Abstract
Enzyme-responsive biomaterials are smart materials designed to interact with the body’s natural processes. Hydrogels, in particular, can change structure in response to specific enzymes, enabling controlled drug delivery, tissue regeneration, and improved healing. By mimicking biological environments, they provide precise, targeted treatments for conditions such as cardiovascular disease, musculoskeletal injuries, and chronic wounds. This review highlights advances in enzyme-responsive biomaterials in the recent decades, focusing on polymeric hydrogels. We examine therapeutic, scaffold, and diagnostic applications across musculoskeletal, cardiovascular, pulmonary, dental, dermal, gastrointestinal, and hepatic systems, and conclude with emerging concepts and future perspectives for clinical translation.
Similar content being viewed by others
Introduction
Biomaterials, whether natural or synthetic, can be tailored chemically or biologically to provide therapeutic and mechanical functions. Hydrogels are a type of biomaterial comprising of polymeric networks that can absorb a large amount of water while maintaining their structure, due to the abundance of hydrophilic groups and the cross-linking structure of polymers1,2,3. With a high swelling ability to hold biological fluids, hydrogel can mimic conditions in living tissues, making them outstanding candidates for biomedical applications4. In addition, hydrogels exhibit remarkable flexibility, biocompatibility, biodegradability, and tunable chemical/mechanical properties, offering opportunities for novel therapeutic strategies. By modifying polymer structures, varying hydrogel precursor concentration and cross-linking agent, hydrogels can be tailored to meet specific requirements1,5,6. The most common applications of hydrogels include but are not limited to drug/cell delivery, artificial tissues, wet adhesives, and surgical tools7.
Stimuli-responsive hydrogels are a subclass of hydrogel that undergo chemical, structural, or mechanical changes in response to triggers4. These triggers fall broadly into two categories: endogenous stimuli from cells or tissues, and exogenous stimuli generated artificially8. Endogenous cues include pH, ionic strength, reactive oxygen species, and enzymes; for example, ischemic tissues exhibit lower pH, ROS are elevated in wounds, and certain enzymes are temporally upregulated in infarcted tissues4,9,10,11,12. Such features can be harnessed for targeted drug release or signaling output through responsive polymer functionalisation13. Exogenous stimuli include temperature, light, and electromagnetic fields; examples include photocleavable moieties for controlled therapeutic release, hydrogels with hydrogen-bond-mediated adhesion at skin temperature, and electric field-responsive systems functioning as bioactuators or artificial muscles14,15,16. Combining both categories enables integrated, multifunctional, and “smart” hydrogel platforms.
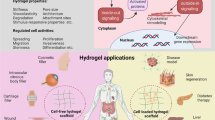
Enzyme-responsive hydrogels are a category of stimuli-responsive materials designed to behave as substrates for exogenous or endogenous enzymatic activity. Due to their role as biological catalysts, enzymes are essential to nearly all cellular processes, acting with high specificity and selectivity toward their substrates17. They operate under mild physiological conditions, making enzyme-responsive biomaterials promising candidates for biomedical applications. As a result of enzyme-substrate specificity, a crucial aspect of enzyme-responsive research is to synthesize biomaterials containing the appropriate peptide sequences to act as substrates for target enzymes18. This approach allows biomaterials to be tailored with high precision for specific diseases and pathological states (Fig. 1). The diversity of enzyme classes and their relevance to biomaterial design are summarized in Box 1. However, this specificity also adds a layer of complexity to the pre-existing challenges of designing suitable biomaterials for use in the body, such as cytocompatibility, biodegradability and mechanical stability.
A major application of enzyme-responsive hydrogels lies in controlled therapeutic delivery via enzymatic degradation. More recently, their use has expanded to include diagnostics and imaging, disease modeling, and tissue engineering. Created in BioRender. Celiz, A. (2025) https://BioRender.com/7bbk1z6.
This review highlights recent developments in enzyme-responsive biomaterials, focusing mainly on polymeric hydrogels, as well as nanoparticles and scaffold-based approaches for delivering enzyme-triggered solutions to various pathologies (Table 1). While many reported systems are based on MMP-responsive and peptide-based hydrogels, these examples are presented as part of the broader landscape rather than the exclusive focus of this review. This review focuses on non-oncology applications; enzyme-responsive hydrogels for cancer are excluded as they are covered extensively elsewhere19,20,21,22. We organize the discussion by organ and tissue systems, including musculoskeletal, cardiovascular, pulmonary, dental, dermal, gastrointestinal, hepatic, and generalized inflammation, followed by emerging concepts beyond tissue-specific uses and future perspectives.
Cardiovascular tissues
Cardiovascular Disease remains the leading global cause of death, making up 32% of all global deaths23. Of all types of cardiovascular disease, myocardial infarction (MI) is the most common type of atherosclerosis and can cause further complications such as heart failure, arrhythmia, and cardiogenic shock24. During MI, thrombotic destruction of blood flow in coronary arteries causes cardiomyocyte death from the subendocardium to the subepicardium, which are subsequently replaced by non-contractile fibrotic tissue.
Enzyme-responsive biomaterial therapies for MI
Currently, most enzyme-responsive biomaterials for MI treatment focus on targeting MMPs, a major family of proteinases upregulated in both serum and the cardiac interstitium at the end of inflammatory phase, due to its role in ECM degradation and myocardial remodeling25. Subtypes of MMPs are categorized based on their structural similarity and substrate specificity, among which MMP-1, -2, -3, -9, -13, -14 are anti-inflammatory and particularly MMP-2/9 are responsible for degrading ECM collagen, laminin, elastin, and fibronectin26,27. Several peptide sequences cleavable by either all MMPs or certain subtypes such as MMP-2/9 have been identified and incorporated into hydrogels, nanoparticles, lipid micellar vehicles for targeted accumulation, prolonged tissue retention, and on-demand therapeutic delivery28,29,30,31. The site-specific enzymatic cleavage leads to biomaterial morphological changes at the desired site for therapeutic release.
Targeted accumulation and prolonged tissue retention have become increasingly demanded properties of biomaterials for MI treatment, as direct injection of therapies such as drugs, stem cells, and growth factors tend to exhibit poor retention in the tissue and can be rapidly degraded, thus impairing therapeutic effect and efficiency32,33,34. A recent work by Carlini et al. developed a biomaterial strategy with MMP-2/9-cleavable peptide sequence (PLGLAG), in the form of self-assembling peptides, where cyclic progelator peptides became linear upon MMP-2/9 cleavage and subsequently cross-link to form self-healing viscoelastic hydrogels at 37 °C (Fig. 2a)29. The in vivo analysis showed successful activation of peptides in respond to MMP-2/9 and subsequent gelation in a rat MI model and no adverse side effects such as inflammation and cardiomyocyte apoptosis. The low viscosity, in-situ gelling property and hemocompatible nature of the self-assembling peptide hydrogel potentially facilitates non-invasive delivery methods such as injection via catheters.
a (i) Cyclic progelator peptides, comprising a gelling sequence (green), MMP/elastase cleavage site (red), and disulfide bridge (black), resist self-assembly due to conformational constraints. Upon enzymatic cleavage, they linearize into self-assembling peptides (SAPs) that form β-sheet fibrillar viscoelastic hydrogels. For in vivo tracking, 5 mol% rhodamine-labeled SAPs (pink ellipse) were included to enable ex vivo imaging. (ii) Representative heart section showing hydrogel assembly at the site of MI. Scale bars are 1 mm (inset 1) and 100 µm (inset 2), respectively. Adapted from Nat. Commun., Carlini et al.29. b (i) Schematic illustration of the preparation and application of an MMP-responsive hydrogel for drug release in a myocardial infarction (MI) rat model. GST-TIMP-bFGF is produced via recombinant expression and conjugated to a GSH-loaded hydrogel (Gel-GSH) through GST–GSH bonding. The resulting hydrogel is then injected intramyocardially into the infarcted area to enable localized therapeutic delivery. (ii) Gel/GST-TIMP-bFGF hydrogel significantly alleviated ventricular wall thinning, decreased collagen deposition and MMP-2/9 expression, and increased in vivo wound vascularization on day 30 after MI. Adapted from Adv. Mat., Fan et al.28. Created in BioRender. Pi, P. (2025) https://BioRender.com/ogamsab.
More recently, enzyme-responsive biomaterials have leveraged MMP upregulation in myocardium to enhance tissue retention and achieve controlled therapeutic release of growth factors, proteins, and small molecules. Purcell et al. developed a HA hydrogel incorporating an MMP-sensitive cross-linker (GGRMSMPV) for delivering a recombinant tissue inhibitor of MMPs (rTIMP-3), where electrostatically sequestered rTIMP-3 was released upon MMP-catalyzed degradation31. This approach successfully inhibited MMP activity in a porcine model, with therapeutic effects persisting 28 days post-injection. Fan et al. developed a dual-function, MMP-2/9 responsive hydrogel for on-demand basic fibroblast growth factor (bFGF) delivery to promote angiogenesis and inhibit cardiac remodeling (Fig. 2b)28. A glutathione (GSH)-modified collagen hydrogel was synthesized by linking the amine groups of collagen with the sulfhydryl groups of GSH. Additionally, a recombinant protein, GST-TIMP-bFGF, was incorporated, in which bFGF is fused with glutathione-S-transferase (GST) and a MMP-2/9-sensitive peptide sequence, PLGLAG (TIMP). The dual function of inhibiting MMP-2/9 activity and promoting angiogenesis was achieved by cleavage of TIMPs occupying upregulated MMP-2/9 and the simultaneous release of bFGF. In vivo studies showed that the hydrogel effectively improved cardiac function, reduced ventricular remodeling, inhibited MMP expression, and enhanced angiogenesis in MI model rats. Beyond therapeutic proteins, Wang et al. demonstrated protease-triggered siRNA delivery using HA hydrogels cross-linked via hydrazide and aldehyde, containing protease-cleavable peptides and β-cyclodextrin-sequestered cholesterol-modified siRNA against MMPs30. Cholesterol-modified-siRNA against MMPs was further sequestered by modifying HA with β-cyclodextrin. This platform effectively delivered siRNA to the heart and significantly improved hemodynamic function.
NO is an important messenger in signaling pathways involved in cardiac protection and can also promote the expression of pro-angiogenic cytokines in stem cells35,36. Many biomaterials that release NO in infarcted myocardium have been developed due to such nature. For example, adipose derived-MSCs (AD-MSCs) were encapsulated in a hydrogel (NapFF-NO), where naphthalene was conjugated to both FFGGG peptide and β-galactose caged NO donor37. The intramyocardial injectable hydrogel releases NO molecules and AD-MSCs in response to local β-galactosidase. In vivo mouse MI model results showed that (1) cell retention was improved when AD-MSCs were co-transplanted with the hydrogel, and (2) the secretion of VEGF and Stromal cell-derived factor 1 by AD-MSCs was significantly higher in the presence of NapFF-NO hydrogel. However, since both the hydrogels with or without NO molecules demonstrated improved functional parameters, the observed increase in angiogenesis could not be fully translated to cardiac function and collagen content.
Enzyme-responsive biomaterial for therapeutic angiogenesis
In ischemic disease, which involves occluded vessels, vascular insufficiency has been a challenge and could potentially cause further complications. Strategies for bypassing diseased vessels and stimulating angiogenesis have been addressed to cope with such problems. Particularly, enzyme-responsive biomaterials have been applied to release growth factors, inorganic molecules, and drugs in a controlled manner.
One such group of biomaterials are conjugated with MMP-cleavable peptides, as MMP activity is high in many ischemic diseases. In a study by Hove et al., PEG hydrogels that release pro-angiogenic peptide drugs in response to MMP were explored38. The two peptides, SPARC113 and SPARC118, from the Secreted Protein Acidic and Rich in Cysteine, were incorporated into the hydrogel as cross-linkers flanked by MMP-cleavable substrates. In vitro tests showed that peptide bioactivity was retained after hydrogel degradation by MMP, and interestingly, the degradation rates of hydrogels containing the two peptides, respectively, were significantly different. Contrarily, in vivo modeling showed a similar degradation rate, and significant improvement in angiogenesis was observed. Although 4-day delivery of peptides was achieved, which was much faster than previously reported protein delivery, the ideal delivery profile for these two peptides was yet to be investigated39,40,41.
Multidomain peptides (MDPs) are self-assembling peptides that form nanofibrous β-sheet architectures. Moore et al. highlighted their potential as bioengineering scaffolds due to the flexibility of peptide sequence design42. By incorporating bioactive domains, MDPs can communicate with the microenvironment via specific molecular interactions. For example, an MMP-2-responsive MDP hydrogel incorporated the cleavable sequence LGR in the core and a VEGF-mimic growth factor at the C-terminus. Injection in a hind-limb ischemia model increased blood perfusion, indicating enhanced ischemia resolution. More recently, da Silva et al. engineered gellan gum-based hydrogels functionalized with divinyl sulfone (GGDVS) to support endothelial cell adhesion and angiogenesis43. Thiol-terminated, cell-adhesive peptides (T1 or C16) were immobilized and cross-linked via an MMP-1-sensitive sequence (CRDGPQGIWGQDRC). Human umbilical vein endothelial cells colonized the MMP-1-degradable hydrogels, showing polarized and elongated morphology in T1-containing gels versus rounded morphology in C16 gels, highlighting the enhanced potential of T1-functionalized, MMP-responsive hydrogels for vascularization.
Besides the studies above, other studies also focus on NO-releasing chitosan hydrogel for angiogenesis, yet in combination with different cell types. For example, VEGFs are promising for angiogenesis and for perfusing ischemic tissues in patients with peripheral artery disease, were co-transplanted with chitosan-NO hydrogel in a hindlimb ischemia mouse model, where NO could provide the pro-angiogenic microenvironment for MSC-based treatment44. It was found that hydrogel containing NO increased the survival of transplanted MSCs, which showed phenotypic and functional features of endothelial cells that possibly contributed to the enhanced pro-angiogenesis observed with bioluminescence imaging technology.
Musculoskeletal system
The musculoskeletal system broadly consists of four main tissue types: cartilage, tendon, bone and muscle. Musculoskeletal conditions affect approximately 1.71 billion people worldwide, making them one of the leading causes of pain and disability according to the World Health Organization45. Biomaterials research in this field is extensive and often focuses on developing biomaterials for musculoskeletal tissue regeneration. The following section highlights solutions for musculoskeletal regeneration which focus on harnessing specific endogenous enzymes for injury repair, while also considering the architecture of each tissue type in their scaffold design.
Cartilage
Osteoarthritis is a degenerative disease of the articular cartilage surrounding synovial joints and is a leading cause of disability, currently affecting an estimated 250 million people worldwide46. It is characterized by the progressive degradation of articular cartilage47. The smooth, lubricated surface becomes inflamed, swollen and often necrotic. This leads to severe pain and loss of mobility in affected patients.
Articular cartilage lacks the ability to self-repair, due to the avascularity of the tissue and the isolation of chondrocytes in lacunae48. The physiological response to articular cartilage damage is to initiate the formation of coarse, dense and fibrous fibrocartilage, an unfavorable candidate for replacing articular cartilage49. This has presented a barrier for cartilage tissue engineering strategies, as chondrogenic stimulation techniques have often resulted in unwanted fibrocartilage production50.
Enzyme-responsive hydrogels are well suited to mimic the soft, highly hydrated nature of articular cartilage51. For several decades, polyethylene glycol (PEG)-based hydrogels have been researched as scaffolds for cartilage growth, repair, and regeneration52,53,54. In more recent years, hyaluronic acid (HA) and collagen-based polymers have been used, due to their in vivo degradability12,55. Biodegradability is a beneficial characteristic for cartilage tissue engineering, as the scaffold can give way to neocartilage over time. However, the synthetic cross-linking agents used to create these hydrogels often do not degrade, which can affect the overall degradation behavior and even the toxicity of the scaffold56. A potential solution is the use of enzymatic cross-linking, an exmaple of which is a double-modified alginate hydrogel functionalized with sulfate groups to mimic glycosaminoglycans and tyramine groups that enable strong adhesion to native cartilage57. Such hydrogels enhanced chondrogenic gene expression and cartilage matrix deposition compared to alginate modified with tyramine only, supporting both in vitro chondrocyte re-differentiation and in vivo stability. Another example of the use of biodegradable cross-linkers is the HA hydrogel developed by Parmar et al., which makes use of MMP-7-sensitive peptides as cross-linking agents, as shown in Fig. 3a58. These cross-links are MMP7-cleavable and therefore can be harnessed to deliver cargo. In this instance, MSCs were delivered to the scaffold site to promote differentiation of MSCs into chondrocytes to stimulate cartilage healing.
a (i) Scheme of peptide-functionalized Scl2 protein hydrogels. The terminal ε-amines of lysines on the Scl2 proteins were modified with a heterobifunctional linker using NHS-ε-amine conjugation chemistry to generate acrylate-functionalized Scl2 proteins. (ii) Total collagen content, sGAG content, DNA content per construct, elastic modulus of compression of hydrogels, anda histological examination of hydrogels after 4 weeks of in vitro culture. Hydrogels are stained with haematoxylin and eosin, alcian blue for sGAG, and by immunohistochemistry for collagen type I, collagen type II and collagen type X. All scale bars are 200 μm. Adapted from Biomaterials, Parmar et al.58. b (i) Fabrication process of the siRNA@microsphere@hyaluronic acid hydrogel–-electrospun antiadhesion barrier for peritendinous antiadhesion. (ii) In vivo evaluation of the anti-adhesion effect of MMP-2-responsive barriers. Gross images show peritendinous adhesion at the injury site 21 days post-surgery. Haematoxylin and eosin-stained sections illustrate different barrier formulations, highlighting the physical peritendinous space (green), adhesion tissue (yellow), sutured site (S), repaired site (R), and anti-adhesion material (M). Adapted from Adv. Mat., Cai et al.64. Created in BioRender. Pi, P. (2025) https://BioRender.com/11ymzcf.
Tendon
Tendinous tissue is responsible for force transmission from muscles to bones. It is commonly damaged in exercise or loading-related injuries, which triggers the tendon healing process that occurs in two stages: intrinsic and extrinsic59. The intrinsic stage consists of obliteration of the tendon and the tendon sheath. The extrinsic stage occurs in three phases: inflammation, repair, and remodeling, and is characterized by the chemotaxis of the specialized fibroblasts into the defect from the ends of the tendon sheath60. Complete regeneration of the tendon is not achieved, with the tissue remaining hypercellular and lacking its original biomechanical strength60,61.
While tendon injuries can be treated with surgical intervention or tissue grafts, a common complication is peritendinous adhesion62. This occurs when injured tendon tissue undergoes fibrosis and adheres to surrounding tissues, limiting motion in the process63. This is a major negative outcome of tendon surgery that has the potential to be resolved by using enzyme-responsive biomaterials. Cai et al. recently developed two different enzyme-responsive hydrogel systems that harness MMP-2 to release small interfering RNAs (siRNA) to the peritendinous adhesion site (Fig. 3b)64,65. The siRNA silences the genes transforming growth factor 1 and Smad3, both of which are normally upregulated in peritendinous adhesion. This enzyme-controlled drug release mechanism led to a significant decrease in adhesion in both cases. However, these techniques both rely on forming composite hydrogel patches, which are an invasive means of drug delivery, and may therefore function best as a preventative measure if inserted into the body at the time of tendon surgery. For greater translational relevance, further research into less invasive strategies is required, such as the development of injectable systems.
Bone
Bone defects can be caused by infections, trauma (surgical or other) or diseases such as osteoarthritis66. Unlike cartilage and tendon tissue, bone is capable of regeneration and can heal itself through primary or secondary wound healing mechanisms, depending on the size and severity of the defect67. More severe defects require secondary healing, where an extracellular matrix (ECM)-rich structural component is necessary to stabilize the defect before healing can take place66. This process requires osteogenesis, osteoinduction, and osteoconduction68. MSCs provide osteoprogenitor cells that differentiate into osteoblasts and osteoclasts, while osteoinductive factors such as transforming growth factor-β1 and -β2 and vascular endothelial growth factor (VEGF) stimulate differentiation. An osteoconductive ECM scaffold is also necessary and is first provided by the hematoma and then by the cartilage callus68.
The process of bone healing is heavily reliant on adequate blood supply to the injury site, as such it is often impeded by the avascularity caused by large defects above a critical size threshold of 5 cm69,70. In addition, comorbidities such as diabetes, obesity and arthritis can negatively affect bone healing71. In cases such as these, the bone may likely never heal without surgical intervention. While allografts are often used in such cases, biomaterial scaffolds with high stiffness have also been extensively researched as alternatives. However, there are very few hydrogel-based solutions due to their intrinsically softer properties. An example is the PEG-diacrylate hydrogel scaffolds by Hsu et al., which incorporates cathepsin K-degradable peptide sequences to allow for the induction of degradation72. This enzyme-specificity allows for degradation when the bone is nearing the end of the remodeling stage, rather than at an arbitrary time point, thus allowing for reliable bone formation before scaffold degradation. It also allows for greater patient-specificity, as healing takes place at different rates in different individuals.
Skeletal muscle
Skeletal muscle is a complex tissue that enables movement and depends on blood vessels and nerves to produce contractile force. While it can self-repair through the activation of satellite cells after minor injuries, severe damage impairs this ability, highlighting the need for tissue engineering73. This is especially important given that muscle injuries account for over 50% of Department of Defense disabilities and 35–55% of sports injuries, leading to around 4.5 million reconstructive surgeries annually in the U.S.74,75,76.
To the authors’ knowledge, there are still no enzyme-responsive biomaterials targeting skeletal muscle. As there are several muscle-injury specific enzymes, such as creatine kinase, lysyl oxidase (LOX), and gamma glutamyl transpeptidase, this presents an interesting avenue for further research.
Pulmonary
Despite the prevalence of pulmonary infectious diseases such as tuberculosis, pneumonia, and COVID-19, there is a lack of effective targeted therapies. These infections, along with chronic inflammatory conditions like asthma, cystic fibrosis, and COPD, are characterized by neutrophil infiltration and the release of proteases such as neutrophil elastase (NE) and MMP-9, which can be harnessed for targeted drug delivery77. Pulmonary administration enables high local drug concentration, rapid action, and bypasses the first-pass effect, making enzyme-responsive micro-/nanoparticles an attractive strategy for site-specific treatment in the inflamed lung78.
PEG-based hydrogel microparticles degradable by MMPs overexpressed in pulmonary diseases were developed by Secret et al.79. PEG diacrylate (PEGDA) precursors were synthesized via an inverse emulsion technique, yielding particles 2.8–4 μm in diameter, within the optimal aerodynamic range (0.5–5 μm) for deep-lung delivery. Enzyme-responsiveness was achieved by incorporating an MMP-specific peptide into the polymer backbone, with degradability confirmed in vitro; further studies are needed to assess biocompatibility, mucoadhesion, and in vivo performance. More recently, Mejías et al. designed a “Nano-in-Microgel” system via maleimide-thiol Michael addition during a water-in-oil emulsion, avoiding UV, heat, or organic solvents that can denature biologics80. Trypsin- and neutrophil elastase-responsive microgels were fabricated using different enzyme-sensitive cross-linkers, with particle diameters tunable from approximately 4–8 μm by adjusting macromer concentration. Trypsin exposure triggered rapid degradation within 30 min, and in vitro studies showed that particles >6 μm with hydrophilic surfaces avoided rapid clearance by alveolar macrophages. In mice, these particles exhibited sustained lung retention before clearance, demonstrating promising pulmonary stability. The platform was further validated by loading Nexinhib20, a neutrophil degranulation inhibitor, and incorporating an NE-degradable peptide; in vitro and in vivo results showed particle degradation in NE-rich airway fluid within 30 min and effective attenuation of neutrophilic inflammation77.
Dental
Dental tissues are prone to infection, and it is estimated that tooth decay affects half of the worldwide population81. If tooth extraction can be avoided, a well-established alternative is the removal of the decayed tissues and substitution with a biomaterial. These treatments, depending on the severity of the injury, range from pulp capping, in which only dentine and enamel are removed, to pulpotomies and pulpectomies, in which partial and complete removal of the pulp tissue are performed, respectively. A wide variety of different biomaterials can be used to replace dental tissues, ranging from bioinert cements and resins to bioactive silicate-based materials82. As infection is a major concern, many of these biomaterials incorporate enzyme-responsive characteristics to facilitate on-demand release of antibacterial agents while minimizing the potential negative side effects of burst release of antibacterials.
Ribeiro et al. synthesized a gelatin methacryloyl hydrogel that degrades in response to endodontic infection-associated MMPs (MMP-1, -2, -8, and -9)83. The hydrogel was functionalized with chlorhexidine-loaded nanotubes for the delivery of antibiotics upon contact with elevated MMP concentrations. Another example of bacterial enzyme-specific degradation is the gingipain-degradable hydrogel synthesized by Liu et al84. The hydrogel consisted of a PEGDA scaffold, dithiothreitol cross-linker, and a functional peptide module. The functional peptide module was designed with a short antimicrobial peptide core, such that after gingipain-specific cleavage, the short antimicrobial peptide was released from the hydrogel for antibacterial effect.
Dermal
Biomaterials designed for dermal applications focus on wound healing, with most research emphasizing the need for treatment of chronic wounds resulting from diabetes mellitus or vascular diseases. In these conditions, tissue repair is hindered by a prolonged state of inflammation. Instead of progressing through the phases of inflammation, new tissue formation, and remodeling, there is an imbalance of proteases and reactive oxygen species which leads to degradation of essential growth factors, impaired angiogenesis, and insufficient cellular recruitment to the wound site. This eventually results in skin ulcer formation and often leads to infection85. As such, much of the research in dermal biomaterials focuses on employing enzyme-responsive hydrogels, scaffolds, and nanomaterials to accelerate the body’s innate healing mechanisms.
Protease-degradable hydrogels for release of growth factors or inflammation inhibitors have been widely reported. For instance, a strategy by Tokatlian et al. used MMP-degradable HA hydrogels to release non-viral pro-angiogenic plasmids to upregulate VEGF at the wound site86. This solution aims to circumvent any issues with burst-release of growth factors that may arise from direct encapsulation of VEGF. However, further work is needed to optimize their approach, as the transfection efficiency of the VEGF plasmids was not high enough to show significant differences in angiogenesis. A similar approach by Zhao et al. utilized MMP-9 activity to deliver Gp1a (a cannabinoid receptor agonist) sequestered in self-assembling triglycerol monostearate hydrogels. This showed promise in skin wound healing to limit inflammation, inhibit fibrogenesis, and promote re-epithelialization87.
A different strategy was used by Wang et al. in 2017, in which the presence of the enzyme itself promoted wound-healing. They synthesized a wound healing patch consisting of an ε-poly-lysine (EPL) based hydrogel able to self-heal in the presence of plasma amine oxidase (PAO)88. The EPL network was first cross-linked by radical polymerization using 1-vinyl-2-pyrrolidinone, then PAO was added to induce the formation of Schiff base pairs by oxidizing free amines on the EPL network. The formation of a dual-cross-linked system with a combination of permanent and dynamic bonds to make a wound healing patch was found to accelerate skin healing due to PAO promoting collagen fiber formation. This novel approach was shown to be effective for in vivo acceleration of wound closure in mice.
Other organs
Colon
Inflammatory bowel disease (IBD) refers to symptoms such as severe stomach pain and diarrhea, the main types of which include Crohn’s disease and ulcerative colitis89. Although the cause of IBD is still unclear, the molecular insight of local inflammation has been explored for discovery of inflammation-targeting therapies. Similar to many applications discussed, biomaterials that are responsive to inflamed-colon-specific enzymes have been designed for targeted drug delivery. Zhang et al. designed a hydrogel microfiber that can preferentially adhere to the inflamed tissue and deliver anti-inflammatory corticosteroid dexamethasone (Dex) upon enzyme digestion90. From in vitro to in vivo mouse colitis model results, Dex-loaded hydrogels led to a significant decrease in inflammation and lower Dex peak serum concentrations, indicating less systemic drug exposure. This platform shows promise for targeted enema-based therapies in colonic IBD patients.
Intestine
As mentioned above, IBD involves inflammation in parts of gastrointestinal (GI) tract. Drug delivery vehicles can be locally delivered to the inflamed intestine relatively easily by rectal administration yet these result in poor patient compliance90,91,92. Oral administration, on the contrary, showed the best patient compliance. However, the successful delivery of drugs to the inflamed site through the GI tract, particularly the prevention of premature drug release, remains extremely challenging93.
Knipe et al. designed a multi-responsive poly(methacrylic acid-co-N-vinylpyrrolidone) hydrogel cross-linked with an arginine- and lysine-rich oligopeptide selectively degraded by trypsin in the small intestine94. The hydrogel remained collapsed under acidic gastric conditions, retaining loaded insulin, and swelled at neutral pH to facilitate release. Trypsin-specific, concentration-dependent degradation was confirmed by turbidity assays and incubation with rat gastrointestinal fluids, with no response to pepsin. The pH-responsive backbone modulated enzyme access, enabling controlled intestinal degradation, and the system showed high biocompatibility with negligible cytotoxicity. Similarly, Szilágyi et al. developed a poly(aspartic acid) hydrogel with trypsin-responsive tetrapeptide cross-links and a pH-sensitive backbone; degradation products were non-cytotoxic, and encapsulated FITC-dextran was released upon trypsin digestion95. Li et al. reported a HA-functionalized porous silicon nanoparticle (AP@PSi-HA@HPMCAS) combining enzyme-responsive hydrogel and pH-responsive polymer for local delivery to inflamed intestinal tissue93. The hierarchical “nano-in-nano-in-micro” structure protected embedded drug-loaded hydrogels, while negatively charged nanoparticles targeted inflamed areas. Ascorbyl palmitate and budesonide were co-loaded via in situ gelation in mesopores, then encapsulated in hydroxypropyl methylcellulose acetate succinate. In vitro and in vivo studies demonstrated reduced systemic exposure and prolonged, enzyme-triggered local release, suggesting the potential of hierarchical nanoparticle-hydrogel systems for treating gastrointestinal inflammation.
Liver
Liver diseases are a main cause of mortality and morbidity worldwide, affecting over 1.3 billion people globally96. Early and exact diagnosis or identification are critical to prevent and treat hepatic diseases, as well as control their progression, thereby reducing illness and death.
A preliminary example of enzyme-responsive hepatic disease modeling is the LOX responsive PEG hydrogel developed by Tirella et al.69. As LOX plays a key role in several ECM-stiffening processes, it was harnessed to stiffen PEGDA hydrogels, which could then act as 3D hepatic disease models, as they matched the stiffness and creep behavior of native healthy and diseased liver tissues. As such, these functionalized hydrogels could be used to mimic changes in the tissue microenvironment which arise upon inflammation and other pathological changes at early stages of fibrosis and cancer. Furthermore, they can be used to study cell mechanobiology and responses to dynamic variation of hydrogels stiffness.
A therapeutic approach developed by Stevens et al. also made use of PEG hydrogels, this time in the form of PEG-diacrylamide with a pan-MMP degradable linkage in the hydrogel backbone97. The construct was used to encapsulate hepatocytes to form a 3D implantable TE scaffold for liver regeneration. By replacing hydrolytically unstable esters in PEGDA with amides, PEG-diacrylamide hydrogels resisted non-specific hydrolysis, while still allowing for MMP-mediated hydrogel degradation.
Generalized inflammation and tissue nonspecific applications
Enzyme-responsive biomaterials have also been developed for generalized inflammation beyond disease- or tissue-specific applications. Inflammation, whether acute or chronic, is a critical immune response that supports tissue repair, remodeling, and regeneration98. Although inflammatory processes vary by pathology, they share common features, such as upregulated inflammation-related enzymes, that can be exploited as therapeutic targets.
During both acute and chronic inflammation, enzymes such as hydrolases, elastases, cathepsins, and MMPs are released by monocytes and macrophages64. Despite their different sources, the enzyme targets employed by enzyme-responsive biomaterials due to their overexpression in tissues fall into two major categories: proteases and hydrolases. MMPs, a family of proteases, are the most studied enzymatic cues in stimuli-responsive material design. In a study by Lueckgen et al., an alginate-based hydrogel for promoting cell spreading and in vivo tissue infiltration was fabricated by modifying alginate with norbornene moieties, which were subsequently cross-linked with two MMP-cleavable peptide cross-linkers using UV-initiated thiol-ene chemistry (Fig. 4a)99. 3D encapsulation of embryonic fibroblasts in hydrogels showed extensive filopodia formation, and implantation of hydrogels showed high cell infiltration, suggesting potential application in tissue regeneration. Another interesting MMP-responsive hydrogel is a drug-eluting hydrogel developed by Fries et al., which features depots of tacrolimus encapsulated within an MMP-degradable triglycerol monostearate scaffold100. Tacrolimus is an immunosuppressant used to prevent implant rejection in vascularized composite allotransplantation. Due to its unpredictable absorption when taken orally, on-demand release of tacrolimus at the graft site may prove to be a useful strategy for controlled drug delivery.
a (i) Fabrication scheme of MMP-degradable, peptide-cross-linked alginate hydrogel. Bis-cysteine peptides cross-link with norbornene-modified alginate via thiol-ene chemistry upon UV exposure in the presence of a photoinitiator. Cysteine-conjugated RGD peptides can be co-incorporated, and the cross-linkers can be engineered for enzymatic degradability. (ii) In vivo cell infiltration into enzymatically degradable peptide-cross-linked alginate hydrogels after 8 weeks of subcutaneous implantation. Hydrogels cross-linked with 3 mg/mL degradable or control cross-linkers were stained with Hematoxylin and Eosin. Scale bar = 100 μm. Cell counts were conducted within the total hydrogel area (including both fiber-rich and fiber-free regions) and within fiber-free regions alone. Adapted from Biomaterials, Lueckgen et al.99. b (i) EGCG conjugated hyaluronic acids and tyramine conjugated hyaluronic acids cross-link via tyrosinase catalysis to form tissue adhesives that can be injected in wound areas. (ii) In vitro and in vivo sealing test results of the HA_TE hydrogel compared with commercial cyanoacrylate and fibrin glue. Adapted from Biomaterials, Kim et al.103. Created in BioRender. Pi, P. (2025) https://BioRender.com/oc1tu63.
In addition to MMPs, other enzymes were also harnessed to deliver encapsulated drugs from hydrogels. For example, Ma et al. developed a polypeptide-based hydrogel whose amide bonds can be degraded by trypsin, an enzyme that may activate inflammatory cells, such as eosinophils during inflammation101. They loaded DOX and diclofenac as model drugs in the hydrogel, their release was observed in response to both pH and enzyme. The hydrogels not only showed high resistance to nonspecific protein adsorption and attachment of Human umbilical vein endothelial cells but also delivered model drugs doxorubicin hydrochloride and diclofenac sodium in a temporal manner. Another example of therapeutic delivery from a hydrogel is an elastase-responsive, H2S-releasing hydrogel developed by Zhou et al.102. The hydrogel is composed of carboxymethylcellulose and PEG, which are cross-linked with an elastase-degradable peptide functionalized with an H2S-releasing S-aroylthiooxime unit. The hydrogel can undergo a sol-gel transition with elastase catalysis, exposing S-aroylthiooxime and releasing H2S. They investigated the H2S-releasing profiles of hydrogels with varying stiffnesses and the cytoprotective property from doxorubicin, which suggests the potential application in chronic inflammation.
Besides drug delivery, enzymes were also harnessed to induce desired mechanical properties of hydrogel such as adhesiveness and stiffness. In a study by Kim et al., a tissue adhesive and anti-inflammatory hydrogel was developed by forming high-affinity enzymatic cross-linking of polyphenolic Epigallocatechin gallates (EGCGs) (Fig. 4b)103. HA was functionalized with EGCGs and tyramine, respectively, to form HA_E and HA_T, which can rapidly cross-link in the presence of tyrosinase. Specifically, the 1,2,3-trihydroxyphenyl group in EGCG has a high affinity to tyrosinase, which allows HA_E to be oxidized and cross-linked with HA_T to form an adhesive hydrogel (HA_TE). Through both in vitro adhesion test with mouse dorsal skin tissues and in vivo mouse skin incision wound closure model, they showed that compared to commercial products such as cyanoacrylate and fibrin glue, which lose their adhesiveness under wet conditions, HA_TE hydrogel displayed stronger adhesion. In vitro tissue necrosis factor-alpha level and in vivo monocytes and macrophage levels also indicated that HA_TE hydrogel induced less immunogenicity than the commercial products.
Novel concepts
Two complementary directions have emerged in recent years: the design of non-tissue-specific enzyme-responsive biomaterials and the engineering of cells that secrete enzymes in response to defined triggers. Applications span biosensors for infection detection, platforms to prolong graft and implant lifetime, and cell-based systems capable of dynamic, environment-guided therapeutic responses. Several enzyme-responsive platforms have been developed as probes. For example, Guo et al. reported a luminescent nanoplatform responsive to elevated myeloperoxidase in inflammation, enabling real-time monitoring of disease progression104. Similarly, Ebrahimi et al. constructed a chitosan-based hydrogel bearing fluorogenic, enzyme-cleavable substrates to sensitively detect α-chymotrypsin, with a detection limit <10 nM105. Chromogenic probes responsive to lysozyme or β-glucuronidase have also been reported for infection detection106,107.
In addition to sensing applications, enzyme-instructed supramolecular hydrogels offer programmable assemblies well-suited for therapeutic use. A study by Webber et al. demonstrated that peptide amphiphiles incorporating a protein kinase A substrate sequence undergo phosphorylation-induced nanofibre disassembly and reassembly upon dephosphorylation, enabling enzymatically triggered drug release with cancer selectivity108. A subsequent study by Zhou et al. expanded enzyme-instructed self-assembly strategies: phosphoserine-containing peptides were shown to undergo phosphatase-mediated dephosphorylation, driving hydrogelation, while dual incorporation of phosphotyrosine provided additional triggers and enhanced stability109. More recently, Shy et al. reported that stereoisomeric phosphopentapeptides form biocompatible supramolecular hydrogels upon enzymatic dephosphorylation, with stereochemistry and sequence shown to govern dephosphorylation kinetics, proteolytic resistance, and cell compatibility110. To address the limited strength of such soft assemblies, Wei et al. developed a dual-enzyme responsive hybrid hydrogel that integrates supramolecular self-assembly with enzyme-mediated covalent cross-linking, yielding a tougher and more porous network111. Collectively, these examples underscore the versatility of supramolecular strategies in engineering enzyme-responsive hydrogels with tunable assembly, stability, and mechanical performance.
Another relevant emerging concept is engineering cells to secrete enzymes in response to triggers, ranging from biomaterial to molecular or cellular signals. SynNotch receptors, pioneered by Morsut et al., established a modular framework to couple ligand recognition to customized outputs such as secretion, and Roybal et al. showed this could be applied in primary T cells to drive local release of therapeutic factors112,113. Garibyan et al. extended the approach by presenting synNotch ligands on biomaterials, pointing to the possibility of material-guided secretion, while Synthetic Enzyme-Armed KillER Chimeric Antigen Receptor-T platforms demonstrated how engineered immune cells can conditionally secrete prodrug-activating enzymes within the tumor microenvironment114,115. Looking ahead, integrating these strategies with responsive biomaterials could enable programmable therapeutic systems in which enzyme release is precisely coordinated with local environment.
Outlook
The majority of strategies discussed in this review focus on enzyme-cleavable hydrogels for delivery of therapeutics, with a particular emphasis on MMP-degradable hydrogels. While effective and adaptable, the novelty of such approaches is limited by the high volume of existing research. By contrast, the lesser-investigated fields of enzyme-responsive biomaterials as probes or for modeling disease states show considerable promise and are expected to expand. Moreover, relatively little work exploits the intrinsic biological and chemical activity of biomaterials themselves as therapeutics, making this an interesting area for future exploration.
Irrespective of application, several overarching challenges remain. A key issue is the overlapping substrate specificity among enzyme families, which can lead to off-target responses. More rational and selective designs are needed to achieve tissue- and pathology-specific activation. In addition to evaluating biocompatibility and long-term cytotoxicity, future studies should also consider the broader impact of enzyme-responsive materials on local tissue homeostasis, particularly in chronic or dynamic disease environments. Another critical challenge lies in the limited understanding of enzyme activity under physiological and pathological conditions, which hampers the design of materials with predictable response kinetics. As discussed in Box 2, sparse quantitative data on enzyme concentrations and activity in target tissues, especially in acute settings such as MI, make it difficult to replicate relevant conditions in vitro and to control degradation in vivo.
Looking forward, progress is likely to come from more sophisticated strategies, including multi-step responses and feedback-controlled mechanisms beyond single-trigger systems. Multi-step cascades, such as tandem GOx/HRP reactions within hydrogels shown in a previous study, demonstrate how sequential enzyme activity could generate more precise and tunable responses116. Feedback-controlled systems, where material degradation exposes new sites or alters local conditions to modulate subsequent enzyme activity, could provide a route to auto-regulated kinetics. Artificial cells or synthetic compartments loaded with enzymes could add another layer of sophistication, integrating sensing, processing, and actuation. Such strategies directly address the challenges outlined in Box 2, offering ways to achieve predictable and adaptive behavior in vivo.
References
Sánchez-Cid, P., Jiménez-Rosado, M., Romero, A. & Pérez-Puyana, V. Novel trends in hydrogel development for biomedical applications: a review. Polymers 14, 3023 (2022).
Bustamante-Torres, M. et al. Hydrogels classification according to the physical or chemical interactions and as stimuli-sensitive materials. Gels 7, 182 (2021).
Ahmed, E. M. Hydrogel: preparation, characterization, and applications: a review. J. Adv. Res. 6, 105–121 (2015).
Li, Z., Zhou, Y., Li, T., Zhang, J. & Tian, H. Stimuli-responsive hydrogels: fabrication and biomedical applications. VIEW 3, 20200112 (2022).
Xie, R., Zheng, W., Guan, L., Ai, Y. & Liang, Q. Engineering of hydrogel materials with perfusable microchannels for building vascularized tissues. Small 16, 1902838 (2020).
Yahia, L. H. History and applications of hydrogels. J. Biomed. Sci. https://doi.org/10.4172/2254-609X.100013 (2015).
Correa, S. et al. Translational applications of hydrogels. Chem. Rev. 121, 11385–11457 (2021).
Hatakeyama, H. Recent advances in endogenous and exogenous stimuli-responsive nanocarriers for drug delivery and therapeutics. Chem. Pharm. Bull. 65, 612–617 (2017).
Stavrou, B. M., Beck, C. & Flores, N. A. Changes in extracellular pH and myocardial ischaemia alter the cardiac effects of diadenosine tetraphosphate and pentaphosphate. Br. J. Pharm. 134, 639–647 (2001).
Shaibani, P. M., Etayash, H., Naicker, S., Kaur, K. & Thundat, T. Metabolic study of cancer cells using a pH sensitive hydrogel nanofiber light addressable potentiometric sensor. ACS Sens. 2, 151–156 (2017).
Liu, Q., Yuan, Z., Guo, X. & Esch, J. H. Dual-functionalized crescent microgels for selectively capturing and killing cancer cells. Angew. Chem. Int. Ed. 59, 14076–14080 (2020).
Zhao, H. et al. ROS-scavenging hydrogel to promote healing of bacteria infected diabetic wounds. Biomaterials 258, 120286 (2020).
Sobczak, M. Enzyme-responsive hydrogels as potential drug delivery systems—state of knowledge and future prospects. Int. J. Mol. Sci. 23, 4421 (2022).
Jiang, H. et al. Tough and electro-responsive hydrogel actuators with bidirectional bending behavior. Nanoscale 11, 2231–2237 (2019).
Liu, W. et al. A temperature responsive adhesive hydrogel for fabrication of flexible electronic sensors. NPJ Flex. Electron. 6, 68 (2022).
Xing, Y., Zeng, B. & Yang, W. Light responsive hydrogels for controlled drug delivery. Front. Bioeng Biotechnol. https://doi.org/10.3389/fbioe.2022.1075670 (2022).
McDonald, A. G. & Tipton, K. F. Enzyme nomenclature and classification: the state of the art. FEBS J. 290, 2214–2231 (2023).
Ulijn, R. V. Enzyme-responsive materials: a new class of smart biomaterials. J. Mater. Chem. 16, 2217–2225 (2006).
Solanki, R. & Bhatia, D. Stimulus-responsive hydrogels for targeted cancer therapy. Gels 10, 440 (2024).
Omer, A. M. et al. Advances in stimuli-responsive polymeric hydrogels for anticancer drug delivery: a review. J. Drug Deliv. Sci. Technol. 102, 106394 (2024).
Zhou, X.-Y. et al. Recent research progress on tumour-specific responsive hydrogels. J. Mater. Chem. B 12, 7246–7266 (2024).
Qin, J. et al. Stimuli-responsive hydrogels for cancer immunotherapy. Polym. Chem. 14, 793–802 (2023).
World Health Organization. Cardiovascular Diseases (CVDs) (2025).
Pamukcu, B. In Myocardial Infarction (IntechOpen, 2019).
Phatharajaree, W., Phrommintikul, A. & Chattipakorn, N. Matrix metalloproteinases and myocardial infarction. Can. J. Cardiol. 23, 727–733 (2007).
NAGASE, H., VISSE, R. & MURPHY, G. Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc Res. 69, 562–573 (2006).
Matsumura, S. et al. Targeted deletion or pharmacological inhibition of MMP-2 prevents cardiac rupture after myocardial infarction in mice. J. Clin. Investig. 115, 599–609 (2005).
Fan, C. et al. Myocardial-infarction-responsive smart hydrogels targeting matrix metalloproteinase for on-demand growth factor delivery. Adv. Mater. 31, 1902900 (2019).
Carlini, A. S. et al. Enzyme-responsive progelator cyclic peptides for minimally invasive delivery to the heart post-myocardial infarction. Nat. Commun. 10, 1–14 (2019).
Wang, L. L. et al. Injectable and protease-degradable hydrogel for siRNA sequestration and triggered delivery to the heart. J. Contr. Release 285, 152–161 (2018).
Purcell, B. P. et al. Injectable and bioresponsive hydrogels for on-demand matrix metalloproteinase inhibition. Nat. Mater. 13, 653–661 (2014).
Paulis, L. E. et al. Distribution of lipid-based nanoparticles to infarcted myocardium with potential application for MRI-monitored drug delivery. J. Contr. Release 162, 276–285 (2012).
Suarez, S., Almutairi, A. & Christman, K. L. Micro- and nanoparticles for treating cardiovascular disease. Biomater. Sci. 3, 564–580 (2015).
Chang, M.-Y. et al. Functionalized nanoparticles provide early cardioprotection after acute myocardial infarction. J. Contr. Release 170, 287–294 (2013).
Berardi, G. R. M. et al. Transplantation of SNAP-treated adipose tissue-derived stem cells improves cardiac function and induces neovascularization after myocardium infarct in rats. Exp. Mol. Pathol. 90, 149–156 (2011).
Tsai, E. J. & Kass, D. A. Cyclic GMP signaling in cardiovascular pathophysiology and therapeutics. Pharm. Ther. 122, 216–238 (2009).
Yao, X. et al. Nitric oxide releasing hydrogel enhances the therapeutic efficacy of mesenchymal stem cells for myocardial infarction. Biomaterials 60, 130–140 (2015).
Van Hove, A. H., Burke, K., Antonienko, E., Brown, E. & Benoit, D. S. W. Enzymatically-responsive pro-angiogenic peptide-releasing poly(ethylene glycol) hydrogels promote vascularization in vivo. J. Contr. Release 217, 191–201 (2015).
Silva, E. A. & Mooney, D. J. Effects of VEGF temporal and spatial presentation on angiogenesis. Biomaterials 31, 1235–1241 (2010).
Murphy, W. L., Peters, M. C., Kohn, D. H. & Mooney, D. J. Sustained release of vascular endothelial growth factor from mineralized poly(lactide-co-glycolide) scaffolds for tissue engineering. Biomaterials 21, 2521–2527 (2000).
Phelps, E. A., Landázuri, N., Thulé, P. M., Taylor, W. R. & García, A. J. Bioartificial matrices for therapeutic vascularization. Proc. Natl. Acad. Sci. USA 107, 3323–3328 (2010).
Moore, A. N. & Hartgerink, J. D. Self-assembling multidomain peptide nanofibers for delivery of bioactive molecules and tissue regeneration. Acc. Chem. Res. 50, 714–722 (2017).
da Silva, L. P. et al. Gellan gum hydrogels with enzyme-sensitive biodegradation and endothelial cell biorecognition sites. Adv. Healthc. Mater. https://doi.org/10.1002/ADHM.201700686 (2018).
Zhang, K. et al. A nitric oxide-releasing hydrogel for enhancing the therapeutic effects of mesenchymal stem cell therapy for hindlimb ischemia. Acta Biomater. 113, 289–304 (2020).
World Health Organization. Musculoskeletal conditions. https://www.who.int/news-room/fact-sheets/detail/musculoskeletal-conditions (2022).
Hunter, D. J. & Bierma-Zeinstra, S. Osteoarthritis. Lancet 393, 1745–1759 (2019).
Buckwalter, J. A., Mankin, H. J. & Grodzinsky, A. J. Articular Cartilage and Osteoarthritis (2005).
Redman, S. N., Oldfield, S. F., Archer, C. W., Roughley, P. J. & Lee, C. Current strategies for articular cartilage repair. Eur. Cell Mater. 9, 23–32 (2005).
Armiento, A. R., Alini, M. & Stoddart, M. J. Articular fibrocartilage—Why does hyaline cartilage fail to repair?. Adv. Drug Deliv. Rev. 146, 289–305 (2019).
Francis, S. L., Di Bella, C., Wallace, G. G. & Choong, P. F. M. Cartilage tissue engineering using stem cells and bioprinting technology—barriers to clinical translation. Front. Surg. 5, 70 (2018).
Peppas, N. A. & Kavimandan, N. J. Nanoscale analysis of protein and peptide absorption: insulin absorption using complexation and pH-sensitive hydrogels as delivery vehicles. Eur. J. Pharm. Sci. 29, 183–197 (2006).
Salinas, C. N. & Anseth, K. S. The enhancement of chondrogenic differentiation of human mesenchymal stem cells by enzymatically regulated RGD functionalities. Biomaterials 29, 2370–2377 (2008).
Bryant, S. J., Bender, R. J., Durand, K. L. & Anseth, K. S. Encapsulating chondrocytes in degrading PEG hydrogels with high modulus: Engineering gel structural changes to facilitate cartilaginous tissue production. Biotechnol. Bioeng. 86, 747–755 (2004).
Appelman, T. P., Mizrahi, J., Elisseeff, J. H. & Seliktar, D. The influence of biological motifs and dynamic mechanical stimulation in hydrogel scaffold systems on the phenotype of chondrocytes. Biomaterials 32, 1508–1516 (2011).
Rubert Pérez, C. M., Panitch, A. & Chmielewski, J. A collagen peptide-based physical hydrogel for cell encapsulation. Macromol. Biosci. 11, 1426–1431 (2011).
Ifkovits, J. L. & Burdick, J. A. Photopolymerizable and degradable biomaterials for tissue engineering applications. Tissue Eng. 13, 2369–2385 (2007).
Oztürk, E. et al. Tyrosinase-crosslinked, tissue adhesive and biomimetic alginate sulfate hydrogels for cartilage repair. Biomed. Mater. https://doi.org/10.1088/1748-605X/ab8318 (2020).
Parmar, P. A. et al. Collagen-mimetic peptide-modifiable hydrogels for articular cartilage regeneration. Biomaterials 54, 213 (2015).
Wang, E. D. Tendon repair. J. Hand Ther. 11, 105–110 (1998).
Maffulli, N., Moller, H. D. & Evans, C. H. Tendon healing: can it be optimised?. Br. J. Sports Med. 36, 315–316 (2002).
Parry, D. A. D., Barnes, G. R. G. & Craig, A. S. A comparison of the size distribution of collagen fibrils in connective tissues as a function of age and a possible relation between fibril size distribution and mechanical properties. Proc. R. Soc. Lond. B Biol. Sci. 203, 305–321 (1978).
Lim, W. L., Liau, L. L., Ng, M. H., Chowdhury, S. R. & Law, J. X. Current progress in tendon and ligament tissue engineering. Tissue Eng. Regen. Med. 16, 549–571 (2019).
Dogramaci, Y. & Duman, I. G. Reinforcement of the flexor tendon repair using human amniotic membrane a biomechanical evaluation using the modified kessler method of tendon repair. J. Am. Podiatr. Med. Assoc. 106, 319–322 (2016).
Cai, C. et al. Self-healing hydrogel embodied with macrophage-regulation and responsive-gene-silencing properties for synergistic prevention of peritendinous adhesion. Adv. Mater. 34, 2106564 (2021).
Cai, C. et al. MMP-2 responsive unidirectional hydrogel-electrospun patch loading TGF-β1 siRNA polyplexes for peritendinous anti-adhesion. Adv. Funct. Mater. https://doi.org/10.1002/ADFM.202008364 (2021).
Roddy, E., DeBaun, M. R., Daoud-Gray, A., Yang, Y. P. & Gardner, M. J. Treatment of critical-sized bone defects: clinical and tissue engineering perspectives. Eur. J. Orthop. Surg. Traumatol. https://doi.org/10.1007/s00590-017-2063-0 (2018).
Stroncek, J. D. & Reichert, W. M. In Indwelling Neural Implants: Strategies for Contending with the In Vivo Environment 3–38 (2008).
Dimitriou, R., Tsiridis, E. & Giannoudis, P. V. Current concepts of molecular aspects of bone healing. Injury 36, 1392–1404 (2005).
Tirella, A., Mattei, G., La Marca, M., Ahluwalia, A. & Tirelli, N. Functionalized enzyme-responsive biomaterials to model tissue stiffening in vitro. Front. Bioeng. Biotechnol. https://doi.org/10.3389/FBIOE.2020.00208/FULL (2020).
Stevenson, S. Enhancement of fracture healing with autogenous and allogene. Clin. Orthop. Relat. Res. https://journals.lww.com/clinorthop/Fulltext/1998/10001/Enhancement_of_Fracture_Healing_With_Autogenous.24.aspx (2022).
Gaston, M. S. & Simpson, A. H. R. W. Inhibition of fracture healing. J. Bone Joint Surg. Br. 89, 1553–1560 (2007).
Hsu, C. W., Olabisi, R. M., Olmsted-Davis, E. A., Davis, A. R. & West, J. L. Cathepsin K-sensitive poly(ethylene glycol) hydrogels for degradation in response to bone resorption. J. Biomed. Mater. Res. A 98, 53–62 (2011).
Morgan, J. E. & Partridge, T. A. Muscle satellite cells. Int. J. Biochem. Cell Biol. 35, 1151–1156 (2003).
Brancaccio, P., Lippi, G. & Maffulli, N. Biochemical markers of muscular damage. Clin. Chem. Lab Med. 48, 757–767 (2010).
Cai, L., Xiong, X., Kong, X. & Xie, J. The role of the lysyl oxidases in tissue repair and remodeling: a concise review. Tissue Eng. Regen. Med. 14, 15 (2017).
Grasman, J. M., Zayas, M. J., Page, R. L. & Pins, G. D. Biomimetic scaffolds for regeneration of volumetric muscle loss in skeletal muscle injuries. Acta Biomater. 25, 2–15 (2015).
Mejías, J. C. et al. Neutrophil-targeted, protease-activated pulmonary drug delivery blocks airway and systemic inflammation. JCI Insight. https://doi.org/10.1172/jci.insight.131468 (2019).
He, S. et al. A roadmap to pulmonary delivery strategies for the treatment of infectious lung diseases. J. Nanobiotechnol. 20, 101 (2022).
Secret, E., Kelly, S. J., Crannell, K. E. & Andrew, J. S. Enzyme-responsive hydrogel microparticles for pulmonary drug delivery. ACS Appl. Mater. Interfaces 6, 10313–10321 (2014).
Mejías, J. C. & Roy, K. In-vitro and in-vivo characterization of a multi-stage enzyme-responsive nanoparticle-in-microgel pulmonary drug delivery system. J. Contr. Release 316, 393–403 (2019).
James, S. L. et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 Diseases and Injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392, 1789–1858 (2018).
Medina-Fernandez, I. & Celiz, A. D. Acellular biomaterial strategies for endodontic regeneration. Biomater. Sci. 7, 506–519 (2019).
Ribeiro, J. S. et al. Injectable MMP-responsive nanotube-modified gelatin hydrogel for dental infection ablation. ACS Appl. Mater. Interfaces 12, 16006–16017 (2020).
Liu, S. et al. Gingipain-responsive thermosensitive hydrogel loaded with SDF-1 facilitates in situ periodontal tissue regeneration. ACS Appl. Mater. Interfaces 13, 36880–36893 (2021).
Blakytny, R. & Jude, E. The molecular biology of chronic wounds and delayed healing in diabetes. Diabet. Med. 23, 594–608 (2006).
Tokatlian, T., Cam, C. & Segura, T. Porous hyaluronic acid hydrogels for localized non-viral DNA delivery in a diabetic wound healing model. Adv. Health. Mater. 4, 1084 (2015).
Zhao, C. et al. An enzyme-responsive Gp1a-hydrogel for skin wound healing. J. Biomater. Appl. 36, 714–721 (2021).
Wang, R. et al. Enzyme-induced dual-network ε-poly-L-lysine-based hydrogels with robust self-healing and antibacterial performance. Chem. Commun. 53, 4803–4806 (2017).
Saeid Seyedian, S., Nokhostin, F. & Dargahi Malamir, M. A review of the diagnosis, prevention, and treatment methods of inflammatory bowel disease. J. Med. Life 12, 113–122 (2019).
Zhang, S. et al. An inflammation-targeting hydrogel for local drug delivery in inflammatory bowel disease. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aaa5657 (2015).
Date, A. A. et al. Local enema treatment to inhibit FOLH1 /GCPII as a novel therapy for inflammatory bowel disease. J. Contr. Release 263, 132–138 (2017).
Herrera Estrada, L. et al. Bioengineering bacterially derived immunomodulants: a therapeutic approach to inflammatory Bowel disease. ACS Nano 11, 9650–9662 (2017).
Li, W. et al. Hierarchical structured and programmed vehicles deliver drugs locally to inflamed sites of intestine. Biomaterials 185, 322–332 (2018).
Knipe, J. M., Chen, F. & Peppas, N. A. Enzymatic biodegradation of hydrogels for protein delivery targeted to the small intestine. Biomacromolecules 16, 962–972 (2015).
Szilágyi, B. Á et al. Amino acid based polymer hydrogel with enzymatically degradable cross-links. React. Funct. Polym. 133, 21–28 (2018).
Wang, F. S., Fan, J. G., Zhang, Z., Gao, B. & Wang, H. Y. The global burden of liver disease: the major impact of China. Hepatology 60, 2099–2108 (2014).
Stevens, K. R., Miller, J. S., Blakely, B. L., Chen, C. S. & Bhatia, S. N. Degradable hydrogels derived from PEG-diacrylamide for hepatic tissue engineering. J. Biomed. Mater. Res. A 103, 3331–3338 (2015).
Hotamisligil, G. S. Inflammation, metaflammation and immunometabolic disorders. Nature 542, 177–185 (2017).
Lueckgen, A. et al. Enzymatically-degradable alginate hydrogels promote cell spreading and in vivo tissue infiltration. Biomaterials 217, 119294 (2019).
Fries, C. A. nton et al. Graft-implanted, enzyme responsive, tacrolimus-eluting hydrogel enables long-term survival of orthotopic porcine limb vascularized composite allografts: a proof of concept study. PLoS ONE 14, e0210914 (2019).
Ma, G. et al. Development of ionic strength/pH/enzyme triple-responsive zwitterionic hydrogel of the mixed L-glutamic acid and L-lysine polypeptide for site-specific drug delivery. J. Mater. Chem. B 5, 935–943 (2017).
Zhou, M., Qian, Y., Zhu, Y. & Matson, J. Elastase-triggered H 2 S delivery from polymer hydrogels. Chem. Commun. 56, 1085–1088 (2020).
Kim, S. H. et al. Fabrication of polyphenol-incorporated anti-inflammatory hydrogel via high-affinity enzymatic crosslinking for wet tissue adhesion. Biomaterials 242, 119905 (2020).
Guo, J. et al. A myeloperoxidase-responsive and biodegradable luminescent material for real-time imaging of inflammatory diseases. Mater. Today 20, 493–500 (2017).
Sadat Ebrahimi, M. M. & Schönherr, H. Enzyme-sensing chitosan hydrogels. Langmuir 30, 7842–7850 (2014).
Tallian, C. et al. Lysozyme-responsive spray-dried chitosan particles for early detection of wound infection. ACS Appl. Bio Mater. 2, 1331–1339 (2019).
Das, D. et al. Enzyme-responsive biopolymeric nanogel fibers by extrusion: engineering of high-surface-area hydrogels and application in bacterial enzyme detection. ACS Appl. Mater. Interfaces 13, 12928–12940 (2021).
Webber, M. J., Newcomb, C. J., Bitton, R. & Stupp, S. I. Switching of self-assembly in a peptide nanostructure with a specific enzyme. Soft Matter. 7, 9665 (2011).
Zhou, J., Du, X., Wang, J., Yamagata, N. & Xu, B. Enzyme-instructed self-assembly of peptides containing phosphoserine to form supramolecular hydrogels as potential soft biomaterials. Front. Chem. Sci. Eng. 11, 509–515 (2017).
Shy, A. N., Li, J., Shi, J., Zhou, N. & Xu, B. Enzyme-instructed self-assembly of the stereoisomers of pentapeptides to form biocompatible supramolecular hydrogels. J. Drug Target 28, 760–765 (2020).
Wei, Q. et al. Printable hybrid hydrogel by dual enzymatic polymerization with superactivity. Chem. Sci. 7, 2748–2752 (2016).
Morsut, L. et al. Engineering customized cell sensing and response behaviors using synthetic Notch receptors. Cell 164, 780–791 (2016).
Roybal, K. T. et al. Engineering T cells with customized therapeutic response programs using synthetic notch receptors. Cell 167, 419–432.e16 (2016).
Garibyan, M. et al. Engineering programmable material-to-cell pathways via synthetic notch receptors to spatially control differentiation in multicellular constructs. Nat. Commun. 15, 5891 (2024).
Gardner, T. J. et al. Engineering CAR-T cells to activate small-molecule drugs in situ. Nat. Chem. Biol. 18, 216–225 (2022).
Tan, H. et al. Heterogeneous multi-compartmental hydrogel particles as synthetic cells for incompatible tandem reactions. Nat. Commun. 8, 663 (2017).
Cai, D. et al. Chitosan-capped enzyme-responsive hollow mesoporous silica nanoplatforms for colon-specific drug delivery. Nanoscale Res. Lett. 15, 123 (2020).
Das, D., Alhusaini, Q. F. M., Kaur, K., Raoufi, M. & Schönherr, H. Enzyme-responsive biopolymeric nanogel fibers by extrusion: engineering of high-surface-area hydrogels and application in bacterial enzyme detection. ACS Appl. Mater. Interfaces 13, 12928–12940 (2021).
Wang, S. et al. Nanoenzyme-reinforced injectable hydrogel for healing diabetic wounds infected with multidrug resistant bacteria. Nano Lett. 20, 5149–5158 (2020).
Acknowledgements
A.D.C. acknowledges a UKRI Future Leaders Fellowship (MR/S034757/1).
Author information
Authors and Affiliations
Contributions
A.D.C. conceptualized this work and supervised writing and revising of Y.P. and K.G. Y.P. and K.G. contributed equally to this work, including initial writing, graphing, and revising.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Materials thanks Mohit Kumar and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pi, Y., Ganabady, K. & Celiz, A.D. Enzyme-responsive biomaterials for biomedical applications. Commun Mater 6, 263 (2025). https://doi.org/10.1038/s43246-025-00983-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43246-025-00983-0