Abstract
Psoriasis is a chronic inflammatory skin condition that is associated with endocrine imbalances and immune system dysregulation. Anhydroicaritin (ANH), derived from Bushen Huayu Decoction, possesses a variety of bioactivities. However, the therapeutic potential of ANH and the underlying molecular mechanisms in psoriasis remain poorly understood. In this study, we evaluate the efficacy of ANH in an imiquimod (IMQ)-induced psoriasis female mouse model and investigated its effects on human keratinocytes along with the mechanistic pathways involved. Our data show that ANH spray significantly improves skin lesions by reducing abnormal proliferation, cytokine release, and infiltration of Th1/Th17 cells in both lipopolysaccharide (LPS)-stimulated HaCaT cells and IMQ-induced lesional model mice. Moreover, extracellular vesicle (EV)-ANH demonstrates enhanced therapeutic effects. Furthermore, RNA-seq indicates that the therapeutic effect of ANH is achieved through the inhibition of acyl-CoA ligase family member 4 (ACSL4)-dependent ferroptosis. These results suggest that ANH holds promise as a therapeutic agent for psoriasis treatment.

Similar content being viewed by others
Introduction
Psoriasis, alternatively called psoriasis vulgaris, presents with common clinical manifestations such as pruritus, erythema, and scaling1. Research has shown that the onset of psoriasis is related to infection, genes, and immune dysregulation2,3. Immunologically, a key feature is abnormal immune activation: overactivated T cells generate various cytokines, including interleukin-23 (IL-23) and tumor necrosis factor-α (TNF-α)4, which cause excessive proliferation and abnormal differentiation of keratinocytes, leading to the formation of classic psoriatic skin lesions5. In addition, the disequilibrium between oxidative stress and antioxidation within the skin microenvironment serves as a crucial foundation for the onset and progression of skin diseases, with oxidative stress being present in numerous skin conditions6.
Approximately 60 million people are estimated to be affected by psoriasis globally, with the global prevalence ranging from 0.14% to 1.99%. In East Asia, the incidence of psoriasis has reached 0.14%7,8. Topical medications, phototherapy, and biologics are available for psoriasis, but they may induce side effects2,3. Thus, there is an immediate need to develop new, well-tolerated treatments capable of alleviating symptoms and modifying the disease pathology of psoriasis. The advantages of traditional Chinese medicine (TCM) have progressively manifested themselves, with its principles of a holistic approach and treatment tailored to syndrome differentiation. TCM has played a significant role in the prevention and management of psoriasis9,10. Epimedium is a traditional Chinese medicinal herb whose core active ingredients include icariin, anhydroicaritin, and various flavonoids11. It is traditionally used to tonify the kidneys and yang, strengthen tendons and bones, and exhibits anti-inflammatory and antioxidant properties. Modern research has shown that epimedium has significant effects on improving male sexual dysfunction, preventing osteoporosis, regulating immunity, combating fatigue, and protecting the cardiovascular system12.
Anhydroicaritin (ANH), a flavonoid, has biological and pharmacological properties, such as antioxidant, antibacterial, antiviral, and many other functions, and has been widely applied in health foods because of its physiologically active functions13,14. ANH is added to antifatigue drinks, men’s health products, antiaging health foods, and sports nutrition supplements to increase immunity, improve body endurance, and delay aging15,16. Extracellular vesicle (EV), nanoscale small-molecule biovesicles, play a significant role in wound healing17. Remarkably, extracellular vesicle secreted by mesenchymal stem cells (MSC-EV) have biological functions comparable to those of MSCs and offer exclusive advantages over cell therapy18. Acting as bilayer lipid nanovesicles that facilitate intercellular communication, EV can deliver endogenous bioactive substances to target recipient cells and, as a result, exert therapeutic effects19,20. Additionally, EV are able to transport monomers from traditional Chinese medicine with low water solubility, improving drug bioavailability17.
Ferroptosis is a type of iron-dependent and regulated cell death triggered by lipid peroxidation. It plays a role in diverse pathophysiological processes in various diseases, such as tumor immunity, neuropathy, and psoriasis21,22,23. Recently, an analysis of psoriatic skin lesions revealed that the expression level of glutathione peroxidase 4 (GPX4), a crucial regulator of ferroptosis, is lower than that in uninvolved skin and healthy skin samples22. In addition, calcipotriol inhibits the proliferation of HaCaT cells by regulating GPX4-mediated ferroptosis24. ACSL4 (acyl-CoA synthetase long-chain family member 4) is a key positive regulator of ferroptosis that catalyzes the esterification of long-chain polyunsaturated fatty acids (such as arachidonic acid), converting them into acyl-CoA derivatives and thereby facilitating the synthesis of polyunsaturated phospholipids that are highly susceptible to lipid peroxidation. These phospholipids serve as the primary substrates for lipid peroxidation during ferroptosis. The expression level of ACSL4 directly determines cellular sensitivity to ferroptosis, and its absence or inhibition can significantly confer resistance to ferroptosis, highlighting its importance in cancer therapy, nervous system diseases, and liver diseases25,26. Furthermore, during the development of psoriasis, ferroptosis is significantly activated and fuels inflammatory responses by upregulating the expression of ACSL4, implying the occurrence of ferroptosis activation in psoriatic skin lesions27.
Moreover, a number of natural compounds, such as Paris saponin VII, celastrol, and L-menthol, have been applied to treat psoriasis via the regulation of pyroptosis, fibroblast-macrophage crosstalk, and the inflammatory response28,29,30. In the present study, we obtained evidence indicating that ANH, both in its free form and when encapsulated within MSC-EV, mitigates the progression of psoriasis. These compounds improve skin lesions by suppressing the abnormal proliferation of keratinocytes, the secretion of cytokines, and the infiltration of Th1/Th17 cells. In addition, we validated the essential role that ferroptosis plays in the progression of psoriasis. Furthermore, our findings revealed that ANH can effectively prevent IMQ-triggered psoriasis by inhibiting ACSL4 and reducing ferroptosis-induced cell death. Taken together, these results demonstrate that ANH has considerable potential as a promising therapeutic agent for psoriasis.
Results
Detection of promising targets of ANH in psoriasis
Using the GeneCard database and UniProt database, 85 overlapping targets between ANH and psoriasis were obtained (Fig. 1A, B). These 85 candidate targets of ANH for psoriasis treatment were submitted to the STRING database to construct a protein-protein interaction (PPI) network (Fig. 1C). Next, through visualization with Cytoscape 3.9.1, HSP90AA1, AKT1, CTNNB1, PPARG, FOS, EGFR, GSK3B, HIF1A, NF-KB1, and JUN were found to lie in the central area of the PPI network (Fig. 1D, E; Supplementary Table 1). Moreover, the drug-target-pathway-disease network provided a direct depiction of the potential targets of ANH in psoriasis, revealing its enrichment in the AKT1/GSK3B/NF-KB1/AP-1 signaling pathways (Fig. 1F).
A The chemical structure of ANH. B Venn diagram displaying overlapping targets between ANH and psoriasis. C, D PPI networks depicting the antipsoriatic mechanisms of ANH. E The top 10 key targets on the basis of degree centrality ranking. F Diamond symbols represent major psoriasis-related signaling pathways, whereas circles represent common targets shared by ANH and psoriasis patients. G GO enrichment analyses encompassing biological processes, cellular components, and molecular functions. H Outcomes of the KEGG enrichment analysis.
Metascape was used to explore the relationships among these potential targets of ANH in psoriasis through analyses of Gene Ontology (GO) biological process (BP), cellular component (CC), molecular function (MF), and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment. The GO enrichment results included the inflammatory response, the ficolin-1-rich granule lumen, and protein kinase activity (Fig. 1G). For KEGG, the potential targets involved in ANH treatment were significantly associated with the “HIF-1 signaling pathway”, “IL-17 signaling pathway”, “Th17 cell differentiation”, and “PI3K-AKT signaling pathway” (Fig. 1H). These data indicate that ANH modulates inflammatory and immune processes.
ANH exerts a suppressive effect on psoriasis
HaCaT cells were utilized to investigate how ANH affects cell proliferation and the inflammatory response. The cells were treated with ANH (0, 10, 20, or 40 μM) in the presence or absence of LPS for 24 or 48 h. The CCK-8 assay revealed that the cell viability of the ANH-treated group was comparable to that of the control (CON) group. Notably, ANH potently inhibited the abnormal proliferation of keratinocytes induced by LPS (Fig. 2A, B). Furthermore, LPS led to significant increases in the levels of the key inflammatory markers interleukin-6 (IL-6), TNF-α, interleukin-1β (IL-1β), and IL-23, whereas ANH treatment downregulated their expression (Fig. 2C). These observations demonstrate that ANH modulates the activity of LPS-stimulated HaCaT cells, suggesting a promising therapeutic strategy to counteract excessive cell proliferation and the release of proinflammatory cytokines.
A, B A CCK-8 assay was used to assess the viability of HaCaT cells treated with ANH (0, 10, 20, and 40 µM) with or without LPS (1 µg/mL) for 24 and 48 h (n = 3). C qRT-PCR was conducted to quantify the mRNA expression levels of IL-6, TNF-α, IL-1β, and IL-23 in HaCaT cells stimulated with ANH (0, 10, 20, and 40 µM) with or without LPS (1 µg/mL) for 12 h (n = 3). D After the dorsal hair was shaved, the mice (excluding those in the control group) were given 62.5 mg of IMQ daily from day 1 to day 6. The DXMS group was treated with 26 mg of dexamethasone, and the ANH group was administered 2 mg/mL ANH. E Images of the dorsal skin from each group (n = 5) on day 6. F Calculated psoriasis area severity index (PASI) scores. G–I Daily evaluations of scaling, epidermal thickening, and erythema severity. J Spleen indices of all the groups. The p-values were calculated via one-way ANOVA followed by Tukey’s multiple comparisons test for (A–C, J). P-values were calculated via two-way ANOVA followed by Tukey’s multiple comparisons test for (F–I). (Mean ± SD; CON, #P < 0.05, ##P < 0.01, ###P < 0.001, ####P < 0.0001; VS. LPS, *P < 0.05, ***P < 0.001, ****P < 0.0001 VS. CON, #### P < 0.0001; VS. IMQ, **P < 0.01, ***P < 0.001; ns= not significant).
To evaluate the therapeutic potential of ANH in vivo, we constructed an IMQ-induced psoriasis-like mouse model and treated the mice with ANH (Fig. 2D). IMQ application successfully induced psoriasis-like lesions in C57BL/6 mice, with characteristic pathological features evident in the model group (Fig. 2E). Compared with the model group, the ANH group presented significantly improved key psoriasis-like symptoms, including scale formation, increased skin thickness, and erythema (Fig. 2F–I). Furthermore, combined treatment with ANH and dexamethasone (DXMS) effectively reduced spleen enlargement and markedly decreased the spleen index (Fig. 2J). Collectively, these data indicate that ANH treatment alleviates cutaneous and systemic symptoms in IMQ-induced psoriatic mice.
ANH restrains IMQ-induced keratinocyte proliferation and immune cell accumulation
The inflammatory response is one of the key drivers of psoriasis pathogenesis. Consequently, inhibiting cytokine production is also a major strategy for alleviating psoriasis31. To better understand how ANH affects psoriasis, we conducted an in-depth investigation into the release of these cytokines. As expected, the gene and protein expression levels of interleukin-17 (IL-17), TNF-α, IL-1β, and IL-23 were elevated in skin extracts. Moreover, ANH was able to reduce the levels of these cytokines (Supplementary Fig. 1), which was consistent with the in vitro findings.
The interplay between cytokine clusters and immune cells promotes the development of psoriasis32. As shown in Fig. 3A, B, epidermal thickening, parakeratosis, and acanthosis were markedly reduced following treatment with DXMS and ANH (Supplementary Table 2). Furthermore, compared with the IMQ group, ANH treatment significantly decreased the elevated levels of proliferation markers such as Ki67 and proliferating cell nuclear antigen (PCNA) (Fig. 3C–F). The results also indicated that ANH treatment strongly suppressed the overexpression of K17 (Fig. 3G, H).
A, B Hematoxylin-eosin (HE) staining of psoriatic skin lesions with a quantitative measurement of epidermal thickness. C, D Ki67 immunostaining was performed, and positive cells were counted. E, F PCNA immunostaining with enumeration of positive cells. G, H K17 immunohistochemistry in which positive cells were counted. I Immunofluorescence analysis of CD4, IFN-γ, and IL-17 colocalization in skin tissue. J Quantitative analysis of the fluorescence intensity for cytokine expression. K Proportions of splenic CD4+IFN-γ+ cells. Counts of splenic CD4+IL-17A+ cells. The p - values were calculated via one-way ANOVA followed by Tukey’s multiple comparisons test for (B, D, F, H, J, K). (Scale bar: 0.1 mm, n = 5, mean ± SD; VS. CON, ###P < 0.001, ####P < 0.0001; VS. IMQ, **P < 0.01, ***P < 0.001, ****P < 0.0001).
To increase our understanding of the alterations in immune cells, we accurately assessed the degree of inflammatory cell infiltration in skin and spleen samples. Our data clearly revealed that ANH significantly reduced the infiltration of Th1 and Th17 cells in both the spleen and skin lesions (Fig. 3I–K). These results provide further strong evidence that ANH effectively reduces abnormal keratinocyte proliferation and the accumulation of immune cells in IMQ-induced nephropathy in mice.
EV-ANH ameliorates psoriasis
Owing to the potential photochemical activity of flavonoids, which may increase skin sensitivity to ultraviolet light and cause phototoxicity or photoallergic reactions33, we employed MSC-EV as drug carriers to improve the safety profile of ANH. MSCs were cultured and identified via FACS (Fig. 4A, B). Moreover, EV were isolated from the cell culture medium (Fig. 4 C) and characterized via transmission electron microscopy (TEM), nanoparticle tracking analysis (NTA), and Western blotting (WB). EV exhibited a median diameter of 155.1 nm, as measured by NTA, and displayed the characteristic cup-shaped morphology under TEM (Fig. 4D). WB analysis confirmed the presence of CD81 and the absence of calnexin (Fig. 4E). ANH was encapsulated in EV through sonication at room temperature. NTA and TEM analysis demonstrated that the structural integrity and morphology of the EV retained after ANH loading (Fig. 4F). The encapsulation efficiency of ANH in EV was quantified by high-performance liquid chromatography (HPLC), and it was found that the drug loading of EV increased proportionally with the ANH concentration, reaching its peak loading efficiency at 1000 μg/mL (Fig. 4G).
A, B Characterization of MSCs, Scale bar: 200 μm and 100 μm. C Isolation workflow for MSC-derived exosomes. D TEM and NTA visualization of EV architecture Scale bar: 200 nm. E WB quantification of EV markers. F TEM and NTA visualization of the EV-ANH architecture. G Concentration-dependent loading capacity and efficiency of ANH.
We next evaluated the effects of EV treatment on cell proliferation and viability. The cells were subjected to EV treatments at concentrations of 0, 1, 2.5, and 5 μg/mL, either in the presence or absence of LPS, for durations of 24 and 48 h. The CCK-8 assay showed that EV treatment alone did not adversely affect HaCaT cell viability. However, EV significantly suppressed the LPS-induced hyperproliferation of these cells (Fig. 5A, B). In line with the preceding data, EV markedly reduced the expression of inflammatory markers (Fig. 5C). Moreover, treatment with either EV alone or EV-ANH alleviated psoriasis-like symptoms. The EV-ANH combination demonstrated superior therapeutic efficacy to EV alone, as shown by greater improvements in skin thickness, erythema, and the spleen index (Fig. 5D–J).
A, B A CCK-8 assay was used to assess the viability of HaCaT cells treated with EV (0, 1, 2.5, or 5 µg/mL) with or without LPS (1 µg/mL) for 24 or 48 h (n = 3). C qRT-PCR was conducted to quantify the mRNA expression levels of IL-6, TNF-α, IL-1β, and IL-23 in HaCaT cells after 12 h of treatment with LPS (1 µg/mL) or EV (0, 1, 2.5, or 5 µg/mL) (n = 3). D After the dorsal hair was shaved, the mice (excluding those in the control group) were given 62.5 mg of IMQ daily from day 1 to day 6. The EV group was treated with 50 µg/mouse of EV, and the EV-ANH group was administered 2 mg/mL of EV-ANH. E Images of the dorsal skin from each group (n = 5) on day 6. F Calculated psoriasis area severity index (PASI) scores. G–I Daily evaluations of scaling, epidermal thickening, and erythema severity. J Spleen indices of all the groups. The p -values were calculated via one-way ANOVA followed by Tukey’s multiple comparisons test for (A–C, J). P-values were calculated via two-way ANOVA followed by Tukey’s multiple comparisons test for (F–I). (Mean ± SD; CON, ###P < 0.001; VS. LPS, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001; VS. CON, ####P < 0.0001; VS. IMQ, ***P < 0.001, ****P < 0.0001; ns = not significant).
To investigate the mechanism underlying the therapeutic effect of the EV-ANH, we conducted tests on the skin lesions of the mice. The results showed that it could inhibit the secretion of cytokines (Supplementary Fig. 2) and the abnormal proliferation of keratinocytes (Fig. 6A–H; Supplementary Table 2). Consistent with these findings, the accumulation of Th1 and Th17 cells in the corresponding spleen and skin lesion tissues also decreased accordingly (Fig. 6I–K).
A, B Hematoxylin-eosin (HE) staining of psoriatic skin lesions with a quantitative measurement of epidermal thickness. C, D Ki67 immunostaining was performed, and positive cells were counted. E, F PCNA immunostaining with enumeration of positive cells. G, H K17 immunohistochemistry was used to count positive cells. I Immunofluorescence analysis of CD4, IFN-γ, and IL-17 colocalization in skin tissue. J Quantitative analysis of the fluorescence intensity for cytokine expression. K Proportions of splenic CD4+IFN-γ+ cells. Counts of splenic CD4+IL-17A+ cells. The p -values were calculated via one-way ANOVA followed by Tukey’s multiple comparisons test for (B, D, F, H, J, K). (Scale bar: 0.1 mm, n = 5, mean ± SD; VS. CON, ### P < 0.001, #### P < 0.0001; VS. IMQ, ** P < 0.01, *** P < 0.001, **** P < 0.0001).
In conclusion, while both EV and EV-ANH treatments effectively suppressed keratinocyte hyperproliferation, inflammatory infiltration, and immune cell accumulation in psoriatic lesions, the EV-ANH combination demonstrated significantly superior therapeutic efficacy.
ANH improves psoriasis via ACSL4
To further investigate the mechanism by which ANH affects psoriasis, RNA sequencing (RNA-seq) was conducted on skin tissues from the IMQ and ANH groups. Principal component analysis (PCA) clearly revealed separation between the groups, confirming the reliability of the transcriptional profiles (Fig. 7A). As shown in Fig. 7B, a total of 2200 differentially expressed genes (DEGs) were identified in the ANH group, with 902 upregulated and 1298 downregulated genes. Additionally, enrichment analysis of the DEGs revealed that, compared with the IMQ group, the ANH group presented reduced expression of key genes such as IL23r, IL17f, IL1b, IL17rb, IL21r, and IL4ra (Fig. 7C). Subsequent KEGG pathway analysis of these DEGs revealed a close association between the ferroptosis signaling pathway and the development of IMQ-induced psoriasis (Fig. 7D). We then examined the changes in the expression of ferroptosis-related genes among the DEGs. This analysis identified ACSL4 and SLC7A11 as the genes whose expression was most significantly altered (Fig. 7E). Subsequent WB experiments revealed that ANH predominantly affected the expression of ACSL4, as opposed to having a substantial influence on the expression of SLC7A11 (Fig. 7F).
A. Dimensionality reduction analysis via PCA. B. Volcano plot showing transcriptomic changes. C. Heatmap illustrating coregulated target genes. D. KEGG pathway analysis for the functional annotation of genes. E. DEG analysis of ferroptosis-related genes. F. Representative western blot bands and quantitative analysis showing the relative levels of ACSL4 and SLC7A11 compared with those of GAPDH. The p -values were calculated via one-way ANOVA followed by Tukey’s multiple comparisons test for F. (n = 3, mean ± SD; VS. CON, ### P < 0.001, #### P < 0.0001; VS. IMQ, * P < 0.05, *** P < 0.001, **** P < 0.0001; ns= no significance).
To further validate the role of ferroptosis, we treated IMQ-induced psoriatic mice with the ferroptosis inhibitor ferrostatin-1 (Fer-1) alongside ANH (Fig. 8A). These findings revealed that the therapeutic effect of ANH was comparable to that of Fer-1. Both treatments were capable of ameliorating skin lesion symptoms in the mice (Fig. 8B-G). Moreover, we also observed a significant reduction in the expression of the ferroptosis markers ACSL4 and 4-HNE following ANH treatment, whereas the expression level of SLC7A11 remained relatively unchanged (Fig. 8H-I).
A. After the dorsal hair was shaved, the mice (excluding those in the control group) were given 62.5 mg of IMQ daily from day 1 to day 6. The Fer-1 group was treated with 0.8 mg/mL Fer-1, and the ANH group was administered 2 mg/mL ANH. B. Images of the dorsal skin from each group (n = 5) on day 6. C. Calculated psoriasis area severity index (PASI) scores. D-F. Daily evaluations of scaling, epidermal thickening, and erythema severity. G. Spleen indices of all groups. H. Immunofluorescence analysis of ACSL4, SLC7A11, and 4-HNE expression in skin tissue Scale bar: 0.1 mm. I. Quantitative analysis of the fluorescence intensity for cytokine expression. The p -values were calculated via two-way ANOVA followed by Tukey’s multiple comparisons test for C, D, E, F. P-values were calculated via one-way ANOVA followed by Tukey’s multiple comparisons test for G and I. (Mean ± SD; VS. CON, #### P < 0.0001; VS. IMQ, * P < 0.05, ** P < 0.01, *** P < 0.001, **** P < 0.0001; ns= not significant).
To validate these findings in vitro, we also utilized LPS-induced HaCaT cells to confirm that ANH inhibited ferroptosis and the expression of ACSL4 (Fig. 9A-D). Through molecular docking experiments, we determined that the binding energy between ANH and ACSL4 was -8.7 kcal/mol (Fig. 9E). To further test our hypothesis, we constructed a cell line in which ACSL4 was knocked down (Fig. 9F). We observed that following the knockdown of ACSL4, the ability of ANH to hinder ferroptosis was significantly weakened (Fig. 9G-I).
A. Relative viability of HaCaT cells exposed to LPS (1 µg/mL) combined with Fer-1 (0.5 µM) or ANH (20 µM) for 72 h. B, C. MDA and GSH levels of HaCaT cells exposed to LPS (1 µg/mL) combined with Fer-1 (0.5 µM) or ANH (20 µM) for 72 h. D. ACSL4 expression in HaCaT cells exposed to LPS (1 µg/mL) combined with Fer-1 (0.5 µM) or ANH (20 µM) for 72 h. E. Computational modeling of the binding interactions between ANH and ACSL4. F. Western blot images showing ACSL4 expression in HaCaT cells after infection with sh-ACSL4 lentivirus. G-I. Cell viability and levels of MDA and GSH in NC and sh-ACSL4 HaCaT cells after treatment with LPS or ANH for 72 h. The p -values were calculated via one-way ANOVA followed by Tukey’s multiple comparisons test for (A–C, F). P-values were calculated via two-way ANOVA followed by Tukey’s multiple comparisons test for (G–I). (n = 3, mean ± SD; VS. NC, **P < 0.01, ***P < 0.001, ****P < 0.0001; ns= not significant).
These results indicate that ANH alleviates the progression of psoriasis by targeting the expression of ACSL4 and inhibiting ferroptosis.
Discussion
Psoriasis is characterized by chronic inflammation and the secretion of various cytokines from immune cells, leading to enhanced keratinocyte proliferation. These activated keratinocytes then secrete cytokines, which serve to maintain the inflammatory cycle. Currently, topical treatments are accompanied by significant side effects, indicating that exploring new therapeutic approaches is critical34. Consequently, traditional Chinese medicines (TCMs), including benzoylaconitine and liquiritin, are gaining increasing attention as treatments for psoriasis35,36. Anhydroicaritin is the key bioactive ingredient found in traditional Chinese medicinal herbs. As a natural flavonoid, it has been shown to possess advantageous pharmacological characteristics, such as anti-inflammatory and anticancer properties13. In this study, we reported that ANH ameliorated psoriasis by curbing abnormal cell proliferation, cytokine synthesis, and immune cell migration.
Mice in the IMQ-induced model group developed obvious psoriasis-like skin lesions. Histopathological examination confirmed features consistent with human psoriasis, thereby validating the successful establishment of the psoriasis-like mouse model, in line with previous reports37,38. The daily topical use of ANH effectively alleviated disease severity, as reflected by a reduction in skin lesions. When drug-induced disease peaks, mice exhibit increased spleen weight, significant body weight loss, and a higher spleen index, which is a critical parameter for evaluating psoriasis progression39. Compared with the control group, the model group presented a significantly greater spleen index. However, ANH treatment reversed body weight loss and spleen enlargement. These results suggest that ANH ameliorates the systemic immune dysregulation associated with psoriasis.
In psoriatic skin, upregulated Ki-67 and PCNA expression serve as crucial diagnostic markers for hyperproliferation, distinguishing psoriatic skin from normal skin. Increased Ki-67 levels are correlated with disease activity, whereas increased PCNA levels indicate increased DNA synthesis in psoriatic skin40. Keratin 17 (K17) is pivotal in psoriasis pathogenesis: its abnormal overexpression drives excessive keratinocyte (KC) proliferation, inhibits apoptosis, and functions as an autoantigen to activate T cells, particularly Th17 cells, to induce the secretion of cytokines such as IL-17 and IL-23. This process forms a proinflammatory feedback loop, and the interaction between K17 and immune cells sustains chronic inflammation in psoriatic lesions, making K17 a possible therapeutic target for psoriasis41,42. Our study results indicated that ANH successfully reduced the expression of Ki67, PCNA, and K17 in lesional skin.
Cytokines, including IL-1β, TNF-α, IL-23, and IL-17, contribute to the intensification of immune cells and intensify keratinocyte proliferation. Thalidomide has been shown to alleviate psoriasis by reducing IL-1β expression and attenuating inflammation43. Th1 cells participate in psoriasis pathogenesis through the secretion of proinflammatory cytokines such as IFN-γ and TNF-α. These cytokines trigger the activation and recruitment of other immune cells, such as dendritic cells and macrophages, which drive abnormal keratinocyte proliferation and inflammatory reactions44. The Th1-mediated disrupted immune response causes epidermal thickening, angiogenesis, and the appearance of characteristic erythematosquamous lesions, and it also maintains a chronic inflammatory microenvironment. TNF-α regulates immune and inflammatory responses during the progression of psoriasis45.
Additionally, increased levels of IL-23, another cytokine, stimulate the differentiation of Th17 cells, resulting in increased production of IL-17, which is recognized as a hallmark of psoriasis46. Given the anti-inflammatory properties of ANH in other disease models, we investigated its effects on this cytokine network in psoriasis47. Our findings demonstrated that ANH reduced the expression of various secreted cytokines and inhibited the infiltration of Th1 and Th17 cells, thus alleviating psoriasis and helping to diminish the inflammatory environment in skin lesions.
Although no direct evidence confirms that ANH itself causes phototoxicity, flavonoids, as a class of compounds, possess photochemical activity that can increase skin sensitivity to ultraviolet light and lead to phototoxic reactions33. As a precautionary measure to mitigate potential side effects and improve therapeutic efficacy, we employed mesenchymal stem cell-derived extracellular vesicle (MSC-EV) as a delivery system. This strategy was designed to enhance transdermal delivery, allow for dose reduction, minimize potential irritation, and achieve targeted therapy. MSC-EV have become a research focus in psoriasis therapy because of their low immunogenicity, robust tissue-penetrating capacity, and potent immunomodulatory properties48,49. By delivering functional miRNAs, proteins, and other bioactive molecules, MSC-Exo can rebalance the Th17/Treg axis, restrain keratinocyte hyperproliferation, and downregulate proinflammatory cytokines, thereby alleviating cutaneous lesions and systemic immune dysregulation50,51. In addition, the targeting ability and membrane protection of exosomes overcome the shortcomings of poor water solubility and easy metabolism of small-molecule compounds. Therefore, loading MSC-Exo with herbal monomers such as schisandrol A is expected to produce synergistic benefits and further increase the immunoregulatory efficacy of exosome-based therapeutic strategies17. Consistent with expectations, ANH-loaded EV demonstrated superior therapeutic effects, including the ability to repair skin lesions, reduce inflammatory cytokine release, and attenuate immune cell infiltration.
Ferroptosis, an iron-dependent, lipid peroxidation-driven modality of regulated necrosis, has emerged as a pathogenic engine in psoriasis. Pharmacologic blockade or genetic silencing of ferroptosis alleviates disease severity, positioning the pathway as a druggable axis6,27. Ferroptosis overactivation induces keratinocyte ferroptosis, triggering a cascade of proinflammatory cytokines, such as IL-17/TNF-α. Experimental studies have demonstrated that the ferroptosis inhibitor ferrostatin-1/liproxstatin-1 significantly alleviates dermatitis in mouse models. Notably, a few studies have proposed that promoting ferroptosis might ameliorate symptoms, but the evidence is limited and contradicts mainstream conclusions. The current consensus holds that inhibiting ferroptosis represents a more reliable therapeutic strategy24,52. In this study, RNA-Seq revealed that ANH conspicuously blunted ferroptosis signatures relative to those in the IMQ group, with the lipid-metabolizing enzyme ACSL4 among the most repressed transcripts. Because ACSL4 catalyzes polyunsaturated fatty acid incorporation into phospholipids and thus sensitizes cells to peroxidation, we asked whether its inhibition underpins the cytoprotective effect of ANH. In silico docking predicted nanomolar ANH-ACSL4 engagement, and functional validation revealed that lentiviral delivery of sh-ACSL4, but not empty pLL3.7, phenocopied ANH-mediated suppression of ferroptosis and psoriasiform changes, whereas ACSL4 overexpression reversed protection. Overall, ANH attenuated psoriasis by downregulating ACSL4, thereby regulating lipid peroxidation and subsequent ferroptosis.
Our investigation revealed that EV-ANH serve as multitarget agents against psoriasis: they concurrently curb excessive keratinocyte proliferation and reestablish balance in dysregulated Th1/Th17 immune responses by targeting ACSL4 to inhibit ferroptosis. These findings position EV-ANHs as highly promising therapeutic candidates.
Materials and methods
Identifying the possible targets of ANH in psoriasis
Potential therapeutic targets linked to psoriasis were identified via searches of the GeneCards database with the keyword “psoriasis”. These targets were subsequently submitted to the UniProt database (http://www.uniprot.org). The prospective protein interactors of ANH were initially profiled through SwissTargetPrediction (http://www.swisstargetprediction.ch/) and SuperPred (https://prediction.charite.de/) with the keyword “Anhydroicaritin”, where nonhuman genes were filtered out, redundant entries were eliminated, and standardized gene nomenclature was applied. Next, we presented their intersection via Venn analysis. Overlapping targets between ANH and the disease were uploaded to the STRING database (https://cn.string-db.org/) to construct a protein-protein interaction (PPI) network. These targets were further imported into Cytoscape 3.9.1, where core targets were determined on the basis of specific degree values. Major components were obtained from the RCSB PDB (http://www.rcsb.org/) and PubChem databases. After receptor proteins were hydrogenated and charges were assigned, AutoDockTools 1.5.6 was used to create docking grid boxes at the active sites. Finally, data visualization was carried out via PyMOL and LigPlot+ software53.
Cell culture and viability
The HaCaT and HEK-293T cell lines were obtained from the Type Culture Collection Committee of the Chinese Academy of Sciences in Shanghai, China. These cells were grown in Dulbecco’s modified Eagle’s medium (DMEM) (Catalogue numbers: 11965092 and 15140122; Gibco, USA) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin and streptomycin (P/S) (Catalogue numbers: A5670701 and 15140122; Gibco, USA). The cultures were incubated in a 37 °C environment with a 5% CO₂ atmosphere (Thermo371, Thermo Fisher Scientific, USA). The corresponding volume of DMSO was added to the coincubation system as a control. HaCaT cells (8×103) were seeded into 96-well plates and incubated with various concentrations of ANH (0, 10, 20, and 40 µM) or LPS (1 μg/mL) for 24 and 48 h. CCK8 (Cat. No. HY-K0301, MCE, USA) was added to each well and incubated for 2 h before the absorbance at 450 nm was read on a microplate reader (SpectraMax M2, Molecular Devices, USA). All cell line authentication was performed upon receipt, and the presence of mycoplasma was tested via PCR.
Animals
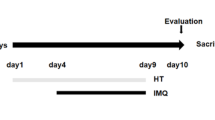
Six- to eight-week-old C57BL/6 female mice were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China). The mice were housed in cages in SPF-rated rooms with free access to food and water. Mice were anaesthetized with isoflurane before handling and euthanized by CO₂ asphyxiation at the end of the experiment. With the exception of those in the control group (CON), 62.5 mg of IMQ cream (5% Aldara, 3 M Pharmaceuticals) was topically applied to the back skin of all the mice once daily for six consecutive days. The mice were randomly divided into four experimental groups and assigned to drug or control groups via sealed envelopes containing an Excel-generated random sequence (n = 5 per group): (1) the control group (CON); (2) the model group (IMQ); (3) the dexamethasone (DXMS) group; and (4) the ANH group. After random allocation, the CON group of mice was treated with an equal quantity of Vaseline ointment. A 5% IMQ cream was applied to the model group of mice. The DXMS group of mice received 26 mg dexamethasone acetate cream. The ANH group of mice was exposed to 2 mg/mL ANH spray (Cat. No. S9080, Selleck, USA) after treatment with IMQ. Dorsal skin alterations and body mass were monitored daily. For EV treatment, the mice were randomly divided into four experimental groups (n = 5 per group): (1) the control group (CON), (2) the model group (IMQ), (3) the EV group, and (4) the EV-ANH group. After random allocation, the mice (excluding those in the control group) were given 62.5 mg of IMQ daily from days 1 to 6. The EV group was treated with 50 µg/mouse of EV, and the EV-ANH group was administered 2 mg/mL of EV-ANH. For Fer-1 treatment, the mice were randomly divided into four experimental groups (n = 5 per group): (1) the control group (CON), (2) the model group (IMQ), (3) the Fer-1 group, and (4) the ANH group. After random allocation, the mice (excluding those in the control group) were given 62.5 mg of IMQ daily from days 1 to 6. The Fer-1 group was treated with 0.8 mg/mL Fer-1 (Cat. No. 347174-05-4; TargetMol, USA), and the ANH group was administered 2 mg/mL ANH. Disease progression was evaluated via PASI scores54. A blinded 0-4 PASI system was used: 0, no erythema or scaling; 1, mild redness with a fine scale; 2, moderate erythema and readily visible scale, plaque elevated; 3, strong erythema, thick scale across most of the lesion, clear plaque height; and 4, severe erythema, coalescent lamellar scale, pronounced thickening. This research was approved by the Ethics Committee of Zhengzhou University (Approval No. 2025-YYY-012). We have complied with all relevant ethical regulations for animal use.
Skin histopathological examination
Tissue samples from the back skin were fixed with a 4% paraformaldehyde (PFA) solution, followed by staining via the hematoxylin and eosin (HE) approach. For the immunohistochemical analyses, the primary antibodies used were rabbit anti-Ki67 (Cat. No. ab15580; Abcam, UK), anti-PCNA (Cat. No. #13110; CST, USA), and anti-K17 (Cat. No. #4543; CST, USA) antibodies. In contrast, the secondary antibody used was polymer-horseradish peroxidase-labeled goat anti-rabbit IgG (Cat. No. ab150077; Abcam, UK).
Immunofluorescence staining
Skin tissue samples were processed via paraffin embedding, tissue sectioning, and other related steps. The obtained skin tissue sections were incubated overnight with anti-CD4 (Cat. No. GB15064, Servicebio, China), anti-IL-17 (GB11110, Servicebio, China), anti-IFN-γ (GB11107, Servicebio, China), 4-HNE (GB150073, Servicebio, China), ACSL4 (abs106075, Absin), and SLC7A11 (abs174692, Absin) antibodies. After this incubation period, suitable secondary antibodies were used to detect and visualize the expression levels of the target proteins.
qRT-PCR
Total RNA was isolated from the samples via TRIzol reagent (Cat. No. 15596018CN; Invitrogen, USA). cDNA synthesis was subsequently conducted via the PrimeScript™ RT Kit (Takara, Japan). Following this step, the qPCR was run on an Applied Biosystems 7500 Real-Time PCR System strictly according to the manufacturer’s guidelines. The primers used for qRT-PCR are listed in Supplementary Table 3.
ELISA
The quantification of inflammatory factors in back skin extracts was performed via ELISA kits from Thermo Fisher Scientific, USA. In particular, the concentrations of mouse IL-1β (88-7013A-88, Thermo Fisher Scientific, USA), mouse IL-17A (88-7371-88, Thermo Fisher Scientific, USA), mouse IL-23 (BMS6017, Thermo Fisher Scientific, USA), and mouse TNF-α (BMS607-3, Thermo Fisher Scientific, USA) were determined following the manufacturers’ instructions (SpectraMax M2, Molecular Devices, USA).
Flow cytometry
Spleens are aseptically collected, minced, and gently pressed through 70 µm strainers into RPMI-1640. Cells are pelleted, erythrocytes lysed with ACK buffer, washed twice, and resuspended in complete medium23. First, the cells were stained with various cell-surface markers: Alexa Fluor 700 anti-CD45 mAb (147715, BioLegend, USA), FITC anti-CD3 mAb (100204, BioLegend, USA), APC anti-CD4 mAb (116013, BioLegend, USA), FITC anti-CD34 hAb (343503, BioLegend, USA), FITC anti-CD45 hAb (982316, BioLegend, USA), FITC anti-CD11b hAb (982614, BioLegend, USA), FITC anti-CD19 hAb (982420, BioLegend, USA), FITC anti-HLA-DR hAb (327006, BioLegend, USA), FITC anti-CD90 hAb (389803, BioLegend, USA), FITC anti-CD105 hAb (323204, BioLegend, USA), and FITC anti-CD73 hAb (344015, BioLegend, USA). For intracellular staining with a PE-CY7 anti-IL-17 mAb (506921, BioLegend, USA) and a PE anti-IFN-γ mAb (163503, BioLegend, USA), the cells were treated with phorbol 12-myristate 13-acetate/ionomycin for 4 h, including GolgiPlug (BD Biosciences). The samples were then assessed on an LSRFortessa flow cytometer (BD Biosciences) and analyzed via FlowJo software v10.8 (TreeStar).
Isolation of MSCs
Once informed consent had been obtained from the 30 years old parturient, 15-20 cm umbilical cords (UCs) were obtained and washed with 4 °C phosphate-buffered saline (PBS). Next, the samples were cut into 10 mm³ fragments and digested in a mixture comprising collagenase with CaCl₂ (3 mM), type I collagenase, and hyaluronidase (0.1%) for 4 h at 37 °C. Following digestion, the samples were cultured with 30 mL low-glucose Dulbecco’s modified Eagle’s medium (DMEM, 1 g/L, Gibco, Life Technologies, NY, USA) enriched with 10% fetal bovine serum (exosome-depleted FBS, PAN-Biotech, Germany) in 175 cm2 culture vessels and incubated in a humidified environment with 5% CO₂ at 37 °C. The culture medium was replaced every three days to eliminate nonadherent cells. All operations were sanctioned by the Research Ethics Committee of Zhengzhou Central Hospital Affiliated with Zhengzhou University and performed under sterile conditions following standard procedures (Approval No. 2024-002).
Characterization and identification of EV
When the confluency of the MSCs reached approximately 60%, the original culture medium was removed. The cells were then rinsed three times with PBS, and serum-free medium was added to replace the original medium. The culture mixture was incubated for an additional 48 h, after which the culture supernatant was harvested. The culture medium was centrifuged step by step. Initially, the mixture was centrifuged at 3200×g for 10 min at 4 °C to eliminate any remaining cells. Next, the samples were subjected to ultracentrifugation at 10,000×g for 30 min at 4 °C, after which the cellular fragments were discarded. Two successive ultracentrifugation procedures at 100,000×g for 2 h at 4 °C were subsequently performed to purify the MSC-EV. The concentration of EV was measured via a BCA assay kit (Beyotime, China). Transmission electron microscopy (TEM, JEM-ARM300F2) and nanoparticle tracking analysis (NTA, Zetaview-PMX120-Z) were employed to analyze the morphological characteristics and dimensional properties of the EV. Western Blot was used to determine the expression of CD81 and Calnexin. Isolated EVs were resuspended in 1× PBS containing 5% trehalose, aliquoted (50 µL) into LoBind tubes, snap-frozen in liquid nitrogen, and stored at –80 °C for ≤ 6 months; single-use aliquots were thawed at 37 °C for < 90 s, kept on ice, and used within 30 min.
Preparation of EV-ANH
The collected EV were blended with ANH at various concentrations (the EV had a concentration of 1 mg/mL, and the ANH concentrations ranged from 0 to 2000 μg/mL). An ultrasonic treatment with a power output of 200 W was applied for 5 s and then paused for 10 s; this process was repeated six times in total. Next, the mixture was placed on ice to cool for 5 min before being subjected to another round of ultrasonic treatment. After the ultrasound treatments, the EV and ANH mixture was incubated at 37 °C for 1 h. Afterwards, the EV extraction procedure was repeated to eliminate any unbound ANH. High-performance liquid chromatography (HPLC, Shimadzu) was utilized to measure the loading efficiency of EV onto ANH. Detection was performed via a C18 column, the mobile phase was a mixture of methanol and water (65:35), and the detection wavelength was 250 nm.
RNA sequencing
Skin tissue samples were lysed with TRIzol and then submitted to Sangon Biotech Co., Ltd. (Shanghai, China) for RNA-seq analysis23. Total RNA quality control was performed on an Agilent 2100 Bioanalyzer prior to library construction with the NEBNext® Ultra™ RNA Library Prep Kit for Illumina®. The prepared libraries were sequenced via the Illumina NovaSeq 6000 platform (USA). Raw reads were processed and aligned to the human reference genome (hg38) with HISAT2, followed by gene count extraction via featureCounts. For data visualization, PCA was implemented via FactoMineR (v2.8) on global mRNA expression profiles. Statistical assessment of differential expression integrated edgeR (v3.42.4) and limma (v3.56.2) in R (v4.3.1), defining DEGs by criteria of a fold change exceeding 1 and an FDR below 0.05. Heatmap visualization was performed with ClusterGVis (v0.1.1). Multiple comprehensive analyses, such as Gene Ontology (GO) analysis, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis, and target gene cluster analysis, were performed. The raw sequence data contained in this paper have been deposited in the Genome Sequence Archive (GSA: CRA028075).
Western blotting
Skin tissue protein samples were extracted with RIPA lysis buffer and then centrifuged at 12,000 rpm for 10 min. After denaturation, equal amounts of the protein samples were separated on 10% SDS protein gels. After separation, the proteins were transferred to PVDF membranes. The membranes were incubated in 5% nonfat skim milk, followed by overnight incubation at 4 °C with primary antibodies. The primary antibodies used were ACSL4 (abs106075, Absin), SLC7A11 (abs174692, Absin), CD81 (abs182484, Absin), Calnexin (abs131462, Absin), and GAPDH (Cat. No. #5174, CST, USA). Thereafter, the membranes were individually incubated with the corresponding secondary antibodies at room temperature and then analyzed via Bio-Rad ChemiDoc Touch Imaging.
Knockdown of ACSL4
Using an online shRNA design tool (http://rnaidesigner.thermofisher.com/rnaiexpress/sort.do), we obtained the interference sequences for human ACSL4, which are detailed in Supplementary Table 4. The siRNAs were produced by Sangon Biotech Co., Ltd. (Shanghai, China). Next, shRNA and an empty vector were transduced into HEK293T cells for the purpose of lentivirus packaging. The resulting lentivirus was incubated with the target cells for 48 h. Subsequently, the cells were collected for subsequent analysis.
Statistics and reproducibility
GraphPad Prism 6.0 software was used to analyze the complete dataset. Statistical evaluations relied on one-way ANOVA or two-way ANOVA followed by Tukey’s multiple comparisons test where appropriate, and the normality test was conducted with the Shapiro–Wilk (SW) test. The results are reported as the means ± SDs. Statistically significant differences are indicated by p values as follows: *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, #P < 0.05, ##P < 0.01, ###P < 0.001, ####p < 0.0001. For every experimental design, each experimental condition had a minimum of three repeated trials.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The RNA-seq data have been deposited in the Genome Sequence Archive (GSA: CRA028075). The uncropped and unedited blot images are shown in Supplementary information. Numerical source data for graphs can be obtained in Supplementary Data. All other data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Kong, Y. et al. Narciclasine inhibits phospholipase A2 and regulates phospholipid metabolism to ameliorate psoriasis-like dermatitis. Front. Immunol. 13, 1094375 (2022).
Garzorz-Stark, N. & Eyerich, K. Psoriasis pathogenesis: keratinocytes are back in the spotlight. J. Invest. Dermatol. 139, 995–996 (2019).
Chen, W. Y. et al. Annoying psoriasis and atopic dermatitis: a narrative review. Int. J. Mol. Sci. 23, 4898 (2022).
Stoffel, E. et al. Analysis of anti-tumour necrosis factor-induced skin lesions reveals strong T helper 1 activation with some distinct immunological characteristics. Br. J. Dermatol. 178, 1151–1162 (2018).
Raiol, T. K. et al. Leprosy associated with psoriasis. Lepr. Rev. 86, 368–373 (2015).
Weng, Z. et al. Shikonin promotes ferroptosis in HaCaT cells through Nrf2 and alleviates imiquimod-induced psoriasis in mice. Chem. Biol. Interact. 27, 110788 (2023).
Puig, L. et al. Generalized pustular psoriasis: a global Delphi consensus on clinical course, diagnosis, treatment goals and disease management. J. Eur. Acad. Dermatol. Venereol. 37, 737–752 (2023).
Parisi, R. et al. National, regional, and worldwide epidemiology of psoriasis: systematic analysis and modelling study. BMJ 369, m1590 (2020).
Lu, H. et al. Effect of traditional chinese medicine on psoriasis vulgaris: a meta-analysis and systematic review. Complement. Med. Res. 30, 63–77 (2023).
Feng, W. et al. Immunoregulatory effects of traditional Chinese medicine and its ingredients on psoriasis. Int. Immunopharmacol. 159, 114896 (2025).
Ding, J. et al. Cancer-related therapeutic potential of epimedium and its extracts. Nutr. Cancer. 76, 885–901 (2024).
Zhang, X. et al. Advancements in the biotransformation and biosynthesis of the primary active flavonoids derived from epimedium. Molecules 28, 7173 (2023).
Wang, H. et al. Anhydroicaritin suppresses tumor progression via the PI3K/AKT signaling pathway in hepatocellular carcinoma. Aging (Albany, NY) 15, 7831–7843 (2023).
Li, F. et al. Anhydroicaritin inhibits EMT in breast cancer by enhancing GPX1 expression: a research based on sequencing technologies and bioinformatics analysis. Front. Cell Dev. Biol. 9, 764481 (2021).
Zheng, Z. G. et al. Anhydroicaritin, a SREBPs inhibitor, inhibits RANKL-induced osteoclastic differentiation and improves diabetic osteoporosis in STZ-induced mice. Eur. J. Pharmacol. 809, 156–162 (2017).
Shi, Y. et al. Identifying the anti-metastasis effect of Anhydroicaritin on breast cancer: Coupling network pharmacology with experimental validation. J. Ethnopharmacol. 293, 115326 (2022).
Hao, M. et al. Schisandrol A-loaded mesenchymal stem cell exosomes modulate FTO/miR-21/WNT5A axis to promote skin healing. Phytomedicine 146, 157088 (2025).
Shao, Y. et al. MSC-EVs alleviate sepsis-induced acute lung injury through inhibiting NET-triggered damage to adherens junctions of endothelial cells. Mol. Immunol. 185, 6–17 (2025).
Li, L. et al. Epigenetic mechanism of iPSC-MSC-EVs in colonic epithelial cell pyroptosis in ulcerative colitis cell models via modulation of ELF3/miR-342-3p/KDM6B axis and histone methylation. Int. Immunopharmacol. 157, 114704 (2025).
Zhao, R. et al. Inhalation of MSC-EVs is a noninvasive strategy for ameliorating acute lung injury. J. Control. Release. 345, 214–230 (2022).
Li, R. et al. MSC-EVs and UCB-EVs promote skin wound healing and spatial transcriptome analysis. Sci. Rep. 15, 4006 (2025).
Zhou, X. et al. Engineered mesenchymal stem cell-derived extracellular vesicles scavenge self-antigens for psoriasis therapy via modulating metabolic and immunological disorders. Adv. Sci. 12, e2410067 (2025).
Gao, Y. et al. CAR T Cells Engineered to secrete ifnkappa induce tumor ferroptosis via an IFNAR/STAT1/ACSL4 axis. Cancer. Immunol. Res. 12, 1691–1702 (2024).
Yang, L. et al. Calcipotriol inhibits the proliferation of psoriasis HaCaT cells by activating the ferroptosis pathway. Acta. Histochem. 127, 152274 (2025).
Doll, S. et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat. Chem. Biol. 13, 91–98 (2017).
Quan, J. et al. ACSL family: the regulatory mechanisms and therapeutic implications in cancer. Eur. J. Pharmacol. 909, 174397 (2021).
Liu, L. & Kang, X. X. ACSL4 is overexpressed in psoriasis and enhances inflammatory responses by activating ferroptosis. Biochem. Biophys. Res. Commun. 623, 1–8 (2022).
Zhou, X. et al. Paris saponin VII attenuates psoriasiform inflammation by regulating STAT3/NFkappaB signaling pathway and Caspase-1-induced pyroptosis. Mol. Med. 31, 200 (2025).
Zhu, Y. et al. Celastrol directly targets LRP1 to inhibit fibroblast-macrophage crosstalk and ameliorates psoriasis progression. Acta. Pharm. Sin B 15, 876–891 (2025).
Wang, Z. et al. Targeting the transcription factor HES1 by L-menthol restores protein phosphatase 6 in keratinocytes in models of psoriasis. Nat. Commun. 13, 7815 (2022).
Liu, X. Q. et al. Circulating inflammatory cytokines and psoriasis risk: A systematic review and meta-analysis. PLoS ONE 18, e0293327 (2023).
Tashiro, T. & Sawada, Y. Psoriasis and systemic inflammatory disorders. Int. J. Mol. Sci. 23, 4457 (2022).
Filho, J. M. T. D. A. et al. Flavonoids as photoprotective agents: a systematic review. J. Med. Plants Res. 10, 848–864 (2016).
Leng, H. et al. Effects of aloe polysaccharide, a polysaccharide extracted from Aloe vera, on TNF‑alpha‑induced HaCaT cell proliferation and the underlying mechanism in psoriasis. Mol. Med. Rep. 18, 3537–3543 (2018).
Li, Y. et al. Benzoylaconitine alleviates progression of psoriasis via suppressing STAT3 phosphorylation in keratinocytes. Molecules 28, 10 (2023).
Guo, D. et al. Liquiritin targeting Th17 cells differentiation and abnormal proliferation of keratinocytes alleviates psoriasis via NF-kappaB and AP-1 pathway. Phytother. Res. 38, 174–186 (2024).
Mota, C. M. D. & Madden, C. J. Neural control of the spleen as an effector of immune responses to inflammation: mechanisms and treatments. Am. J. Physiol. Regul. Integr. Comp. Physiol. 323, R375–R384 (2022).
Sangaraju, R. et al. Galangin ameliorates Imiquimod-Induced psoriasis-like skin inflammation in BALB/c mice via down regulating NF-kappaB and activation of Nrf2 signaling pathways. Int. Immunopharmacol. 96, 107754 (2021).
Chen, Y. H. et al. Lactobacillus pentosus GMNL-77 inhibits skin lesions in imiquimod-induced psoriasis-like mice. J. Food. Drug. Anal. 25, 559–566 (2017).
Sugumaran, D. et al. Advances in psoriasis research: From pathogenesis to therapeutics. Life. Sci. 355, 122991 (2024).
Song, C. et al. Deciphering the mechanism of Fang-Ji-Di-Huang-Decoction in ameliorating psoriasis-like skin inflammation via the inhibition of IL-23/Th17 cell axis. J. Ethnopharmacol. 281, 114571 (2021).
Lin, Y. et al. Keratin 17 in psoriasis: current understanding and future perspectives. Semin. Cell. Dev. Biol. 128, 112–119 (2022).
Shannon, E. et al. Thalidomide suppressed IL-1beta while enhancing TNF-alpha and IL-10, when cells in whole blood were stimulated with lipopolysaccharide. Immunopharmacol. Immunotoxicol. 30, 447–457 (2008).
Wu, R. et al. MicroRNA-210 overexpression promotes psoriasis-like inflammation by inducing Th1 and Th17 cell differentiation. J. Clin. Invest. 128, 2551–2568 (2018).
Tang, R. et al. ErbB4 affects Th1/Th17 cell differentiation and promotes psoriasis progression. Cell. Mol. Biol. (Noisy-le-grand) 69, 1–5 (2023).
Lu, Y. et al. Polyethylene glycol ointment alleviates psoriasis-like inflammation through down-regulating the function of Th17 Cells and MDSCs. Front. Med. (Lausanne) 7, 560579 (2020).
Ci, X. et al. Regulation of inflammatory mediators in lipopolysaccharide-stimulated RAW 264.7 cells by 2”-hydroxy-3”-en-anhydroicaritin involves down-regulation of NF-kappaB and MAPK expression. Int. Immunopharmacol. 10, 995–1002 (2010).
Alshahrani, M. Y. et al. A comprehensive insight into the immunomodulatory role of MSCs-derived exosomes (MSC-Exos) through modulating pattern-recognition receptors (PRRs). Cell. Biochem. Funct. 42, e4029 (2024).
Xu, Y. et al. Multiple strategies enhance the efficacy of MSC-Exos transplantation for spinal cord injury. Exp. Neurol. 383, 115038 (2025).
Zhao, J. et al. Erratum: Human gingiva tissue-derived MSC ameliorates immune-mediated bone marrow failure of aplastic anemia via suppression of Th1 and Th17 cells and enhancement of CD4+Foxp3+ regulatory T cells differentiation. Am. J. Transl. Res. 12, 1167 (2020).
Tang, D. et al. Thermally engineered MSC-derived extracellular vesicles ameliorate colitis in mice by restoring the imbalanced Th17/Treg cell ratio. Int. Immunopharmacol. 125, 111077 (2023).
Shou, Y. H. et al. Inhibition of keratinocyte ferroptosis suppresses psoriatic inflammation. Cell Death Dis 12, 1009 (2021).
Xie, L. et al. Protective effects of Wenqingyin on sepsis-induced acute lung injury through regulation of the receptor for advanced glycation end products pathway. Phytomedicine 129, 155654 (2024).
Gao, Y. et al. GPR97 depletion aggravates imiquimod-induced psoriasis pathogenesis via amplifying IL-23/IL-17 axis signal pathway. Biomed. Pharmacother. 179, 117431 (2024).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 82503727), the Key Science and Technology Program of Henan Province (No. 242102310344, 222102310349), the Medical Science and Technology Research Project of Henan Province (No. LHGJ20240243, LHGJ20230786), the Natural Science Foundation of Henan Province (No. 252300420523), the Incubation Project of Advanced Medical Research Center (No. XJYXZX2021007), and the Doctoral Research Initiation Fund Project (No. KYQDJJ2025004). Figure 4C and Graphical abstract were created with Biorender.
Author information
Authors and Affiliations
Contributions
Yaoxin Gao and Ranran Jin: Writing-original draft, Validation, Methodology, Data curation. Yaoxin Gao: Software, Data curation. Xiaoyue Qi, Yiming Shao, and Qiurui He: Software, Methodology, Data curation. Xiaoyue Qi, Baodong Ma, and Ranran Jin: Formal analysis. Yaoxin Gao, Baodong Ma, and Luoming Zhang: Visualization, Funding acquisition. Qinjun Chu and Zhengyuan Xia: Writing-review & editing, Supervision, Project administration.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Jin Zhang and yumei Li for their contribution to the peer review of this work. Primary Handling Editors: Dr Chunfu Zheng and Dr Ophelia Bu. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gao, Y., Ma, B., Jin, R. et al. Anhydroicaritin-loaded mesenchymal stem cell exosomes ameliorate psoriasis via ACSL4-mediated ferroptosis in mice. Commun Biol 9, 306 (2026). https://doi.org/10.1038/s42003-026-09575-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42003-026-09575-1