Abstract
Carbon quantum dots (CQDs) have recently received a lot of attention due to their unique physical properties, and their environmentally friendly features such as low toxicity and high biocompatibility. Supercritical fluids, which possess unusual properties such as high solubility, high diffusivity, low viscosity and zero surface tension, are now commonly used particularly in the fields of electronic, chemical and materials science and engineering. Here, we synthesise carbon nano/microparticles in supercritical acetone, in which neither external molecules nor starting materials are dissolved/dispersed. We find that carbon microparticles and nano structures such as graphene quantum dots (GQDs), carbon nano onions (CNOs) and elongated carbon nano onions (eCNOs) are self-assembled via thermal decomposition of acetone under its supercritical conditions. We also find that the carbon microparticles are in fact formed by GQDs, CNOs and eCNOs, the microparticles being physically resolved into GQDs, CNOs and eCNOs with sonication. The fluorescence features of the carbon nano structures are clarified, noting that no photobleaching was observed for at least one month. The present result may well lead to the development of facile bottom-up methodologies for synthesising nano materials in solvents under their supercritical conditions without using any external precursors/starting materials.
Similar content being viewed by others
Introduction
Fundamental carbon nanostructures such as fullerenes, carbon nanobelts, carbon nanorings, carbon nanotubes, and graphene are of great interest and importance as zero-, one- and two-dimensional ones1,2,3,4,5, whereas they are also important from a practical point of view considering a wide range of applications in the fields of nano/micro mechatronics, electronics, chemistry, biochemistry, biotechnology, biomedicine, and environmental science and technology6,7,8,9. Carbon quantum dots (CQDs) have recently received a lot of attention due to their unique physical properties, and their environmentally friendly features, such as low toxicity and high biocompatibility10,11. The synthetic methods of CQDs are classified into two; i.e., (a) top–down ultrafine fabrication using starting materials and (b) bottom-up self-assembly using precursors/catalysts10,11.
Second-order phase transitions occur once the temperature; \(T\), pressure; \(P\), and molar volume; \(V\), reach their critical values; \({T}_{c}\), \({P}_{c}\) and \({V}_{c}\). It is known that gas-liquid transition is one of second-order phase transitions and that large molecular clusters are formed as the fluid systems approach their critical points. As a result, “Critical opalescence” is induced, and the specific heat and compressibility diverge as described by the universal scaling laws12. Supercritical fluids, the state of which is located above the critical points, possess unique properties such as high solubility, high diffusivity, low viscosity and zero surface tension, being neither gas nor liquid, and they are nowadays quite commonly utilised in various fields; e.g., for the extraction of chemicals, the purification of materials/devices, the encouragement of reactions and the synthesis of materials13,14,15. One of the efficient top-down methods for producing CQDs is, utilising the above unique properties; particularly high compressibility, exfoliation of graphite in supercritical fluids16. It is also known that the formation of carbon-carbon bonding is encouraged and that graphene oxide (GO) is efficiently transformed into reduced GO (rGO) in supercritical fluids14,17.
In this study, we synthesise carbon particles in acetone under its supercritical conditions without dissolving/dispersing any external precursors or starting materials in acetone. We find that both carbon microparticles and carbon nanostructures such as graphene quantum dots (GQDs), carbon nano onions (CNOs), and elongated CNOs (eCNOs) are self-assembled via thermal decomposition of acetone, and what is more, surprisingly, the microparticles are in fact formed by GQDs, CNOs, and eCNOs, noting that the microparticles are physically resolved into GQDs, CNOs and eCNOs with sonication.
Results and discussion
First, the experimental methods are briefly explained here (see also Fig. 1). The details of the experimental and characterisation procedures are summarised in the “Methods” section. We confined acetone in a supercritical fluid chamber, and the fluid temperature was regulated using a heater surrounding the chamber and a proportional-integral-differential (PID) controller (see Fig. 1a, b). The temperature and molar volume of acetone were set at (400 °C, 2.75 × 10−4 m3 mol−1), (425, 2.75 × 10−4), (450, 2.75 × 10−4), (450, 3.67 × 10−4), (450, 5.50 × 10−4) or (450, 8.46 × 10−4). Note that the above conditions lie in the supercritical state of acetone, Tc, Pc, and Vc of acetone being 508 K (235 °C), 4.8 MPa, and 2.16 × 10−4 m3 mol−118 (see Fig. S1 for the experimental conditions plotted on the pressure-molar volume phase diagram). Once the temperature had reached a target value, it was kept constant for 0, 0.5, 1, 1.5, 2, or 6 h, and then lowered down to room temperature (Fig. 1c). The materials produced in the chamber were dispersed in ethanol, replacing acetone with ethanol in the chamber (Fig. 1d), and the solution of the materials dispersed in ethanol was filtered with a 100 nm mesh to separate sub-micro/microscale materials (“Sub-micro/microscale materials I”) from the solution (“Filtered solution I”) (Fig. 1e, f). “Sub-micro/microscale materials I” collected with filtration were dispersed in ethanol again (Fig. 1g), having been washed four times with ethanol, and then the solution was divided into two (Fig. 1h, i). The first solution was filtered with a 100 nm mesh to separate the sub-micro/microscale materials (“Sub-micro/microscale materials II”) from the solution (“Filtered solution II”) (Fig. 1h), noting that “Sub-micro/microscale materials II” would be basically the same as “Sub-micro/microscale materials I” and that no materials would be dissolved/dispersed in “Filtered solution II”. The second solution was sonicated for 1 h (Fig. 1i) and then filtered with a 100 nm mesh to separate sub-micro/microscale materials (“Sub-micro/microscale materials III”) from the solution (“Filtered solution III”) (Fig. 1j), noting that “Sub-micro/microscale materials I” could be physically resolved into fragments with sonication, in which case those fragments would be dissolved/dispersed in “Filtered solution III”. The structures of “Sub-micro/microscale materials I” were observed and characterised with scanning electron microscopy (SEM), X-ray diffractometry (XRD), and Raman spectroscopy. The fluorescence spectra of the original acetone and “Filtered solution I, II and III” were measured with fluorescence spectroscopy, whereas the nanostructures dissolved/dispersed in “Filtered solution I and III” were observed with transmission electron microscopy (TEM) and the absorption spectrum of “Filtered solution III” was measured with ultraviolet-visible (UV-Vis) spectroscopy.
a, b Supercritical fluid chamber. Acetone was confined in the chamber. The temperature was regulated by a heater surrounding the chamber and a PID controller. c Time variation of the temperature. Once the temperature had reached a target value, it was kept constant for 0, 0.5, 1, 1.5, 2, or 6 h and then lowered down to room temperature. d Replacement of acetone with ethanol in the chamber. e, f Filtration of the solution with a 100 nm mesh. Sub-micro/microscale materials (“Sub-micro/microscale materials I”) were separated from the solution (“Filtered solution I”). g Dispersion of “Sub-micro/microscale materials I” in ethanol. h Filtration of the solution with a 100 nm mesh. Sub-micro/microscale materials (“Sub-micro/microscale materials II”) were separated from the solution (“Filtered solution II”). i Ultrasonication of the solution. j Filtration of the solution with a 100 nm mesh. Sub-micro/microscale materials (“Sub-micro/microscale materials III”) were separated from the solution (“Filtered solution III”).
We found that sub-micro/microscale particles were produced in supercritical acetone under certain conditions. SEM images, X-ray diffraction pattern, and Raman spectrum of sub-micro/microscale particles (“Sub-micro/microscale materials I”) are shown in Fig. 2 (see also Fig. 1e, f). The effect of the synthetic time on the production of sub-micro/microscale particles is shown in Fig. 2a−c, where the temperature and molar volume of acetone were set at 450 °C and 2.75 × 10−4 m3 mol−1. It is clearly shown that the quantity of spherical particles increased with an increase in the synthetic time. Sub-micro/microscale particles were most efficiently produced, in terms of the quantity of the particles, when the synthetic temperature was 450 °C, the synthetic time was longer than or equal to 2 h, and the molar volume was 2.75 × 10−4 m3 mol−1. Note that, in general, the amount of sub-micro/microscale particles produced in supercritical acetone increased with an increase in the temperature and synthetic time, and with a decrease of the molar volume (see Figs. S2–4 in the Supplementary Information for the effect of the synthetic conditions on the production of sub-micro/microscale particles). There were no sub-micro/microscale particles assembled under the following conditions; the molar volume was 8.46 × 10−4 m3 mol−1, the temperature was lower than or equal to 425 °C and the synthetic time was shorter than or equal to 0.5 h. The X-ray diffraction pattern and Raman spectrum of the sub-micro/microscale particles corresponding to Fig. 2c are shown in Fig. 2d, e, where the wavelength of the X-ray and excitation laser was, respectively, 0.1392 and 515 nm. It is supposed, from the (002) diffraction peak in the XRD pattern and the peaks of D and G bands in the Raman spectrum, that the sub-micro/microscale particles were composed of graphitic layers. The primary peak corresponding to the (002) crystal plane was located at \(2\theta\) = 25.35° and the full width at half maximum (FWHM); \(\beta\), was 0.056 rad (Fig. 2d). Therefore, the distance between neighbouring graphitic layers; \(d\), the height of layers; \({L}_{c}\), and the number of layers; \({n}_{L}\), are estimated to be \(d\) = 0.317 nm, \({L}_{c}\) = 2.395 nm and \({n}_{L}\) = 7.56 based on Eqs. (1)–(3) shown in the “Method” section19,20. In the case of the Raman spectrum, peaks corresponding to the D and G bands appeared at 1361 and 1596 cm−1, whereas those of broad 2D and D + G bands at 2706 and 2922 cm−121,22,23. The ratio of the intensity of the D band peak to that of the G band peak; \({I}_{D}/{I}_{G}\), was 0.728, from which the lateral length of a fragment of graphitic layers; \({L}_{D}\), is estimated to be \({L}_{D}\) = 13.19 nm (see Eq. (4) in the “Method” section)24. The broad 2D band and low ratio \({I}_{2D}/{I}_{G}\) also indicate that the fragments are composed of several layers of graphene22. It is therefore supposed, based on the XRD and Raman spectra, that the sub-micro/microscale particles were formed by fragments composed of graphitic layers, the average lateral size, interlayer distance, and number of layers of which were 13.19 nm, 0.317 nm, and 7 or 8.
a SEM image of particles. The synthetic temperature, synthetic time, and molar volume were 450 °C, 0.5 h, and 2.75 × 10−4 m3 mol−1. The scale bar represents 10 µm. b SEM image of particles. The synthetic temperature, synthetic time, and molar volume were 450 °C, 1 h, and 2.75 × 10−4 m3 mol−1. The scale bar represents 10 µm. c SEM image of particles. The synthetic temperature, synthetic time, and molar volume were 450 °C, 2 h, and 2.75 × 10−4 m3 mol−1. The scale bar represents 10 µm. d XRD pattern of the particles. The particles were synthesised under the conditions of 450 °C, 2 h, and 2.75 × 10−4 m3 mol−1 (Fig. 2c). The wavelength of the X-ray was 0.1392 nm. The (002) diffraction peak was located at \(2\theta\) = 25.35°. e Raman spectrum of particles corresponding to Fig. 2c. The wavelength of the excitation laser was 515 nm. The D and G band peaks appeared at 1361 and 1596 cm−1. The 2D and D + G bands were also detected at 2706 and 2922 cm−1.
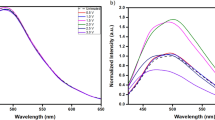
Sub-micro/microscale particles dispersed in ethanol were sonicated and then filtered so that the particles (“Sub-micro/microscale materials III”) were separated from the solution (“Filtered solution III”) (see Fig. 1i, j and the procedure (g) in the “Method” section). Now, let us focus on the structures of fragments dissolved/dispersed in “Filtered solution III”. TEM images of fragments dissolved/dispersed in “Filtered solution III” are shown in Fig. 3a−c. Interestingly, the sub-micro/microscale particles were physically resolved into fragments with sonication, and the structures of those fragments dissolved/dispersed in “Filtered solution III” were either planer layers (GQDs) (Fig. 3a) or closed layers such as carbon nano onions (CNOs) (Fig. 3b) and eCNOs (Fig. 3c). The gap between neighbouring layers measured from the TEM images was 0.32 nm in both GQDs and CNOs/eCNOs cases, which coincided with the interlayer distance estimated by the above XRD analysis (see Fig. 3d−f). It can, therefore, be concluded that the sub-micro/microscale particles were formed by GQDs and CNOs/eCNOs, the sub-micro/microscale particles having been resolved into fragments composed of GQDs and CNOs/eCNOs with sonication. TEM images of fragments dissolved/dispersed in “Filtered solution I” are shown in Fig. S5 in the Supplementary Information. Those fragments dissolved/dispersed in “Filtered solution I” were also either planer graphene layers or closed layers, the average interlayer distance of which was 0.32 nm. It is, therefore, supposed that those nanostructures, such as GQDs and CNOs/eCNOs, were first formed, and then sub-micro/microscale particles were self-assembled by GQDs and CNOs/eCNOs. The photographs of acetone before the synthesis of particles and “Filtered solution I, II, and III” are shown in Fig. 4a, where the left and right photographs in each pair of them were, respectively, taken under irradiation of white light and UV light of 365 nm wavelength in a dark box. Acetone and “Filtered solution II” were not fluorescent, which shows that there were no nanostructures dissolved/dispersed in acetone in advance of the experiments and in “Filtered solution II” as expected, whereas “Filtered solution I and III” were fluorescent. This indicates that sub-micro/microscale particles (“Sub-micro/microscale materials I”) were self-assembled by those nanostructures such as GQDs and CNOs/eCNOs as explained above and that an excess amount of GQDs and CNOs/eCNOs, which had not participated in the formation of “Sub-micro/microscale materials I”, were dissolved/dispersed in “Filtered solution I”, while GQDs and CNOs/eCNOs, which had been physically resolved into from the sub-micro/microscale particles with sonication, were dissolved/dispersed in “Filtered solution III”. The fluorescence spectra of those nanostructures; i.e., GQDs and CNOs/eCNOs, dissolved/dispersed in “Filtered solution III” are shown in Fig. 4b, where the wavelength of the excitation laser was changed from 300 to 490 nm at 10 nm intervals. The primary emission frequency shifted gradually from around 450 nm to 500 nm, and the peak intensity decreased with an increase in the excitation wavelength16,25. There was no apparent change in the fluorescence spectra of the solution (“Filtered solution III”) for at least one month. The fluorescence spectrum of dried CQDs is shown in Fig. S6 in the Supplementary Information, where the excitation wavelength was 360 nm. The primary fluorescence peak of those nanostructures dissolved/dispersed in “Filtered solution III” shifted to 550 nm after the solvent had been dried. Photobleaching was not observed for at least one month in the case of dried CQDs as well. Let us note that the fluorescence spectra of those nanostructures dissolved/dispersed in “Filtered solution I” were more or less the same as those of nanostructures dissolved in “Filtered solution III” (see Fig. S7 in the Supplementary Information for the fluorescence spectra of nanostructures dissolved/dispersed in “Filtered solution I”) and that “Filtered solution II” was not fluorescent (see Fig. 4a and Fig. S8 in the Supplementary Information for the fluorescence spectra of “Filtered solution II”), which also confirms that an excess amount of GQDs and CNOs/eCNOs, which had not participated in the formation of “Sub-micro/microscale materials I”, were dissolved/dispersed in “Filtered solution I” and that GQDs and CNOs/eCNOs, which had been physically resolved into from the sub-micro/microscale particles, were dissolved/dispersed in “Filtered solution III”. The dependence of the fluorescence spectra of “Filtered solution I” on the synthetic time is shown in Fig. S9 in the Supplementary Information, where the synthetic time was changed; 0, 1, 2, and 6 h. There were no sub-micro/microscale particles produced when the synthetic time was 0 h, but the “Filtered solution I” was still fluorescent. The position of the primary peak corresponding to the 0 h synthesis was the same as that corresponding to the 1, 2, and 6 h syntheses when the excitation frequency was over 320 nm, which confirms that carbon nanostructures such as GQDs and CNOs/eCNOs were first created and then sub-micro/microscale particles were formed by GQDs and CNOs/eCNOs when the number of GQDs and CNOs/eCNOs was large enough. The effect of the synthetic temperature on the fluorescence spectra of “Filtered solution I” is shown in Fig. S10 in the Supplementary Information, where the synthetic temperature was changed; 400, 425, and 450 °C, noting that sub-micro/microscale particles were produced only at 450 °C. This also confirms that there were nanostructures such as GQDs and CNOs/eCNOs created even when no sub-micro/microscale particles were formed, although the fluorescence intensity in the case of 400 and 425 °C was much lower than that in the case of 450 °C. The absorption spectrum of “Filtered solution III” is shown in Fig. 4c. It is supposed that the shoulder at 225 nm represents the \(\pi -{\pi }^{* }\) transitions of C–C bonds in GQDs and CNOs/eCNOs, whereas the one at 256 nm the \(n-{\pi }^{* }\) transitions of C = O bonds16,25.
Sub-micro/microscale particles were synthesised in supercritical acetone under the conditions of 450 °C (synthetic temperature), 2 h (synthetic time), and 2.75 × 10−4 m3 mol−1 (molar volume of acetone), and the solution of the sub-micro/microscale materials dispersed in ethanol was ultrasonicated. a TEM image of graphene layers composing the sub-micro/microscale particles. The scale bar represents 5 nm. b TEM image of a carbon nano onion composing the sub-micro/microscale particles. The scale bar represents 5 nm. c TEM image of an elongated carbon nano onion composing the sub-micro/microscale particles. The scale bar represents 5 nm. d Grey level distribution along the red line across the graphene shown in Fig. 3a. e Grey level distribution along the red line across the carbon nano onion shown in Fig. 3b. f Grey level distribution along the red line across the elongated carbon nano onion shown in Fig. 3c.
a Colour of acetone before the experiment and “Filtered solution I, II and III”. The left and right photographs in each pair of them were, respectively, taken under irradiation of white light and UV light of 365 nm wavelength in a dark box. b Fluorescence spectra of “Filtered solution III”. The excitation wavelength was changed from 300 to 490 nm at 10 nm intervals. c Absorption spectrum of “Filtered solution III”.
Let us discuss the mechanism of the formation of carbon nanostructures such as GQDs and CNOs/eCNOs and carbon sub-micro/microscale particles in supercritical acetone. It is known that byproducts such as benzene, 1-methylnaphthalene, and 2-methylnaphthalene are created via the thermal decomposition of acetone26, and fragments of graphene layers are self-assembled by methylnaphthalene27. The formation of carbon-carbon bonding is encouraged in supercritical fluid14 and GO is efficiently reduced to rGO in supercritical fluid17. It is also known that CNOs are formed by fragments of graphene layers28. It is therefore supposed in the present case that (a) acetone was thermally decomposed and byproducts such as benzene and methylnaphthalene were formed while the temperature was increased and kept constant; (b) fragments of graphene layers were formed by those byproducts; (c) some of the graphene layers were converted to closed carbon layers such as CNOs and eCNOs; and (d) what is more, sub-micro/microscale particles were assembled by those carbon nanostructures during the cooling period when the number of the fragments was high enough. Sub-micro/microscale particles may well be quite easily assembled by nanostructures in supercritical fluids in general due to their unique properties such as high solubility, high diffusivity, low viscosity, and zero surface tension under high pressure29,30,31.
Conclusions
We developed a methodology for producing CQDs utilising supercritical fluid. We successfully synthesized carbon nanostructures such as graphene carbon dots (GCDs) and carbon nano onions (CNOs)/elongated carbon nano onions (eCNOs) in acetone under its supercritical conditions without dissolving/dispersing any starting materials/precursors/catalysts in acetone. The fluorescence features of those nanostructures were clarified, noting that no photobleaching was observed for at least one month. We also found that sub-micro/microscale particles were self-assembled by GCDs and CNOs/eCNOs. It is, in general, much easier to handle/manipulate microparticles than nanoparticles, and in the present case, CQDs can be obtained at any time with sonication to the solution of the carbon microparticles. We will be investigating the effect of different organic solvents on the production of carbon nano/microstructures under their supercritical conditions as part of the development and establishment of facile one-pot bottom-up methodologies for creating carbon nano/micro materials utilizing supercritical fluids without dissolving any external molecules/materials in the fluids.
Methods
Experimental procedure
The experimental procedure is summarised below (see also Fig. 1).
-
(a)
Acetone was introduced into a supercritical fluid chamber made of stainless steel and the chamber was sealed (see Fig. 1a, b). The inner volume of the chamber was 3.72 × 10−5 m3.
-
(b)
The temperature of acetone was regulated by a heater surrounding the chamber and a PID controller (LT47A, Chino Co.) (Fig. 1a, b). The temperature and molar volume of acetone were set at (400 °C, 2.75 × 10−4 m3 mol−1), (425, 2.75 × 10−4), (450, 2.75 × 10−4), (450, 3.67 × 10−4), (450, 5.50 × 10−4) or (450, 8.46 × 10−4) (see Fig. S1 in the Supplementary Information for the experimental conditions plotted on the pressure-molar volume phase diagram, where isotherms and the critical point are shown based on the van der Waals equation, noting that the values of the actual critical temperature, pressure and molar volume of acetone; that is, 508 K (235 °C), 4.8 MPa and 2.16 × 10−4 m3 mol−118, are different from those derived by the van der Waals equation as well known). The chamber was heated, and once the temperature had reached a target temperature, it was kept constant for 0, 0.5, 1, 1.5, 2, or 6 h. The chamber was then cooled down to room temperature (Fig. 1c).
-
(c)
After the above experiment, the chamber was opened, and acetone was replaced with ethanol in the chamber (Fig. 1d).
-
(d)
The solution of the synthesised materials dispersed in ethanol was filtered with a 100 nm mesh (H010A047A, Advantec Toyo Kaisha Ltd.) so that sub-micro/microscale materials (“Sub-micro/microscale materials I”) were separated from the solution (“Filtered solution I”) (Fig. 1e, f). Note that nanoscale materials, which had not been trapped by the filter, would have been dissolved/dispersed in “Filtered solution I”.
-
(e)
“Sub-micro/microscale materials I” were washed four times with ethanol and dispersed in ethanol. The solution was divided into two (Fig. 1g–i). The structures of “Sub-micro/microscale materials I” were characterised with SEM, XRD, and Raman spectroscopy (see “Procedure (k), (l), and (m)” in the “Characterisation procedure” below).
-
(f)
The first solution was filtered with a 100 nm mesh so that the sub-micro/microscale materials (“Sub-micro/microscale materials II”) were separated from the solution (“Filtered solution II”) (Fig. 1h). Note that it is supposed that no materials were dissolved/dispersed in “Filtered solution II”, which was confirmed with fluorescence spectroscopy (see “Procedure (h)” in the “Characterisation procedure” below).
-
(g)
The second solution was sonicated at 38 kHz and 0.18 kW (US-5KS, SND Co. Ltd.) for 1 h and then filtered with a 100 nm mesh so that the sub-micro/micrometre materials (“Sub-micro/microscale materials III”) were separated from the solution (“Filtered solution III”) (Fig. 1i, j). Note that nanoscale materials would have been dissolved/dispersed in “Filtered solution III” if the sub-micro/microscale materials had been resolved into nanoscale fragments with sonication.
Characterisation procedure
The characterisation procedure is summarised below.
-
(h)
The fluorescence spectra of the original acetone and “Filtered solution I, II, and III” were measured with fluorescence spectroscopy (FP-6500, JASCO Co.). The excitation wavelength was changed from 300 to 490 nm at 10 nm intervals.
-
(i)
The structures of nanoscale materials dissolved/dispersed in “Filtered solution I and III” were observed with TEM (ARM200F, JEOL Ltd.), setting the acceleration voltage at 80 kV. The distances between the graphitic layers in the nanoscale materials were determined from the grey-level distributions across the layers using ImageJ.
-
(j)
The absorption spectra of “Filtered solution III” were measured with UV-vis spectroscopy (DU730, Beckman Coulter Inc.).
-
(k)
The morphology of “Sub-micro/microscale materials I” was observed with SEM (SU8030, Hitachi Ltd.).
-
(l)
The crystallinity of “Sub-micro/microscale materials I” was measured with XRD (SmartLab, Rigaku Corp.). The wavelength of the X-ray was 0.1392 nm. The distance between neighbouring graphitic layers; \(d\), the height of layers; \({L}_{c}\), and the number of layers; \({n}_{L}\), were estimated by the following equations refs. 19,20.
$$2d\sin \theta =n{\lambda }_{X}$$(1)$${L}_{C}=K{\lambda }_{X}/\left(\beta \cos \theta \right)$$(2)$${n}_{L}={L}_{C}/d$$(3)where \(d\), \(\theta\), \({\lambda }_{X}\), \({L}_{C}\), \(K\) and \(\beta\) are the distance between neighbouring graphitic layers, the angle corresponding to the (002) diffraction peak, the wavelength of the X-ray, the height of layers, the shape factor, and the FWHM of the (002) diffraction peak. In the present study, 0.94 was assigned to \(K\)19,20.
-
(m)
The Raman spectra of “Sub-micro/microscale materials I” were measured with laser Raman spectroscopy (HR-800UV, Horiba Ltd.). The wavelength of the incident laser was 515 nm. The lateral length of graphitic layers; \({L}_{D}\), was estimated by the following equation24.
where \({I}_{D}/{I}_{G}\) and \({\lambda }_{L}\) are the ratio of the intensity of the D band peak to that of the G band peak and the wavelength of the laser.
Data availability
All of the data supporting this work are available from the corresponding author upon reasonable request.
References
Kroto, H. W., Heath, J. R., O’Brien, S. C., Curl, R. F. & Smalley, R. E. C60: buckminsterfullerene. Nature 318, 162–163 (1985).
Povie, G., Segawa, Y., Nishihara, T., Miyauchi, Y. & Itami, K. Synthesis of a carbon nanobelt. Science 356, 172–175 (2017).
Omachi, H., Nakayama, T., Takahashi, E., Segawa, Y. & Itami, K. Initiation of carbon nanotube growth by well-defined carbon nanorings. Nat. Chem. 5, 572–576 (2013).
Iijima, S. Synthesis of carbon nanotubes. Nature 354, 56–58 (1991).
Geim, A. K. & Novoselov, K. S. The rise of graphene. Nat. Mater. 6, 183–191 (2007).
Acquah, S. F. A. et al. The beautiful molecule: 30 years of C60 and its derivatives. ECS J. Solid State Sci. Technol. 6, M3155 (2017).
Rao, R. et al. Carbon nanotubes and related nanomaterials: critical advances and challenges for synthesis toward mainstream commercial applications. ACS Nano 12, 11756–11784 (2018).
Zurutuza, A. & Marinelli, C. Challenges and opportunities in graphene commercialization. Nat. Nanotechnol. 9, 730–734 (2014).
Zhang, R., An, D., Zhu, J., Lu, X. & Liu, Y. Carbon nanorings and nanobelts: material syntheses, molecular architectures, and applications. Adv. Funct. Mater. 33, 2305249 (2023).
Li, M., Chen, J., Gooding, J. J. & Liu, J. Review of carbon and graphene quantum dots for sensing. ACS Sens 4, 1732–1748 (2019).
Wareing, T. C., Gentile, P. & Phan, A. N. Biomass-based carbon dots: current development and future perspectives. ACS Nano 15, 15471−15501 (2021).
Stanley, H. E. Introduction to phase transitions and critical phenomena (Oxford University Press, 1987).
Dias, A. L. B., de Aguiar, A. N. & Rostagno, M. A. Extraction of natural products using supercritical fluids and pressurized liquids assisted by ultrasound: Current status and trends. Ultrason. Sonochem. 74, 105584 (2021).
Prajapati, D. & Gohain, M. Recent advances in the application of supercritical fluids for carbon–carbon bond formation in organic synthesis. Tetrahedron 60, 815–833 (2004).
Pang, Y. X. et al. Application of supercritical fluid in the synthesis of graphene materials: a review. J. Nanopart. Res. 23, 204 (2021).
Pang, Y. X. et al. The synthesis of carbon-based quantum dots: a supercritical fluid approach and perspective. Mater. Today Phys. 27, 100752 (2022).
Groshkova, Y. A., Buslaeva, E. Y. & Gubin, S. P. Transformation of graphene oxide in supercritical media. Russ. Chem. Bull. 68, 2028–2032 (2019).
Chen, Z., Yao, Y., Yuan, S. & Yin, H. Measurement of critical temperatures, critical pressures and densities of acetone-water solutions for simulation. J. Solut. Chem. 52, 1331–1351 (2023).
Kaur, A., Kaur, J. & Singh, R. C. Tailor made exfoliated reduced graphene oxide nanosheets based on oxidative-exfoliation approach. Fuller. Nanotub. Carbon Nanostruct. 26, 1–11 (2018).
Lim, D. J., Marks, N. A. & Rowles, M. R. Universal Scherrer equation for graphene fragments. Carbon 162, 475e480 (2020).
Moon, I. K., Lee, J., Ruoff, R. S. & Lee, H. Reduced graphene oxide by chemical graphitization. Nat. Commun. 1, 73 (2010).
Johra, F. T., Lee, J. W. & Jung, W. G. Facile and safe graphene preparation on solution based platform. J. Ind. Eng. Chem. 20, 2883–2887 (2014).
Wu, J. B., Lin, M. L., Cong, X., Liua, H. N. & Tan, P. H. Raman spectroscopy of graphene-based materials and its applications in related devices. Chem. Soc. Rev. 47, 1822 (2018).
Cançado, L. G. et al. Quantifying defects in graphene via Raman spectroscopy at different excitation energies. Nano Lett. 11, 3190–3196 (2011).
Panda, A. et al. Chemical-free sustainable carbon nano-onion as a dual-mode sensor platform for noxious volatile organic compounds. Appl. Surf. Sci. 537, 147872 (2021).
Tange, K., Nomura, S. & Mukasa, S. Formation of two kinds of carbon with different properties by acetone decomposition using in-liquid plasma method. Int. J. Hydrog. Energy 44, 23912–23920 (2019).
Price, R. J., Ladislaus, P. I., Smith, G. C. & Davies, T. J. A novel ‘bottom-up’ synthesis of few- and multi-layer graphene platelets with partial oxidation via cavitation. Ultrason. Sonochem. 56, 466–473 (2019).
Guo, A. et al. Soft-chemistry synthesis, solubility and interlayer spacing of carbon nano-onions. RSC Adv. 11, 6850 (2021).
Chattopadhyay, P. & Gupta, R. B. Supercritical CO2-based production of fullerene nanoparticles. Ind. Eng. Chem. Res. 39, 2281–2289 (2000).
Fukuda, T., Ishii, K., Kurosu, S., Whitby, R. & Maekawa, T. Formation of clusters composed of C60 molecules via self-assembly in critical fluids. Nanotechnology 18, 145611 (2007).
Fukuda, T. et al. Low temperature synthesis of carbon fibres and metal-filling carbon nanoparticles with laser irradiation into near-critical benzene. RSC Adv. 5, 12671–12677 (2015).
Acknowledgements
The present study was carried out as part of the “Bio-Nano Innovation Programme: Development of advanced functional nanomaterials and devices, and their application to biotechnology” organised by the Bio-Nano Electronics Research Centre, Toyo University. We would like to thank Toyo University for raising a fund for the above programme since April 2021. We would like to thank Mr. Naoya Ochi, Technical Manager of the Mechanical Factory, Toyo University, for his technical support for the design and manufacturing of supercritical fluid chambers. Yuri Tanuma acknowledges the financial support programme “Overseas Research Fellowships Scheme” organised by the Japanese Society for the Promotion of Science since April 2023.
Author information
Authors and Affiliations
Contributions
S.K. performed experiments, characterisations, and data analyses, and drew graphs; Y.K. performed experiments, characterisations and data analyses, and drew graphs; K.Z. performed experiments and characterisations; H.Y. performed experiments and characterisations; R.H. performed experiments and characterisations; K.Y. performed SEM and TEM observations and data analyses; H.S. performed TEM observations and data analyses, Y.T. performed data analyses, H.M. performed data analyses, and T.M. organised the present research project, raised the fund, performed data analyses and wrote the manuscript. All the authors checked the manuscript, images, and graphs and agreed with the contents of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Chemistry thanks Ryan Schultz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kurosu, S., Kaizuka, Y., Zhou, K. et al. Precursor-free synthesis of carbon quantum dots and carbon microparticles in supercritical acetone. Commun Chem 7, 283 (2024). https://doi.org/10.1038/s42004-024-01367-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s42004-024-01367-0