Abstract
Background
The population distribution of age-related functional impairments (ARFIs) and their associations with mortality and life expectancy (LE) among Chinese adults remain poorly understood.
Methods
We included 12,906 participants (mean age: 62.6 years) from the China Health and Retirement Longitudinal Study. Visual impairment, hearing impairment, cognitive impairment, sleep disorder, depressive symptoms, and disability in activities of daily living (ADL) were assessed. Cox proportional hazards models were used to estimate the associations of ARFIs with all-cause mortality. Life expectancy at age 50 was estimated by the presence and number of key ARFIs.
Results
The six ARFIs exhibit distinct distributions by ages and provinces across China. During the 9-year follow-up, ADL disability, cognitive impairment, and depressive symptoms are independently associated with 64% (95% confidence interval [CI]: 48%–80%), 41% (21%–64%), and 20% (8%–33%) higher risks of mortality, corresponding to LE losses of 4.45, 3.08, and 1.59 years at the age of 50 years. A greater number of key ARFIs is associated with higher mortality risk in a dose-response manner (hazard ratios [95% CI] vs. none: 1.23 [1.11–1.36] for one, 1.42 [1.26–1.61] for two, and 1.86 [1.47–2.36] for three) and greater LE loss (1.63 [1.35–1.90] years for one, 3.37 [3.02–3.71] for two, and 4.96 [4.22–5.71] for three, with three ARFIs accounting for 17% of total LE).
Conclusions
The study highlights the important roles of co-existing ARFIs in mortality and LE loss. Integrated prevention strategies and management systems for multiple functional impairments are warranted in the context of rapid population aging.
Plain language summary
Age-related functional impairments (ARFIs) are prevalent in aging populations, yet how they relate to death or longevity is uncertain. Global population aging poses major challenges to the public health systems, but the prevalences of ARFIs and their joint associations with life expectancy remained unclear. We studied Chinese adults from age 50 onward and found that inability to carry out normal daily living activities, cognitive impairment, and depressive symptoms are independently associated with an increased risk of death. The number of specific ARFIs was associated with loss in life expectancy in a dose-response manner. Our findings highlight the importance of addressing multiple functional impairments simultaneously. Given the disproportionate burden of ARFIs in socioeconomically disadvantaged regions, integrated and equity-oriented prevention strategies are urgently needed to mitigate health disparities.
Similar content being viewed by others
Introduction
Global population aging poses major challenges to the public health systems, especially for China with one of the largest and growing aging populations. In China, the population aged over 60 years is expected to increase from 254 million (18% of the population) in 2019 to 402 million (28%) in 2040,1,2 which necessitates effective strategies to improve the quality of life in later life. Aging is characterized by multiple functional impairments,3,4 and these age-related functional impairments (ARFIs), such as sensory, physical, mental, and cognitive impairments, often co-exist and are bidirectionally associated with each other.5 While the age-standardized prevalence of some ARFIs remained stable over the past decades,6,7,8 accelerated population aging resulted in increased burdens of these ARFIs (e.g., the prevalence of hearing impairment increased by 27.8% from 1990 to 2019)6,7,8 and highlighted the need to investigate these conditions within an integrated context to facilitate effective prevention. Prior evidence from the Global Burden of Disease also documented a higher prevalence of ARFIs in lower-income regions.6,7 Given China’s rapidly aging population and substantial regional disparities in socioeconomic development, it is critical to understand the distribution and burden of ARFIs and examine regional variations within China to identify vulnerable subpopulations and inform targeted interventions. Importantly, several ARFIs, such as depression, visual impairments, and hearing loss, can be prevented or treated.6,7,9 For instance, depression in older adults can be effectively managed through both behavioral and pharmacotherapeutic approaches.10 Additionally, regular screenings and corrective measures, such as glasses and hearing aids, can help mitigate the progression of visual and hearing impairments and reduce the risk of subsequent ARFIs.
However, the independent and joint roles of the ARFIs in long-term healthy aging remained unclear. First, while it has been widely acknowledged that ARFIs often co-exist, their combined contribution to loss in life expectancy has not been quantified. In previous studies, individual ARFIs have been respectively associated with higher risk of mortality11,12,13,14,15,16,17, but whether all ARFIs are independent contributors is unknown. Understanding their independent and joint associations with mortality risk and loss in life expectancy will help prioritize key ARFIs for targeted prevention in health policy to maximize the public benefits. Second, existing evidence on the associations of ARFIs with healthy aging was limited and mostly based on Western populations18,19, and evidence from Asian populations, especially the aging Chinese population, is rare. Given the differences in genetics, lifestyle, and healthcare accessibility, the distributions of these ARFIs may vary among groups with different sociodemographic and geographic characteristics, and a nuanced examination will provide a basis for targeted intervention implementation. In China, which resides the largest aging population, several studies have characterized the prevalence of and associations between aging phenotypes20,21,22. However, few have compared their prevalence and associations in the same population to facilitate comparison across ARFIs.
In this large-scale, prospective cohort study, we used data from the nationally representative China Health and Retirement Longitudinal Study (CHARLS). Among 12,906 Chinese middle-aged and older adults, we identify clear geographic disparities in the prevalence of six ARFIs, with higher ARFI burdens in economically disadvantaged provinces. We find that ADL disability, cognitive impairment, and depressive symptoms are independently associated with increased mortality risk. The accumulation of specific ARFIs shows a dose-response relation with life expectancy loss. People with co-existing ADL disability, cognitive impairment, and depressive symptoms experience an average of 4.96-year reduction in life expectancy at age 50 compared with those with none.
Methods
Study population
CHARLS is a longitudinal study among middle-aged and older adults in China with national representativeness23. The multistage stratified sampling design based on population size and urbanicity within provinces facilitates reasonable representation at the provincial level. From June 2011 to March 2012, CHARLS recruited 17,708 respondents from 150 counties in 28 provinces of mainland China (overall response rate = 80.5%). In 2013, 2015, 2018, and 2020, the CHARLS conducted four rounds of in-person follow-ups. A detailed description could be found in the cohort profile24. The CHARLS was approved by the Biomedical Ethics Review Committee of Peking University (IRB Protocol: IRB00001052-11015). Written informed consent was obtained from all participants prior to data collection. The present secondary analysis was approved by the Institutional Review Board of the School of Public Health, Zhejiang University (ZGL202101-8). De-identified CHARLS data were obtained from the official CHARLS website after completion of user registration (http://charls.pku.edu.cn).
Among the 17,708 participants recruited in 2011–2012, we excluded 4622 participants aged <50 years at baseline because cognitive assessments were only administered to participants aged 50 years or older. We further excluded 180 participants who had missing data on the six ARFIs of interest (Supplementary Fig. S1). The formal analysis included 12,906 participants at the baseline of CHARLS. For each step of analyses, different numbers of participants were excluded because of the data availability or prevalent cases.
Assessment of age-related functional impairments
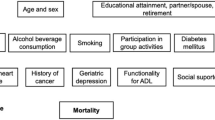
In the current study, we included six ARFIs, including visual impairment, hearing impairment, cognitive impairment, sleep disorder, depressive symptoms, and activities of daily living (ADL) disability, which were prevalent in older populations, potentially associated with mortality in later life, and reliably measured in CHARLS.5 Visual impairment and hearing impairment were defined using self-reports. Participants were asked how good their vision and hearing are in their usual state, with assistive devices if normally used. Participants who reported either fair or poor were classified as having visual or hearing impairment, respectively25,26. Cognitive impairment was defined using the cognitive function score, with a score (range: 0–31)27 set at less than the population mean minus 1.5 times the standard deviation (SD), based on the normative distribution in each wave. The cognitive function test included the serial 7’s test (subtracting 7 from 100 for 5 times, range: 0–5 points), orientation (describing the day, month, year, and the day of the week, range: 0–4 points), picture drawing (copying an assigned picture, range: 0–1 point), and immediate and delayed word recall (10 words for each, range: 0–20 points). According to a previous study, individuals sleeping less than 7 h a night and feeling unrested more than three nights per week were defined as having a sleep disorder28. The score of 10-item Centre for Epidemiologic Studies-Depression (CES-D) scale ≥10 indicated depressive symptoms29. ADL disability was defined as self- or proxy-reported need for help with bathing, dressing, toileting, transferring, or eating30. Details of the definitions were presented in Supplementary Table S1.
Ascertainment of mortality
Survival status of all participants was collected at each follow-up visit of the CHARLS, and all-cause mortality was documented from baseline to the end of follow-up (September 2020). If a respondent’s death was reported, the CHARLS team conducted a comprehensive exit interview with the informant to obtain detailed information. The exact dates of death were only available in waves 2 and 5. For participants who died between these waves, we used the interview date of the reporting wave as the time of death, consistent with definitions used in previous CHARLS studies31,32.
Covariates
We included socioeconomic status, lifestyle factors, and health conditions for confounding adjustments. Age was calculated from the date of birth (ascertained via the registry) to the date of recruitment. Sex was self-reported in the baseline questionnaire. Body mass index (BMI) is derived by dividing the respondent’s measured weight (kg) by the squared value of their measured height.33 Hukou regions (agricultural or non-agricultural, according to household registration), living areas (urban or rural), marital status (married/partnered or not), education level (illiterate, primary school, middle school, or high school or above), annual houshold income ( <10,000, 10,000 to <20,000, 20,000 to <30,000, or ≥30,000 CNY), smoking status (never, former, or current), and alcohol drinking status (never, former, or current) were all self-reported in a structural questionnaire. Participants reported the frequency of moderate-to-vigorous physical activity (MVPA) they did and were classified into three groups (<1 time per month, 1–4 times per month, >4 times per month). Physician diagnoses of health conditions, including stroke, cancer, memory-related diseases, psychiatric conditions, hypertension, diabetes, heart diseases, lung diseases, arthritis, dyslipidaemia, liver diseases, kidney diseases, digestive diseases, and asthma, were all self-reported. Self-reported diagnoses of the health conditions showed moderate-to-high accuracy in previous validation studies in CHARLS and similar cohorts, and the rigorous quality control procedures in CHARLS improved the reliability.34,35,36 The missing rates of covariates were presented in Supplementary Table 2.
Statistics and reproducibility
Baseline characteristics of the study participants (overall and by sex) were described in mean (SD) for continuous variables and number (percentage) for categorical variables. We calculated the prevalence at baseline and incidence rates during the follow-up of the six ARFIs by age and sex, and presented their prevalence by province in 2020. Penalized splines were utilized to assess both linear and non-linear relationships between age and the prevalence and incidence of ARFIs while controlling for overfitting37. We conducted likelihood ratio tests to assess whether the standardized prevalence of ARFIs significantly differs across provinces with adjustments for age and sex. To explore the threshold below which the prevalence of ARFIs disproportionately increases, we used segmented regression approaches, adjusting for the percentage of the population aged over 50 for each province. The Sankey diagram was used to visualize the flow of participants across various categories (number) of functional impairments and mortality status over time, providing an intuitive depiction of the associations38. All descriptive data were sampling-weighted to account for the multi-stage sampling procedure and ensure representativeness of the Chinese population.
We assessed the pairwise longitudinal associations of ARFIs using Cox proportional hazard models adjusted for age, sex, education, married status, Hukou regions, living areas, family income, BMI, smoking status, drinking status, history of stroke, cancer, hypertension, diabetes, and heart diseases, as well as prevalence and incidence of other ARFIs to eliminate mediation or confounding by other ARFIs. Person-time was calculated to the earliest event of interested outcomes, mortality, or loss-to-follow-up. To assess the associations of specific ARFIs with mortality, we first entered all ARFIs into the Cox proportional hazard models with adjustment for covariates as in the main analyses, and ARFIs showing independent associations with mortality were defined as key ARFIs. To demonstrate the associations of co-existing pairs of ARFIs with mortality, we assessed the relations of dyad combinations of ARFIs at baseline with mortality with a similar approach. Furthermore, we assessed associations of the number of key ARFIs (which were significantly associated with mortality) at baseline with mortality risk (from 2011 to 2020).
Using the HRs estimated from the Cox models, we further calculated the life expectancy at the age of 50 and life expectancy loss associated with the specific ARFIs, combinations of ARFIs, and the number of key ARFIs.39 We used mortality incidence data of China from the Global Burden of Disease 2019 40, the age-specific prevalence of co-existing ARFIs generated from the current study, and the HRs associated with the number of ARFIs to construct a life table to calculate the life expectancy and the life expectancy loss. Briefly, we constructed a life table to assume a survival probability of 1 at the age of 50 and calculated the age-specific survival probability using the mortality incidence through 100 years, at which point the survival probability converged to 0. Life expectancy at each age was calculated as the area under the survival probability-age curve after a specific age, and life expectancy loss was calculated as the difference in life expectancy at the age of 50. Parametric Monte Carlo methods were used to calculate the confidence intervals in life expectancy loss, assuming a log-normal distribution of HR and normal distributions of mortality incidence and ARFI prevalence.
We conducted several sensitivity analyses when investigating the ARFI-mortality associations: (1) we additionally adjusted the models for baseline self-rated general health, lung diseases, arthritis, dyslipidaemia, liver diseases, kidney diseases, digestive diseases, and asthma; (2) we additionally adjusted the models for baseline psychiatric conditions and memory-related diseases, which might be correlated with depressive symptoms and cognitive impairment, respectively; and (3) we additionally adjusted the models for MVPA as it might be influenced by ADL disability.
All statistical analyses were performed using R. We handled missing values of the covariates using multiple imputations with chained equations using the R package “mice”41. We conducted 50 iterations with 10 imputed datasets (i.e., 50 × 10) and presented the pooled estimates. Two-sided P-values < 0.05 indicate statistical significance. We applied Bonferroni correction to P-values in each step of analysis.
Results
Baseline characteristics of participants and prevalence of ARFIs
In this study, we analysed data from 12,906 CHARLS participants aged 50 years or older, with complete data on all six ARFIs from 150 counties in 28 provinces of mainland China in 2011–2012. Of these participants, the weighted mean age was 63.2 years (standard deviation [SD]: 9.0 years), 50.6% were women, 30.9% were illiterate, 29.2% were current smokers, 27.8% were current drinkers, and 40.7% were classified as overweight or obese (Table 1). The study population reflects the geographical distribution of Chinese adults aged 50 years or older, as shown in Supplementary Table S3. There were no substantial differences in characteristics between included and excluded participants, except for a higher proportion of urban residents among those excluded (Supplementary Table S4). Among the participants included in the study, those who were lost to follow-up had a greater number of baseline ARFIs (mean: 1.17 vs. 0.98, Supplementary Table S5).
The baseline prevalence was 35.8% for depressive symptoms, 26.2% for sleep disorder, 19.0% for ADL disability, 10.7% for hearing impairment, 8.8% for cognitive impairment, and 6.9% for visual impairment. At baseline, compared with men, women had a higher prevalence of visual impairment (7.8% versus 5.9%), cognitive impairment (12.0% versus 6.1%), sleep disorder (31.9% versus 20.3%), depressive symptoms (43.2% versus 28.2%), and ADL disability (21.6% versus 16.2%), but not hearing impairment (10.6% versus 10.9%). The prevalence of all ARFIs increased with age and was the highest among individuals aged 85 years and older, except for visual impairment, which peaked in individuals aged 81–85 (Supplementary Table S6).
The prevalence of ARFIs in 2020 showed distinct disparities across provinces (Fig. 1 and Supplementary Table S7, P-values for heterogeneity <0.001 for all ARFIs). Visual and hearing impairments were more prevalent in Northern than Southern China. This trend was exemplified by the prevalence of visual impairment of 7.4% in Gansu versus 0.9% in Jiangsu, similar to that of hearing impairment (7.9% in Inner Mongolia versus 1.9% in Zhejiang). Sleep disorder (40.0% in Hubei versus 22.6% in Fujian), depressive symptoms (59.3% in Gansu versus 29.8% in Zhejiang), and ADL disability (30.9% in Guangxi versus 12.4% in Zhejiang) demonstrated the higher prevalence in less developed regions. Segmented regression analysis revealed potential threshold effects of GDP per capita on the prevalence of ARFIs (Supplementary Table S8 and Fig. S2). Statistically significant breakpoints (Bonferroni corrected P-values < 0.05) were identified for cognitive impairment (~85.1 thousand CNY), depressive symptoms (~43.7 thousand CNY), and ADL disability (~64.6 thousand CNY). The prevalence of cognitive impairment, depressive symptoms, and ADL disability showed different slopes before and after these breakpoints.
ADL: activities of daily living. All descriptive data were sampling-weighted to account for the multi-stage sampling procedure and ensure representativeness of the Chinese population. Panels A to F presented the national distribution of the prevalence for visual impairment (A), hearing impairment (B), cognitive impairment (C), sleep disorders (D), depressive symptoms (E), and ADL disability (F). All age-related functional impairment demonstrated statistically significant differences across provinces (P-values for heterogeneity were 1.3 × 10−7, 2.8 × 10−7, 8.8 × 10−55, 5.3 × 10−18, 2.3 × 10−69, and 5.9 × 10−16 for visual impairment, hearing impairment, cognitive impairment, sleep disorders, depressive symptoms, and ADL disability) under likelihood ratio test. Gray indicated no or insufficient data (<200 participants) for the province in 2020. The map was colored based on the standard map provided as GS (2022)1061 without shape modification.
Incidence and co-existing progression of ARFIs
During the study follow-up from 2011 to 2020 (median [interquartile], 9 [4, 9] years), depressive symptoms had the highest incidence rate (70.7 cases per 1000 person-years), followed by ADL disability (61.8), sleep disorder (58.7), hearing impairment (34.7), visual impairment (24.3), and cognitive impairment (21.9) (Supplementary Fig. S2). Before the age of 70, the incidence rate of depressive symptoms was the highest, but in older age groups, it was surpassed by ADL disability, a trend observed in both women and men. In men, the incidence rates of sleep disorders and depressive symptoms exhibited less prominent or non-significant increases with increasing age (P-values for linear trend = 0.035 and 0.775, respectively), whereas incidence of other ARFIs significantly increased with age (P-value for linear trend<0.05, Supplementary Table S9). Women participants had higher incidence rates of visual impairment (27.8 versus 20.7 cases per 1000 person-years), cognitive impairment (26.8 versus 18.2), sleep disorder (72.1 versus 47.6), depressive symptoms (86.6 versus 58.6), and ADL disability (75.1 versus 49.3, P-value < 0.001 for all aforementioned tests) compared with men participants, while the gender-difference in incidence rates of hearing impairment was not significant (35.6 versus 33.9, P-value = 0.487). Meanwhile, the incidence of hearing impairment (81–85 versus >85 years: 126.0 versus 102.5), cognitive impairment (70.4 versus 35.5), sleep disorder (58.9 versus 40.4), declined after the age of 80 years, while other ARFIs still maintained an upward trend. An increasing proportion of participants had co-existing ARFIs during follow-up (Supplementary Fig. S3), and co-existing patterns became more diverse (Supplementary Fig. S4). For example, at baseline (2011-2012), 29.8% (3054) of the participants had co-existing ARFIs, which increased to 68.0% among survivors in 2020. The most prevalent co-existing combination in 2011 was depressive symptoms and sleep disorder, whereas by 2020, the triad of depressive symptoms, sleep disorders, and ADL disability became the top pattern due to the incidence of ADL disability at older age.
Pairwise associations of ARFIs
We examined the pairwise longitudinal associations among ARFIs, as shown in Fig. 2, to assess whether the presence of a specific ARFI at baseline was associated with the risk of developing another ARFI during follow-up. After adjustments for age, sex, education, married status, regions according to household registration, living areas, family income, BMI, smoking status, drinking status, history of stroke, cancer, hypertension, diabetes, and heart diseases, and the other four ARFIs, depressive symptoms were significantly associated with increased risks of all other ARFIs, with hazard ratios (HRs) ranging from 1.43 for cognitive impairment (95% CI: 1.26, 1.62) to 1.74 for sleep disorder (95% CI: 1.61, 1.88). Likewise, baseline ADL disability was associated with higher risks of all other ARFIs: visual impairment (HR: 1.48; 95% CI: 1.32, 1.66), depressive symptoms (1.38; 1.24, 1.53), hearing impairment (1.25; 1.13, 1.39), and sleep disorder (1.23; 1.11, 1.35), with all Bonferroni-corrected P-values < 0.05, except for cognitive impairment (1.06; 0.92, 1.23). Sleep disorders were also predictive of increased risks of subsequent depressive symptoms (1.49; 1.36, 1.64), visual impairment (1.23; 1.10, 1.38), and ADL disability (1.20; 1.11, 1.29). After Bonferroni correction, baseline visual, hearing, and cognitive impairments were not independently associated with the development of other ARFIs during follow-up.
ARFI age-related functional impairment, PY person-year, HR hazard ratio, CI confidence interval, ADL activities of daily living. Error bars indicate two-sided 95% confidence interval. Bold fonts indicate statistical significance after Bonferroni correction. All models were adjusted for age, sex, education, married status, Hukou region, living areas, family income, smoking status, drinking status, BMI, history of stroke, cancer, hypertension, diabetes, and heart diseases, and presence of other ARFIs.
Associations of ARFIs with mortality
We evaluated the independent associations of baseline ARFIs with all-cause mortality during follow-up. After multivariable adjustments, three key ARFIs were significantly associated with increased mortality risks independent of other ARFIs: ADL disability was related to 66% (95% CI: 51%, 83%) higher mortality risk, followed by cognitive impairment (40%; 95% CI: 20%, 63%), and depressive symptoms (18%; 95% CI: 6%, 30%). In contrast, the other ARFIs, i.e., visual impairment, hearing impairment, and sleep disorder, were not significantly associated with mortality risk, with HRs (95% CIs) being 1.08 (0.94, 1.24), 0.96 (0.85, 1.07), and 0.97 (0.87, 1.08) (Fig. 3).
ARFI age-related functional impairment, PY person-year, HR hazard ratio, CI confidence interval, ADL activities of daily living. Error bars indicate two-sided 95% confidence interval. Bold fonts indicate statistical significance after Bonferroni correction. All Cox proportional hazards models were adjusted for age, sex, education, married status, Hukou region, living areas, family income, smoking status, drinking status, BMI, history of stroke, cancer, hypertension, diabetes, and heart diseases. The models for the specific ARFIs with mortality were additionally adjusted for the presence of other ARFIs.
We further examined the combined associations of ARFIs with mortality risk to identify the most impactful combinations of impairments, as presented in Supplementary Table S10 and Fig. S5. ARFI dyads involving cognitive impairment and ADL disability had the strongest associations. Compared with participants with neither, those with co-existing cognitive impairment and ADL disability had 104% higher risk (95% CI: 63%, 154%) of mortality and 9.90 years (95% CI: 8.10, 11.70) loss in life expectancy, followed by the combinations of ADL disability and visual impairment (HR: 1.70; 95% CI: 1.42, 2.04), ADL disability and hearing impairment (1.62; 1.39, 1.89), ADL disability and depressive symptoms (1.62; 1.42, 1.83), and cognitive impairment and depressive symptoms (1.53; 1.25, 1.86).
The number of key ARFIs (ADL disability, cognitive impairment, and depressive symptoms) showed dose-response associations with higher risk of mortality and loss in life expectancy. Each additional key ARFI was associated with a 21% (95% CI: 15%, 27%) increased risk of mortality. Compared to individuals with no key ARFIs, those with one, two, and three key impairments had 20% (8%, 33%), 46% (30%, 65%), and 75% (37%, 123%) higher risks of mortality, respectively (Fig. 3). In the sensitivity analyses additionally adjusting the models for self-rated general health, lung diseases, arthritis, dyslipidaemia, liver diseases, kidney diseases, digestive diseases, and asthma, the associations for depression symptoms attenuated to null, while those for cognitive impairment and ADL disability remained. Additionally, adjusting for psychiatric and memory-related conditions did not significantly change the findings. When we additionally adjusted the models for moderate-to-vigorous physical activity, the associations were not substantially changed (Supplementary Table S11).
Associations of key ARFIs with life expectancy at the age of 50 years
Based on the findings on the above-mentioned ARFI-mortality associations, we calculated the life expectancy at the age of 50 for populations with different ARFI profiles (Fig. 4A, B and Supplementary Table S12). The CHARLS participants had an average life expectancy of 28.18 (95% CI: 27.02, 29.40) years at the age of 50. Participants with cognitive impairment had a life expectancy of 25.89 (25.51, 26.27) years, which was 3.02 (95% CI: 2.61, 3.43; corresponding to a 10.4% reduction) less than those without cognitive impairment (28.91, 95% CI: 28.75, 29.06). Similarly, participants with depressive symptoms and ADL disability had life expectancy (95% CI) of 27.35 (27.16, 27.53) and 25.33 (25.14, 25.53) years, respectively, which was 1.44 (1.20, 1.68; corresponding to a 5.0% reduction) and 4.65 (4.40, 4.89; corresponding to a 15.5% reduction) years less than non-impaired populations.
ARFI age-related functional impairment, LE life expectancy, HR hazard ratio, ADL activities of daily living. Panel A demonstrated the LE of overall population, with or without key ARFIs at the age of 50 years. Panel B showed the association of number of ARFIs with LE and the LE at the age of 50 to 100 years.
Similarly, participants with a larger number of key ARFIs had less life expectancy at the age of 50 (Fig. 4C and Supplementary Table S12). Participants with 0 key ARFI had a life expectancy of 29.57 (95% CI: 29.41, 29.72) years, which was 1.63 (1.35, 1.90) years longer than those with exactly one key ARFI (27.94; 95% CI: 27.71, 28.17), 3.37 (3.02, 3.71) years longer than those with two (26.20; 95% CI: 25.90–26.51), and 4.96 (4.22, 5.71) years longer than those with three (24.60; 95% CI: 23.88, 25.33). Namely, compared with 0 key ARFI at baseline, the percentage difference in life expectancy was 5.5% for one, 11.4% for two, and 16.8% for three key ARFIs.
Discussion
In this prospective study among Chinese middle-aged and older adults, we systematically examined the population distributions of the prevalence and incidence of six age-related functional impairments. We highlighted the central roles of sleep disorders, depressive symptoms, and ADL disability, which demonstrated independent associations with other functional impairments and identified ADL disability, cognitive impairment, and depressive symptoms as independent key predictors of all-cause mortality. The number of these impairments exhibited a dose-response relationship with mortality risk and contributed to an estimated loss of up to 16.8% in life expectancy at age 50. These findings highlight the need for integrated prevention strategies and management for multiple functional impairments in the context of rapid population aging and widening health disparities.
In our analysis, the prevalence of the ARFIs increased with age, which is consistent with previous studies42,43,44,45,46,47. Notably, women had a relatively higher prevalence of all ARFIs except for hearing impairments, particularly at younger ages, which is consistent with previous studies in Western populations48. Existing literature has established that incidence rates of visual impairment, cognitive impairment, sleep disorder, and ADL disability increased with age49,50,51,52, while the incidence of hearing impairment and depressive symptoms was less understood in the Chinese population. In this study, we showed that the incidence of visual, hearing, cognitive, and ADL impairments increased with age, while the reduction in the incidence rate of certain ARFIs after the age of 85 years could be due to survivor bias. Importantly, many of the ARFIs may be under-reported or underdiagnosed when relying solely on self-reported diagnoses. This limitation was mitigated by the use of standardized assessments, such as CES-D for depressive symptoms, which were uniformly administered to the study population. Our finding of regional disparities in the burden of ARFIs, with a higher prevalence in lower-income provinces, aligns with evidence from the Global Burden of Disease, which documented higher prevalence of ARFIs, such as hearing impairment6, visual impairment7, and certain mental conditions53, in lower-income countries. These findings underscore the importance of tailoring public health strategies to promote health equity among older adults across China’s vast and socioeconomically diverse regions.
Our findings on the interrelationships among ARFIs build upon existing knowledge54,55,56,57 by offering a more comprehensive and integrated perspective. For example, while previous research among older Chinese adults has shown that accelerated vision decline is associated with higher odds of ADL disability and cognitive impairment54, few studies have systematically assessed the bidirectional associations of multiple ARFIs within the same context, especially among Chinese older adults58,59. In our earlier work in 17,914 participants from the Health and Retirement Study followed for 20 years5, all ARFIs were found to have bidirectional associations. Extending this investigation to the Chinese cohort, we confirmed that sleep disorders, depressive symptoms, and ADL disability were each independently associated with subsequent development of other ARFIs, and visual impairment and sleep disorders were also associated with ADL disability. While the differences between the two cohorts might be attributable to the genetic, economic, and cultural differences between the study populations, the US-based cohort had longer follow-up of >20 years, which may enable the detection of weaker associations. In aggregate, while ARFIs are broadly interconnected, certain impairments exhibit stronger and more consistent associations with others. Of note, ADL disability may be an overall indicator of severe functional impairments, reflecting underlying deterioration in physical, cognitive, and neurological systems that predisposes individuals to subsequent impairments in other functional domains.
Recognizing that not all ARFIs were independently associated with mortality risk, we further identified ADL disability, cognitive impairment, and depressive symptoms as independent key predictors of mortality, whereas visual impairment, hearing impairment, and sleep disorders were not, after adjusting for the presence and incidence of other ARFIs. In previous studies, the associations for visual impairment60, hearing impairment61, cognitive impairment62, depressive symptoms63, and ADL disability64 were each extensively reported, but those for sleep disorders65 were less conclusive. Our findings suggest that some previously observed associations may be mediated or confounded by co-occurring impairments. When investigating the dyads of ARFIs, combinations involving cognitive impairment and ADL disability were most strongly associated with increased mortality risk, which confirmed our previous results in US adults5. As such, the number of the key ARFIs showed a dose-response association with risk of mortality, which suggested their synergistic roles in predicting mortality5. These key ARFIs may synergistically predict mortality via interrelated biological and behavioral pathways such as chronic inflammation, neurodegeneration, and stress-response dysregulation, which all accelerate functional decline66. For example, depression involves HPA-axis dysfunction, elevating cortisol and driving neuroinflammation and hippocampal atrophy, which overlaps with cognitive impairment arising from amyloid/tau pathology and neuronal loss and ADL disability stemming from frailty9.
We further quantified the health impacts of key ARFIs by estimating their associated loss in life expectancy. Compared with individuals without any of the three key impairments, those with all three experienced a reduction of ~5 years in life expectancy at age 50, equivalent to a 17% decrease. In a previous study in the Chinese population39, adopting a healthy lifestyle was associated with 8 years longer life expectancy at the age of 30 years. Our study further extends the evidence and underscores the importance of integrated prevention strategies (potentially through lifestyle improvement) and management systems for multiple functional impairments that are warranted to achieve healthy aging. Notably, we adjusted the models using baseline covariates because our primary aim was to assess the long-term associations of baseline ARFIs with mortality, and future studies could explore the role of time-varying health status to gain a more comprehensive understanding of the underlying mechanisms.
As one of the few studies to systematically investigate and compare the health impacts of multiple ARFIs within a single cohort, our findings highlight the importance of early identification and intervention in these impairments to mitigate their long-term consequences and improve the overall health of the aging population. In both public health and clinical settings, prioritizing key ARFIs may help prevent downstream impairments and reduce the risk of premature mortality. Additionally, our research provides information for designing and implementing strategies targeted at individuals with specific functional statuses, with a particular focus on cognitive function and the ability to perform daily activities. Although some ARFIs were not directly associated with mortality and longevity, they may be important predictors of the key ARFIs and help identify at-risk populations for early intervention to promote long-term well-being.
The major strengths of this study include its use of a nationally representative sample and a comprehensive and systematic evaluation of multiple ARFIs. The longitudinal design also enabled the exploration of the temporal sequence of ARFIs that are crucial for establishing potential causality. However, our study has several limitations. First, this study, by its observational nature, does not rule out residual confounding and reverse causation, despite multiple sensitivity analyses confirming the robustness of the main findings. Specifically, while the temporal sequence of ARFIs at baseline remains undetermined, our longitudinal analyses provide important insights into the progression of ARFIs and their joint associations with mortality. Second, most ARFIs are defined according to self-reports in questionnaires, which, while extensively validated and widely used, are subject to recall bias and subjectivity. While visual and hearing impairments are often considered age-related, these conditions can also occur at younger ages, which should be taken into account when interpreting the results. Additionally, other ARFIs, such as motor function decline and muscle strength reduction, were not included in this study because they are not consistently measured in CHARLS and could directly lead to ADL disability that has already been included in this study. Third, individuals residing in nursing homes were excluded at the study baseline, given that few older adults live in such institutions (~0.73% according to China’s Seventh National Census in 2020), and only 0.6% of participants entered nursing homes during the follow-up period. Although this does not directly influence the validity of our findings, the generalizability of our findings warrants further study. Fourth, the lack of exact death dates for mortality events occurring between 2015 and 2018 may have led to an overestimation of survival time and, consequently, an underestimation of mortality risk, as is noted by previous studies31,32. Nevertheless, this limitation would be more likely to attenuate, rather than exaggerate, the observed associations. Finally, the 9-year follow-up, while being one of the longest of its kind in Chinese middle aged and older adults, may not be sufficiently long to fully capture the longer-term associations and warrants future efforts with a longer follow-up. Despite the overall attrition rate being relatively low, those lost to follow-up had a higher mean number of co-existing ARFIs, potentially attenuating the strength of the observed associations with mortality.
Conclusions
The current study provides a detailed examination of the incidence and interrelations of ARFIs in a large cohort of middle-aged and older adults in China. We observed that depressive symptoms, ADL disability, and sleep disorder were independent predictors of future ARFIs. ADL disability, cognitive impairment, and depressive symptoms are independently associated with a higher risk of mortality and loss of life expectancy. The numbers of co-existing ARFIs were associated with higher mortality in a dose-response manner and resulted in substantial loss of life expectancy. These findings highlight the relative importance of ARFIs in the identification of populations with high mortality risk and call for integrated prevention strategies and management systems for multiple ARFIs to achieve healthy aging.
Data availability
Data used in the study are publicly available from the CHARLS website: http://charls.pku.edu.cn upon registration. The source data for the figures are provided in Supplementary Data 1.
Code availability
All statistical analyses were conducted using R (version 4.3.0). The analysis code has been deposited in a publicly accessible repository at Github67 and can be accessed at: https://github.com/YuanLabZJU/arfis-charls-share. No proprietary code was used.
References
Hu, K. Challenges facing the Chinese health care system. Asia Pac. J. Public Health 33, 160–161 (2021).
Ageing and health—China. https://www.who.int/china/health-topics/ageing.
Wagner, K.-H., Cameron-Smith, D., Wessner, B. & Franzke, B. Biomarkers of aging: from function to molecular biology. Nutrients 8, 338 (2016).
Scullin, M. K. & Bliwise, D. L. Sleep, cognition, and normal aging: integrating a half century of multidisciplinary research. Perspect. Psychol. Sci. 10, 97–137 (2015).
Chen, H. et al. Progression and trajectory network of age-related functional impairments and their combined associations with mortality. iScience 26, 108368 (2023).
Haile, L. M. et al. Hearing loss prevalence and years lived with disability, 1990–2019: findings from the Global Burden of Disease Study 2019. Lancet 397, 996–1009 (2021).
Steinmetz, J. D. et al. Causes of blindness and vision impairment in 2020 and trends over 30 years, and prevalence of avoidable blindness in relation to VISION 2020: the Right to Sight: an analysis for the Global Burden of Disease Study. Lancet Glob. Health 9, e144–e160 (2021).
Nichols, E. et al. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health 7, e105–e125 (2022).
Byers, A. L. & Yaffe, K. Depression and risk of developing dementia. Nat. Rev. Neurol. 7, 323–331 (2011).
Irwin, M. R. et al. Prevention of incident and recurrent major depression in older adults with insomnia: a randomized clinical trial. JAMA Psychiatry 79, 33–41 (2022).
Ng Yin Ling, C. et al. Visual impairment, major eye diseases, and mortality in a multi-ethnic asian population and a meta-analysis of prospective studies. Am. J. Ophthalmol. 231, 88–100 (2021).
Shukla, A. et al. Hearing loss, loneliness, and social isolation: a systematic review. Otolaryngol. Head. Neck Surg. 162, 622–633 (2020).
Mahalingam, G. et al. Social connections and risk of incident mild cognitive impairment, dementia, and mortality in 13 longitudinal cohort studies of ageing. Alzheimers Dement. 19, 5114–5128 (2023).
Chang, S.-F. & Lin, P.-L. Frail phenotype and mortality prediction: a systematic review and meta-analysis of prospective cohort studies. Int. J. Nurs. Stud. 52, 1362–1374 (2015).
Wang, Y.-H. et al. Depression and anxiety in relation to cancer incidence and mortality: a systematic review and meta-analysis of cohort studies. Mol. Psychiatry 25, 1487–1499 (2020).
Grandner, M. A. Sleep, health, and society. Sleep. Med. Clin. 17, 117–139 (2022).
Kuper, H., Rotenberg, S., Azizatunnisa, L., Banks, L. M. & Smythe, T. The association between disability and mortality: a mixed-methods study. Lancet Public Health 9, e306–e315 (2024).
Rutherford, B. R., Brewster, K., Golub, J. S., Kim, A. H. & Roose, S. P. Sensation and psychiatry: linking age-related hearing loss to late-life depression and cognitive decline. Am. J. Psychiatry 175, 215–224 (2018).
Parada, H., Laughlin, G. A., Yang, M., Nedjat-Haiem, F. R. & McEvoy, L. K. Dual impairments in visual and hearing acuity and age-related cognitive decline in older adults from the Rancho Bernardo Study of Healthy Aging. Age Ageing 50, 1268–1276 (2021).
Cao, X. et al. Contribution of life course circumstances to the acceleration of phenotypic and functional aging: a retrospective study. EClinicalMedicine 51, 101548 (2022).
Bian, L. et al. Associations of combined phenotypic aging and genetic risk with incident cancer: a prospective cohort study. Elife 13, RP91101 (2024).
Zhao, W. et al. Comorbid depressive and anxiety symptoms and frailty among older adults: findings from the West China health and aging trend study. J. Affect Disord. 277, 970–976 (2020).
National School of Development, Peking University. China Health and Retirement Longitudinal Study (CHARLS). http://charls.pku.edu.cn.
Zhao, Y., Hu, Y., Smith, J. P., Strauss, J. & Yang, G. Cohort profile: the China health and retirement longitudinal study (CHARLS). Int J. Epidemiol. 43, 61–68 (2014).
He, J., Tang, X., Jiang, M. & Zheng, X. Associations between vision and hearing impairment and cardiovascular diseases: a longitudinal cohort of middle-aged and older adults in China. J. Am. Heart Assoc. 13, e034851 (2024).
Wang, Q., Zhang, S., Wang, Y., Zhao, D. & Zhou, C. Dual sensory impairment as a predictor of loneliness and isolation in older adults: national cohort study. JMIR Public Health Surveill. 8, e39314 (2022).
Cao, L., Zhao, Z., Ji, C. & Xia, Y. Association between solid fuel use and cognitive impairment: a cross-sectional and follow-up study in a middle-aged and older Chinese population. Environ. Int. 146, 106251 (2021).
Liu, F. et al. Effects of air pollution and residential greenness on sleep disorder: a 8-year nationwide cohort study. Environ. Res. 220, 115177 (2023).
Andresen, E. M., Malmgren, J. A., Carter, W. B. & Patrick, D. L. Screening for depression in well older adults: evaluation of a short form of the CES-D (Center for Epidemiologic Studies Depression Scale). Am. J. Prev. Med. 10, 77–84 (1994).
Lee, A. K., Diaz-Ramirez, L. G., Boscardin, W. J., Smith, A. K. & Lee, S. J. A comprehensive prognostic tool for older adults: Predicting death, ADL disability, and walking disability simultaneously. J. Am. Geriatr. Soc. 70, 2884–2894 (2022).
Ou, H., Wei, M., Li, X. & Xia, X. C-reactive protein-triglyceride glucose index in evaluating cardiovascular disease and all-cause mortality incidence among individuals across stages 0-3 of cardiovascular-kidney-metabolic syndrome: a nationwide prospective cohort study. Cardiovasc. Diabetol. 24, 296 (2025).
Hao, M. et al. Metrics of physiological network topology are novel biomarkers to capture functional disability and health. J. Gerontol. A Biol. Sci. Med. Sci. 80, glae268 (2024).
Kong, W. et al. Association between BMI and asthma in adults over 45 years of age: analysis of Global Burden of Disease 2021, China Health and Retirement Longitudinal Study, and National Health and Nutrition Examination Survey data. EClinicalMedicine 82, 103163 (2025).
Glymour, M. M. & Avendano, M. Can self-reported strokes be used to study stroke incidence and risk factors?: Evidence from the health and retirement study. Stroke 40, 873–879 (2009).
Mullins, M. A. et al. Validation of self-reported cancer diagnoses using Medicare diagnostic claims in the US Health and Retirement Study, 2000-2016. Cancer Epidemiol. Biomark. Prev. 31, 287–292 (2022).
Ning, M., Zhang, Q. & Yang, M. Comparison of self-reported and biomedical data on hypertension and diabetes: findings from the China Health and Retirement Longitudinal Study (CHARLS). BMJ Open 6, e009836 (2016).
Hurvich, C. M., Simonoff, J. S. & Tsai, C.-L. Smoothing parameter selection in nonparametric regression using an improved Akaike information criterion. J. R. Stat. Soc. Ser. B Stat. Methodol. 60, 271–293 (1998).
Lamer, A. et al. Exploring patient path through Sankey diagram: a proof of concept. in Digital Personalized Health and Medicine 218–222 (IOS Press, 2020).
Sun, Q. et al. Healthy lifestyle and life expectancy at age 30 years in the Chinese population: an observational study. Lancet Public Health 7, e994–e1004 (2022).
GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1204–1222 (2020).
Buuren, S. van & Groothuis-Oudshoorn, K. mice: multivariate Imputation by Chained Equations in R. J. Stat. Softw. 45, 1–67 (2011).
Zou, M. et al. Prevalence of visual impairment among older Chinese population: a systematic review and meta-analysis. J. Glob. Health 11, 08004 (2021).
Yang, D. et al. Hearing impairment prevalence and risk factors among adults in rural China: a population-based cross-sectional study. Postgrad. Med. 133, 369–376 (2021).
Chen, H. et al. Trends in the prevalence of cognitive impairment at old age in China, 2002-2018. Alzheimers Dement. 20, 1387–1396 (2024).
Gulia, K. K. & Kumar, V. M. Sleep disorders in the elderly: a growing challenge. Psychogeriatrics 18, 155–165 (2018).
Zhang, L., Xu, Y., Nie, H., Zhang, Y. & Wu, Y. The prevalence of depressive symptoms among the older in China: a meta-analysis. Int J. Geriatr. Psychiatry 27, 900–906 (2012).
Ma, L., Chhetri, J. K. & Chan, P. Frailty in China: from research to practice. J. Nutr. Health Aging 25, 479–483 (2021).
Jiang, C.-Y. et al. Global, regional, and national prevalence of hearing loss from 1990 to 2019: A trend and health inequality analyses based on the Global Burden of Disease Study 2019. Ageing Res. Rev. 92, 102124 (2023).
Chen, X. et al. Prevalence and causes of visual impairment in adults in Binhu District, Wuxi, China. Med. Sci. Monit. 24, 317–323 (2018).
Gao, M. et al. The time trends of cognitive impairment incidence among older Chinese people in the community: based on the CLHLS cohorts from 1998 to 2014. Age Ageing 46, 787–793 (2017).
Fong, J. H. & Feng, J. Patterns of functional disability in the oldest adults in China. J. Am. Geriatr. Soc. 64, 1890–1894 (2016).
Wang, J. et al. Prevalence of sleep disturbances and associated factors among Chinese residents: a web-based empirical survey of 2019. J. Glob. Health 13, 04071 (2023).
Global, regional, and national burden of 12 mental disorders in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Psychiatry 9, 137–150 (2022).
Cao, G. et al. Visual trajectories and risk of physical and cognitive impairment among older Chinese adults. J. Am. Geriatr. Soc. 69, 2877–2887 (2021).
Wang, Q. et al. The effect of dual sensory impairment and multimorbidity patterns on functional impairment: a longitudinal cohort of middle-aged and older adults in China. Front. Aging Neurosci. 14, 807383 (2022).
Yuan, Y., Peng, C., Burr, J. A. & Lapane, K. L. Frailty, cognitive impairment, and depressive symptoms in Chinese older adults: an eight-year multi-trajectory analysis. BMC Geriatr. 23, 843 (2023).
Zhou, L., Ma, X. & Wang, W. Relationship between cognitive performance and depressive symptoms in Chinese older adults: the China Health and Retirement Longitudinal Study (CHARLS). J. Affect Disord. 281, 454–458 (2021).
Han, S., Gao, T., Mo, G., Liu, H. & Zhang, M. Bidirectional relationship between frailty and cognitive function among Chinese older adults. Arch. Gerontol. Geriatr. 114, 105086 (2023).
Wu, C. Bidirectional association between depression and hearing loss: evidence from the china health and retirement longitudinal study. J. Appl. Gerontol. 41, 971–981 (2022).
Ehrlich, J. R. et al. Association between vision impairment and mortality: a systematic review and meta-analysis. Lancet Glob. Health 9, e418–e430 (2021).
Loughrey, D. G., Kelly, M. E., Kelley, G. A., Brennan, S. & Lawlor, B. A. Association of age-related hearing loss with cognitive function, cognitive impairment, and dementia: a systematic review and meta-analysis. JAMA Otolaryngol. Head. Neck Surg. 144, 115–126 (2018).
Dewey, M. E. & Saz, P. Dementia, cognitive impairment and mortality in persons aged 65 and over living in the community: a systematic review of the literature. Int. J. Geriatr. Psychiatry 16, 751–761 (2001).
Pan, A. et al. Depression and risk of stroke morbidity and mortality: a meta-analysis and systematic review. JAMA 306, 1241–9 (2011).
Kojima, G., Iliffe, S. & Walters, K. Frailty index as a predictor of mortality: a systematic review and meta-analysis. Age Ageing 47, 193–200 (2018).
Lovato, N. & Lack, L. Insomnia and mortality: a meta-analysis. Sleep. Med. Rev. 43, 71–83 (2019).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Hallmarks of aging: an expanding universe. Cell 186, 243–278 (2023).
Github Repository. https://github.com/YuanLabZJU/arfis-charls-share (2025).
Acknowledgements
We thank the participating individuals and staff of the CHARLS study who made the study possible. The CHARLS data collection received financial support from the Behavioral and Social Research division of the National Institute on Aging, the Natural Science Foundation of China, the World Bank, and Peking University. The study was supported by the National Key Research and Development Program of China (2023YFC3606300 and 2022YFC3600600 to AP and 2022YFC2010106 to CY) and the Zhejiang University Global Partnership Fund for this study. The funding organizations had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
Hui Chen, Binghan Wang, and Changzheng Yuan had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Changzheng Yuan, An Pan. Acquisition, analysis, or interpretation of data: Hui Chen, Binghan Wang. Drafting of the manuscript: Hui Chen, Binghan Wang. Critical revision of the manuscript for important intellectual content: Hui Chen, Binghan Wang, Minqing Yan, Ting Shen, Mengjia Zhao, Yanping Li, Xiaolin Xu, Klodian Dhana, Xiaobo Yang, An Pan, and Changzheng Yuan. Statistical analysis: Binghan Wang, Hui Chen. Obtained funding: Changzheng Yuan, An Pan. Administrative, technical, or material support: Changzheng Yuan, An Pan. Supervision: Changzheng Yuan, An Pan. Corresponding authors: Changzheng Yuan or An Pan.
Corresponding authors
Ethics declarations
Competing interests
No disclosures. Xiaolin Xu is an Editorial Board member for Communications Medicine but was not involved in the editorial review or peer review, nor the decision to publish this article.
Peer review
Peer review information
This manuscript has been previously reviewed at another Nature Portfolio journal. The manuscript was considered suitable for publication without further review at Communications Medicine.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, H., Wang, B., Yan, M. et al. A prospective study of age-related functional impairments, mortality, and life expectancy of Chinese adults aged 50. Commun Med 6, 103 (2026). https://doi.org/10.1038/s43856-025-01350-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-025-01350-3