.c.m.) and isoflurane (2 s.c.c.m.) using the MIDMARK Matrx VIP 3000 Calibrated Vaporizer. Within 15 min, the mouse is sufficiently sedated. Extended Data Fig. 2 shows a mouse placed on a Kopf stereotaxic apparatus with intra-aural positioning studs, a tooth bar and a heating pad (37 °C). The eyes of the mouse are protected with ophthalmic ointment (Puralube Vet Ointment). Subcutaneous injections of dexamethasone (0.1 ml mg−1, 2 µl g−1 of mouse body weight), ketoprofen (2 mg ml−1, 2.5 µl g−1 of mouse body weight) and glycopyrrolate (0.1 mg ml−1, 2 µl g−1 of mouse body weight) are performed for anti-inflammatory, analgesic and anticholinergic purposes, respectively.
Mouse hair is shaved off using a hair clipper (Extended Data Fig. 2), followed by a hair removal gel (Veet Sensitive Hair Remover Gel Cream Pink; Extended Data Fig. 2). The exposed head skin is sanitized with iodine (Betadine Solution Povidone Iodine, 5%) and 70% ethanol, and the skin above the cranial site of interest is selectively removed using a surgical scissor. The exposed skull is then sanitized again with 70% ethanol, and the edge of the skin is glued using a vet bond (3M Vetbond Tissue Adhesive) to prevent skin regrowth. The transcranial site is outlined with respect to the exposed bregma (5-mm diameter centred at (−1 mm, 3 mm)), and a biopsy punch (INTEGRA MILTEX Sterile Disposable Biopsy Punch, 5-mm diameter) is used to make an initial transcranial trench. Then, using a surgical drill (Meisinger Dental HM1 005 REF 500 104 HP), the skull window is isolated and removed and the dura remains fully intact.
Once the skull is removed (Extended Data Fig. 2d), MOTEs are ready for insertion (Extended Data Fig. 2e) using a scheme similar to a previously reported one based on a nanoinjector (DRUMMOND SCIENTIFIC COMPANY NANOINJECT III), X–Y–Z stage (SUTTER INSTRUMENT MP-285A and Luigs & Neumann GmbH SM5) and pulled pipettes28 (Supplementary Video 3). In between MOTE insertion, the exposed brain area is kept moist using medical-grade saline-soaked sponges (Sugi Eyespear pointed tip) to minimize potential damage to the site. Once the insertions are completed, a quartz window (POTOMAC; custom made) is placed over the surgical site with gentle but sustained pressure (Extended Data Fig. 2f). The window is first secured using a vet bond, and then, a custom stainless steel head bar is gently placed before a dental cement (C&B Metabond Adhesive Cement S396 Radiopaque L-Powder) secures both the window and the head bar (Extended Data Fig. 2g). A typical surgery lasts 3 h and after the first 30 min, the isoflurane flow rate is often reduced to 1 s.c.c.m. (the oxygen remains the same at 1 s.c.c.m.) to prevent overdose. An hourly glucose (5% by weight, 10 µl g−1 of the mouse body weight) is also administered subcutaneously.
After the procedures (Extended Data Fig. 2h), the mouse is moved to a temperature-controlled environment using a heat pad (37 °C) and given wet food, as well as ketoprofen (2 mg ml−1, 2.5 µl g−1 of mouse body weight) every 24 h for the next 72 h for analgesic and anti-inflammatory purposes. All animal experiments and housing procedures were conducted in accordance with the Cornell University Institutional Animal Care and Use Committee Guidance, protocol number 2010-0031.
All animal experimentation and housing procedures were conducted in accordance with Cornell University Institutional Animal Care and Use Committee guidance. Mice were housed in a conventional room in the animal core facility of Cornell University; 22.3–22.7 °C and 38–40% humidity; ad libitum access to food and water; 12 h dark and light cycle. Male and female (2–10 months old) mice of strains C57BL/6J and B6.Cg-Tg(CAG–DsRed*MST)1Nagy/J (The Jackson Laboratory) were used.
MOTE measurement in awake mice
As in transcranial surgery, a mouse is placed inside a rodent-anaesthetizing box with a gas mixture of oxygen (1 s.c.c.m.) and isoflurane (2 s.c.c.m.) using the MIDMARK Matrx VIP 3000 Calibrated Vaporizer. Once the mouse is sufficiently sedated (typically within 15 min), the mouse is removed from the anaesthetizing box and fastened to a custom head-fixed stage before waking up (it takes less than 5 min for the mouse to fully wake up). These measurement sessions are abbreviated to accommodate each mouse, with continuous monitoring of its stress throughout to ensure its well-being. Extended Data Fig. 6a shows a top-down view after a mouse is placed on the head-fixed stage in which the transcranial window is visible. Extended Data Fig. 6b,c shows a typical measurement session seen from the side and front, respectively, where the head-fixed stage was tilted to maximize the amount of output pulsed light from the MOTE that reaches the photodiode. A similar setup was used to take MPM images to track the MOTEs after insertion (Extended Data Fig. 6d).
Intravital three-photon microscopy (MPM)
To image the MOTEs implanted in mouse brains, intravital three-photon microscopy was performed, as described in a previous paper53. We used a three-photon microscope (Thorlabs Bergamo II) with a non-collinear optical parametric amplifier (Spectra Physics Sprit) pumped by an amplifier (Spectra Physics Spirit 1030-70) and an objective with a numerical aperture of 1.05 (XLPLN25XWMP2, Olympus). The excitation wavelength and pulse repetition rate were 1,300 nm and 326 kHz, respectively. We used 447 ± 30 nm, 525 ± 25 nm, 585 ± 15 nm and 623 ± 16 nm band-pass filters (Semrock FF01-447/60, FF03-525/50, FF01-585/29 and FF01-623/32, respectively) to detect third harmonic generation, green autofluorescence, DsRed and second harmonic generation, respectively. To separate out four different colours, we used 488 nm, 562 nm and 594 nm of dichroic beamsplitters (Semrock Di02-R488, Di02-R561 and Di02-R594, respectively). IMARIS software (Version 10.2.0; Bitplane) was used for three-dimensional image reconstruction.
PPM decoding algorithm
A silicon-avalanche-photodiode-based photodetector (Thorlabs APD410A) detects the MOTE’s PPM light pulses, and an oscilloscope (PicoScope 6424E), with an 8-bit resolution and a 12.8-ns sampling interval, records the resulting voltage pulses. For real-time and continuous recording despite the limited PicoScope internal buffer size, we continuously pipelined (using 64-bit PicoSDK (v. 10.7.25.290)) the PPM data in 500-ms segments in the binary format (.bin) onto a laptop that is connected to the PicoScope via a USB 3.0 cable. Our decoding algorithm based on Python (v. 3.10.4) then takes the binary files and translates the spacing between the PPM pulses to the corresponding ‘decoded’ voltage values (Fig. 4b).
The decoder recognizes the PPM pulses through thresholding in which the first point above the threshold is registered as the ‘arrival’ time of that pulse, and our code discerns the ‘reference pulses’ from the ‘encoding pulses’ based on their statistical distributions (reference pulses maintain a constant spacing between each other, whereas encoding pulses do not and are dependent on the differential input voltage that a MOTE sees). We then utilize a priori knowledge on pulse spacing, from the CMOS circuit design, to set the minimum and maximum time delays between the PPM pulses. The combination of statistical distribution and a priori circuit knowledge enables us to reconstruct all reference pulses, even in a noisy environment in which several reference peaks might become lost or mistaken. In the case of missing or false encoding peaks, either the immediate neighbour points or the averages of the neighbours patch the gaps.
Histology
Mice are anaesthetized by an intraperitoneal injection of a ketamine/xylazine cocktail (75–90 mg kg−1 of ketamine and 5–8 mg kg−1 of xylazine) or by the inhalation of isoflurane to maintain a breath rate of around once per second. Anaesthesia is monitored by the absence of palpebral reflex and foot withdrawal when pressing the footpad; the breathing rate (around one breath per second) and foot colour (pink) are also closely monitored to avoid over-anaesthesia. The mice are kept on a heat pad throughout the procedure, with the head immobilized and levelled on a Kopf stereotaxic apparatus using the intra-aural positioning studs and tooth bar.
Once stably anaesthetized, the mice are euthanized through an interperitoneal injection of a lethal overdose of pentobarbital (250 mg kg−1 of rodent weight). Perfusion is then performed before the brains of the killed mice are harvested for subsequent analysis: on the observation that animals show no response to a foot pinch, an incision is made laterally along the inferior surface of the rib cage and the thoracic cavity is opened to expose the heart. A 25G butterfly needle is inserted into the left ventricle, with a small incision made in the right atrium. The heart is then infused with 20 ml of phosphate-buffered saline at a rate of 10 ml min−1, followed by 20 ml of 4% paraformaldehyde solution at 10 ml min−1.
To evaluate the impact of MOTE implantation on the health of neuronal tissue, we used a specific marker, Fox-3/Rbfox3/NeuN (Alexa647)54, for neuronal nuclei, GCaMP as a calcium indicator to monitor neuronal activity55, and Iba1 to label microglia as a neuroinflammation marker56, assessing various cellular responses around the implant site. We found that neurons below the MOTEs appear unaffected whereas the area above, directly under the transcranial windows, exhibits microglial activation, suggesting a localized inflammatory response to implantation. Microglia play a crucial role in the response to injury; therefore, their activation in this context is not unexpected. However, Fig. 6a shows that the above-MOTE microglia reaction is similar to that of the control area, which is also under the window, but not in an area in which a MOTE was inserted. This implies that the design and placement of MOTEs effectively minimizes distress to brain tissue. Although the control area actually seems to show more microglia reaction, we believe that this stems from measurement variations. Figure 6a also compares histological findings on a MOTE that had been implanted for six months against an optical fibre, which had been implanted for 3 months (by which time no neural recording could be made). Although we observed capsule formations from foreign body reactions to the 6-month MOTE, we did not observe noticeable capsule formations in the MOTE that had been implanted for 1 month.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data shown in the paper and supporting the findings of this study are available via figshare at https://figshare.com/s/e22c2dc2f6111f0e632d.
Code availability
The decoder used in this study is available via figshare at https://figshare.com/s/e22c2dc2f6111f0e632d.
References
Szarowski, D. H. et al. Brain responses to micro-machined silicon devices. Brain Res. 983, 23–35 (2003).
Song, H. J., Allee, D. R. & Speed, K. T. Single chip system for bio-data acquisition, digitization and telemetry. In Proc. IEEE International Symposium on Circuits and Systems (ISCAS) (ed. Lai, J. K. L.) 1848–1851 (IEEE, 1997).
Nieder, A. Miniature stereo radio transmitter for simultaneous recording of multiple single-neuron signals from behaving owls. J. Neurosci. Methods 101, 157–164 (2000).
Takeuchi, S. & Shimoyama, I. A radio-telemetry system with a shape memory alloy microelectrode for neural recording of freely moving insects. IEEE Trans. Biomed. Eng. 51, 133–137 (2004).
Harrison, R. R. et al. A low-power integrated circuit for a wireless 100-electrode neural recording system. IEEE J. Solid-State Circuits 42, 123–133 (2007).
Biederman, W. et al. A fully-integrated, miniaturized (0.125 mm2) 10.5 µW wireless neural sensor. IEEE J. Solid-State Circuits 48, 960–970 (2013).
Borton, D. A., Yin, M., Aceros, J. & Nurmikko, A. An implantable wireless neural interface for recording cortical circuit dynamics in moving primates. J. Neural Eng. 10, 026010 (2013).
Muller, R. et al. A minimally invasive 64-channel wireless μECoG implant. IEEE J. Solid-State Circuits 50, 344–359 (2015).
Atzeni, G. et al. A 260 × 274 μm2 572 nW neural recording micromote using near-infrared power transfer and an RF data uplink. In Proc. IEEE Symposium on VLSI Technology (eds Jurczak, G. M. & Nikolić, B.) 64–65 (IEEE, 2022).
Lee, J. et al. Neural recording and stimulation using wireless networks of microimplants. Nat. Electron. 4, 604–614 (2021).
Seo, D. et al. Wireless recording in the peripheral nervous system with ultrasonic neural dust. Neuron 91, 529–539 (2016).
Ghanbari, M. M. et al. A sub-mm3 ultrasonic free-floating implant for multi-mote neural recording. IEEE J. Solid-State Circuits 54, 3017–3030 (2019).
Piech, D. K. et al. A wireless millimetre-scale implantable neural stimulator with ultrasonically powered bidirectional communication. Nat. Biomed. Eng. 4, 207–222 (2020).
Kahn, J. M., Katz, R. H. & Pister, K. S. J. Emerging challenges: mobile networking for ‘Smart Dust’. J. Commun. Netw. 2, 188–196 (2000).
Lee, S. et al. A 250 μm × 57 μm microscale opto-electronically transduced electrodes (MOTEs) for neural recording. IEEE Trans. Biomed. Circuits Syst. 12, 1256–1266 (2018).
Lim, J. et al. A light-tolerant wireless neural recording IC for motor prediction with near-infrared-based power and data telemetry. IEEE J. Solid-State Circuits 57, 1061–1074 (2022).
Burton, A. et al. Wireless, battery-free, and fully implantable electrical neurostimulation in freely moving rodents. Microsyst. Nanoeng. 7, 62 (2021).
Shi, C. et al. Application of a sub–0.1-mm3 implantable mote for in vivo real-time wireless temperature sensing. Sci. Adv. 7, eabf6312 (2021).
Weber, M. J. et al. A miniaturized single-transducer implantable pressure sensor with time-multiplexed ultrasonic data and power links. IEEE J. Solid-State Circuits 53, 1089–1101 (2018).
Sonmezoglu, S., Fineman, J. R., Maltepe, E. & Maharbiz, M. M. Monitoring deep-tissue oxygenation with a millimeter-scale ultrasonic implant. Nat. Biotechnol. 39, 855–864 (2021).
Khalifa, A. et al. The microbead: a highly miniaturized wirelessly powered implantable neural stimulating system. IEEE Trans. Biomed. Circuits Syst. 12, 521–531 (2018).
Laursen, K. et al. Ultrasonically powered compact implantable dust for optogenetics. IEEE Trans. Biomed. Circuits Syst. 14, 583–594 (2020).
Lee, A. H., Lee, J., Leung, V., Larson, L. & Nurmikko, A. Patterned electrical brain stimulation by a wireless network of implantable microdevices. Nat. Commun. 15, 10093 (2024).
Zipfel, W. R., Williams, R. M. & Webb, W. W. Nonlinear magic: multiphoton microscopy in the biosciences. Nat. Biotechnol. 21, 1369–1377 (2003).
Horton, N. G. et al. In vivo three-photon microscopy of subcortical structures within an intact mouse brain. Nat. Photon. 7, 205–209 (2013).
Barretto, R. P. J., Messerschmidt, B. & Schnitzer, M. J. In vivo fluorescence imaging with high-resolution microlenses. Nat. Methods 6, 511–512 (2009).
Lee, S. et al. A 330 μm × 90 μm opto-electronically integrated wireless system-on-chip for recording of neural activities. In Proc. IEEE International Solid-State Circuits Conference (ISSCC) (eds Mercier, P. P. & Yan, L.) 292–294 (IEEE, 2018).
Lee, S. et al. Fabrication of injectable micro-scale opto- electronically transduced electrodes (MOTEs) for physiological monitoring. J. Microelectromech. Syst. 29, 720–726 (2020).
Cortese, A. J. et al. Microscopic sensors using optical wireless integrated circuits. Proc. Natl Acad. Sci. USA 117, 9173–9179 (2020).
Molnar, A. C. et al. Nanoliter-scale autonomous electronics: advances, challenges, and opportunities. In Proc. IEEE Custom Integrated Circuits Conference (CICC) (eds Chakraborty, S. & Sankaran, S.) 1–6 (IEEE, 2021).
Haydaroglu, I. & Mutlu, S. Optical power delivery and data transmission in a wireless and batteryless microsystem using a single light emitting diode. J. Microelectromech. Syst. 24, 155–165 (2015).
Tedford, C. E., DeLapp, S., Jacques, S. & Anders, J. Quantitative analysis of transcranial and intraparenchymal light penetration in human cadaver brain tissue. Lasers Surg. Med. 47, 312–322 (2015).
Podgorski, K. & Ranganathan, G. Brain heating induced by near-infrared lasers during multiphoton microscopy. J. Neurophysiol. 116, 1012–1023 (2016).
Jain, A. et al. Minimizing residues and strain in 2D materials transferred from PDMS. Nanotechnology 29, 265203 (2018).
Fan, B., Wolfrum, B. & Robinson, J. T. Impedance scaling for gold and platinum microelectrodes. J. Neural Eng. 18, 056025 (2021).
Homma, Y. & Tsunekawa, S. Planar deposition of aluminum by RF/DC sputtering with RF bias. J. Electrochem. Soc. 132, 1466 (1985).
Shannon, C. E. A mathematical theory of communication. Bell Syst. Tech. J. 27, 379–423 (1948).
Hemmati, H., Biswas, A. & Djordjevic, I. B. Deep-space optical communications: future perspectives and applications. Proc. IEEE 99, 2020–2039 (2011).
van der Ziel, A. Noise: Sources, Characterization, Measurement (Prentice-Hall, 1954).
Wallace, M. et al. Nuclear damage in LMNA mutant iPSC-derived cardiomyocytes is associated with impaired lamin localization to the nuclear envelope. Mol. Biol. Cell 34, ar113 (2023).
Sandhu, L. C., Warters, R. L. & Dethlefsen, L. A. Fluorescence studies of Hoechst 33342 with supercoiled and relaxed plasmid pBR322 DNA. Cytometry 6, 191–194 (1985).
Dou, W. et al. A carbon-based biosensing platform for simultaneously measuring the contraction and electrophysiology of iPSC-cardiomyocyte monolayers. ACS Nano 16, 11278–11290 (2022).
Petersen, C. C. H. Sensorimotor processing in the rodent barrel cortex. Nat. Rev. Neurosci. 20, 533–546 (2019).
Wang, T. et al. Quantitative analysis of 1,300-nm three-photon calcium imaging in the mouse brain. eLife 9, e53205 (2020).
Yecies, D. et al. Speckle modulation enables high-resolution wide-field human brain tumor margin detection and in vivo murine neuroimaging. Sci. Rep. 9, 10388 (2019).
Augustinaite, S. & Kuhn, B. Chronic cranial window for imaging cortical activity in head-fixed mice. STAR Protoc. 1, 100194 (2020).
Senzai, Y., Fernandez-Ruiz, A. & Buzsáki, G. Layer-specific physiological features and interlaminar interactions in the primary visual cortex of the mouse. Neuron 101, 500–513.e5 (2019).
Ghajari, S., Lee, S., Norris, S. L., McEuen, P. L. & Molnar, A. C. Redox-enabled microscale opto-electronically transduced electrodes (ReMOTEs). In Proc. IEEE International Symposium on Circuits and Systems (ISCAS) (ed. Murmann, B.) 1–5 (IEEE, 2023).
Bevilacqua, F. et al. In vivo local determination of tissue optical properties: applications to human brain. Appl. Opt. 38, 4939–4950 (1999).
Wang, M., Kim, M., Xia, F. & Xu, C. Impact of the emission wavelengths on in vivo multiphoton imaging of mouse brains. Biomed. Opt. Express 10, 1905–1918 (2019).
Li, X. et al. Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 324, 1312–1314 (2009).
Sharma, A., Li, G., Rajarajan, K., Hamaguchi, R., Burridge, P. W. & Wu, S. M. Derivation of highly purified cardiomyocytes from human induced pluripotent stem cells using small molecule-modulated differentiation and subsequent glucose starvation. J. Vis. Exp. 97, e52628 (2015).
Hontani, Y. et al. Deep-tissue three-photon fluorescence microscopy in intact mouse and zebrafish brain. J. Vis. Exp. 186, e63213 (2022).
Gusel’nikova, V. V. & Korzhevskiy, D. E. NeuN as a neuronal nuclear antigen and neuron differentiation marker. Acta Naturae 7, 42–47 (2015).
Nakai, J., Ohkura, M. & Imoto, K. A high signal-to-noise Ca2+ probe composed of a single green fluorescent protein. Nat. Biotechnol. 19, 137–141 (2001).
Ohsawa, K., Imai, Y., Sasaki, Y. & Kohsaka, S. Microglia/macrophage-specific protein Iba1 binds to fimbrin and enhances its actin-bundling activity. J. Neurochem. 88, 844–856 (2004).
Acknowledgements
We acknowledge the Cornell NanoScale Facility staff, particularly T. Pennell, J. Clark, C. Alpha, G. Bordonaro and V. Genova, for their guidance and support throughout the integration fabrication processes, Y. Yang and X. Gong of Nanyang Technological University (NTU) for optoelectrical characterization of the PVLEDs, and J. Fetcho of Cornell University; S. Chae, K. A. Khor and L. Wei of NTU; J. Chua of National University of Singapore; A. Kohan of University of Pittsburgh; H. Kim of Hanyang University; and J. C. Shin of EPI Solution for helpful discussions. This work was supported in part by the National Institutes of Health under grants R21-EY027581 (S.L., J.H.G., P.L.M., C.X. and A.C.M.), U01-NS107687 (S.L., S.G., S.S., A.J.C., K.C., A.M., C.W., P.M., J.U.P., S.Z., C.S., T.W., J.H.G., P.L.M., C.X. and A.C.M.) and R01-HL082792 (H.Z., M.W. and J.L.), Nanyang Technological University, Singapore, under Nanyang Assistant Professorship (S.L., Y.Z. and R.J.), Imperial/NTU CYber Protection for HEalthcaRe (IN-CYPHER) programme of the National Research Foundation, Prime Minister’s Office, Singapore, under its Campus for Research Excellence and Technological Enterprise (CREATE) programme (S.L., Y.Z. and R.J.), by the Ministry of Education, Singapore, under its Academic Research Fund Tier 1 (RG141/23) (S.L., Y.Z. and R.J.) and Tier 2 (MOE-T2EP50124-0021) (S.L., Y.Z. and R.J.), and the ASPIRE League Partnership Seed Fund 2024 (S.L.). The integration fabrication work was performed in part at the Cornell NanoScale Facility, a member of the National Nanotechnology Coordinated Infrastructure (NNCI), which is supported by the National Science Foundation (grant NNCI-2025233), and at the Nanyang NanoFabrication Centre, NTU, Singapore.
Author information
Authors and Affiliations
Contributions
S.L., J.H.G., P.L.M., C.X. and A.C.M. conceived the experiments. S.L. and A.C.M. designed the CMOS circuits. A.J.C. designed and fabricated the AlGaAs PVLEDs and transferred them onto CMOS. S.L. designed and performed the integration, fabrication, measurement setup, transcraniotomy and MOTE insertion surgery. S.L., S.G. and S.S. fabricated the MOTEs. H.Z., M.W. and J.L. provided the cardiomyocytes and guided in their use and experimental interpretations. S.L., H.Z., S.S., J.C.W. and T.A.C. conducted the in vitro experiments. S.L., S.G., A.M., C.W., T.W., S.Z. and C.S. conducted the in vivo experiments. S.L., Y.Z., R.J., P.M. and J.U.P. developed the in vivo measurement and data acquisition setup. Y.Z. and R.J. developed the mouse whisker stimulation setup and the adaptive decoding algorithm. W.G., S.L. and M.R.W. performed histological analyses. K.C. and A.M. took the MPM images of in-brain MOTEs. All authors reviewed the final manuscript and contributed to the editing. T.W. and W.F. performed the Monte Carlo simulations for the optical link efficiency characterization across the link angles.
Corresponding author
Ethics declarations
Competing interests
A.J.C., A.C.M. and P.L.M. are the co-founders of OWIC Technologies Inc., a company developing microscopic optical smart ID tags that use similar CMOS posting processes for etching, shielding and release. A.J.C., A.C.M., P.L.M. and S.L. are inventors of a US patent (number 12/039,389) that covers wireless, optically powered optoelectronic sensors based on the heterogeneously integrated CMOS platform. The other authors declare no competing interests.
Peer review
Peer review information
Nature Electronics thanks José A. Plaza and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Staining of human-induced pluripotent stem cell derived cardiomyocytes (iPSC-CM) used.
a, Cardiac troponin T. b, Actin. c, Lamin B1. d, 4’,6-diamidino-2-phenylindole (DAPI). e, Merged. This iPSC-CM has been used in multiple projects of which findings from staining have been similar.
Extended Data Fig. 2 Transcranial surgery flow.
a, Before surgery. b, After shaving. c, Skin removal. d, Skull removal. e, Pre-insertion. f, Insertion. g, Post insertion. h, Window installation. i, Installed window (mouse 2). j, After surgery.
Extended Data Fig. 3 Angular dependency of MOTE link efficiency.
a, Monte Carlo simulation33,44 results of the collection efficiency, PV cell flux, and link efficiency of a MOTE at varying depths and angles within the brain. To better illustrate the angular dependency of the link efficiency, the bottom plots have been normalized by the 0 ° values. b, MOTEs under the agarose-beads mixture (200 µm thick). c, The angle-dependent link efficiency measurement setup. d, Summary of experimental results, where the link output is defined as the average height of the MOTE PPM pulse.
Extended Data Fig. 4 Mouse 4’s ECoG recording over 116 days.
a, Overlay plot on day 4 where MOTE’s ECoG measurement shows sustained fluctuations with a noticeable association with the stimuli: the purple arrow denotes the stimulation rod moving downward towards the whiskers and the green arrow denotes the opposite movement (the stimulation rod moves upward towards the whiskers). b, Overlay plot on day 4 for an ECoG measurement without stimulation. c, Overlay plot on day 116 where the signals have become smaller and the association to the stimuli has also become weakened, although the overall amplitude is stronger than in the control case shown in d. d, Overlay plot on day 116 for an ECoG measurement without stimulation. e, Stereoscopic images over 116 days to show how the MOTE drifts over time – once post-surgery inflammation subsides (which typically lasts 1-3 weeks46), the MOTE is fairly stationary.
Extended Data Fig. 5 Mouse 5’s ECoG recording on day 51.
a, Overlay plot on day 51 where the ECoG shows a strong association with a ‘downward’ stimulus – the purple arrow denotes the stimulation rod moving downwards towards the whiskers and the green arrow denotes the opposite movement (stimulation rod moves upwards towards the whiskers). b, Overlay plot on day 51 without stimulation, of which the ECoG recording is much quieter than in a. c, Stereoscopic image of the MOTE in mouse 5 on day 51.
Extended Data Fig. 6 MOTE in vivo measurements in awake mice.
a, Head-fixed stage. b, Awake measurement in action (side). c, Measurement (front). d, Multi-photon microscope (MPM) imaging setup.
Extended Data Fig. 7 Intravital three-photon microscopy images of MOTEs in mouse brains.
a, MOTE 1 inside mouse 1 on day 128, b, MOTE 1 inside mouse 2 on day 132, c, MOTE 2 inside mouse 3 on day 8 (left) and day 127 (right). Auto-fluorescence was detected in 525 ± 25 nm by using 1300 nm excitation source. The black arrows (on the sides of the bottom figures) indicate the brain surface where the top of the z-axis is the bottom of the implanted cover glass; sometimes there is a space between the cover glass and the brain surface (for example, MOTE 2 inside mouse 3 on day 8 shown in c). All scale bars (yellow horizontal lines) are 100 µm and we have measured six different MOTEs over three different dates with similar findings (also see Supplementary Videos 4–7).
Extended Data Fig. 8 LFP recording of mouse 1 on day 365.
a, Overlay plot (40 traces with the average sound in black) on day 365 where the LFP shows, although weak, an association to a ‘downward’ stimulus – as in previous figures, the purple arrow denotes the stimulation rod moving downward towards the whiskers and the green arrow denotes the opposite movement. b, Overlay plot (40 traces with the average sound in black) on day 365 without stimulation, of which the LFP recording is much quieter than in a. c, Stereoscopic image of the MOTE in mouse 5 on day 365.
Supplementary information
Supplementary Information (download PDF )
Supplementary Sections 1–8, Figs. 1–7 and Tables 1 and 2.
Supplementary Video 1 (download MP4 )
Beating cardiac cells over MOTEs.
Supplementary Video 2 (download MOV )
Whisker stimulation during the in vivo validation.
Supplementary Video 3 (download MP4 )
MOTEs insertion during a transcranial surgery.
Supplementary Video 4 (download MP4 )
Multiphoton Microscopy Imaging on MOTE on mouse 2’s MOTE 1.
Supplementary Video 5 (download MP4 )
Multiphoton Microscopy Imaging on MOTE on mouse 1’s MOTE 1.
Supplementary Video 6 (download MP4 )
Multiphoton Microscopy Imaging on MOTE on mouse 3’s MOTE 1.
Supplementary Video 7 (download MP4 )
Multiphoton Microscopy Imaging on MOTE on mouse 3’s MOTE 2.
Supplementary Video 8 (download MOV )
PVLED emission when driven as an LED.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, S., Ghajari, S., Sadeghi, S. et al. A subnanolitre tetherless optoelectronic microsystem for chronic neural recording in awake mice. Nat Electron 8, 1259–1271 (2025). https://doi.org/10.1038/s41928-025-01484-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41928-025-01484-1