Abstract
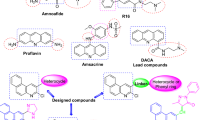
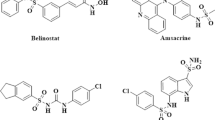
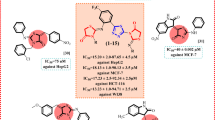
The tetrahydroisoquinoline scaffold is an important structural motif in many natural products and pharmaceuticals, known for its broad biological activities. Fused tetrahydroisoquinoline polycyclic heterocyclic structures have increasingly attracted attention, yet their synthetic pathways remain limited. Herein, we developed an efficient, metal-free strategy for assembling tetrahydroisoquinoline-fused polycycles via Vilsmeier-reagent promoted decarbonylative cyclization reaction. The protocol features mild conditions, excellent functional-group tolerance, and high efficiency. Furthermore, biological evaluations demonstrated that the constructed derivatives exhibit antiproliferative activity in cancer cell lines, thereby providing potential starting points for further optimization.

Similar content being viewed by others
Data availability
Detailed experimental procedures and characterizations of new compounds are available in Supplementary Information and Supplementary Data 1–4. The X-ray crystallographic coordinates for structures reported in this Article have been provided as Supplementary Data 1 and 2 and have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers CCDC 2446305 (2a) and 2446312 (4r). These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via http://www.ccdc.cam.ac.uk/data_request/cif.
References
Pyne, M. E. et al. A yeast platform for high-level synthesis of tetrahydroisoquinoline alkaloids. Nat. Commun. 11, 3337–3346 (2020).
Qin, Z., Tang, R., Liang, J. & Jia, X. Berberine, a natural alkaloid: advances in its pharmacological effects and mechanisms in the treatment of autoimmune diseases. Int. Immunopharmacol. 137, 112422–112430 (2024).
Coscia, C. J. et al. Occurrence of a new class of tetrahydroisoquinoline alkaloids in L-DOPA-treated Parkinsonian patients. Nature. 269, 617–619 (1977).
Wen, W. H. et al. Reductive inactivation of the hemiaminal pharmacophore for resistance against tetrahydroisoquinoline antibiotics. Nat. Commun. 12, 7085–7095 (2021).
Faheem et al. 1,2,3,4-tetrahydroisoquinoline (THIQ) as privileged scaffold for anticancer de novo drug design. Expert Opin. Drug Discov. 16, 1119–1147 (2021).
Gao, Y. et al. Progress in the total synthesis of antitumor tetrahydroisoquinoline alkaloids. Chem. Biodivers. 20, 172–186 (2023).
Saito, N. Chemical research on antitumor isoquinoline marine natural products and related compounds. Chem. Pharm. Bull. 69, 155–177 (2021).
Luo, G. et al. Development of novel tetrahydroisoquinoline-hydroxamate conjugates as potent dual SERDs/HDAC inhibitors for the treatment of breast cancer. Eur. J. Med. Chem. 226, 113870–113884 (2021).
Morishita, K. et al. 2-Acyl-3-carboxyl-tetrahydroisoquinoline derivatives: mixed-type PTP1B inhibitors without PPARγ activation. Chem. Pharm. Bull. 66, 1131–1152 (2018).
Klopfenstein, S. R. et al. 1,2,3,4-Tetrahydroisoquinolinyl sulfamic acids as phosphatase PTP1B inhibitors. Bioorg. Med. Chem. Lett. 16, 1574–1578 (2006).
Kubo, M. et al. Insulin sensitization by a novel partial peroxisome proliferator-activated receptor γ agonist with protein tyrosine phosphatase 1B inhibitory activity in experimental osteoporotic rats. J. Pharmacol. Sci. 124, 276–285 (2014).
Otake, K. et al. Novel (S)-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acids: peroxisome proliferator-activated receptor γ selective agonists with protein-tyrosine phosphatase 1B inhibition. Bioorg. Med. Chem. 20, 1060–1075 (2012).
Qiu, G. & Wu, J. Generation of N-heterocycles via tandem reactions of N‘-(2-Alkynylbenzylidene)hydrazides. Chem. Rec. 16, 19–34 (2016).
Cheng, P. et al. 1-Aryl-tetrahydroisoquinoline analogs as active anti-HIV agents in vitro. Bioorg. Med. Chem. Lett. 18, 2475–2478 (2008).
Kashiwada, Y. et al. Anti-HIV benzylisoquinoline alkaloids and flavonoids from the leaves of Nelumbo nucifera, and structure-activity correlations with related alkaloids. Bioorg. Med. Chem. 13, 443–448 (2005).
Chander, S., Ashok, P., Singh, A. & Murugesan, S. De-novo design, synthesis and evaluation of novel 6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline derivatives as HIV-1 reverse transcriptase inhibitors. Chem. Cent. J. 9, 33–45 (2015).
Iwasa, K. et al. Simple isoquinoline and benzylisoquinoline alkaloids as potential antimicrobial, antimalarial, cytotoxic, and anti-HIV agents. Bioorg. Med. Chem. 9, 2871–2884 (2001).
Wang, X. et al. Novel tetrahydroisoquinoline-based heterocyclic compounds efficiently inhibit SARS-CoV-2 infection in vitro. Viruses 15, 502–514 (2023).
Suzuki, K. et al. Tetrazomine, a new antibiotic produced by an actinomycete strain. Taxonomy, fermentation, isolation and characterization. J. Antibiot. 44, 479–485 (1991).
Wipf, P. & Hopkins, C. R. Enantioselective synthesis of the AB-ring system of the antitumor antibiotic tetrazomine. J. Org. Chem. 66, 3133–3139 (2001).
Olianas, M. C. & Onali, P. PD 102807, a novel muscarinic M4 receptor antagonist, discriminates between striatal and cortical muscarinic receptors coupled to cyclic AMP. Life Sci. 65, 2233–2240 (1999).
Tompkins, E. et al. PD 102807 induces M3 mAChR-dependent GRK-/arrestin-biased signaling in airway smooth muscle cells. Am. J. Respir. Cell Mol. Biol. 67, 550–561 (2022).
Böhme, T. M. et al. Synthesis and pharmacology of benzoxazines as highly selective antagonists at M(4) muscarinic receptors. J. Med. Chem. 45, 3094–3102 (2002).
Trivedi, G. et al. Muscarinic acetylcholine receptor regulates self-renewal of early erythroid progenitors. Sci. Transl. Med. 11, eaaw3781 (2019).
Kitaichi, K., Day, J. C. & Quirion, R. A novel muscarinic M(4) receptor antagonist provides further evidence of an autoreceptor role for the muscarinic M(2) receptor sub-type. Eur. J. Pharmacol. 383, 53–56 (1999).
Valuskova, P. et al. Autoradiography of (3)H-pirenzepine and (3)H-AFDX-384 in mouse brain regions: possible insights into M(1), M(2), and M(4) muscarinic receptors distribution. Front. Pharmacol. 9, 124 (2018).
Zhou, J. et al. Identification and biological evaluation of fused tetrahydroisoquinoline derivatives as Wnt/β-catenin signaling inhibitors to suppress colorectal cancer. Eur. J. Med. Chem. 276, 116664 (2024).
Katz, R. J. et al. Serotonergic (5-HT2) mediation of anxiety-therapeutic effects of serazepine in generalized anxiety disorder. Biol. Psychiatry 34, 41–44 (1993).
Li, K. et al. Natural staurosporine derivatives with fluorescence for cancer theranostics. ChemMedChem 20, e202500629 (2025).
Xu, L. et al. Cytotoxic and antifungal staurosporine derivatives from marine-derived actinomycete Streptomyces sp. ZS-A121. Chem. Biodivers. 21, e202301712 (2024).
Lai, R. et al. Novel berberine derivatives as p300 histone acetyltransferase inhibitors in combination treatment for breast cancer. Eur. J. Med. Chem. 266, 116116 (2024).
Li, J. et al. A multicomponent reaction for modular assembly of indole-fused heterocycles. Chem. Sci. 15, 5211–5217 (2024).
Bhunia, A., Porwal, D., Gonnade, R. G. & Biju, A. T. Multicomponent reactions involving arynes, quinolines, and aldehydes. Org. Lett. 15, 4620–4623 (2013).
Gao, Y. et al. Elucidation and biosynthesis of tetrahydroisoquinoline alkaloids: Recent advances and prospects. Biotechnol. Adv. 79, 108524–108544 (2025).
Scott, J. D. & Williams, R. M. Total synthesis of (-)-tetrazomine. Determination of the stereochemistry of tetrazomine and the synthesis and biological activity of tetrazomine analogues. J. Am. Chem. Soc. 124, 2951–2956 (2002).
Jiang, B. et al. ‘One-pot’ synthesis of dihydrobenzo[4,5][1,3]oxazino[2,3-a] isoquinolines via a silver(I)-catalyzed cascade approach. Molecules 18, 814–831 (2013).
Deb, M. L., Pegu, C. D., Borpatra, P. J. & Baruah, P. K. Metal-free intramolecular α-sp3 C–H oxygenation of tert-amine: an efficient approach to 1,3-oxazines. Tetrahedron Lett. 57, 5479–5483 (2016).
Borpatra, P. J., Deb, M. L. & Baruah, P. K. Copper-catalyzed tandem multi-component approach to 1,3-oxazines at room temperature by cross-dehydrogenative coupling using methanol as C1 feedstock. Synlett. 29, 1171–1175 (2018).
Yang, R., Chen, Q., Sheng, W. & Zhang, K. Oxazine ring-containing polycyclic monomers: a class of benzoxazine thermosetting resins with intrinsically low curing temperature. Macromolecules. 57, 5608–5619 (2024).
Szatmári, I. et al. Unexpected isomerization of new naphth[1,3]oxazino[2,3-a]isoquinolines in solution, studied by dynamic NMR and supported by theoretical DFT computations. Tetrahedron 69, 7455–7465 (2013).
Saikia, B. S., Deb, M. L. & Baruah, P. K. Green synthesis of 1,3-oxazines by visible light-promoted catalyst-free C–H activation/cyclization of tertiary amines. Environ. Chem. Lett. 20, 109–118 (2022).
Richers, M. T. et al. Redox-neutral α-oxygenation of amines: reaction development and elucidation of the mechanism. J. Am. Chem. Soc. 136, 6123–6135 (2014).
Deb, M. L. et al. Catalyst-free multi-component cascade C–H-functionalization in water using molecular oxygen: an approach to 1,3-oxazines. Green. Chem. 19, 4036–4042 (2017).
Deb, M. L. et al. Copper-catalyzed regioselective intramolecular oxidative α-functionalization of tertiary amines: an efficient synthesis of dihydro-1,3-oxazines. Angew. Chem. Int. Ed. 52, 9791–9795 (2013).
Lu, H., Yu, T. Y., Xu, P. F. & Wei, H. Selective decarbonylation via transition-metal-catalyzed carbon-carbon bond cleavage. Chem. Rev. 121, 365–411 (2021).
Bhuyan, K., Borang, L. & Jamatia, R. Visible light-mediated radical decarboxylative strategies for the synthesis of heterocyclic compounds. Asian J. Org. Chem. 14, e202400493 (2024).
Lian, F., Tan, Z. & Xu, K. Electrochemical decarboxylative functionalizations through carbocation intermediates. Adv. Synth. Catal. 367, e70137 (2025).
Zeng, R. & Dong, G. Rh-catalyzed decarbonylative coupling with alkynes via C-C activation of isatins. J. Am. Chem. Soc. 137, 1408–1411 (2015).
Zhang, X. et al. Visible-light-induced decarboxylative cyclization of 2-alkenylarylisocyanides with α-oxocarboxylic acids: access to 2-acylindoles. J. Org. Chem. 85, 9503–9513 (2020).
Guo, D. et al. Design, synthesis, and biological evaluation of novel tetrahydroprotoberberine derivatives (THPBs) as selective α(1A)-adrenoceptor antagonists. J. Med. Chem. 59, 9489–9502 (2016).
Zhou, S. et al. Design, synthesis and biological evaluation of 4,7,12,12a-tetrahydro-5H-thieno[3’,2’:3,4]pyrido[1,2-b]isoquinolines as novel adenosine 5’-monophosphate-activated protein kinase (AMPK) indirect activators for the treatment of type 2 diabetes. Eur. J. Med. Chem. 140, 448–464 (2017).
Gu, Z. et al. Design, synthesis, and structure-activity relationships of novel 4,7,12,12a-tetrahydro-5H-thieno[3′,2′:3,4]pyrido[1,2-b]isoquinoline and 5,8,12,12a-tetrahydro-6H-thieno[2′,3′:4,5]pyrido[2,1-a]isoquinoline derivatives as cellular activators of adenosine 5′-monophosphate-activated protein kinase (AMPK). Bioorg. Med. Chem. 26, 2017–2027 (2018).
Wu, C. et al. Design, synthesis, and biological evaluation of novel tetrahydroprotoberberine derivatives (THPBs) as proprotein convertase subtilisin/kexin type 9 (PCSK9) modulators for the treatment of hyperlipidemia. Acta Pharm. Sin. B 9, 1216–1230 (2019).
Zhang, R. et al. Design, synthesis, and biological evaluation of tetrahydroisoquinoline derivatives as novel, selective PDE4 inhibitors for antipsoriasis treatment. Eur. J. Med. Chem. 211, 113004 (2021).
Ding, S. & Jiao, N. N. N-dimethylformamide: a multipurpose building block. Angew. Chem., Int. Ed. 51, 9226–9237 (2012).
Le Bras, J. & Muzart, J. Recent uses of N,N-dimethylformamide and N,N-dimethylacetamide as reagents. Molecules 23, 1939 (2018).
Chevrier, Q. et al. Synthesis of N-sulfonyl formamidines by direct condensation between sulfonamide and formamide enabled by a photogenerated Vilsmeier-type reagent. J. Org. Chem. 89, 15282–15288 (2024).
Zhang, Z. et al. Synthesis of 4-ynamides and cyclization by the Vilsmeier reagent to dihydrofuran-2(3H)-ones. Org. Lett. 17, 6190–6193 (2015).
Liang, F. et al. Photo-on-demand synthesis of Vilsmeier reagents with chloroform and their applications to one-pot organic syntheses. J. Org. Chem. 86, 6504–6517 (2021).
Thota, N. et al. Reaction of carbohydrates with Vilsmeier reagent: a tandem selective chloro O-formylation of sugars. Org. Biomol. Chem. 7, 1280–1283 (2009).
Srivastava, V., Negi, A. S., Kumar, J. K. & Gupta, M. M. A simple, convenient, and chemoselective formylation of sterols by Vilsmeier reagent. Steroids 71, 632–638 (2006).
Chung, C. Y. et al. Structural identification between phthalazine-1,4-diones and N-aminophthalimides via vilsmeier reaction: nitrogen cyclization and tautomerization study. Molecules 26, 2907 (2021).
Li, J. et al. Facile and efficient access to Androsten-17-(1’,3’,4’)-pyrazoles and Androst-17β-(1’,3’,4’)-pyrazoles via Vilsmeier reagents, and their antiproliferative activity evaluation in vitro. Eur. J. Med. Chem. 130, 1–14 (2017).
Wang, M., Han, F., Yuan, H. & Liu, Q. Tandem Nazarov cyclization-halovinylation of divinyl ketones under Vilsmeier conditions: synthesis of highly substituted cyclopentadienes. Chem. Commun. 46, 2247–2249 (2010).
Yan, X., Zheng, J. & Li, W.-D. Z. Concise total syntheses of berberine and its analogues enabled by trifluoroacetic anhydride-promoted decarbonylative-elimination reaction. Tetrahedron Lett. 132, 154826–154830 (2023).
Koskinen, A. M. & Rapoport, H. Synthetic and conformational studies on anatoxin-a: a potent acetylcholine agonist. J. Med. Chem. 28, 1301–1309 (1985).
Weinstein, B. & Craig, A. R. A synthetic approach to the cephalotaxine skeleton. J. Org. Chem. 41, 875–878 (1975).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (Nos. 22377138 and 22337003).
Author information
Authors and Affiliations
Contributions
Conceptualization: Jian Li, Kaixian Chen, Jing Li, and Hong Liu; methodology: Mengdi Yan, Umit Mukatay, Hui Shen, Jiming Sun, and Jina Sun; investigation: Mengdi Yan, Umit Mukatay, Hui Shen, Jiming Sun, and Ruohan Zhang; writing—original draft: Mengdi Yan and Jian Li; writing—review and editing: Kaixian Chen, Jian Li, and Hong Liu; funding acquisition: Kaixian Chen and Hong Liu; resources: Jina Sun, Jian Li, and Hong Liu; supervision: Jian Li and Hong Liu.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Chemistry thanks Suven Das, Sandip Murarka and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yan, M., Mukatay, U., Shen, H. et al. Synthesis of tetrahydroisoquinoline-fused polycyclic heterocyclic skeletons via Vilsmeier-reagent promoted decarbonylative annulation. Commun Chem (2026). https://doi.org/10.1038/s42004-026-01982-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-026-01982-z