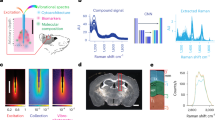
Abstract
Early detection of retinal molecular biomarkers is crucial for addressing the unmet clinical need to prevent irreversible neural tissue damage in ophthalmic and neurodegenerative diseases. Among emerging molecular sensing techniques, non-resonant Raman spectroscopy stands out as a naturally label-free and noninvasive method, offering rich biochemical information. However, in vivo detection of non-resonant Raman spectra from retinal tissue has proven to be challenging so far. Previous studies have reported conflicting results, likely due to overwhelming pigment autofluorescence. In this study, we identified the optic nerve head as the optimal retinal location for acquiring non-resonant Raman spectra in the molecular fingerprint region. Through longitudinal intra-subject measurements, we revealed dynamic changes in the molecular composition. Furthermore, a comparative study across age groups enabled the identification of molecular alterations associated with aging. These findings establish a critical foundation for utilizing non-resonant Raman spectroscopy as an early diagnostic tool for the detection of molecular biomarkers associated with ophthalmic and neurodegenerative diseases.
Similar content being viewed by others
Data availability
The source data underlying graphs can be obtained from Supplementary Data 1. All other data are available from the corresponding author (or other sources, as applicable) on reasonable request.
Code availability
The raw data and analysis code to generate the results in this study is available at https://doi.org/10.6084/m9.figshare.3123027475.
References
Capozzi, M. E., Gordon, A. Y., Penn, J. S. & Jayagopal, A. Molecular imaging of retinal disease. J. Ocul. Pharmacol. Ther. 29, 275–286 (2013).
Nguyen, V. P., Zhe, J., Hu, J., Ahmed, U. & Paulus, Y. M. Molecular and cellular imaging of the eye. Biomed. Opt. Expr. 15, 360 (2024).
Wang, Y. et al. Global incidence, progression, and risk factors of age-related macular degeneration and projection of disease statistics in 30 years: A modeling study. Gerontology 68, 721–735 (2022).
Kiruthika, M. & Malathi, G. A comprehensive review on early detection of drusen patterns in age-related macular degeneration using deep learning models. Photodiagn. Photodyn. Ther. 51, 104454 (2025).
London, A., Benhar, I. & Schwartz, M. The retina as a window to the brain—from eye research to CNS disorders. Nat. Rev. Neurol. 9, 44–53 (2013).
Helboe, K. S., Eddelien, H. S. & Kruuse, C. Visual symptoms in acute stroke – A systematic review of observational studies. Clin. Neurol. Neurosurg. 229, 107749 (2023).
Pleșa, A. et al. Eyes as windows: Unveiling neuroinflammation in multiple sclerosis via optic neuritis and uhthoff’s phenomenon. Diagnostics 14, 2198 (2024).
Archibald, N. K., Clarke, M. P., Mosimann, U. P. & Burn, D. J. The retina in Parkinson’s disease. Brain 132, 1128–1145 (2009).
Wagner, S. K. et al. Retinal optical coherence tomography features associated with incident and prevalent Parkinson disease. Neurology 101, e1581–e1593 (2023).
Zhang, J., Shi, L. & Shen, Y. The retina: A window in which to view the pathogenesis of Alzheimer’s disease. Ageing Res. Rev. 77, 101590 (2022).
Costanzo, E. et al. Ocular biomarkers for Alzheimer disease dementia: An umbrella review of systematic reviews and meta-analyses. JAMA Ophthalmol. 141, 84 (2023).
Bouma, B. E. et al. Optical coherence tomography. Nat. Rev. Methods Prim. 2, 79 (2022).
Everett, M., Magazzeni, S., Schmoll, T. & Kempe, M. Optical coherence tomography: From technology to applications in ophthalmology. Transl. Biophotonics 3, e202000012 (2021).
Patel, N. B., Lim, M., Gajjar, A., Evans, K. B. & Harwerth, R. S. Age-associated changes in the retinal nerve fiber layer and optic nerve head. Invest. Ophthalmol. Vis. Sci. 55, 5134 (2014).
Vujosevic, S. et al. Optical coherence tomography as retinal imaging biomarker of neuroinflammation/neurodegeneration in systemic disorders in adults and children. Eye 37, 203–219 (2023).
Doustar, J., Torbati, T., Black, K. L., Koronyo, Y. & Koronyo-Hamaoui, M. Optical coherence tomography in Alzheimer’s disease and other neurodegenerative diseases. Front. Neurol. 8, 701 (2017).
Leitgeb, R. A. Multimodal optical medical imaging concepts based on optical coherence tomography. Front. Phys. 6, 17 (2018).
Krafft, C. et al. Label-free molecular imaging of biological cells and tissues by linear and nonlinear Raman spectroscopic approaches. Angew. Chem. Int. Ed. 56, 4392–4430 (2017).
Talari, A. C. S., Movasaghi, Z., Rehman, S. & Rehman, I. ur. Raman spectroscopy of biological tissues. Appl. Spectrosc. Rev. 50, 46–111 (2015).
Guo, S., Popp, J. & Bocklitz, T. Chemometric analysis in Raman spectroscopy from experimental design to machine learning–based modeling. Nat. Protoc. 16, 5426–5459 (2021).
Shipp, D. W., Sinjab, F. & Notingher, I. Raman spectroscopy: Techniques and applications in the life sciences. Adv. Opt. Photonics 9, 315 (2017).
Khristoforova, Y., Bratchenko, L. & Bratchenko, I. Raman-based techniques in medical applications for diagnostic tasks: A review. IJMS 24, 15605 (2023).
Chen, C. et al. Applications of Raman spectroscopy in the diagnosis and monitoring of neurodegenerative diseases. Front. Neurosci. 18, 1301107 (2024).
Erckens, R. J. et al. Raman spectroscopy in ophthalmology: From experimental tool to applications in vivo. Lasers Med Sci. 16, 236–252 (2001).
Salbreiter, M. et al. Lighting the Path: Raman spectroscopy’s journey through the microbial maze. Molecules 29, 5956 (2024).
Synytsya, A., Judexova, M., Hoskovec, D., Miskovicova, M. & Petruzelka, L. Raman spectroscopy at different excitation wavelengths (1064, 785 and 532 nm) as a tool for diagnosis of colon cancer. J. Raman Spectrosc. 45, 903–911 (2014).
Bernstein, P. S. et al. Resonance Raman measurement of macular carotenoids in normal subjects and in age-related macular degeneration patients. Ophthalmology 109, 1780–1787 (2002).
Li, B. et al. Imaging lutein and zeaxanthin in the human retina with confocal resonance Raman microscopy. Proc. Natl. Acad. Sci. USA 117, 12352–12358 (2020).
Cakir, B. et al. In vivo noninvasive mitochondrial redox assessment of the optic nerve head to predict disease. PNAS Nexus 2, pgad148 (2023).
Beattie, J. R., Brockbank, S., McGarvey, J. J. & Curry, W. J. Effect of excitation wavelength on the Raman spectroscopy of the porcine photoreceptor layer from the area centralis. Mol. Vis. 11, 825–832 (2005).
Beattie, J. R., Brockbank, S., McGarvey, J. J. & Curry, W. J. Raman microscopy of porcine inner retinal layers from the area centralis. Mol. Vis. 13, 1106–1113 (2007).
Evans, J. W. et al. Optical coherence tomography and Raman spectroscopy of the ex-vivo retina. J. Biophotonics 2, 398–406 (2009).
Patil, C. A. et al. Integrated system for combined Raman spectroscopy–spectral domain optical coherence tomography. J. Biomed. Opt. 16, 011007 (2011).
Wang, Q. et al. Exploring Raman spectroscopy for the evaluation of glaucomatous retinal changes. J. Biomed. Opt. 16, 107006 (2011).
Wang, Q. et al. Detection and characterization of glaucoma-like canine retinal tissues using Raman spectroscopy. J. Biomed. Opt. 18, 067008 (2013).
Banbury, C. et al. Development of the Self Optimising Kohonen Index Network (SKiNET) for Raman Spectroscopy Based Detection of Anatomical Eye Tissue. Sci. Rep. 9, 10812 (2019).
Stiebing, C. et al. Biochemical characterization of mouse retina of an Alzheimer’s disease model by Raman spectroscopy. ACS Chem. Neurosci. 11, 3301–3308 (2020).
Banbury, C. et al. Spectroscopic detection of traumatic brain injury severity and biochemistry from the retina. Biomed. Opt. Express 11, 6249 (2020).
Banbury, C. et al. Window into the mind: Advanced handheld spectroscopic eye-safe technology for point-of-care neurodiagnostic. Sci. Adv. 9, eadg5431 (2023).
Glenn, J. V. et al. Confocal Raman microscopy can quantify advanced glycation end product (AGE) modifications in Bruch’s membrane leading to accurate, nondestructive prediction of ocular aging. FASEB J. 21, 3542–3552 (2007).
Stiebing, C. et al. Nonresonant Raman spectroscopy of isolated human retina samples complying with laser safety regulations for in vivo measurements. Neurophoton 6, 1 (2019).
Keilhauer, C. N. & Delori, F. C. Near-infrared autofluorescence imaging of the fundus: Visualization of ocular melanin. Invest. Ophthalmol. Vis. Sci. 47, 3556 (2006).
Zhang, P. et al. Feasibility study of Raman spectroscopy for investigating the mouse retina in vivo. In Ophthalmic Technologies XXVIII (eds Manns, F., Söderberg, P. G. & Ho, A.) 54 (SPIE, San Francisco, United States, https://doi.org/10.1117/12.2288790. 2018).
Alba-Arbalat, S. et al. In vivo molecular changes in the retina of patients with multiple sclerosis. Invest. Ophthalmol. Vis. Sci. 62, 11 (2021).
Sentosa, R. et al. Development of a label-free, functional, molecular and structural imaging system combining optical coherence tomography and Raman spectroscopy for in vivo measurement of rat retina. Biomed. Opt. Expr. 16, 566 (2025).
Wang, Y. X., Panda-Jonas, S. & Jonas, J. B. Optic nerve head anatomy in myopia and glaucoma, including parapapillary zones alpha, beta, gamma and delta: Histology and clinical features. Prog. Retinal Eye Res. 83, 100933 (2021).
Lieber, C. A. & Mahadevan-Jansen, A. Automated method for subtraction of fluorescence from biological Raman spectra. Appl Spectrosc. 57, 1363–1367 (2003).
Afseth, N. K. & Kohler, A. Extended multiplicative signal correction in vibrational spectroscopy, a tutorial. Chemometr. Intell. Lab. Syst. 117, 92–99 (2012).
Krafft, C. Raman spectroscopy of proteins and nucleic acids: From amino acids and nucleotides to large assemblies. In Encyclopedia of Analytical Chemistry (ed. Meyers, R. A.) 1–15 (Wiley, 2018).
Krafft, C., Neudert, L., Simat, T. & Salzer, R. Near infrared Raman spectra of human brain lipids. Spectrochimica Acta Part A: Mol. Biomol. Spectrosc. 61, 1529–1535 (2005).
Czamara, K. et al. Raman spectroscopy of lipids: a review. J. Raman Spectrosc. 46, 4–20 (2015).
Wiercigroch, E. et al. Raman and infrared spectroscopy of carbohydrates: A review. Spectrochimica Acta Part A: Mol. Biomol. Spectrosc. 185, 317–335 (2017).
Pezzotti, G. Raman spectroscopy in cell biology and microbiology. J. Raman Spectrosc. 52, 2348–2443 (2021).
Hernández, B., Pflüger, F., Adenier, A., Kruglik, S. G. & Ghomi, M. Vibrational analysis of amino acids and short peptides in hydrated media. VIII. Amino acids with aromatic side chains: l -Phenylalanine, l -Tyrosine, and l -Tryptophan. J. Phys. Chem. B 114, 15319–15330 (2010).
Rygula, A. et al. Raman spectroscopy of proteins: a review: Raman spectroscopy of proteins. J. Raman Spectrosc. 44, 1061–1076 (2013).
Vaughan, D. W. & Hansen, L. A Learning System in Histology: CD-ROM and Guide. (Oxford University Press, Oxford, 2002).
J. Salazar, J. et al. Anatomy of the Human Optic Nerve: Structure and Function. in Optic Nerve (ed. Ferreri, F. M.) (IntechOpen, 2019).
Muench, N. A. et al. The influence of mitochondrial dynamics and function on retinal ganglion cell susceptibility in optic nerve disease. Cells 10, 1593 (2021).
Liu, H., Liu, H. & Prokosch, V. The relationship between mitochondria and neurodegeration in the Eye: A review. Appl. Sci. 11, 7385 (2021).
Dias, M. S., Luo, X., Ribas, V. T., Petrs-Silva, H. & Koch, J. C. The role of axonal transport in glaucoma. IJMS 23, 3935 (2022).
Guo, J. et al. Aging and aging-related diseases: From molecular mechanisms to interventions and treatments. Sig Transduct. Target Ther. 7, 391 (2022).
Li, Y. et al. Molecular mechanisms of aging and anti-aging strategies. Cell Commun. Signal 22, 285 (2024).
Coleman-Belin, J. et al. Aging effects on optic nerve neurodegeneration. Int J. Mol. Sci. 24, 2573 (2023).
Kim, J. H., Yang, D. & Park, S. Raman spectroscopy in cellular and tissue aging research. Aging Cell e14494 https://doi.org/10.1111/acel.14494. (2025)
Harayama, T. & Riezman, H. Understanding the diversity of membrane lipid composition. Nat. Rev. Mol. Cell Biol. 19, 281–296 (2018).
Skowronska-Krawczyk, D. & Budin, I. Aging membranes: Unexplored functions for lipids in the lifespan of the central nervous system. Exp. Gerontol. 131, 110817 (2020).
Wagner, H., Fink, B. A. & Zadnik, K. Sex- and gender-based differences in healthy and diseased eyes. Optom. - J. Am. Optometric Assoc. 79, 636–652 (2008).
Kobayashi, T. et al. Influence of age and gender on the pulse waveform in optic nerve head circulation in healthy men and women. Sci. Rep. 9, 17895 (2019).
Niehorster, D. C. et al. The best fixation target revisited: New insights from retinal eye tracking. Behav. Res 58, 2 (2025).
Bergholt, M. S., Serio, A. & Albro, M. B. Raman Spectroscopy: Guiding Light for the Extracellular Matrix. Front. Bioeng. Biotechnol. 7, 303 (2019).
Tominaga, Y., Fujiwara, A. & Amo, Y. Dynamical structure of water by Raman spectroscopy. Fluid Phase Equilibria 144, 323–330 (1998).
Lang, X., Shi, L., Zhao, Z. & Min, W. Probing the structure of water in individual living cells. Nat. Commun. 15, 5271 (2024).
Miyamori, D. et al. A Raman algorithm to estimate human age from protein structural variations in autopsy skin samples: A protein biological clock. Sci. Rep. 11, 5949 (2021).
Kostamovaara, J. et al. Fluorescence suppression in Raman spectroscopy using a time-gated CMOS SPAD. Opt. Expr. 21, 31632 (2013).
Sentosa, R. & Leitgeb, R. Analysis code for: Label-free in vivo molecular profiling of the human retina by non-resonant Raman spectroscopy. 10071887 Bytes figshare https://doi.org/10.6084/M9.FIGSHARE.31230274 (2026).
Acknowledgements
The authors acknowledge: A. Amelink and P. Bussink from TNO, P. de Bettignies from HORIBA France SAS for technical support; C. Stiebing from Leibniz-IPHT and A. Krause from the Medical University of Vienna for discussions on Raman signal processing; H. Sattmann, A. Hodul and B. Rosenauer from the Medical University of Vienna for electrical and mechanical support; S. Rentz-Chorherr, M. Martin, T. Schneider and K. Memarpour from the Medical University of Vienna for support related to the medical device approval; M.C. Nguyen for the illustration in Fig. 1A. This work was carried out in the framework of project MOON. The project has received funding from: European Union’s Horizon 2020 research and innovation program under grant agreement No 732969 (It is an initiative of the Photonics Public Private Partnership. www.photonics21.org), CD-Labor OPTRAMED (CD10260501), Carl Zeiss Labor (UE60506007) and Medical Scientific Fund of the Mayor of the City of Vienna.
Author information
Authors and Affiliations
Contributions
R.A.L., W.D., M.Kem., M.Sch. and J.P. conceptualized the project. R.S. designed, built and integrated the OCT/RS module. M.Sal. developed the OCT acquisition software. M.E. and W.Dj. conceptualized and assisted the integration between the IR fundus imaging module and the OCT/RS module. V.S. conceptualized and assisted the integration of the RS module. R.S., M.Ken. and R.A.L. carried out the investigation and experimental work. R.S. performed the data analysis, visualization and interpretation. R.A.L., M.A. and A.U. assisted the data analysis. H.S. and A.P. recruited the subjects and provided clinical support. R.A.L., T.S. and W.D. supervised the study. R.S. wrote the original manuscript. All authors reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
M.E. and M.Kem. are employed by Carl Zeiss Meditec AG; V.S. was employed by HORIBA France SAS; T.S. is employed by Carl Zeiss Meditec, Inc; All other authors declare that they have no competing interests.
Peer review
Peer review information
Communications Biology thanks Philippe Leproux, Yasuaki Kumamoto and Adriana Adamczyk for their contribution to the peer review of this work. Primary Handling Editor: Ophelia Bu. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sentosa, R., Kendrisic, M., Salas, M. et al. Label-free in vivo molecular profiling of the human retina by non-resonant Raman spectroscopy. Commun Biol (2026). https://doi.org/10.1038/s42003-026-09744-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-026-09744-2