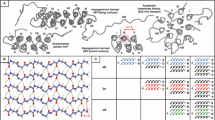
Abstract
Collagen’s structural integrity and biological function derive from its characteristic triple helix, governed by a unique amino acid sequence containing the rare residue (2S,4R)-hydroxyproline (4R-Hyp). Substituting 4R-Hyp with its diastereomer 4S-Hyp markedly reduces triple-helix thermal stability and impairs collagen function. The origins of this destabilization have mostly been ascribed to stereoelectronic effects such as the gauche effect and n → π* interactions. Here, we dissect the role of different stabilizing interactions on molecular conformation using linear infrared and two-dimensional infrared spectroscopy, aided by density functional theory calculations. We investigate the structure of N-Boc-(2S,4S)-4-hydroxyproline-methyl ester (Boc-4S-Hyp-OMe) and N-Boc-(2S,4R)-4-hydroxyproline-methyl ester (Boc-4R-Hyp-OMe) in chloroform solution, a simplified model for the water-deficient environment in collagen tissues where triple helices assemble into fibrils. We find that, while stereoelectronic effects indeed influence molecular conformation, n → π* interactions only moderately alter conformational equilibria. Conversely, our results show that hydrogen bonding is pivotal: the conformation of Boc-4S-Hyp-OMe is stabilized by an intramolecular hydrogen bond, whereas Boc-4R-Hyp-OMe primarily forms intermolecular hydrogen bonds, leading to a greater intermolecular affinity. These findings highlight hydrogen bonding as a key determinant of hydroxyproline conformation at the single-residue level.

Similar content being viewed by others
Data availability
All raw data generated and analyzed throughout this work are available from the corresponding authors upon reasonable request. The source data for Figs. 3, 4, and 5 are provided as Supplementary Data 1. Coordinate files of the DFT optimized geometries are provided as Supplementary Data 2. Additional DFT results using explicit solvent molecules, calculated absorption coefficients, fits of the linear IR spectra, and 2D-IR spectra with parallel polarization combinations are available in the Supplementary Material file attached to this paper.
References
Shoulders, M. D. & Raines, R. T. Collagen structure and stability. Annu. Rev. Biochem. 78, 929–958 (2009).
Li, X., Zhang, Q., Yu, S. M. & Li, Y. The chemistry and biology of collagen hybridization. J. Am. Chem. Soc. 145, 10901–10916 (2023).
Ramshaw, J. A., Shah, N. K. & Brodsky, B. Gly-xy tripeptide frequencies in collagen: a context for host–guest triple-helical peptides. J. Struct. Biol. 122, 86–91 (1998).
Kessler, J. L. et al. Peptoid residues make diverse, hyperstable collagen triple-helices. J. Am. Chem. Soc. 143, 10910–10919 (2021).
Fratzl, P. Collagen: Structure and mechanics, an introduction (Springer US, 2008).
Peterkofsky, B. Ascorbate requirement for hydroxylation and secretion of procollagen: relationship to inhibition of collagen synthesis in scurvy. Am. J. Clin. Nutr. 54, 1135S–1140S (1991).
Myllyharju, J. & Kivirikko, K. I. Collagens, modifying enzymes and their mutations in humans, flies and worms. Trends Genet. 20, 33–43 (2004).
Beck, K. et al. Destabilization of osteogenesis imperfecta collagen-like model peptides correlates with the identity of the residue replacing glycine. Proc. Nat. Acad. Sci. 97, 4273–4278 (2000).
Burjanadze, T. V. Hydroxyproline content and location in relation to collagen thermal stability. Biopolymers 18, 931–938 (1979).
Holmgren, S. K., Bretscher, L. E., Taylor, K. M. & Raines, R. T. A hyperstable collagen mimic. Chem. Biol. 6, 63–70 (1999).
Inouye, K., Sakakibara, S. & Prockop, D. J. Effects of the stereo-configuration of the hydroxyl group in 4-hydroxyproline on the triple-helical structures formed by homogeneous peptides resembling collagen. Biochim. Biophys. Acta 420, 133–141 (1976).
Vitagliano, L., Berisio, R., Mazzarella, L. & Zagari, A. Structural bases of collagen stabilization induced by proline hydroxylation. Biopolymers 58, 459–464 (2001).
Jiravanichanun, N. et al. Unexpected puckering of hydroxyproline in the guest triplets, hyp-pro-gly and pro-allohyp-gly sandwiched between pro-pro-gly sequence. ChemBioChem 6, 1184–1187 (2005).
Jiravanichanun, N., Nishino, N. & Okuyama, K. Conformation of allohyp in the y position in the host–guest peptide with the pro–pro–gly sequence: Implication of the destabilization of (pro–allohyp–gly) 10. Biopolymers 81, 225–233 (2006).
Bella, J., Eaton, M., Brodsky, B. & Berman, H. M. Crystal and molecular structure of a collagen-like peptide at 1.9 å resolution. Science 266, 75–81 (1994).
Nishi, Y. et al. Different effects of 4-hydroxyproline and 4-fluoroproline on the stability of collagen triple helix. Biochem 44, 6034–6042 (2005).
Shoulders, M. D., Kotch, F. W., Choudhary, A., Guzei, I. A. & Raines, R. T. The aberrance of the 4s diastereomer of 4-hydroxyproline. J. Am. Chem. Soc. 132, 10857–10865 (2010).
Holmgren, S. K., Taylor, K. M., Bretscher, L. E. & Raines, R. T. Code for collagen’s stability deciphered. Nature 392, 666–667 (1998).
Bretscher, L. E., Jenkins, C. L., Taylor, K. M., DeRider, M. L. & Raines, R. T. Conformational stability of collagen relies on a stereoelectronic effect. J. Am. Chem. Soc. 123, 777–778 (2001).
Shoulders, M. D., Satyshur, K. A., Forest, K. T. & Raines, R. T. Stereoelectronic and steric effects in side chains preorganize a protein main chain. Proc. Nat. Acad. Sci. 107, 559–564 (2010).
Hulgan, S. A. & Hartgerink, J. D. Recent advances in collagen mimetic peptide structure and design. Biomacromolecules 23, 1475–1489 (2022).
DeRider, M. L. et al. Collagen stability: Insights from nmr spectroscopic and hybrid density functional computational investigations of the effect of electronegative substituents on prolyl ring conformations. J. Am. Chem. Soc. 124, 2497–2505 (2002).
Gerig, J. & McLeod, R. Conformations of cis-and trans-4-fluoro-yl-proline in aqueous solution. J. Am. Chem. Soc. 95, 5725–5729 (1973).
Renner, C. et al. Fluoroprolines as tools for protein design and engineering. Angew. Chem., Int. Ed. 40, 923–925 (2001).
Newberry, R. W. & Raines, R. T. The n → π* interaction. Acc. Chem. Res. 50, 1838–1846 (2017).
Bartlett, G. J., Newberry, R. W., VanVeller, B., Raines, R. T. & Woolfson, D. N. Interplay of hydrogen bonds and n → π* interactions in proteins. J. Am. Chem. Soc. 135, 18682–18688 (2013).
Singh, S. K., Mishra, K. K., Sharma, N. & Das, A. Direct spectroscopic evidence for an n → π* interaction. Angew. Chem., Int. Ed. 55, 7801–7805 (2016).
León, I., Alonso, E., Cabezas, C., Mata, S. & Alonso, J. Unveiling the n → π* interactions in dipeptides. Commun. Chem. 2, 3 (2019).
Newberry, R. W., VanVeller, B., Guzei, I. A. & Raines, R. T. n → π* interactions of amides and thioamides: implications for protein stability. J. Am. Chem. Soc. 135, 7843–7846 (2013).
Deb, P. et al. Interconverting hydrogen-bonding and weak n → π* interactions in aqueous solution: A direct spectroscopic evidence. J. Phys. Chem. Lett. 9, 5425–5429 (2018).
Kuemin, M. et al. Tuning the cis/trans conformer ratio of xaa–pro amide bonds by intramolecular hydrogen bonds: The effect on ppii helix stability. Angew. Chem., Int. Ed. 122, 6468–6471 (2010).
Haris, P. I. & Chapman, D. The conformational analysis of peptides using fourier transform ir spectroscopy. Biopolymers 37, 251–263 (1995).
Woutersen, S. et al. Peptide conformational heterogeneity revealed from nonlinear vibrational spectroscopy and molecular-dynamics simulations. J. Chem. Phys. 117, 6833–6840 (2002).
Ham, S., Kim, J.-H., Lee, H. & Cho, M. Correlation between electronic and molecular structure distortions and vibrational properties. ii. amide i modes of nma–nd2o complexes. J. Chem. Phys. 118, 3491–3498 (2003).
Woutersen, S. & Hamm, P. Structure determination of trialanine in water using polarization sensitive two-dimensional vibrational spectroscopy. J. Phys. Chem. B 104, 11316–11320 (2000).
Zheng, J. et al. Ultrafast dynamics of solute-solvent complexation observed at thermal equilibrium in real time. Science 309, 1338–1343 (2005).
Kwak, K., Rosenfeld, D. E., Chung, J. K. & Fayer, M. D. Solute- solvent complex switching dynamics of chloroform between acetone and dimethylsulfoxide- two-dimensional ir chemical exchange spectroscopy. J. Phys. Chem. B 112, 13906–13915 (2008).
DeCamp, M. et al. Amide i vibrational dynamics of n-methylacetamide in polar solvents: The role of electrostatic interactions. J. Phys. Chem. B 109, 11016–11026 (2005).
Herschlag, D. & Pinney, M. M. Hydrogen bonds: simple after all? Biochem 57, 3338–3352 (2018).
Improta, R., Benzi, C. & Barone, V. Understanding the role of stereoelectronic effects in determining collagen stability. 1. a quantum mechanical study of proline, hydroxyproline, and fluoroproline dipeptide analogues in aqueous solution. J. Am. Chem. Soc. 123, 12568–12577 (2001).
Hamm, P. & Zanni, M. Concepts and methods of 2D infrared spectroscopy (Cambridge University Press, 2011).
Hamm, P., Lim, M., DeGrado, W. F. & Hochstrasser, R. M. The two-dimensional ir nonlinear spectroscopy of a cyclic penta-peptide in relation to its three-dimensional structure. Proc. Natl. Acad. Sci. 96, 2036–2041 (1999).
Giubertoni, G., Sofronov, O. O. & Bakker, H. J. Observation of distinct carboxylic acid conformers in aqueous solution. J. Phys. Chem. Lett. 10, 3217–3222 (2019).
Golonzka, O. & Tokmakoff, A. Polarization-selective third-order spectroscopy of coupled vibronic states. J. Chem. Phys. 115, 297–309 (2001).
Buck, U. & Huisken, F. Infrared spectroscopy of size-selected water and methanol clusters. Chem. Rev. 100, 3863–3890 (2000).
Khare, E. et al. Discovering design principles of collagen molecular stability using a genetic algorithm, deep learning, and experimental validation. Proc. Nat. Acad. Sci. 119, e2209524119 (2022).
Neese, F. The orca program system. WIRES Comput. Mol. Sci. 2, 73–78 (2012).
Pracht, P., Bohle, F. & Grimme, S. Automated exploration of the low-energy chemical space with fast quantum chemical methods. Phys. Chem. Chem. Phys. 22, 7169–7192 (2020).
Bannwarth, C., Ehlert, S. & Grimme, S. Gfn2-xtb–an accurate and broadly parametrized self-consistent tight-binding quantum chemical method with multipole electrostatics and density-dependent dispersion contributions. J. Chem. Theory Comput. 15, 1652–1671 (2019).
Zhao, Y. & Truhlar, D. G. The m06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four m06-class functionals and 12 other functionals. Theor. Chem. Acc. 120, 215–241 (2008).
Dunning Jr, T. H. Gaussian basis sets for use in correlated molecular calculations. i. the atoms boron through neon and hydrogen. J. Chem. Phys. 90, 1007–1023 (1989).
Barone, V. & Cossi, M. Quantum calculation of molecular energies and energy gradients in solution by a conductor solvent model. J. Phys. Chem. A 102, 1995–2001 (1998).
Acknowledgements
We acknowledge funding by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)—SFB1551—Project No. 464588647. G. Giubertoni acknowledges that this publication is part of the project (with Project Number VI.Veni.212.240) of the research program NWO Talent Program Veni 2021, which is financed by the Dutch Research Council (NWO).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
F.M.: Conceptualization, Investigation, Formal Analysis, Writing—Original Draft Preparation, Writing—Review & Editing. P.G.A.: Formal Analysis, Writing—Review & Editing. M.B.: Conceptualization, Supervision, Writing—Review & Editing. G.G.: Conceptualization, Formal Analysis, Supervision, Writing—Review & Editing. J.H.: Conceptualization, Formal Analysis, Supervision, Writing—Review & Editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Chemistry thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Matsumura, F., Gómez Argudo, P., Bonn, M. et al. Stereoelectronic and hydrogen-bonding effects on hydroxyproline conformation. Commun Chem (2026). https://doi.org/10.1038/s42004-026-01984-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-026-01984-x