Abstract
Nitrene transfer chemistry is an effective strategy for introducing C–N bonds, which are ubiquitous in pharmaceuticals, agrochemicals and diverse bioactive natural products. The development of chemical methodology that can functionalize unique sites within natural products through nitrene transfer remains a challenge in the field. Herein, we developed copper catalyzed chemoselective allylic C–H amination and catalyst-free visible-light induced aziridination of alkenes through nitrene transfer. In general, both reactions tolerate a wide range of functional groups and occur with predictable regioselectivity. Furthermore, combination of these two methods enable the intermolecular chemo-selective late-stage amination of biologically active natural products, leading to C–H amination or C=C aziridination products in a tunable way. A series of control experiments indicate two-step radical processes were involved in both reaction systems.
Similar content being viewed by others
Introduction
Natural products play an important role in chemistry and drug discovery1,2,3. In the past few decades, chemists have made enormous efforts to create structurally complex, diverse molecules resembling natural products4,5,6,7,8. Since traditional synthesis of natural product derivatives from simple starting materials can be laborious and ineffective, an alternative approach is to derivatize natural products directly via selective reactions. With this approach, synthetic routes may be shortened and more effective means can be provided to produce diverse natural products derivatives9,10,11,12,13,14,15. Since nitrogen-containing molecules are the key structural constituent of many pharmaceutical compounds that play a pivotal role in drug development, the development of methodology for the late-stage introduction of nitrogen-containing functionalities would be valuable to medicinal chemistry16,17,18,19,20.
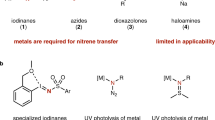
Transformations of C–H and C=C bonds to C–N bonds through transition-metal catalyzed nitrene transfer are powerful synthetic tools to prepare valuable amine building blocks21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41. Control over the chemoselectivity of nitrene insertion into either C–H or C=C bond was primarily limited to intramolecular transformation (Fig. 1a)42,43,44,45,46,47,48. More recently, it has been shown that the chemoselectivity of intermolecular nitrene transfer can be controlled by changing the metal identity or altering the ligands (Fig. 1b)49,50,51. Although several methods have been reported, the chemoselective amination of unsaturated natural products by these reactions has been limited to examples containing a minimum of functional groups. A rare example of selective intermolecular nitrene insertion for functionalizing unique sites within unsaturated natural products employs the rhodium (II)-based system developed by Du Bois52. With the rhodium (II) catalyst, chemoselectivity for C–H amination and alkene aziridination is highly substrate-dependent. For example, geraniol and parthenin gave site-selective aziridination products, while allylic C–H amination occurred selectively with eupalmerolide and Eupalmerin acetate. So, developing efficient, inexpensive reaction systems for predictable, tunable and intermolecular chemoselective nitrene transfer for late-stage amination of unsaturated natural products remains to be addressed.
a Intramolecular nitrene transfer reaction of unsaturated hydrocarbon compounds. b Intermolecular nitrene transfer reaction of unsaturated hydrocarbon compounds. c Combining copper and photo catalysis for nitrene transfer reaction of unsaturated hydrocarbon compounds.
Schomaker and co-workers have demonstrated that silver(I) catalysis offers unique opportunities to tune the chemo- and site-selectivity of intramolecular or intermolecular nitrene transfer53,54. Copper, which is in the same group as silver, has been frequently employed for aziridination of olefins and presented promising catalytic activity for C–H amination of sp2 C–H bonds, benzylic C–H bonds and inactivated sp3 C–H bonds in saturated hydrocarbons through nitrene transfer28,55,56,57,58. However, copper-catalyzed selective amination of allylic C–H bonds within unsaturated hydrocarbons through nitrene insertion has been underexplored, probably owing to a preferred reactivity for aziridination with copper catalysis28,57. Considering the low cost of copper and its compatibility with biology, developing tunable copper-based catalytic system for selective late-stage C–H amination of unsaturated natural products would be helpful for performing arming or Structure-Activity Relationship (SAR) studies of unsaturated natural products. On the other hand, the selective aziridination of natural products is also of great importance because aziridines are found in diverse biologically active compounds59. Also, the ring constraint of aziridines render them valuable intermediates as building blocks for a plethora of chemicals by means of stereo- and regiospecific transformations, including ring opening, ring expansion, and rearrangement, and thus lead to more complex nitrogen-containing molecules60,61. The transition-metal catalyzed aziridination of alkene with nitrene precursors is the most popular and commonly used approach to construct an aziridine subunit26,28,34. Recently, metal-free and catalyst-free aziridination of alkenes have emerged as greener alternatives, but only limited approaches are available62,63,64,65. Among them, photo-induced aziridination of alkenes with iminoiodinanes seems to be a promising tool, but high energy ultraviolet (UV)-irradiation was used in this protocol64. An ideal approach is to develop visible-light induced selective aziridination of alkenes in the absence of any catalysts. Herein, we developed copper catalyzed chemoselective allylic C–H amination and catalyst-free visible light-induced aziridination of alkenes through nitrene transfer (Fig. 1c). Furthermore, combination of these two methods enable the intermolecular chemoselective late-stage amination of biologically active natural products, leading to C–H amination or C=C aziridination products in a tunable way.
Results and discussion
Reaction optimization
Inspired by the rapidly developed photochemistry, we initially investigated the feasibility of utilizing visible light as an energy source for selective aziridination of alkenes. Iminoiodinanes stabilized by ortho-substituents were found to be activated under ultraviolet (UV) light irradiation and were used as nitrene precursor for amination reactions64. Nemoto and coworkers demonstrated that the hypervalent iodine could also be activated with light from the apparently non-absorbing region via a direct S0-Tn transition, which has been considered a forbidden process66. Notably, in some cases the generation of an excited triplet state via a direct S0-Tn transition is more efficient than that via S0-S1-T1 intersystem crossing. Inspired by this, we envisioned that such ortho-substituted iminoiodinanes could be similarly activated under light of a longer wavelength, such as visible light, and acted as a nitrene precursor for the aziridination of alkenes. As shown in Table 1, we studied the reaction of cyclohexene 2 with ortho-methoxymethyl iminoiodinanes 1 under blue LED irradiation and indeed obtain the aziridination product in good yield (entry 1). Control reaction confirms that no reaction occurred in the absence of visible light (entry 2). When the reaction was carried out in air, the desired product 3 was obtained in 72% yield, indicating that air is tolerated in this reaction (entry 3). In the absence of ortho substituent, iminoiodinane PhI = NTs also reacted to give the corresponding aziridination product, albeit in a lower yield (entry 4). Utilizing (1,2-dichloroethane) DCE instead of CH3CN as the solvent led to a lower yield (entry 5). Other polar solvents, such as (tetrahydrofuran) THF and (N, N-dimethylformamide) DMF, were notably less effective, mostly giving a mixture of side products (entries 6–7). Besides blue LED, we also tried to use white LED as the light source and a comparable yield was observed (entry 3 in Supplementary Table 1). We also investigated the impact of substrate ratio and found that reducing the amount of alkene to one or two equivalent both led to a slight decrease in product yield (entries 9 and 10 in Supplementary Table 1). No improvement in yield was observed when the reaction time was prolonged to 12 h. (entry 11 in Supplementary Table 1). These results are described in the Supplementary Results and Discussion (Supplementary Table 1).
For the selective allylic C–H amination, copper was selected as the central metal for catalyst screening in view of its low cost and biological compatibility. As previously shown, catalyst control of the nitrene transfer could be achieved by altering the nature of ligands53,54. So, we initially investigated the effects of different ligands (Table 2, Supplementary Table 2). In general, phenanthroline and bipyridine ligands favor aziridination (entries 1–5) and oxazolines favor allylic C–H amination (entries 6–12), except for L6 and L10. β-Diketiminate ligand (L13 and L14) and PyBox ligand (L15) prefer the aziridination of C=C bond to deliver aziridines as the major products (entries 13–15). Altering the solvent to DCE enhanced the site selectivity in favor of the allylic C–H amination (entry 16). By contrast, (dimethyl sulfoxide) DMSO, (dimethylacetamide) DMA, MeOH and (N-methyl–2-pyrrolidone) NMP were all inefficient solvents for both nitrene transfer reactions (entries 17–20). Finally, a bidentate oxazoline ligand L9 offers advantages in terms of yield and chemoselectivity using DCE as the solvent (entry 22). Notably, the addition of (4 Å Molecular Sieves) 4ÅMS increased the reaction yield substantially and did not affect the chemoselectivity (entry 23). As shown in the Supplementary Results and Discussion, we also conducted parallel experiments of copper-catalyzed C–H amidation with different metal:ligand ratios, from 1:1 to 1:2.5. Compared with the chemoselectivity in 1:1.5 metal/ligand ratio, the yield and chemoselectivity for C–H amination is poorer when the catalyst system was used in 1:1 metal/ligand ratio (entry 10 in Supplementary Table 3). Further increasing the metal/ligand ratio from 1:1.5 to 1:2 or 1:2.5 improved the overall yield to some extent, but failed to improve the chemoselectivity for C–H amination (entries 11 and 12 in Supplementary Table 3). Slightly lower yields were obtained when the amount of alkene was reduced to one or two equivalents, while the chemoselectivity was unaffected (entries 13 and 14 in Supplementary Table 3). According to the reactivity of iminoiodinanes reported by Takemoto and coworkers, we prepared another two iminoiodinanes and compared their activities in copper-catalyzed C-H amination (Supplementary Scheme 1). In general, the corresponding C–H amination product was successfully obtained in moderate yields using ortho-methoxymethyl or ortho-nitro iminoiodinanes of p-toluenesulfonamide as nitrogen sources, respectively. Both yield and chemoselectivity for C–H amination with ortho-methoxymethyl iminoiodinane was slightly higher than that with ortho-nitro iminoiodinane. ortho-Nitro iminoiodinane of ortho-nitrosulfonamide gave only trace amounts of C–H amination and aziridination products, probably due to its poor solubility in DCE.
Substrate scope
With the optimal reaction conditions in hand, we set out to explore the substrate scope of the selective aziridination/allylic C–H amination of various alkenes. Under blue LED irradiation, aliphatic aziridines 3–16 were all smoothly generated in moderate to good yields from the corresponding cyclic, 1,1-disubstituted, terminal and internal olefins (Fig. 2a). Electron-poor alkene and diene are also compatible with the reaction conditions, giving 17 and 18 in 23 and 37% yield, respectively. Styrenes having different substituents on the aromatic rings were all tolerated to afford the corresponding aziridines 19–23 in acceptable yields. The disubstituted aromatic olefin provided anti-isomer as the main product (24). Importantly, alkyl halides and aryl halides that can undergo further coupling reactions were all well-tolerated (13 and 21). Under visible-light irradiation, pinene also underwent aziridination smoothly to give the corresponding products in 66% yield, with 25a as the major isomer. α-Terpinylacetate gives exclusively selective aziridination product with a anti/syn ratio of 3.8:1 (26a and 26b). Only aziridination of limonene is observed with photo induction, with the endocyclic alkene preferred slightly over the exocyclic alkene by a ratio of 1:0.8 (27a and 27b).
a The reaction scope of blue light-induced aziridination of alkenes. Reaction conditions: 1 (0.05 mmol), 5 (0.25 mmol), in CH3CN (0.5 mL) under 12 W blue LEDs (435–445 nm) at room temperature for 7 h. Isolated yield. b The reaction scope of copper-catalyzed allylic C–H amination. Reaction conditions: 1 (0.1 mmol), 5 (0.5 mmol), [Cu(OTf)]2·toluene (10 mol%), L9 (15 mol%), 4ÅMS (100 mg), DCE (0.9 mL), r.t., under N2 for 16 h. Isolated yield. The ratio of isomers determined by 1H NMR is given in the parenthesis. The ratio of C–H amination/C = C aziridination product (I:II) determined by 1H NMR is given in the parenthesis.
Then olefins containing both C=C and C–H bonds were selected to examine the robustness of copper-catalyzed allylic C–H amination (Fig. 2b). Five- and six-membered rings selectively gave C–H amination products (4 and 28). However, reaction of cycloheptene showed less selectivity for C–H amination (1.5:1 I:II) (29), probably due to poor stabilization of an allylic radical, which were induced by competing transannular interactions. This provides indirect evidence that copper-catalyzed C–H amination may operate via a stepwise nitrene transfer pathway. The reaction of (E)-1,3-diarylpropenes and pent-2-ene gave the corresponding C–H amination products mainly and no isomerization was observed (30–32). (E)-hex-2-ene gave good selectivity for C–H amination and showed rearranged product, which also indicates a potential stepwise pathway (33a and 33b). Good chemoselectivity for C–H amination was also noted for 1-methylcyclohexene and 1-phenylcyclohexene (34–35). Amination at the less hindered allylic C–H bonds is favored by a ratio of 2.8:1 and 3.2:1, respectively. Under copper catalysis, C–H amination of α-terpinylacetate occurred to deliver 36c and 36d in a 53% overall yield, where activation of less hindered C–H bond is preferred by a ratio of 4.5:1. The presence of terminal alkene in limonene results in poor selectivity for copper-catalyzed C–H amination, favoring less hindered C–H bond by a ratio of 4.0:1 (37c and 37d). Pinene showed excellent chemoselectivity for C–H amination (38b and 38c), providing the corresponding products in modest yields with syn-isomer (38b) as the major product. The absolute configuration of 38b was determined by X-ray crystallography (see Supplementary Fig. 105 and Supplementary Data 1). In contrast, copper-catalyzed aziridination of terminal alkenes (39 and 40) and 1,2-dihydronaphthalene (41) is favored over C–H amination.
Late-stage functionalization of biologically active natural products
The developed C–H amination/C=C aziridination methods were applied to a diverse set of natural products and their derivatives, containing multiple alkene and allylic C–H functionalities. In most cases, chemo-site selectivity can be tuned by altering the reaction systems (photo-induction or copper catalysis) (Fig. 3). Aziridination of β-ionone 42 occurred exclusively on the cyclic C=C bond a, perhaps due to the electron poorness of C=C bond b, giving 42a in 42% yield. Copper-catalyzed C–H amination of β-ionone 42 showed excellent chemo- and regio-selectivity, providing 42c as the sole product in 51% yield. As expected, aziridination of α-bisabolol acetate 43 gave a 0.82:1 mixture of aziridine products (43a to 43b) in 59% yield, with exo product preferred slightly over endo product. Switching to copper catalysis resulted in site-selective C–H amination (43c and 43d), but the aziridination process were also favored because of the presence of two electron-rich alkenes in 43. However, site-selectivity for amination of the less hindered C–H bond is excellent with a ratio of >20:1. Both cholesterol acetate 44 and pregnenolone acetate 45 underwent facially selective aziridination with photo irradiation, leading to 44a and 45a as single diastereomers in acceptable yields, respectively. Pregnenolone acetate 45 presented higher aziridination efficiency (74% yield) than cholesterol acetate 44 (49% yield), probably due to the better solubility of pregnenolone acetate 45 in CH3CN. Switching to copper catalysis, both cholesterol acetate and pregnenolone acetate gave C–H aminated diene products (44c and 45c) in good chemo- and site-selectivity. The diene structure may result from elimination of acetate group in the presence of copper catalyst, which has been reported previously67. In the cases of sclareol and andrographolide acetate (46 and 47), aziridinations were observed exclusively at the terminal alkenes under both photo induction and copper catalysis (46a and 47a), which is consistent with the reactivity of terminal alkenes in Fig. 2b. The absolute configuration of 47a was characterized by X-ray diffraction (see Supplementary Fig. 106 and Supplementary Data 1). We also compared the aziridination efficiency between photo induction and metal catalysis. Surprisingly, photo induced aziridination of andrographolide acetate gave the best yield and selectivity. With copper catalysis, the aziridine product was obtained in 34% yield, along with some side products derived from copper-catalyzed ring-opening reaction of aziridine. In contrast, Rh2(esp)2 gave only trace amount of the desired aziridination product with most of the natural product intact.
a Late-stage aziridination of biologically active natural products. Reaction conditions: 1 (0.05 mmol), natural products (0.1 mmol), in CH3CN (0.5 mL) under 12 W blue LEDs (435–445 nm) at room temperature for 7 h. Isolated yield. b Late-stage allylic C–H amidation of biologically active products. Reaction conditions: 1 (0.1 mmol), natural products (0.2 mmol), [Cu(OTf)]2·toluene (10 mol%), L9 (15 mol%), 4ÅMS (100 mg), DCE (0.9 mL), r.t., under N2 for 16 h. Isolated yield. The ratio of isomers is given in the parenthesis. The ratio of C–H amidation/aziridination product (I:II) is given in the parenthesis.
Mechanistic study
To get a better understanding of the reaction mechanism, control experiments were carried out. As shown in Fig. 4a, subjecting 1 and 2 to aziridination conditions in the presence of (2,2,6,6-tetramethylpiperidinooxy) TEMPO as a radical inhibitor showed a substantial decrease in yield, which suggests the presence of radical intermediates. Additionally, compound 48, which may derive from coupling of nitrene precursor with TEMPO, was obtained in 16% yield. Then we used cis- and trans- alkene to get stereochemical insight into the visible light-induced aziridination of alkenes (Fig. 4b). The photo-induced reaction of cis-49 gave the corresponding aziridine 50 in 70% overall yield with syn/anti ratio of 15:85, whereas trans-49 gave 50 in 77% yield with 7:93 syn/anti ratio. These results strongly suggest a two-step mechanism, which allows isomerization to proceed, rather than a concerted nitrogen atom transfer. Finally, in order to get insight into photo-induced activation of substrates, we investigated the conversion of iminoiodinane and alkene 49 under blue LED irradiation, separately. As shown in Fig. 4c, iminoiodinane was decomposed into 52% yield of sulfonamide 51 and 61% yield of 52 after irradiating for 7 h. In contrast, both the cis- and trans- isomers of 49 remained unchanged. These results suggest iminoiodinanes, rather than alkene substrates were activated under photo irradiation. Based on the studies above, a possible mechanism for visible light-induced aziridination was proposed as in Fig. 4f. The reaction started with photo-promoted conversion of iminoiodinane to an active intermediate 55 in the triplet state. The intermediate was rapidly trapped by alkene to furnish a radical intermediate 56, which underwent intramolecular cyclization to provide the aziridine product 57. For copper-catalyzed C–H amination, the addition of TEMPO led to a 25% decrease in yield (Fig. 4d). In addition, we note that copper-catalyzed C–H amination of 3-methylhexene generates a mixture of 53c, 53a and 53b in a ratio of 1:2:9. A transposed double bond was observed in 53b. All these results supported the presence of radical intermediates in the copper-catalyzed C–H amination. Base on this, a stepwise nitrene transfer pathway involving a copper-nitrene intermediate was proposed as in Fig. 4g.
a Influence of the radical inhibitor TEMPO on the aziridination. b Study the aziridination of cis- or trans- alkenes. c Study the activation of substrates by blue LED irradiation. d Influence of TEMPO on the allylic C–H amination. e Allylic isomerization studies. f, g The proposed mechanisms.
In conclusion, we have developed visible light-induced aziridination and copper-catalyzed selective C–H amination. Both reaction systems demonstrate complementary chemoselectivities in alkenes, largely independent of substrate identities. Furthermore, combination of the above two methods allow for selective functionalization of biologically active natural products containing sites for both aziridination and C–H amination in nitrene transfer. A series of mechanistic studies indicate a two-step radical process was involved in both reaction systems.
Methods
General procedure for photo-induced aziridination of alkenes
In a glovebox, a solution of 1 (0.05 mmol, 21 mg) in CH3CN (0.5 mL) was added into a 2.0 mL vial. Then alkene (0.25 mmol of simple alkenes or 0.1 mmol of complex natural products) was added and the resulting mixture was stirred under 12 W blue LED (435–445 nm) at room temperature for 7 h. The resultant reaction mixture was purified by preparative TLC (hexane/ ethyl acetate 10:1 to 5:1).
General procedure for copper-catalyzed allylic C–H amidation of alkenes
In a glovebox, a solution of [Cu(OTf)]2·toluene (10 mol%) and L9 (15 mol%) in DCE (0.9 mL) was added into a 2.0 mL vial. The mixture was stirred for 30 min. Then 4ÅMS (100 mg), alkene (0.5 mmol of simple alkenes or 0.2 mmol of complex natural products) and 1 (0.1 mmol) were added successively. The resulting mixture was stirred at room temperature for 16 h. The resultant reaction mixture was purified by preparative TLC (hexane/ethyl acetate 5:1).
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information files. For experimental details and compound characterization data see Supplementary Methods and Supplementary Notes 1, 2. For 1H NMR and 13C NMR spectra see Supplementary Figs. 1–104. For the X-ray crystallographic data see Supplementary Fig. 105 for 38b, Supplementary Fig. 106 for 47a and Supplementary Data 1. The X-ray crystallographic coordinates for structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition number CCDC 2125791 for 38b and 2125792 for 47a. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. Extra data are available from the corresponding authors upon request.
References
Paterson, I. & Anderson, E. A. The renaissance of natural products as drug candidates. Science 310, 451–453 (2005).
Danishefsky, S. On the potential of natural products in the discovery of pharma leads: A case for reassessment. Nat. Prod. Rep. 27, 1114–1116 (2010).
Nelson, A. L., Dhimolea, E. & Reichert, J. M. Development trends for human monoclonal antibody therapeutics. Nat. Rev. Drug Discov. 9, 767–774 (2010).
Hong, B., Luo, T. & Lei, X. Late-stage diversification of natural products. ACS Cent. Sci. 6, 622–635 (2020).
Bebbington, M. W. P. Natural product analogues: Towards a blueprint for analogue-focused synthesis. Chem. Soc. Rev. 46, 5059–5109 (2017).
Kim, K. E., Kim, A. N., McCormick, C. J. & Stoltz, B. M. Late-stage diversification: A motivating force in organic synthesis. J. Am. Chem. Soc. 143, 16890–16901 (2021).
Romero, E. et al. Enzymatic late-stage modifications: Better late than never. Angew. Chem., Int. Ed. 60, 16824–16855 (2021).
Karageorgis, G., Foley, D. J., Laraia, L., Brakmann, S. & Waldmann, H. Pseudo natural products-chemical evolution of natural product structure. Angew. Chem., Int. Ed. 60, 15705–15723 (2021).
Pelish, H. E., Westwood, N. J., Feng, Y., Kirchhausen, T. & Shair, M. D. Use of biomimetic diversity-oriented synthesis to discover galanthamine-like molecules with biological properties beyond those of the natural product. J. Am. Chem. Soc. 123, 6740–6741 (2001).
Goess, B. C., Hannoush, R. N., Chan, L. K., Kirchhausen, T. & Shair, M. D. Synthesis of a 10,000-membered library of molecules resembling carpanone and discovery of vesicular traffic inhibitors. J. Am. Chem. Soc. 128, 5391–5403 (2006).
Lewis, C. A. & Miller, S. J. Site-selective derivatization and remodeling of erythromycin a by using simple peptide-based chiral catalysts. Angew. Chem., Int. Ed. 45, 5616–5619 (2006).
Pathak, T. P. & Miller, S. J. Chemical tailoring of teicoplanin with site-selective reactions. J. Am. Chem. Soc. 135, 8415–8422 (2013).
Morrison, K. C. & Hergenrother, P. J. Natural products as starting points for the synthesis of complex and diverse compounds. Nat. Prod. Rep. 31, 6–14 (2014).
Huigens III, R. W. et al. A ring-distortion strategy to construct stereochemically complex and structurally diverse compounds from natural products. Nat. Chem. 5, 195–202 (2013).
Karimov, R. R. & Hartwig, J. F. Transition-metal-catalyzed selective functionalization of C(sp3)-H bonds in natural products. Angew. Chem. Int. Ed. 57, 4234–4241 (2018).
Vitaku, E., Smith, D. T. & Njardarson, J. T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among US FDA approved pharmaceuticals. J. Med. Chem. 57, 10257–10274 (2014).
Park, Y., Kim, Y. & Chang, S. Transition metal-catalyzed C–H amination: Scope, mechanism, and applications. Chem. Rev. 117, 9247–9301 (2017).
Hartwig, J. F. & Larsen, M. A. Undirected, homogeneous C–H bond functionalization: Challenges and opportunities. ACS Cent. Sci. 2, 281–292 (2016).
Chiappini, N. D., Mack, J. B. C. & Du Bois, J. Intermolecular C(sp3)-H amination of complex Molecules. Angew. Chem. Int. Ed. 57, 4956–4959 (2018).
Karimov, R. R., Sharma, A. & Hartwig, J. F. Late stage azidation of complex molecules. ACS Cent. Sci. 2, 715–724 (2016).
Collet, F., Lescot, C. & Dauban, P. Catalytic C–H amination: The stereoselectivity issue. Chem. Soc. Rev. 40, 1926–1936 (2011).
Dequirez, G., Pons, V. & Dauban, P. Nitrene chemistry in organic synthesis: Still in its Infancy? Angew. Chem., Int. Ed. 51, 7384–7395 (2012).
Lebel, H., Spitz, C., Leogane, O., Trudel, C. & Parmentier, M. Stereoselective rhodium-catalyzed amination of alkenes. Org. Lett. 13, 5460–5463 (2011).
Harvey, M. E., Musaev, D. G. & Du Bois, J. A diruthenium catalyst for selective, intramolecular Allylic C–H Amination: Reaction development and mechanistic insight gained through experiment and theory. J. Am. Chem. Soc. 133, 17207–17216 (2011).
Paradine, S. M. & White, M. C. Iron-catalyzed intramolecular Allylic C–H amination. J. Am. Chem. Soc. 134, 2036–2039 (2012).
Cramer, S. A. & Jenkins, D. M. Synthesis of aziridines from alkenes and aryl azides with a reusable macrocyclic tetracarbene iron catalyst. J. Am. Chem. Soc. 133, 19342–19345 (2011).
Hennessy, E. T., Liu, R. Y., Iovan, D. A., Duncan, R. A. & Betley, T. A. Iron-mediated intermolecular N-group transfer chemistry with olefinic substrates. Chem. Sci. 5, 1526–1532 (2014).
Bagchi, V. et al. A Versatile Tripodal Cu(I) Reagent for C–N construction via nitrene-transfer chemistry: Catalytic perspectives and mechanistic insights on C–H aminations/amidinations and olefin aziridinations. J. Am. Chem. Soc. 136, 11362–11381 (2014).
Maestre, L., Sameera, W. M. C., Díaz-Requejo, M. M., Maseras, F. & Pérez, P. J. A general mechanism for the copper- and silver-catalyzed olefin aziridination reactions: Concomitant involvement of the singlet and triplet pathways. J. Am. Chem. Soc. 135, 1338–1348 (2013).
Li, Z., Capretto, D. A., Rahaman, R. & He, C. Silver-catalyzed intermolecular amination of C-H Groups. Angew. Chem., Int. Ed. 46, 5184–5186 (2007).
Llaveria, J. et al. Chemo-, Regio-, and Stereoselective silver-catalyzed aziridination of dienes: scope, mechanistic studies, and ring-opening reactions. J. Am. Chem. Soc. 136, 5342–5350 (2014).
Yang, M. et al. Silver-catalysed direct amination of unactivated C–H bonds of functionalized molecules. Nat. Commun. 5, 4707–4712 (2014).
Paradine, S. M. et al. A manganese catalyst for highly reactive yet chemoselective intramolecular C(sp3)–H amination. Nat. Chem. 7, 987–994 (2015).
Liang, J.-L., Huang, J.-S., Yu, X.-Q., Zhu, N. & Che, C.-M. Metalloporphyrin-mediated asymmetric nitrogen-atom transfer to hydrocarbons: Aziridination of alkenes and amidation of saturated C-H bonds catalyzed by chiral ruthenium and manganese porphyrins. Chem. Eur. J. 8, 1563–1572 (2002).
Ju, M. & Schomaker, J. M. Nitrene transfer catalysts for enantioselective C–N bond formation. Nat. Rev. Chem. 5, 580–594 (2021).
Ju, M. et al. Silver-catalyzed enantioselective propargylic C−H bond amination through rational ligand design. J. Am. Chem. Soc. 142, 12930–12936 (2020).
Harada, S., Kobayashi, M., Kono, M. & Nemoto, T. Site-selective and chemoselective C−H functionalization for the synthesis of spiroaminals via a silver-catalyzed nitrene transfer reaction. ACS Catal. 10, 13296–13304 (2020).
Knecht, T., Mondal, S., Ye, J.-H., Das, M. & Glorius, F. Intermolecular, branch-selective, and redox-neutral Cp*IrIII - catalyzed Allylic C-H Amidation. Angew. Chem. Int. Ed. 58, 7117–7121 (2019).
Teh, W. P., Obenschain, D. C., Black, B. M. & Michael, F. E. Catalytic metal-free Allylic C−H amination of terpenoids. J. Am. Chem. Soc. 142, 16716–16722 (2020).
Lei, H. & Rovis, T. Ir-catalyzed intermolecular branch-selective allylic C−H amidation of unactivated terminal olefins. J. Am. Chem. Soc. 141, 2268–2273 (2019).
Burman, J. S. & Blakey, S. B. Regioselective intermolecular Allylic C-H amination of disubstituted olefins via Rhodium/π-Allyl Intermediates. Angew. Chem. Int. Ed. 56, 13666–13669 (2017).
Rigoli, J. W. et al. Chemoselective Allene Aziridination via Ag(I) Catalysis. Org. Lett. 15, 290–293 (2013).
Huang, M., Yang, T., Paretsky, J. D., Berry, J. F. & Schomaker, J. M. Inverting steric effects: Using “Attractive” Noncovalent Interactions to direct silver-catalyzed nitrene transfer. J. Am. Chem. Soc. 139, 17376–17386 (2017).
Alderson, J. M., Corbin, J. R. & Schomaker, J. M. Tunable, chemo- and site-selective nitrene transfer reactions through the rational design of Silver(I) Catalysts. Acc. Chem. Res. 50, 2147–2158 (2017).
Weatherly, C., Alderson, J. M., Berry, J. F., Hein, J. E. & Schomaker, J. M. Catalyst-controlled nitrene transfer by tuning metal: Ligand ratios: Insight into the mechanisms of chemoselectivity. Organometallics 36, 1649–1661 (2017).
Rigoli, J. W., Weatherly, C. D., Alderson, J. M., Vo, B. T. & Schomaker, J. M. Tunable, Chemoselective Amination via Silver Catalysis. J. Am. Chem. Soc. 135, 17238–17241 (2013).
Ju, M. et al. Tunable catalyst-controlled syntheses of β- and γ-amino alcohols enabled by silver-catalysed nitrene transfer. Nat. Catal. 2, 899–908 (2019).
Wang, J., Zheng, K., Li, T. & Zhan, X. Mechanism and chemoselectivity of Mn-catalyzed intramolecular nitrene transfer reaction: C–H Amination vs. C=C Aziridination. Catalysts 10, 292–303 (2020).
Dolan, N. S., Scamp, R. J., Yang, T., Berry, J. F. & Schomaker, J. M. Catalyst-controlled and tunable, chemoselective silver-catalyzed intermolecular nitrene transfer: Experimental and computational studies. J. Am. Chem. Soc. 138, 14658–14667 (2016).
Liang, C. et al. Toward a synthetically useful stereoselective C−H Amination of Hydrocarbons. J. Am. Chem. Soc. 130, 343–350 (2008).
Lescot, C., Darses, B., Collet, F., Retailleau, P. & Dauban, P. Intermolecular C−H Amination of Complex Molecules: Insights into the Factors Governing the Selectivity. J. Org. Chem. 77, 7232–7240 (2012).
Li, J. et al. Simultaneous structure–activity studies and arming of natural products by C–H amination reveal cellular targets of eupalmerin acetate. Nat. Chem. 5, 510–517 (2013).
Alderson, J. M., Phelps, A. M., Scamp, R. J., Dolan, N. S. & Schomaker, J. M. Ligand-controlled, tunable silver-catalyzed C−H Amination. J. Am. Chem. Soc. 136, 16720–16723 (2014).
Scamp, R. J., Jirak, J. G., Dolan, N. S., Guzei, I. A. & Schomaker, J. M. A General Catalyst for Site-Selective C(sp3)−H bond amination of activated secondary over Tertiary Alkyl C(sp3)−H Bonds. Org. Lett. 18, 3014–3017 (2016).
Bakhoda, A. et al. Copper-Catalyzed C(sp3)-H Amidation: Sterically driven primary and secondary C-H site-selectivity. Angew. Chem. Int. Ed. 58, 3421–3425 (2019).
Meng, D., Tang, Y., Wei, J., Shi, X. & Yang, M. Copper-catalyzed remote (δ) C(sp3)–H bond amination: A practical strategy to construct pyrrolidine derivatives. Chem. Commun. 53, 5744–5747 (2017).
Dauban, P., Sanière, L., Tarrade, A. & Dodd, R. H. Copper-catalyzed nitrogen transfer mediated by iodosylbenzene PhI=O. J. Am. Chem. Soc. 123, 7707–7708 (2001).
Badiei, Y. M. et al. Copper–nitrene complexes in catalytic C-H Amination. Angew. Chem. Int. Ed. 47, 9961–9964 (2008).
Degennaro, L., Trinchera, P. & Luisi, R. Recent advances in the stereoselective synthesis of aziridines. Chem. Rev. 114, 7881–7929 (2014).
Singh, G. S., D’hooghe, M. & Kimpe, N. D. Synthesis and reactivity of C-heteroatom-substituted aziridines. Chem. Rev. 107, 2080–2135 (2007).
Ohno, H. Synthesis and applications of vinylaziridines and ethynylaziridines. Chem. Rev. 114, 7784–7814 (2014).
Johnson, S. L. & Hilinski, M. K. Organocatalytic olefin aziridination via iminium-catalyzed nitrene transfer: Scope, limitations, and mechanistic insight. J. Org. Chem. 84, 8589–8595 (2019).
Guo, Y., Pei, C., Jana, S. & Koenigs, R. M. Synthesis of trifluoromethylated aziridines via photocatalytic amination reaction. ACS Catal. 11, 337–342 (2021).
Masakado, S., Kobayashi, Y. & Takemoto, Y. Photo-induced aziridination of alkenes with N-Sulfonyliminoiodinanes. Chem. Pharm. Bull. 66, 688–690 (2018).
Minakata, S., Morino, Y., Oderaotoshi Y. & Komatsu, M. Novel aziridination of olefins: direct synthesis from sulfonamides using t-BuOI. Chem. Commun. 31, 3337–3339 (2006).
Nakajima, M. et al. Direct S0→Tn Transition in the Photoreaction of Heavy-Atom-Containing Molecules. Angew. Chem. Int. Ed. 59, 6847–6852 (2020).
Caballero, G. M. & Gros, E. G. On the Mechanism of Cleavage of Thioacetals Promoted by Copper(II) Sulphate Adsorbed on Silica Gel. Z. Naturforsch. 52b, 1147–1151 (1997).
Acknowledgements
This work was financially supported by National Natural Science Foundation of China (21977008, 21603246), Natural Science Foundation of Guangdong Province (2018A0303130052), and Shenzhen Basic Research Project (JCYJ20180503182116931).
Author information
Authors and Affiliations
Contributions
Q.X., C.C., and Z.Z. conceived, designed, and originated this project. Q.X., D.J., M.D., H.C., and J.Z. performed the experiments, obtained all spectroscopic data, and analyzed the results. Q.X., C.C., L.G., and Y.Z. co-wrote the manuscript. T.L. performed the biology experiments and data analysis. We would like to thank professor Zhen Yang (Peking University) for his support and valuable suggestions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Chemistry thanks Allan Watson and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xing, Q., Jiang, D., Zhang, J. et al. Combining visible-light induction and copper catalysis for chemo-selective nitrene transfer for late-stage amination of natural products. Commun Chem 5, 79 (2022). https://doi.org/10.1038/s42004-022-00692-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42004-022-00692-6