Abstract
Background
HIV-tuberculosis (HIV-TB) coinfection poses a significant public health challenge among children in high-burden African regions. Most previous transcriptomic studies have concentrated on adults and non-African populations, primarily analyzing gene-level differential expression. This approach overlooks multi-isoform complexity and may obscure both inherent and pathogen-induced intragenic heterogeneity. This multi-center case-control study aimed to identify and characterize the transcript-level landscape of HIV-TB coinfection in children from different African regions.
Methods
We analyzed whole-blood RNA sequencing data from 97 children with and without tuberculosis from Uganda (East Africa) and from Botswana and Eswatini (Southern Africa). Reads were quality-controlled, and low-abundance transcripts filtered out. Differential transcript expression was estimated using models that adjusted for batch, age, and sex, with multiple testing controlled by the Benjamini–Hochberg procedure. Pathway enrichment was performed on the set of differentially expressed transcripts.
Results
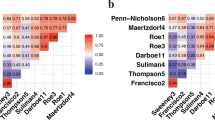
Our analyses show geographic heterogeneity in immune responses; however, the top three gene pathways – immune system, innate immune system, and neutrophil degranulation are consistently conserved across regions. Although there is limited overlap among upregulated transcripts, four of the six shared differentially expressed transcripts (DETs) are enriched in neutrophil degranulation pathways, indicating a conserved transcriptional signature of HIV-TB coinfection. Additionally, we identify five genes with region-specific, non-overlapping isoforms, a distinction not detectable through gene-level analysis.
Conclusions
These findings demonstrate a conserved whole-blood transcriptomic signature in pediatric HIV-TB coinfection, while also highlighting regional variation at the isoform level. This supports the use of transcript-level analyses to identify biomarkers and enhance understanding of host responses in diverse African settings.
Plain language summary
Tuberculosis (TB) is difficult to diagnose in children with HIV, yet they are at a high risk of contracting it. We analyzed gene expression in the blood of children living with HIV with and without TB in Uganda, Botswana, and Eswatini. We focused on transcripts with at least a fourfold increase in abundance in HIV-TB coinfection. While children from different regions had different gene expression, many were involved in similar cellular processes, such as certain parts of the innate immune system, with four of the six shared transcripts. Our findings highlight the conserved nature of the immune response and underscore the need for studies across different regions to better understand these infections in children.
Similar content being viewed by others
Data availability
All source data underlying the graphs and charts presented in the main text and Supplementary Information are available in Figshare under the title “CAfGEN RNA-seq count matrices and supporting processed data for ‘Conserved neutrophil degranulation transcripts in HIV-TB coinfected children across East and Southern Africa’” (https://doi.org/10.6084/m9.figshare.30551264). The RNA-seq datasets generated and analyzed during the current study are being deposited under the project name “CAfGEN RNA-seq Data” into the European Genome-phenome Archive (https://www.ebi.ac.uk/ega/) in accordance with the H3Africa Consortium consensus agreement. Data deposition is being coordinated through H3ABioNet/AfriGen-D, the authorized H3Africa data submission center, in accordance with the NIH H3Africa Data Access and Release Policy. The datasets will become publicly discoverable on the EGA once accession identifiers are issued. Access will be controlled and granted only to bona fide researchers whose proposed use aligns with the participants’ informed consent, subject to approval by the H3Africa Data and Biospecimen Access Committee (DBAC). Requests for access should be submitted via the H3Africa data portal (https://www.h3abionet.org/resources/h3africa-archive). Until the accession number is available, qualified researchers may contact the corresponding author for information on the submission status or to initiate the access request process.
Code availability
Scripts used for data analysis are available at https://github.com/katagirya53.
References
Gelaw, Y. A., Williams, G., Soares Magalhães, R. J., Gilks, C. F. & Assefa, Y. HIV prevalence among tuberculosis patients in sub-Saharan Africa: a systematic review and meta-analysis. AIDS Behav. 23, 1561–1575 (2019).
Kwan, C. & Ernst, J. D. HIV and tuberculosis: a deadly human syndemic. Clin. Microbiol. Rev. 24, 351–376 (2011).
UNAIDS. Global HIV & AIDS Statistics—2024 Fact Sheet. (2024).
World Health Organization. Global Tuberculosis Report. (2024).
Venturini, E. et al. Tuberculosis and HIV co-infection in children. BMC Infect. Dis. 14, 1–10 (2014).
McNerney, R. et al. Tuberculosis diagnostics and biomarkers: needs, challenges, recent advances, and opportunities. J. Infect. Dis. 205, S147–S158 (2012).
Sivakumaran, D. et al. Host blood-based biosignatures for subclinical TB and incipient TB: a prospective study of adult TB household contacts in Southern India. Front. Immunol. 13, 1051963 (2022).
Xu, H., Blair, R. V., Veazey, R. S. & Wang, X. Immunopathogenesis in HIV-associated pediatric tuberculosis. Pediatr. Res. 91, 21 (2021).
Azad, A. K., Lloyd, C., Sadee, W. & Schlesinger, L. S. Challenges of immune response diversity in the human population concerning new tuberculosis diagnostics, therapies, and vaccines. Front. Cell. Infect. Microbiol. 10, 519431 (2020).
Tepekule, B. et al. Transcriptional profile of Mycobacterium tuberculosis infection in people living with HIV. iScience 27, (2024).
Duffy, F. J., Thompson, E. G., Scriba, T. J. & Zak, D. E. Multinomial modelling of TB/HIV co-infection yields a robust predictive signature and generates hypotheses about the HIV+TB+ disease state. PLoS One 14, (2019).
Djebali, S. et al. Landscape of transcription in human cells. Nature 489, 101–108 (2012).
Kalam, H., Singh, K., Chauhan, K., Fontana, M. F. & Kumar, D. Alternate splicing of transcripts upon Mycobacterium tuberculosis infection impacts the expression of functional protein domains. IUBMB Life 70, 845–854 (2018).
Kalam, H., Fontana, M. F. & Kumar, D. Alternate splicing of transcripts shape macrophage response to Mycobacterium tuberculosis infection. PLoS Pathog. 13, e1006236 (2017).
Byun, S., Han, S., Zheng, Y., Planelles, V. & Lee, Y. The landscape of alternative splicing in HIV-1 infected CD4 T-cells. BMC Med. Genomics 13, 1–10 (2020).
Imbeault, M., Giguère, K., Ouellet, M. & Tremblay, M. J. Exon level transcriptomic profiling of HIV-1-infected CD4+ T cells reveals virus-induced genes and host environment favorable for viral replication. PLoS Pathog. 8, e1002861 (2012).
Simmons, J. D. et al. Differentially expressed transcript isoforms associate with resistance to tuberculin skin test and interferon gamma release assay conversion. PLoS One 18, (2023).
Gupta, R. K. et al. Concise whole blood transcriptional signatures for incipient tuberculosis: a systematic review and patient-level pooled meta-analysis. Lancet Respir. Med. 8, 395–406 (2020).
Berry, M. P. R. et al. An interferon-inducible neutrophil-driven blood transcriptional signature in human tuberculosis. Nature 466, 973–977 (2010).
Mboowa, G. et al. Open Peer Review The Collaborative African Genomics Network (CAfGEN): Applying Genomic technologies to probe host factors important to the progression of HIV and HIV-tuberculosis infection in sub-Saharan Africa [version 1; referees: 1 approved]. AAS Open Res. 1, 3 (2018).
Zar, H. J., Workman, L. J., Little, F. & Nicol, M. P. Diagnosis of pulmonary tuberculosis in children: assessment of the 2012 National Institutes of Health Expert Consensus Criteria. Clin. Infect. Dis. 61, S173–S178 (2015).
Graham, S. M. et al. Clinical case definitions for classification of intrathoracic tuberculosis in children: an update. Clin. Infect. Dis. 61, S179–S187 (2015).
Piccini, P., Chiappini, E., Tortoli, E., de Martino, M. & Galli, L. Clinical peculiarities of tuberculosis. BMC Infect. Dis. 14, 1–12 (2014).
WHO. Guidance for national tuberculosis programmes on the management of tuberculosis in children. Second edition. (2014).
Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. http://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (2010).
Ewels, P., Magnusson, M., Lundin, S. & Käller, M. MultiQC: summarize analysis results for multiple tools and samples in a single report. Bioinformatics 32, 3047–3048 (2016).
Chen, S., Zhou, Y., Chen, Y. & Gu, J. Fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34, i884–i890 (2018).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Zhang, Y., Parmigiani, G. & Johnson, W. E. ComBat-seq: Batch effect adjustment for RNA-seq count data. NAR Genomics Bioinforma. 2, (2020).
Szklarczyk, D. et al. The STRING database in 2023: protein-protein association networks and functional enrichment analyses for any sequenced genome of interest. Nucleic Acids Res. 51, D638–D646 (2023).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis. (Springer-Verlag New York, 2016).
Chihota, V. N. et al. Geospatial distribution of Mycobacterium tuberculosis genotypes in Africa. PLoS One 13, (2018).
Nduva, G. M., Nazziwa, J., Hassan, A. S., Sanders, E. J. & Esbjörnsson, J. The role of phylogenetics in discerning hiv-1 mixing among vulnerable populations and geographic regions in sub-Saharan Africa: a systematic review. Viruses 13, 1174 (2021).
Mogashoa, T. et al. Genetic diversity of Mycobacterium tuberculosis strains circulating in Botswana. PLoS One 14, e0216306 (2019).
Micheni, L. N., Kassaza, K., Kinyi, H., Ntulume, I. & Bazira, J. Diversity of Mycobacterium tuberculosis Complex Lineages Associated with Pulmonary Tuberculosis in Southwestern, Uganda. Tuberc. Res. Treat. 2021, 1–6 (2021).
Dlamini, T. C., Mkhize, B. T., Sydney, C., Maningi, N. E. & Malinga, L. A. Molecular investigations of Mycobacterium tuberculosis genotypes among baseline and follow-up strains circulating in four regions of Eswatini. BMC Infect. Dis. 23, 1–13 (2023).
Wampande, E. M. et al. Genetic variability and consequence of Mycobacterium tuberculosis lineage 3 in Kampala-Uganda. PLoS One 14, e0221644 (2019).
Mwesigwa, S. et al. Unmapped exome reads implicate a role for Anelloviridae in childhood HIV-1 long-term non-progression. npj Genom. Med. 6, 1–9 (2021).
Actor, J. K. Lactoferrin: A modulator for immunity against tuberculosis related granulomatous pathology. Mediators Inflamm. 2015, 409596 (2015).
Zupin, L. et al. Association between LTF polymorphism and risk of HIV-1 transmission among Zambian seropositive mothers. Curr. HIV Res. 16, 52–57 (2017).
Hwang, S. A., Arora, R., Kruzel, M. L. & Actor, J. K. Lactoferrin enhances efficacy of the BCG vaccine: comparison between two inbred mice strains (C57BL/6 and BALB/c). Tuberculosis 89, S49–S54 (2009).
Pham, C. T. N. Neutrophil serine proteases: Specific regulators of inflammation. Nat. Rev. Immunol. 6, 541–550 (2006).
Reeves, E. P. et al. Killing activity of neutrophils is mediated through activation of proteases by K+ flux. Nature 416, 291–297 (2002).
Ong, C. W. M. et al. Neutrophil-derived MMP-8 drives AMPK-dependent matrix destruction in human pulmonary tuberculosis. PLoS Pathog. 11, (2015).
Dallenga, T. et al. Targeting neutrophils for host-directed therapy to treat tuberculosis. Int. J. Med. Microbiol. 308, 142–147 (2018).
Remot, A., Doz, E. & Winter, N. Neutrophils and close relatives in the hypoxic environment of the tuberculous granuloma: new avenues for host-directed therapies? Front. Immunol. 10, 439028 (2019).
Scott, N. R. et al. S100A8/A9 regulates CD11b expression and neutrophil recruitment during chronic tuberculosis. J. Clin. Investig. 130, 3098–3112 (2020).
Welin, A. et al. The human neutrophil subsets defined by the presence or absence of OLFM4 both transmigrate into tissue in vivo and give rise to distinct NETs in vitro. PLoS One 8, e69575 (2013).
Kulkarni, V. et al. A two-gene signature for tuberculosis diagnosis in persons with advanced HIV. Front. Immunol. 12, 436 (2021).
Meier, S. et al. Neutrophil degranulation, NETosis and platelet degranulation pathway genes are co-induced in whole blood up to six months before tuberculosis diagnosis. PLoS One 17, e0278295 (2022).
Liu, Y. et al. Impact of alternative splicing on the human proteome. Cell Rep. 20, 1229–1241 (2017).
Liu, H. et al. Correlation between ITGB2 expression and clinical characterization of glioma and the prognostic significance of its methylation in low-grade glioma(LGG). Front. Endocrinol. 13, 1106120 (2023).
Eric, K. CAfGEN DET analysis (v1.0.0). https://doi.org/10.5281/zenodo.17521612 (2025).
Acknowledgements
We would like to acknowledge Nasinghe Emmanuel, Gaseene Sebetso, Thembela Mavuso, Bheki Ntshangase, Buhle Dlamini, Abhilash Sathyamoorthi, Bathusi Mathuba, Yves Mafulu, Gerald Mboowa, Harriet Nakayiza, Edgar Kigozi, Fred Katabazi, Keboletse Mokete, Lesego Ketumile, Kennedy Sichone, Keofentse Mathuba, LeToya Balebetse, Muambi Muyaya, Nancy Zwane, Nicholas Muriithi, Sibongile Mumanga, Thabo Diphoko, Thobile Jele, and Thato Regonamanye. Finally, we acknowledge the families and children who participated in the study. The project described was supported by Award Number U54AI110398, administered by the National Institute of Allergy and Infectious Diseases (NIAID), the Eunice Kennedy Shriver National Institute of Child Health & Human Development (NICHD), and the National Human Genome Research Institute (NHGRI), as part of the NIH Common Fund H3Africa Initiative. Research reported in this publication was supported by the Eunice Kennedy Shriver National Institute of Child Health & Human Development of the National Institutes of Health under Award Number U01HD114479. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Consortia
Contributions
E.K., B.M., S.K., M.W., G.M., A.K., B.N., M.M., G.A., I.K., S.W.M., M.L.J., C.B., G.M.#, and N.H.: conceptualization, writing–review and editing, funding acquisition, investigation, and project administration. E.K., S.K., S.M., M.W., M.A., J.M., A.M., B.N., G.R., B.M., L.W., and K.M.: data curation. E.K., S.K., S.M., and N.H.: formal analysis. G.M., N.H., S.K., M.L.J., D.P.K., C.B., D.K., G.A.: funding acquisition. E.K., S.K., G.K., G.R., B.N., M.M., C.B., G.M., and N.H.: methodology. SK, GK, MD: project administration. G.M., N.H., M.L.J., and E.M.W.: supervision. E.K., S.K., S.M., G.M., M.A., F.A.R., D.K., and N.H.: validation and writing–original draft. The CAfGEN Consortium coordinated participant recruitment, sample processing, and sequencing, and contributed to data curation and quality control. All authors contributed to the article and approved the submitted version. GM = Graeme Mardon and GM# = Gerald Mboowa
Corresponding author
Ethics declarations
Competing interests
Gerald Mboowa is an Editorial Board Member for Communications Medicine but was not involved in the editorial review or peer review, nor in the decision to publish this article.
Peer review
Peer review information
Communications Medicine thanks Gerrit Woltmann and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Katagirya, E., Mlotshwa, B., Kyobe, S. et al. Conserved neutrophil degranulation transcripts in HIV-TB coinfected children across East and Southern Africa. Commun Med (2026). https://doi.org/10.1038/s43856-025-01284-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43856-025-01284-w