Abstract
Background
Polycystic ovary syndrome (PCOS) is a complex endocrine disorder and a leading cause of infertility. When accompanied by anti-thyroid peroxidase antibodies (TPOAb), PCOS patients may face an increased risk of reproductive complications. However, the impact of TPOAb on assisted reproductive technology (ART) outcomes in PCOS remains unclear. Assessing the impact of TPOAb levels on embryologic and pregnancy outcomes in IVF/ICSI cycles among euthyroid PCOS patients is crucial for identifying potential risks and refining clinical management strategies to improve reproductive outcomes.
Methods
This retrospective cohort study includes PCOS patients selected using standard diagnostic codes, strict inclusion and exclusion criteria, and propensity score matching (PSM) at a 1:4 ratio. A total of 62 TPOAb-positive PCOS patients are matched with 216 TPOAb-negative PCOS patients undergoing IVF/ICSI treatment. The primary outcomes assessed are clinical pregnancy rate and live birth rate, while secondary outcomes include other pregnancy-related outcomes and embryo quality parameters.
Results
Here we show that the TPOAb-positive group has significantly higher fertilization rate, normal fertilization (two-pronuclei, 2PN) rate, and 2PN cleavage rate after PSM (all P < 0.05). However, multivariate analyses adjusting for propensity scores reveal no significant associations between TPOAb status and any embryologic indicators. Additionally, there are no statistically significant differences between the groups in pregnancy outcomes (all P > 0.05).
Conclusions
TPOAb positivity is not associated with adverse embryonic or pregnancy outcomes in euthyroid PCOS patients undergoing IVF/ICSI treatment. Therefore, the presence of TPOAb alone should not be overemphasized, nor does it warrant overtreatment in this patient population.
Plain Language Summary
Polycystic ovary syndrome (PCOS) is a common cause of infertility. People with PCOS have irregular periods and over produce male hormones. The presence of anti-thyroid peroxidase antibodies (TPOAb), proteins that neutralise the effect of a component important for producing thyroid hormone, can be used as an indicator of thyroid problems. TPOAb levels have also been associated with an increased risk of PCOS complications. We investigated whether the presence of TPOAb impacted outcomes during in vitro fertilization or intracytoplasmic sperm injection (IVF/ICSI) among euthyroid people with PCOS, who are those that are diagnosed with PCOS and have normal thyroid-stimulating hormone (TSH) levels. The results show that the presence of TPOAb is not associated with adverse impacts on the embryo or pregnancy. This highlights that the presence of TPOAb does not indicate the need for additional medical care for euthyroid people with PCOS during IVF/ICSI.
Similar content being viewed by others
Introduction
Polycystic ovarian syndrome (PCOS) is an endocrine disorder that predominantly affects women of reproductive age1. This condition is characterized by a variety of symptoms, including irregular menstrual cycles, hyperandrogenism (excess male hormones), and polycystic ovaries2. As a common issue in the fields of obstetrics and gynecology, reproductive medicine, endocrinology, and metabolism, PCOS presents a significant challenge for healthcare providers and patients alike. According to estimates, the prevalence rate of PCOS ranges from 9% to 19.9%, which translates to over 100 million women worldwide being affected by this condition3. In addition to its hallmark symptoms, PCOS is associated with a range of metabolic and reproductive complications4.
Women with PCOS have reduced reproductive potential, which may result from abnormal oocyte development5, decreased endometrial competence6, and potentially impaired endometrial-embryo cross-talk7. Research shows that in women with PCOS exhibit dysregulation of sex hormone receptors and co-receptors expression in the endometrium8, as well as increased endometrial insulin resistance with impaired glucose transport and utilization9, leading to chronic low-grade inflammation10, immune dysfunction11, abnormal endometrial gene expression and cellular abnormalities6,12.
Beyond infertility, PCOS is also linked to an elevated risk of pregnancy complications13. Meta-analyses demonstrate that women with PCOS have a significantly increased risk of ovarian hyperstimulation syndrome, miscarriage, pregnancy-induced hypertension, gestational diabetes mellitus, and preterm birth compared with non-PCOS controls14,15. In the context of assisted reproductive technology (ART), including in vitro fertilization and embryo transfer (IVF-ET), PCOS introduces additional challenges. Patients often exhibit either an inadequate or exaggerated response to ovarian stimulation, reduced oocyte quality, abnormal fertilization, and impaired embryo development, all of which hinder optimal ART outcomes16,17. Given the hormonal imbalances and metabolic disturbances inherent to PCOS, individualized treatment strategies are essential to improve the chances of successful conception and pregnancy outcomes18.
In addition, PCOS patients frequently exhibit immune abnormalities, such as thyroid autoimmunity (TAI). The prevalence of TAI in PCOS patients is ~18−40%, about three times higher than in non-PCOS patients19,20,21,22. Compared with either TAI or PCOS alone, TAI combined with PCOS carries a higher risk of metabolic and reproductive complications23,24. Moreover, TAI is associated with a poor response to infertility treatment in patients with PCOS25. Studies investigating the correlation between TAI and pregnancy outcomes find that women with TAI have an increased risk of miscarriage and preterm birth26,27. However, the associations between TAI and pregnancy outcomes in ART remain controversial. Some studies observe that the presence of thyroid autoantibodies negatively influences the clinical pregnancy rate and is associated with higher miscarriage and lower live birth rate, resulting in poorer IVF outcomes28,29. Conversely, other studies show that TAI in infertile patients does not affect fertilization, embryo quality, or pregnancy outcomes, including implantation, clinical pregnancy, miscarriage, preterm delivery, and live birth30,31. In consequence, the interplay between TAI and PCOS in the context of ART outcomes remains unresolved, highlighting the need for further research to clarify these associations and develop targeted interventions to improve reproductive success in this subgroup.
In this study, we evaluate the relationship between anti-thyroid peroxidase antibodies (TPOAb), an marker of TAI, and embryologic or pregnancy outcomes of in vitro fertilization or intracytoplasmic sperm injection (IVF/ICSI) in a cohort of infertile women diagnosed with PCOS and euthyroidism. We show that TPOAb status are not significantly relevant to any of the key embryologic outcomes, including mature oocyte rate, fertilization rate, normal fertilization (two-pronuclei, 2PN) rate, 2PN cleavage rate, high-quality cleavage-stage embryo rate, blastocyst formation rate, high-quality blastocyst rate, or available blastocyst rate, in euthyroid PCOS patients. We also find that TPOAb levels are not significantly associated with biochemical pregnancy, clinical pregnancy, miscarriage (early or late), or live birth in TPOAb-positive and TPOAb-negative PCOS patients.
Methods
Study design and population
This retrospective cohort study was conducted at the Assisted Reproductive Centre of Shandong Provincial Maternal and Child Health Care Hospital affiliated with Qingdao University, and Sichuan Jinxin Xinan Women and Children’s Hospital from October 2014 to September 2022. The study protocol was approved by the Ethics Committees of both hospitals (Approval No. 2023-77; 202104). To ensure patient privacy, all data were anonymized, and the ethics committees waived the requirement for written informed consent due to the minimal risk associated with the use of de-identified data. Clinical and IVF/ICSI characteristic data were retrieved from electronic medical records. The flowchart of inclusion and exclusion of research participants was shown in Fig. 1.
TPOAb: anti-thyroid peroxidase antibodies. TPOAb-positive is defined as a serum anti-thyroid peroxidase antibodies concentration ≥9 U/mL. TPOAb-negative is defined as a serumanti-thyroid peroxidase antibodies concentration <9 IU/mL. n: biologically independent samples. IVF in vitro fertilization, ICSI intracytoplasmic sperm injection, TSH thyroid-stimulating hormone, PCOS polycystic ovary syndrome, IVM oocyte in vitro maturation, PGT preimplantation genetic testing, PSM propensity score matching.
Patients were included in this study if they met the following criteria: (1) females patients aged 20–45 years old; (2) their first oocyte retrieval cycle resulted in clinical pregnancy or not after a fresh embryo transfer using autologous sperm and oocytes; (3) they had undergone TPOAb testing, had thyroid-stimulating hormone (TSH) concentrations ≤4 μIU/mL at the initial evaluation, were classified as euthyroid, did not receive pharmacological treatment, and were managed in accordance with clinical guidelines and consensus for infertility or pregnancy32,33,34; (4) they were diagnosed with PCOS according to the 2003 Rotterdam ESHRE/ASRM PCOS Consensus Workshop Group criteria.
Patients were excluded if they met any of the following: (1) history of thyroid disease, thyroid hormone/antithyroid medication, or thyroid surgery; (2) other autoimmune disease; (3) recurrent spontaneous abortion; (4) either partner had a chromosomal abnormality; (5) diagnosis of endometriosis or uterine abnormality.
Thyroid function measurement
Patients were tested for thyroid function before IVF/ICSI treatment. Thyroid function parameters, including serum TSH, free triiodothyronine (FT3), free thyroxine (FT4), total triiodothyronine (T3), total thyroxine (T4) and TPOAb, were measured by chemiluminescent immunoassay (UniCel DxI 800, Beckman Coulter, CA, USA), as previously described35. The TPOAb level ≥9 IU/mL was defined as TPOAb-positive36.
Controlled ovarian stimulation and embryo transfer
The controlled ovarian stimulation (COS) protocols included the gonadotropin-releasing hormone (GnRH) antagonist protocol, the long follicular phase protocol, and other protocols such as mild and luteal-phase stimulation. Details of these protocols were described in a previous study37. Ovulation induction was carried out using a dual trigger with human chorionic gonadotropin (hCG) or a GnRH agonist (GnRH-a), followed by transvaginal oocyte retrieval. Fertilization was performed via IVF or ICSI, depending on sperm quality. Fresh embryo transfer was conducted on day 3 or day 5 after oocyte retrieval.
Outcome measures and definitions
The primary outcomes were the live birth rate and clinical pregnancy rate. Secondary outcomes included additional pregnancy-related measures, such as biochemical pregnancy rate and miscarriage rate, as well as laboratory indicators of embryologic quality. These embryologic indicators included mature oocyte rate, fertilization rate, 2PN fertilization rate, 2PN cleavage rate, high-quality cleavage-stage embryo rate, blastocyst formation rate, high-quality blastocyst rate, and available blastocyst rate.
Live birth rate was defined as the number of live births divided by the number of embryo transfer cycles. Clinical pregnancy was confirmed by transvaginal ultrasound 28 days after embryo transfer, based on gestational sac or fetal heartbeat. Biochemical pregnancy was defined as serum β-human chorionic gonadotropin (β-hCG) >25 U/L, measured 14 days after embryo transfer. Miscarriages were classified as early or late. Early miscarriage was defined as the proportion of spontaneous pregnancy losses occurring before 12 weeks of gestation relative to the total number of clinical pregnancies. Late miscarriage refers to losses occurring between 12 and 28 weeks of gestation.
Embryo and blastocyst assessments were performed based on the Istanbul Consensus and Gardner blastocyst grading system38,39. Mature oocyte rate was calculated as metaphase II (MII) oocytes divided by retrieved oocytes during ICSI. Fertilization rate was defined as 1PN, 2PN and 3PN embryos, as well as cleavage-stage embryos derived from 0PN zygotes, divided by retrieved oocytes (IVF) or mature oocytes (ICSI). The 2PN fertilization rate was calculated as 2PN embryos divided by retrieved (IVF) or mature oocytes (ICSI). The 2PN cleavage rate was defined as the number of cleavage-stage embryos from 2PN divided by the total 2PNs. High-quality cleavage-stage embryo rate was defined as high-quality cleavage-stage embryos divided by normal fertilized embryos (2PN embryos). Blastocyst formation rate was calculated as total blastocysts formed divided by 2PN embryos. The available blastocyst rate was defined as blastocysts suitable for cryopreservation or transfer divided by 2PN embryos. High-quality blastocyst rate was defined according to the Gardner blastocyst grading system as high-quality blastocysts divided by 2PN embryos40,41.
Statistics and reproducibility
All statistical analyses were performed using SPSS version 27.0 and R version 4.2.3. The Kolmogorov-Smirnov test was used to assess normality assumptions for continuous variables. Baseline patient characteristics across different groups were summarized as follows: categorical data were presented as frequencies (%); normally distributed continuous data were expressed as mean ± standard deviation (SD), while non-normally distributed data were presented as median (interquartile range, IQR). Comparisons in continuous variables were conducted using independent sample t-tests or Mann-Whitney U tests, as appropriate. Categorical variables were compared with the chi-square test. The effect of TPOAb positivity on embryo quality and clinical outcomes was assessed using generalized linear models (GLM). Continuous outcomes were analyzed using an identity link function, while binomial outcomes were evaluated with a logistic link function. Results were reported as β coefficients with corresponding 95% confidence intervals (CI) for continuous outcomes, and odds ratios (OR) with their respective 95% CI for binomial outcomes. A P-value < 0.05 was considered statistically significant for all tests.
Propensity score matching (PSM)
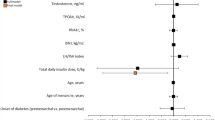
To reduce confounding bias associated with these outcomes, we perform propensity score matching (PSM), a robust method for reducing bias in observational cohort studies. Variables in the model included body mass index (BMI), TSH, T3, basal follicle-stimulating hormone (FSH), Gn dosage and antral follicle count (AFC). TPOAb-positive and TPOAb-negative populations were matched using nearest neighbor matching at a 1:4 ratio, with a caliper value of 0.2.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Results
Baseline characteristics
Patients were categorized based on TPOAb status into TPOAb-positive (n = 64) and TPOAb-negative (n = 497) groups (Supplementary Data 1). Prior to PSM, the TPOAb-positive group showed significantly different baseline characteristics compared to the TPOAb-negative group, including higher levels of BMI, TSH, T3, FSH, and Gn dosage. Following PSM, baseline characteristics were well balanced between the TPOAb-positive (n = 62) and TPOAb-negative (n = 216) groups. No significant differences were observed in female age, BMI, infertility duration, thyroid function parameters, hormone levels, AFC, Gn dosage, COS protocol, infertility type, or fertilization type.
Embryo quality
The comparison of embryo quality between the TPOAb-positive and TPOAb-negative groups was shown in Table 1. After PSM, the TPOAb-positive group showed significantly higher fertilization rate, 2PN fertilization rate, and 2PN cleavage rate compared to the TPOAb-negative group (83.13% vs 79.36%, P = 0.023; 73.55% vs 68.63%, P = 0.010; 98.35% vs 96.70%, P = 0.046). However, there were no significant differences in the mature oocyte rate, high-quality cleavage-stage embryo rate, blastocyst formation rate, high-quality blastocyst rate, or available blastocyst rate between the two groups (all P > 0.05).
To further address potential confounding factors and baseline characteristic imbalances, we adjusted for propensity scores and calculated adjusted β-values for the correlation between TPOAb levels and embryo quality in TPOAb-positive and TPOAb-negative PCOS patients (Table 1). The results indicated that TPOAb levels were not significantly associated with mature oocyte rate, fertilization rate, 2PN fertilization rate, 2PN cleavage rate, high-quality cleavage-stage embryo rate, blastocyst formation rate, high-quality blastocyst rate, or available blastocyst rate in TPOAb-positive and TPOAb-negative PCOS patients (all P > 0.05).
Pregnancy outcomes and live birth rate
The comparison of pregnancy outcomes between the TPOAb-positive and TPOAb-negative groups was presented in Table 2. After PSM, no statistically significant differences were observed between the two groups in terms of biochemical pregnancy rate, clinical pregnancy rate, miscarriage rate (including both early and late miscarriage), or live birth rate (all P > 0.05).
Furthermore, the results of the multivariate analyses, adjusted for propensity scores, were also summarized in Table 2. These analyses confirmed that TPOAb status was not significantly associated with any of the key pregnancy outcomes, including biochemical pregnancy, clinical pregnancy, miscarriage (early or late), or live birth, in PCOS patients (all P > 0.05).
Discussion
PCOS and thyroid disorders are two prevalent endocrine conditions that often coexist, suggesting a complex interrelationship that influences both metabolic and reproductive health19,42. Notably, the prevalence of TAI is higher in women with PCOS compared to those without. Additionally, subclinical hypothyroidism (SCH) is more prevalent in women with PCOS compared to the general female population43. The coexistence of PCOS and thyroid disorders may exacerbate metabolic disturbances, such as insulin resistance, and reproductive issues, including ovulatory dysfunction42.
Epidemiological data indicate that TAI is highly prevalent in PCOS patients, with rates approximately three times higher than those observed in non-PCOS patients21. This high prevalence adds complexity to the treatment and management of infertility in women with PCOS. A recent cross-sectional study has found that, as an indicator of TAI, TPOAb level alterations are more prevalent among mothers and babies with PCOS44. Moreover, among patients with elevated levels of TPOAb, the prevalence of hypothyroidism is higher in those with PCOS than in those without PCOS44. Despite these observations, the interplay between TPOAb and PCOS in the context of ART outcomes remains poorly understood and represents an unsolved problem in reproductive medicine23,24.
In this retrospective cohort study, we investigated the association between TPOAb positivity and reproductive outcomes in euthyroid PCOS patients undergoing IVF/ICSI. After balancing baseline characteristics through PSM, we found that TPOAb-positive patients exhibited significantly higher fertilization rate, 2PN fertilization rate, and 2PN cleavage rate compared to their TPOAb-negative counterparts, which warrants a nuanced discussion, given that the existing literature presents mixed findings on the impact of TPOAb on assisted reproductive outcomes. Some research suggests that TPOAb-positive patients show immune dysregulation during ART treatment for COS, which may affect follicle development and egg quality45. Other studies indicate significantly reduced fertilization and high-quality cleavage-stage embryo rates in TPOAb-positive patients46. However, recent studies have found no significant association between TPOAb and fertilization rates or embryo quality47,48. These discrepancies imply that the influence of TPOAb on reproductive outcomes may be context-dependent and potentially modulated by underlying conditions such as PCOS. One possible explanation is that TPOAb positivity, by modulating the local immune microenvironment, might transiently promote oocyte competence and early fertilization events. This hypothesis aligns with the notion that mild immune activation can enhance folliculogenesis and oocyte maturation. Nonetheless, further mechanistic and molecular studies are needed to clarify these potential immunological effects and determine their clinical relevance49.
Importantly, the observed differences in fertilization-related parameters lost statistical significance after adjusting for confounding variables in multivariate analyses. This indicated that TPOAb status was not an independent predictor of embryo quality in this patient population. Our results were in line with previous studies reporting no adverse effects of TPOAb on oocyte maturation, fertilization, or early embryonic development in euthyroid women undergoing ART47,48. The transient increases noted before adjustment may have reflected residual confounding or biological variability rather than a true effect of TPOAb positivity. Moreover, the absence of significant differences in other key embryologic indicators, such as mature oocyte rate, high-quality cleavage-stage embryo rate, and blastocyst formation rate, further supported the conclusion that TPOAb positivity did not meaningfully influence embryologic competence in IVF/ICSI cycles among euthyroid PCOS patients.
Several studies have consistently linked TPOAb positivity to adverse pregnancy outcomes in the general population. Meta-analyses have demonstrated increased risks of miscarriage and preterm birth among TPOAb-positive women26,27. Other studies have shown that the abortion rate of TPOAb-positive women is significantly higher than that of TPOAb-negative women27,29,50,51,52. However, the impact of TPOAb on pregnancy outcomes in ART remains controversial. Some studies report significantly lower clinical pregnancy and live birth rates among TPOAb-positive infertility women receiving ART28,50,53, while others find no effect on pregnancy outcomes31,54,55,56. Furthermore, researches indicate that TPOAb positivity does not affect clinical pregnancy and live birth rates in ART patients29,30,57. Additionally, certain studies suggest no difference in miscarriage rates between TPOAb-positive and TPOAb-negative women29. In our study, utilizing a strict PSM method to adjust for confounders, we found no significant correlation between TPOAb positivity and adverse pregnancy outcomes in euthyroid PCOS patients. Specifically, our analysis showed no difference in biochemical pregnancy rate, clinical pregnancy rate, miscarriage rate, or live birth rate between TPOAb-positive and TPOAb-negative groups. These findings were particularly noteworthy given the extensive adjustments for potential confounders, including age, BMI, duration of infertility, type of infertility, ovarian stimulation protocols, and other clinical parameters.
While our data suggest that TPOAb alone is not a useful biomarker for predicting pregnancy outcomes in this specific population, this raises important questions about the role of alternative or complementary markers. For instance, the presence of thyroglobulin antibodies (TgAb) has been less extensively studied but may also be implicated in immune-mediated reproductive dysfunction. TgAb, often coexisting with TPOAb, could influence the intra-ovarian immune milieu or endometrial receptivity, thereby affecting implantation and early pregnancy development58. Future studies should explore the combined or independent effects of TgAb in PCOS patients undergoing ART to determine whether it serves as a more sensitive or complementary marker. Additionally, it is plausible that a broader spectrum of autoantibodies, not limited to thyroid-specific antibodies, could contribute to reproductive outcomes in PCOS women, such as antinuclear antibodies (ANA), antiphospholipid antibodies (APA), or even cytokine profiles, which may offer deeper insights into immune dysregulation in this population59,60,61. Therefore, a comprehensive immunological assessment, rather than reliance on a single antibody marker like TPOAb, may better inform individualized treatment strategies and improve reproductive prognostication in PCOS patients.
Given the complex pathogenesis of PCOS, clinicians treating euthyroid PCOS patients may not need to prioritize monitoring TPOAb levels during IVF/ICSI treatments. Instead, attention should be directed towards other endocrine, metabolic, and immune factors that may impact embryonic and pregnancy outcomes.
The strength of our study lay in the stringent inclusion criteria we employed. We excluded women with a history of thyroid disease, thyroid hormone or anti-thyroid medication use, thyroid surgery, as well as those with a history of autoimmune diseases or recurrent miscarriages. To address the significant difference in sample sizes between the TPOAb-positive and TPOAb-negative PCOS patients, we performed PSM at a ratio of 1:4 to reduce the effect of confounding bias between the two groups in this observational cohort study.
However, our study had several limitations. First, the lack of a prospective research design may have introduced certain biases. Second, we did not analyze metabolic factors affecting PCOS, such as insulin metabolism, which may play a role in the development of insulin resistance in patients with PCOS. Third, although the study population was stratified into three ovarian stimulation protocol categories, GnRH antagonist protocol, long follicular phase protocol, and other protocols, the limited sample sizes in the latter two groups after PSM restricted meaningful subgroup comparisons.
Conclusion
TPOAb is not a risk factor for embryonic and pregnancy outcomes in PCOS patients with euthyroidism undergoing IVF/ICSI. Therefore, we suggest that for PCOS patients with euthyroidism undergoing IVF/ICSI, a TPOAb-positive indicator should not be overly emphasized, and does not require excessive treatment.
Data availability
The data that support the findings of this study are available in figshare with the identifier (doi:10.6084/m9.figshare.30126034)62.
References
Su, Y. N. et al. Effects of yulin tong bu formula on modulating gut microbiota and fecal metabolite interactions in mice with polycystic ovary syndrome. Front Endocrinol. (Lausanne) 14, 1122709 (2023).
Yang, J. P. et al. Glycyrrhizin ameliorates impaired glucose metabolism and ovarian dysfunction in a polycystic ovary syndrome mouse model. Biol. Reprod. 109, 83–96 (2023).
Macklon, N. S. Polycystic ovary syndrome. BMJ 343, d6407 (2011).
Williams, T., Mortada, R. & Porter, S. Diagnosis and treatment of polycystic ovary syndrome. Am. Fam. Physician 94, 106–113 (2016).
Palomba, S., Daolio, J. & La Sala, G. B. Oocyte competence in women with polycystic ovary syndrome. Trends Endocrinol. Metab 28, 186–198 (2017).
Palomba, S., Piltonen, T. T. & Giudice, L. C. Endometrial function in women with polycystic ovary syndrome: a comprehensive review. Hum. Reprod. Update 27, 584–618 (2021).
Palomba, S. Is fertility reduced in ovulatory women with polycystic ovary syndrome? An opinion paper. Hum. Reprod. 36, 2421–2428 (2021).
Palomba, S., Homburg, R., Santagni, S., La Sala, G. B. & Orvieto, R. Risk of adverse pregnancy and perinatal outcomes after high technology infertility treatment: a comprehensive systematic review. Reprod. Biol. Endocrinol. 14, 76 (2016).
Orostica L. Poblete C. Romero C. & Vega M. Pro-inflammatory markers negatively regulate IRS1 in endometrial cells and endometrium from women with obesity and PCOS. Reprod. Sci. 27, 290–300 (2020).
Alkhuriji, A. F. et al. Association of IL-1beta, IL-6, TNF-alpha, and TGF-beta1 gene polymorphisms with recurrent spontaneous abortion in polycystic ovary syndrome. Dis. Markers 2020, 6076274 (2020).
Liu, S. et al. Evaluation of the endometrial immune status of polycystic ovary syndrome. J. Reprod. Immunol. 144, 103282 (2021).
Eriksson, G. et al. Single-cell profiling of the human endometrium in polycystic ovary syndrome. Nat. Med. 31, 1925−1938 (2025).
Palomba, S. et al. Pregnancy complications in women with polycystic ovary syndrome. Hum. Reprod. Update 21, 575–592 (2015).
Boomsma, C. M. et al. A meta-analysis of pregnancy outcomes in women with polycystic ovary syndrome. Hum. Reprod. Update 12, 673–683 (2006).
Sha, T., Wang, X., Cheng, W. & Yan, Y. A meta-analysis of pregnancy-related outcomes and complications in women with polycystic ovary syndrome undergoing IVF. Reprod. Biomed. Online 39, 281–293 (2019).
Thakre N. & Homburg R. A review of IVF in PCOS patients at risk of ovarian hyperstimulation syndrome. Expert Rev. Endocrinol. Metab. 14, 315–319 (2019).
Xu, X. et al. Development and validation of a prediction model for suboptimal ovarian response in polycystic ovary syndrome (PCOS) patients undergoing GnRH-antagonist protocol in IVF/ICSI cycles. J. Ovarian Res. 17, 116 (2024).
Kotlyar A. M. & Seifer B. D. Women with PCOS who undergo IVF: a comprehensive review of therapeutic strategies for successful outcomes. Reprod. Biol. Endocrinol. 21, 70 (2023).
Romitti M. Fabris V. C. Ziegelmann P. K. Maia A. L. & Spritzer P. M. Association between PCOS and autoimmune thyroid disease: a systematic review and meta-analysis. Endocr Connect 7, 1158–1167 (2018).
Garelli, S. et al. High prevalence of chronic thyroiditis in patients with polycystic ovary syndrome. Eur. J Obstet Gynecol. Reprod. Biol. 169, 248–251 (2013).
Janssen, O. E., Mehlmauer, N., Hahn, S., Offner, A. H. & Gartner, R. High prevalence of autoimmune thyroiditis in patients with polycystic ovary syndrome. Eur. J. Endocrinol. 150, 363–369 (2004).
Petrikova, J., Lazurova, I. & Yehuda, S. Polycystic ovary syndrome and autoimmunity. Eur. J. Intern. Med. 21, 369–371 (2010).
Skrzynska, K. J., Zachurzok, A. & Gawlik, A. M. Metabolic and hormonal profile of adolescent girls with polycystic ovary syndrome with concomitant autoimmune thyroiditis. Front Endocrinol. (Lausanne) 12, 708910 (2021).
Adamska, A. et al. Ovarian reserve and serum concentration of thyroid peroxidase antibodies in euthyroid women with different polycystic ovary syndrome phenotypes. Front Endocrinol. (Lausanne) 11, 440 (2020).
Ott, J. et al. Elevated antithyroid peroxidase antibodies, indicating Hashimoto’s thyroiditis, are associated with the treatment response in infertile women with polycystic ovary syndrome. Fertil Steril 94, 2895–2897 (2010).
Dong, A. C., Morgan, J., Kane, M., Stagnaro-Green, A. & Stephenson, M. D. Subclinical hypothyroidism and thyroid autoimmunity in recurrent pregnancy loss: a systematic review and meta-analysis. Fertil Steril 113, 587–600 (2020).
Thangaratinam, S. et al. Association between thyroid autoantibodies and miscarriage and preterm birth: meta-analysis of evidence. BMJ 342, d2616 (2011).
Litwicka, K., Arrivi, C., Varricchio, M. T., Mencacci, C. & Greco, E. In women with thyroid autoimmunity, does the administration of low-dose prednisolone, compared with no adjuvant therapy, improve in vitro fertilization clinical results? J. Obstet. Gynaecol. Res. 41, 722–728 (2015).
Mintziori, G. et al. Association of TSH concentrations and thyroid autoimmunity with IVF outcome in women with TSH concentrations within normal adult range. Gynecol. Obstet Invest. 77, 84–88 (2014).
Lukaszuk, K. et al. The impact of the presence of antithyroid antibodies on pregnancy outcome following intracytoplasmic sperm injection (ICSI) and embryo transfer in women with normal thyreotropine levels. J. Endocrinol. Invest. 38, 1335–1343 (2015).
Huang, N. et al. Impact of thyroid autoimmunity on in vitro fertilization/intracytoplasmic sperm injection outcomes and fetal weight. Front Endocrinol.12, 698579 (2021).
Alexander, E. K. et al. 2017 guidelines of the American thyroid association for the diagnosis and management of thyroid disease during pregnancy and the postpartum. Thyroid 27, 315–389 (2017).
Ad Hoc Writing Committee for Guidelines on diagnosis and management of thyroid diseases during pregnancy and postpartum, Chinese Society of Endocrinology, Chinese Medical Association, Chinese Society of Perinatology, Chinese Medical Association. Guideline on diagnosis and management of thyroid diseases during pregnancy and postpartum (2nd edition). Chinese J. Endocrinol. Metab. 35, 636−665 (2019).
The Fourth Committee of the Chinese Society of Reproduction Medicine, Chinese Medical Association. Chinese society for reproductive medicine consensus for subclinical hypothyroidism in the infertile female population. Rep. Contracept. 39, 609–621 (2019).
Rao, M. et al. Subclinical hypothyroidism is associated with lower ovarian reserve in women aged 35 years or older. Thyroid 30, 95–105 (2020).
Webster, G. M., Venners, S. A., Mattman, A. & Martin, J. W. Associations between perfluoroalkyl acids (PFAS) and maternal thyroid hormones in early pregnancy: a population-based cohort study. Environ. Res. 133, 338–347 (2014).
Liu, R. L. et al. Association of ambient air pollutant mixtures with IVF/ICSI-ET clinical pregnancy rates during critical exposure periods. Hum. Reprod. Open 2024, hoae51 (2024).
Alpha Scientists In Reproductive Medicine ESIG The istanbul consensus workshop on embryo assessment: proceedings of an expert meeting. Hum. Reprod. 26, 1270–1283 (2011).
Gardner, D. K. & Schoolcraft, W. B. Culture and transfer of human blastocysts. Curr. Opin. Obstet Gynecol. 11, 307–311 (1999).
Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of Embryology. The Istanbul Consensus Workshop on Embryo Assessment: Proceedings of an Expert Meeting. Hum. Reprod. 26, 1270–1283 (2011).
Sun, Q. et al. CSRM consensus on key indicators for quality control in the IVF laboratory. J. Reprod. Med. 27, 836–851 (2018).
Palomba, S., Colombo, C., Busnelli, A., Caserta, D. & Vitale, G. Polycystic ovary syndrome and thyroid disorder: a comprehensive narrative review of the literature. Front Endocrinol. (Lausanne) 14, 1251866 (2023).
Fan, H., Ren, Q., Sheng, Z., Deng, G. & Li, L. The role of the thyroid in polycystic ovary syndrome. Front Endocrinol. (Lausanne) 14, 1242050 (2023).
Feigl, S. et al. Impact of thyroid function on pregnancy and neonatal outcome in women with and without PCOS. Biomedicines 10, 750 (2022).
Miko, E. et al. Characteristics of peripheral blood NK and NKT-like cells in euthyroid and subclinical hypothyroid women with thyroid autoimmunity experiencing reproductive failure. J. Reprod. Immunol. 124, 62–70 (2017).
Steiner, A. Z. et al. Antimullerian hormone as a predictor of natural fecundability in women aged 30−42 years. Obstet Gynecol 117, 798–804 (2011).
Fleming, R., Seifer, D. B., Frattarelli, J. L. & Ruman, J. Assessing ovarian response: antral follicle count versus anti-mullerian hormone. Reprod. Biomed. Online 31, 486–496 (2015).
Bedenk, J., Vrtacnik-Bokal, E. & Virant-Klun, I. The role of anti-mullerian hormone (AMH) in ovarian disease and infertility. J. Assist. Reprod. Genet 37, 89–100 (2020).
Ameho, S. & Klutstein, M. The effect of chronic inflammation on female fertility. Reproduction 169, e240197 (2025).
Poppe, K. et al. Assisted reproduction and thyroid autoimmunity: an unfortunate combination? J. Clin. Endocrinol. Metab. 88, 4149–4152 (2003).
Negro, R. et al. Levothyroxine treatment in thyroid peroxidase antibody-positive women undergoing assisted reproduction technologies: a prospective study. Hum. Reprod. 20, 1529–1533 (2005).
Toulis, K. A. et al. Risk of spontaneous miscarriage in euthyroid women with thyroid autoimmunity undergoing IVF: a meta-analysis. Eur. J. Endocrinol. 162, 643–652 (2010).
Kilic, S. et al. The effect of anti-thyroid antibodies on endometrial volume, embryo grade and IVF outcome. Gynecol. Endocrinol. 24, 649–655 (2008).
Unuane, D. et al. Impact of thyroid autoimmunity on cumulative delivery rates in in vitro fertilization/intracytoplasmic sperm injection patients. Fertil Steril 106, 144–150 (2016).
Unuane, D. et al. Impact of thyroid autoimmunity in euthyroid women on live birth rate after IUI. Hum. Reprod. 32, 915–922 (2017).
Ke, H. et al. Impact of thyroid autoimmunity on ovarian reserve, pregnancy outcomes, and offspring health in euthyroid women following in vitro fertilization/intracytoplasmic sperm injection. Thyroid 30, 588–597 (2020).
Negro, R. et al. Euthyroid women with autoimmune disease undergoing assisted reproduction technologies: the role of autoimmunity and thyroid function. J. Endocrinol. Invest. 30, 3–8 (2007).
Tanska, K., Gietka-Czernel, M., Glinicki, P. & Kozakowski, J. Thyroid autoimmunity and its negative impact on female fertility and maternal pregnancy outcomes. Front Endocrinol. 13, 1049665 (2022).
Hefler-Frischmuth, K. et al. Serologic markers of autoimmunity in women with polycystic ovary syndrome. Fertil Steril 93, 2291–2294 (2010).
Malinowski, A. et al. Antiphospholipid autoantibodies in women treated for infertility. Ginekol. Pol. 71, 1011–1016 (2000).
Omidvar-Mehrabadi, A., Ebrahimi, F., Shahbazi, M. & Mohammadnia-Afrouzi, M. Cytokine and chemokine profiles in women with endometriosis, polycystic ovary syndrome, and unexplained infertility. Cytokine 178, 156588 (2024).
Liu, J., Yang, H., Ding, Y. The effect of TPOAb on pregnancy outcomes among people with polycystic ovary syndrome with euthyroidism undergoing in vitro fertilization. Figshare https://doi.org/10.6084/m9.figshare.30126034.v2 (2025).
Acknowledgements
This work was supported by the Foundation of Shandong Province Maternal and Child Health Association (YJKY2022-009; YJKY2022-033) and the Foundation of Qingdao University (RZ2400004747).
Author information
Authors and Affiliations
Contributions
Jiao-Jing Liu: Conceptualization, Methodology, Formal analysis, Visualization, Validation, Writing—original draft, Writing—review and editing. Li-Yang Zhou: Conceptualization, Methodology, Data curation, Writing—review and editing. Li-Hong Geng: Validation, Project administration. Fang-Di Dong: Methodology, Investigation, Visualization. Li-Juan Fu: Methodology, Validation. Guang-Yan Gu: Validation, Project administration. Na Yu: Investigation, Visualization. Shi-Yu Liu: Methodology, Data curation. Yu-Bin Ding: Methodology, Data curation, Resources, Supervision, Writing—review and editing. Hui-Jun Yang: Conceptualization, Methodology, Data curation, Resources, Supervision, Project administration, Funding acquisition, Writing —original draft, Writing—review and editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks Mara Simopoulou and the other anonymous reviewer(s) for their contribution to the peer review of this work. [A peer review file is available].
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, JJ., Zhou, LY., Geng, LH. et al. The effect of TPOAb on pregnancy outcomes among people with polycystic ovary syndrome with euthyroidism undergoing in vitro fertilization. Commun Med 5, 485 (2025). https://doi.org/10.1038/s43856-025-01173-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-025-01173-2