Abstract
While Roux-en-Y gastric bypass is an effective treatment for obesity and type 2 diabetes, up to one-third of patients develop post-bariatric hypoglycaemia (PBH). Individuals with PBH exhibit increased postprandial secretion of the intestinal hormone fibroblast growth factor 19 (FGF19, Fgf15 in mice). However, the underlying mechanisms contributing to PBH remain uncertain. Here we demonstrate that faecal and plasma bile acid (BA) profiles are significantly altered in postoperative individuals with PBH versus those without hypoglycaemia. Furthermore, altered BAs in PBH induce FGF19 secretion in intestinal cells in a manner dependent on the apical sodium-dependent BA transporter (ASBT). We demonstrate that ASBT inhibition reduces Fgf15 expression and increases postprandial glucose in hypoglycaemic mice. Our data suggest that dysregulation of luminal BA profiles and transport may contribute to PBH and provide proof of concept that ASBT inhibition could be developed as a new therapeutic strategy for PBH.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this article and its Extended Data and Supplementary Information. Requests for additional information can be made to the corresponding authors. Source data are provided with this paper.
Code availability
No custom code or mathematical algorithms were used in this study.
References
Courcoulas, A. P. et al. Long-term outcomes of medical management vs bariatric surgery in type 2 diabetes. JAMA 331, 654–664 (2024).
Sandoval, D. A. & Patti, M. E. Glucose metabolism after bariatric surgery: implications for T2DM remission and hypoglycaemia. Nat. Rev. Endocrinol. 19, 164–176 (2023).
Flynn, C. R. et al. Bile diversion to the distal small intestine has comparable metabolic benefits to bariatric surgery. Nat. Commun. 6, 7715 (2015).
Patti, M. E. et al. Serum bile acids are higher in humans with prior gastric bypass: potential contribution to improved glucose and lipid metabolism. Obesity 17, 1671–1677 (2009).
Ryan, K. K. et al. FXR is a molecular target for the effects of vertical sleeve gastrectomy. Nature 509, 183–188 (2014).
Cummings, C. et al. Continuous glucose monitoring in patients with post-bariatric hypoglycaemia reduces hypoglycaemia and glycaemic variability. Diabetes Obes. Metab. 25, 2191–2202 (2023).
Ostrovsky, V. et al. Persistent post-bariatric-surgery hypoglycemia: a long-term follow-up reassessment. Nutr. Metab. Cardiovasc. Dis. 33, 1197–1205 (2023).
Davis, D. B. et al. Roux en Y gastric bypass hypoglycemia resolves with gastric feeding or reversal: confirming a non-pancreatic etiology. Mol. Metab. 9, 15–27 (2018).
Bozadjieva, N., Heppner, K. M. & Seeley, R. J. Targeting FXR and FGF19 to treat metabolic diseases-lessons learned from bariatric surgery. Diabetes 67, 1720–1728 (2018).
Mulla, C. M. et al. Plasma FGF-19 levels are increased in patients with post-bariatric hypoglycemia. Obes. Surg. 29, 2092–2099 (2019).
van den Broek, M. et al. Altered bile acid kinetics contribute to postprandial hypoglycaemia after Roux-en-Y gastric bypass surgery. Int. J. Obes. 45, 619–630 (2021).
Kir, S. et al. FGF19 as a postprandial, insulin-independent activator of hepatic protein and glycogen synthesis. Science 331, 1621–1624 (2011).
DePaoli, A. M. et al. FGF19 analog as a surgical factor mimetic that contributes to metabolic effects beyond glucose homeostasis. Diabetes 68, 1315–1328 (2019).
Shin, D. J. & Osborne, T. F. FGF15/FGFR4 integrates growth factor signaling with hepatic bile acid metabolism and insulin action. J. Biol. Chem. 284, 11110–11120 (2009).
Picard, A. et al. A genetic screen identifies hypothalamic Fgf15 as a regulator of glucagon secretion. Cell Rep. 17, 1795–1806 (2016).
Moron-Ros, S. et al. FGF15/19 is required for adipose tissue plasticity in response to thermogenic adaptations. Mol. Metab. 43, 101113 (2021).
Roesch, S. L. et al. Perturbations of fibroblast growth factors 19 and 21 in type 2 diabetes. PLoS ONE 10, e0116928 (2015).
Gerhard, G. S. et al. A role for fibroblast growth factor 19 and bile acids in diabetes remission after Roux-en-Y gastric bypass. Diabetes Care 36, 1859–1864 (2013).
Dutia, R. et al. Temporal changes in bile acid levels and 12alpha-hydroxylation after Roux-en-Y gastric bypass surgery in type 2 diabetes. Int. J. Obes. 39, 806–813 (2015).
Gomez-Ambrosi, J. et al. FGF19 and FGF21 serum concentrations in human obesity and type 2 diabetes behave differently after diet- or surgically-induced weight loss. Clin. Nutr. 36, 861–868 (2017).
Harris, L. L. S. et al. Roux-en-Y gastric bypass surgery has unique effects on postprandial FGF21 but not FGF19 secretion. J. Clin. Endocrinol. Metab. 102, 3858–3864 (2017).
Nemati, R. et al. Increased bile acids and FGF19 after sleeve gastrectomy and Roux-en-Y gastric bypass correlate with improvement in type 2 diabetes in a randomized trial. Obes. Surg. 28, 2672–2686 (2018).
Guo, J. Y. et al. Fibroblast growth factor 19 and fibroblast growth factor 21 regulation in obese diabetics, and non-alcoholic fatty liver disease after gastric bypass. Nutrients https://doi.org/10.3390/nu14030645 (2022).
Dreyfuss, J. M. et al. High-throughput mediation analysis of human proteome and metabolome identifies mediators of post-bariatric surgical diabetes control. Nat. Commun. 12, 6951 (2021).
Ryan, P. M., Hayward, N. E., Sless, R. T., Garwood, P. & Rahmani, J. Effect of bariatric surgery on circulating FGF-19: a systematic review and meta-analysis. Obes. Rev. 21, e13038 (2020).
Lundasen, T., Galman, C., Angelin, B. & Rudling, M. Circulating intestinal fibroblast growth factor 19 has a pronounced diurnal variation and modulates hepatic bile acid synthesis in man. J. Intern. Med. 260, 530–536 (2006).
Inagaki, T. et al. Fibroblast growth factor 15 functions as an enterohepatic signal to regulate bile acid homeostasis. Cell Metab. 2, 217–225 (2005).
Xiang, J. et al. Effect of different bile acids on the intestine through enterohepatic circulation based on FXR. Gut Microbes 13, 1949095 (2021).
Ridlon, J. M., Harris, S. C., Bhowmik, S., Kang, D. J. & Hylemon, P. B. Consequences of bile salt biotransformations by intestinal bacteria. Gut Microbes 7, 22–39 (2016).
Chaudhari, S. N. et al. Bariatric surgery reveals a gut-restricted TGR5 agonist with anti-diabetic effects. Nat. Chem. Biol. 17, 20–29 (2021).
Hegyi, P., Maleth, J., Walters, J. R., Hofmann, A. F. & Keely, S. J. Guts and gall: bile acids in regulation of intestinal epithelial function in health and disease. Physiol. Rev. 98, 1983–2023 (2018).
Zhang, Y. et al. Comparative potency of obeticholic acid and natural bile acids on FXR in hepatic and intestinal in vitro cell models. Pharmacol. Res. Perspect. https://doi.org/10.1002/prp2.368 (2017).
Panzitt, K. & Wagner, M. FXR in liver physiology: multiple faces to regulate liver metabolism. Biochim. Biophys. Acta Mol. Basis Dis. 1867, 166133 (2021).
Jiang, L., Zhang, H., Xiao, D., Wei, H. & Chen, Y. Farnesoid X receptor (FXR): structures and ligands. Comput. Struct. Biotechnol. J. 19, 2148–2159 (2021).
Chaudhari, S. N., McCurry, M. D. & Devlin, A. S. Chains of evidence from correlations to causal molecules in microbiome-linked diseases. Nat. Chem. Biol. 17, 1046–1056 (2021).
Zhan, L. et al. Genome-wide binding and transcriptome analysis of human farnesoid X receptor in primary human hepatocytes. PLoS ONE 9, e105930 (2014).
Katafuchi, T. & Makishima, M. Molecular basis of bile acid–FXR–FGF15/19 signaling axis. Int. J. Mol. Sci. https://doi.org/10.1002/prp2.368 (2022).
Kim, K. H. et al. Hepatic FXR/SHP axis modulates systemic glucose and fatty acid homeostasis in aged mice. Hepatology 66, 498–509 (2017).
Dawson, P. A. Role of the intestinal bile acid transporters in bile acid and drug disposition. Handb. Exp. Pharmacol. https://doi.org/10.1007/978-3-642-14541-4_4 (2011).
Chaudhari, S. N. et al. A microbial metabolite remodels the gut-liver axis following bariatric surgery. Cell Host Microbe 29, 408–424 (2021).
Craddock, A. L. et al. Expression and transport properties of the human ileal and renal sodium-dependent bile acid transporter. Am. J. Physiol. 274, G157–G169 (1998).
Rao, A. et al. The organic solute transporter alpha-beta, Ostalpha-Ostbeta, is essential for intestinal bile acid transport and homeostasis. Proc. Natl Acad. Sci. USA 105, 3891–3896 (2008).
Zhang, J. H. et al. Potent stimulation of fibroblast growth factor 19 expression in the human ileum by bile acids. Am. J. Physiol. Gastrointest. Liver Physiol. 304, G940–G948 (2013).
Ding, L. et al. Vertical sleeve gastrectomy activates GPBAR-1/TGR5 to sustain weight loss, improve fatty liver, and remit insulin resistance in mice. Hepatology 64, 760–773 (2016).
Yang, N. et al. ASBT(SLC10A2): a promising target for treatment of diseases and drug discovery. Biomed. Pharmacother. 132, 110835 (2020).
Miner, P. B. Jr Elobixibat, the first-in-class ileal bile acid transporter inhibitor, for the treatment of chronic idiopathic constipation. Expert Opin. Pharmacother. 19, 1381–1388 (2018).
Soroka, C. J., Ballatori, N. & Boyer, J. L. Organic solute transporter, OSTα-OSTβ: its role in bile acid transport and cholestasis. Semin. Liver Dis. 30, 178–185 (2010).
Bozadjieva-Kramer, N. et al. Intestinal-derived FGF15 protects against deleterious effects of vertical sleeve gastrectomy in mice. Nat. Commun. 12, 4768 (2021).
Salehi, M., Vella, A., McLaughlin, T. & Patti, M. E. Hypoglycemia after gastric bypass surgery: current concepts and controversies. J. Clin. Endocrinol. Metab. 103, 2815–2826 (2018).
Flynn, C. R., Albaugh, V. L. & Abumrad, N. N. Metabolic effects of bile acids: potential role in bariatric surgery. Cell Mol. Gastroenterol. Hepatol. 8, 235–246 (2019).
Zheng, D. et al. Comparative profiling of serum, urine, and feces bile acids in humans, rats, and mice. Commun. Biol. 7, 641 (2024).
Chiang, J. Y. Bile acid metabolism and signaling. Compr. Physiol. 3, 1191–1212 (2013).
Yao, L. et al. A selective gut bacterial bile salt hydrolase alters host metabolism. Elife https://doi.org/10.7554/eLife.37182 (2018).
Qiu, Y. et al. Depletion of gut microbiota induces skeletal muscle atrophy by FXR-FGF15/19 signalling. Ann. Med. 53, 508–522 (2021).
Li, W. et al. A bacterial bile acid metabolite modulates Treg activity through the nuclear hormone receptor NR4A1. Cell Host Microbe 29, 1366–1377 (2021).
Carvalho, M. B. et al. The role of FGF19 in metabolic regulation: insights from preclinical models to clinical trials. Am. J. Physiol. Endocrinol. Metab. 327, E279–E289 (2024).
Young, M. T., Phelan, M. J. & Nguyen, N. T. A decade analysis of trends and outcomes of male vs female patients who underwent bariatric surgery. J. Am. Coll. Surg. 222, 226–231 (2016).
Sardao, D. et al. The impact of cholecystectomy in patients with post-bariatric surgery hypoglycemia. Obes. Surg. 34, 2570–2579 (2024).
van Furth, A. M. et al. Cholecystectomy increases the risk of dumping syndrome and postbariatric hypoglycemia after bariatric surgery. Surg. Obes. Relat. Dis. 16, 1939–1947 (2020).
Mosinska, P., Szczepaniak, A. & Fichna, J. Bile acids and FXR in functional gastrointestinal disorders. Dig. Liver Dis. 50, 795–803 (2018).
Nakajima, A. et al. Impact of elobixibat on serum and fecal bile acid levels and constipation symptoms in patients with chronic constipation. J. Gastroenterol. Hepatol. 37, 883–890 (2022).
Yamauchi, R. et al. Elobixibat, an ileal bile acid transporter inhibitor, ameliorates non-alcoholic steatohepatitis in mice. Hepatol. Int. 15, 392–404 (2021).
Caballero-Camino, F. J. et al. A3907, a systemic ASBT inhibitor, improves cholestasis in mice by multi-organ activity and shows translational relevance to humans. Hepatology https://doi.org/10.1097/HEP.0000000000000376 (2023).
Karpen, S. J., Kelly, D., Mack, C. & Stein, P. Ileal bile acid transporter inhibition as an anticholestatic therapeutic target in biliary atresia and other cholestatic disorders. Hepatol. Int. 14, 677–689 (2020).
Kang, L., Connolly, T. M., Weng, N. & Jian, W. LC-MS/MS quantification of 7α-hydroxy-4-cholesten-3-one (C4) in rat and monkey plasma. J. Chromatogr. B 1064, 49–55 (2017).
Thomas, C. et al. TGR5-mediated bile acid sensing controls glucose homeostasis. Cell Metab. 10, 167–177 (2009).
Acknowledgements
This study was supported by the National Institutes of Health awards R01 DK121995 (to M.-E.P), DK126855 (to E.G.S. and A.S.D.), K99 DK128503 (to S.N.C.) and P30 DK036836 (Diabetes Research Center, Joslin Diabetes Center) and an American Surgical Association Foundation Fellowship Award (to E.G.S.). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. We are grateful to the study participants and clinical research nursing staff.
Author information
Authors and Affiliations
Contributions
Conceptualization and study design was carried out by S.N.C., E.G.S., A.S.D. and M.-E.P. Human samples were collected and banked by C.C., A.S. and M.-E.P. Human blood molecular analyses were performed by R.F.-B. and C.C. All in vitro experiments and BA analyses in in vitro and in vivo samples were performed by S.N.C. C4 analyses were performed by G.D.A. and F.Y. In vivo experiments and assays were performed by R.F.-B., P.C.Q., B.O., H.W. and Y.C. All authors edited and contributed to the critical review of the paper.
Corresponding authors
Ethics declarations
Competing interests
A.S.D. is an ad hoc consultant for Axial Therapeutics. E.G.S. is a consultant for Vicarious Surgical, and received speaker fees at Cine-Med. S.N.C. is an ad hoc consultant for Metis Therapeutics. M.-E.P. serves on the data safety monitoring board for Fractyl, has received investigator-initiated research support from Dexcom and has served as a consultant for Eiger, Hanmi and MBX Pharmaceuticals. The other authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks Rohit Kohli, Antonio Moschetta and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Jean Nakhle and Ashley Castellanos-Jankiewicz, in collaboration with the Nature Metabolism team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
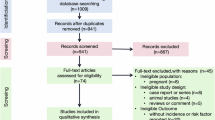
Extended Data Fig. 1 Consort diagram.
a. Consort diagram for participants providing stool samples from NCT04428866 (left) and NCT03385707 (right). b. Consort diagram for participants in NCT04428866 for which mixed meal tolerance test samples were analyzed.
Extended Data Fig. 2 Post-op hypoglycemia in human patients is characterized by a shift in fecal BAs.
Individual BAs whose levels were reduced in the feces of post-op hypoglycemic individuals but for which the difference between groups was not statistically significant (post-op non-hypoglycemic n = 15, post-op hypoglycemic n = 46; two-tailed Welch’s t test). Data not marked are not statistically significant (p > 0.05). All other significant BAs shown in Fig. 1. Patient characteristics and demographics detailed in Supplementary Table 1. All data are represented as mean ± SEM.
Extended Data Fig. 3 Post-op hypoglycemia in human patients is characterized by a shift in post-prandial plasma BAs.
Individual BAs whose levels were reduced in the plasma of post-op hypoglycemic individuals but for which the difference between groups was not statistically significant (post-op non-hypoglycemic n = 9, post-op hypoglycemic n = 10; two-tailed Welch’s t test for each time point). Data not marked are not statistically significant (p > 0.05). All other significant BAs shown in Fig. 2. Patient characteristics and demographics detailed in Supplementary Table 1. All data are represented as mean ± SEM.
Extended Data Fig. 4 BAs shifted in post-op hypoglycemia patients induce FGF19 secretion and gene expression in vitro with differential effects on SHP.
a. FGF19 secretion and expression induced by BA pools totaling physiological concentrations (post-op non-hypoglycemic 1 mM, post-op hypoglycemic 0.6 mM) in undifferentiated Caco-2 cells. Post-op hypoglycemic fecal BAs induced higher FGF19 secretion and expression compared to post-op non-hypoglycemic fecal BAs (DMSO, n = 3; BA pools n = 6; FGF19 expression, n = 6 in each group; One-way ANOVA with Dunnet’s multiple comparison test. b. Post-op hypoglycemic fecal BAs (1 mM) induced higher Fgf15 expression compared to post-op non-hypoglycemic fecal BAs (1 mM) in in ex vivo ileal segments from lean, chow-fed mice. (n = 8 in each group; one-way ANOVA with Dunnet’s multiple comparison test. c. Dose-response curves for human SHP activation measured by using a SHP-luciferase reporter expressed in Caco-2 cells. Values were normalized to DMSO. EC50 values for activating BAs (blue) are indicated within each panel. BAs showing inhibitory (red) or no activity (black) have no EC50 or IC50 values listed. d. Dose-response curves for human SHP inhibition measured with the addition of 10 µM CDCA, a SHP activator. BAs were tested in their ability to inhibit SHP reporter in Caco-2 cells. Values were normalized to the agonist CDCA. EC50 and IC50 values for BAs are indicated within each panel and also listed in Supplementary Table 2 (each dose-response curve data point contains \(\ge\)3 biological replicates). e. SHP luciferase reporter activation (left) and shp expression (right) induced by BA pools totaling 1 mM in Caco-2 cells. No statistically significant difference was observed in SHP activation or expression in cells treated with post-op hypoglycemic fecal BAs compared to post-op non-hypoglycemic fecal BAs (SHP activation, DMSO, n = 3; BA pools n = 6; shp expression, n = 6 in each group; One-way ANOVA with Dunnet’s multiple comparison test. All data has been reproduced at least twice, each time in biological triplicate). All statistically significant p values are indicated. Lines above graphs indicate the groups being compared with p values. ns=not significant. Data not marked are not statistically significant (p > 0.05). All data are represented as mean ± SEM.
Extended Data Fig. 5 Portal BA profiling in chow-fed male mice treated with elobixibat and subjected to an MMTT.
UPLC-MS analysis of portal BAs from mice treated with PBS or elobixibat (ASBTi), followed by an MMTT and sacrificed at 120 min post-meal (n = 8 in each group, two-tailed Welch’s t test). All statistically significant p values are indicated in each graph. Lines above graphs indicate the groups being compared with p values. ns = not significant. All data are represented as mean ± SEM.
Extended Data Fig. 6 Serum BA profiling in chow-fed male mice treated with elobixibat and subjected to an MMTT.
UPLC-MS analysis of serum BAs from mice treated with PBS or elobixibat (ASBTi), followed by an MMTT and sacrificed at 120 min post-meal (n = 8 in each group, two-tailed Welch’s t test). All statistically significant p values are indicated in each graph. Lines above graphs indicate the groups being compared with p values. ns = not significant. All data are represented as mean ± SEM.
Extended Data Fig. 7 Elobixibat treatment prior to MMTT increased glucose, but did not significantly alter insulin or GLP-1 levels.
a. Elobixibat treatment significantly increased post-prandial blood glucose at 30 min after MM gavage compared with PBS (n = 4 in each group, two-way ANOVA). b. Insulin levels during MMTT did not differ between groups (n = 4 in each group, two-way ANOVA. AUC adjacent). c. GLP1 levels during MMTT did not differ between groups (n = 4 in each group, two-way ANOVA. AUC adjacent). Data not marked are not statistically significant (p > 0.05). All data are represented as mean ± SEM.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1–3 and research protocol.
Source data
Source Data Fig. 1 (download XLSX )
FGF19 and BA levels.
Source Data Fig. 2 (download XLSX )
Glucose, GLP-1, insulin and C4 levels.
Source Data Fig. 3 (download XLSX )
BA and FGF19 levels.
Source Data Fig. 4 (download XLSX )
BA and FGF19 levels.
Source Data Fig. 5 (download XLSX )
BA and FGF19 levels.
Source Data Fig. 6 (download XLSX )
Glucose, BA and gene expression measurements.
Source Data Fig. 7 (download XLSX )
Caecal BA.
Source Data Fig. 8 (download XLSX )
Glucose levels.
Source Data Extended Data Fig. 2 (download XLSX )
Human faecal BA levels.
Source Data Extended Data Fig. 3 (download XLSX )
Human plasma BA levels.
Source Data Extended Data Fig. 4 (download XLSX )
FGF19 expression/secretion and Shp expression/activity.
Source Data Extended Data Fig. 5 (download XLSX )
Mouse portal vein BA levels.
Source Data Extended Data Fig. 6 (download XLSX )
Mouse serum BA levels.
Source Data Extended Data Fig. 7 (download XLSX )
Mouse serum glucose, insulin and GLP-1 levels.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chaudhari, S.N., Chen, Y., Ferraz-Bannitz, R. et al. Alterations in intestinal bile acid transport provide a therapeutic target in patients with post-bariatric hypoglycaemia. Nat Metab 7, 792–807 (2025). https://doi.org/10.1038/s42255-025-01262-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s42255-025-01262-5
This article is cited by
-
Bile acids in the crosshairs for hypoglycaemia after gastric bypass
Nature Reviews Endocrinology (2025)