Abstract
Therapies for chronic and end-organ diseases are often associated with systemic toxicity, and such conditions result in difficulty treating patients. RNA nanomedicine has demonstrated unprecedented advantages in the field of therapeutics through its selective organ targeting, personalized treatment option and safety. In this paper, we extensively discuss the current state of nanotechnology in regenerative medicine and how this unique combination of RNA nanomedicine and regenerative medicine can be leveraged for a successful end-organ and chronic disease therapy. More specifically, we have put forward strategies to specifically deliver RNA nanomedicine to target organs/tissues in chronic disease patients, design considerations for regenerative medicine RNA therapy versus RNA vaccines, and barriers to clinical translation. We further discuss the call to action short- and long-term goals in the field of regenerative RNA nanomedicine that could change the face of regenerative medicine in the coming years.
Similar content being viewed by others
Introduction
End-organ diseases refer to the permanent damage to one or several major organs due to varied causes, leading to the shutdown of these organs and the inability to perform adequately for the normal functioning of the body.
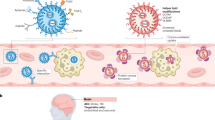
The use of RNA-based therapies has encompassed several advantages in the field of regenerative medicine, including specific organ/cell targeting, a dose-dependent response, and ease of production1,2,3. However, there are significant barriers to the viability of RNA therapies, including nuclease degradation, clearance by the reticuloendothelial system, and inefficient RNA delivery into the cellular cytoplasm due to its negative charge and size4,5,6. Additionally, RNA molecules trigger an antiviral response and accumulate in off-target sites, decreasing therapeutic efficacy in cell and organ regeneration7,8,9 (Fig. 1).
Overview of the applications of RNA nanotherapy for regenerative medicine and ways to overcome the barriers in clinical translation.
To overcome these critical limitations, nanoparticles have been formulated to enable successful RNA delivery. RNA-containing nanoparticles can be designed to avoid macrophage clearance, extend RNA half-life, and target delivery to avoid toxicity at off-target sites10,11. Multiple RNA types can also be potentially incorporated into one nanoparticle to target different genes12. RNA nanomedicine has emerged in recent years as a vehicle by which genetic instructions can be safely delivered into target cells without being degraded. Such nanoparticles have various formulations and can carry RNA into target cells to reprogram them. For example, one could envision packaging RNA that can block the production of harmful and damaging proteins or could stimulate regeneration.
Additionally, the milestone achieved in RNA therapeutics through the success of Pfizer-BioNTech and Moderna mRNA-LNP COVID-19 vaccines has motivated several researchers to explore this field of research for therapeutics beyond vaccines. Evidently, the safety profile of these mRNA vaccines, showing exceptional efficacy against coronavirus with no adverse reactions except transient local and systemic reactions, exhibits RNA nanomedicine as a safe and effective means of therapeutic delivery13,14,15,16. Additionally, mRNA vaccines have shown to improve protein translation, regulate innate and adaptive immunogenicity, and enhance cellular delivery17. Patients with chronic and end-organ disease are a group that would especially benefit from such localized therapy to prevent systemic side effects. End-organ diseases involving the liver, kidneys, heart, lungs, or brain has shown to increase exponentially each year18. The rate of descent into end-organ disease varies from one organ to another. For example, although chronic liver disease can evolve into cirrhosis and liver failure over time, this accumulates over decades due to the liver’s unique capacity to regenerate19,20,21. This allows the liver to heal itself over time despite ongoing injury20,21,22. If we could leverage and harness this unique regenerative capacity of the liver at earlier stages and across organs, we could prevent progression to end-organ disease and associated deaths19.
Considering the high impact of RNA nanomedicine in the current era, we convened a Medicine by Design Convergent Working group on March 27th, 2023, to discuss the challenges and future perspectives in translating RNA nanomedicine into the clinic. By inviting local as well as international pioneers in RNA nanomedicine and clinical regenerative medicine, we have drafted this position paper as a call for action on how we can control delivery of RNA-containing nanoparticles into cells of interest, and how we can make these therapies safe for ultimate clinical translation in patients with chronic and end-organ disease along with the advancements, current challenges, and call to action short-term and long-term goals. This is an opportune time to exploit the growing expertise in RNA nanomedicine and bring it to the bedside to stimulate regeneration in patients with chronic diseases.
Current state of nanotechnology in regenerative medicine
Regenerative medicine allows repair, replacement, or restoration of diseased or damaged tissues. Research has evolved from strategies using tissue engineering to tissue reconstruction, further allowing early-intervention treatments for traumatic injury or degenerative diseases. The field of regenerative medicine has shown promise in treating several diseases that are represented with a large unmet medical need, including blindness, neurodegenerative diseases, diabetes, and various organ repairs.
Researchers have harnessed the power of RNA nanomedicine through Patisiran (Onpattro®), a first-in-class, first-in-human FDA-approved siRNA encapsulated lipid nanoparticle (LNP) formulation currently used for the treatment of hATTR in patients with polyneuropathy due to transthyretin (TTR) amyloidosis23 (https://www.fda.gov/drugs/spotlight-cder-science/new-class-drugs-fulfills-promise-rna-based-medicine, https://www.ema.europa.eu/en/medicines/human/EPAR/onpattro). TTR amyloidosis is a systemic disease with abnormal amyloid protein deposition affecting the heart, peripheral, and autonomic nervous systems. Patisiran acts by inhibiting the TTR protein in the liver by specifically targeting hepatocytes and has shown exceptional efficacy during clinical studies with >80% gene silencing and an excellent safety profile23. LNPs are the most well-established nano carriers today that are composed of an ionizable lipid, a diffusible PEG-lipid, and other lipids such as PEG, with each component playing a significant role in the therapeutic efficacy and specific targeting24. The unique characteristics of lipids used in the development of LNPs allow liver targeting through LDL receptor binding for their internalization into hepatocytes, further releasing the RNA machinery in the cytoplasm24. This is achieved by LNPs binding to ApoE serum proteins that facilitate binding to the LDL receptor expressed on the surface of hepatocytes, enabling selective uptake of LNPs into hepatocytes via receptor-mediated endocytosis24. Upon internalization, LNPs are distributed to endosomal compartments. Endosomal release of LNPs is a critical step in the RNA therapeutic delivery process. Although it remains a complex and less clear process, studies have indicated that the acidic environment in the endosomes could lead to the protonation of ionizable lipids. This destabilizes the endosomal membrane, facilitating the release of the RNA machinery into the cytoplasm24. Overall, the FDA and European Commission approval of Patisiran for TTR amyloidosis reflects the remarkable progress of RNA nanomedicine for genetic diseases that have a systemic impact and suggests significant potential in regenerative medicine (https://www.fda.gov/drugs/spotlight-cder-science/new-class-drugs-fulfills-promise-rna-based-medicine, https://www.ema.europa.eu/en/medicines/human/EPAR/onpattro).
Several types of RNAs, such as small interfering RNA (siRNA), messenger RNA (mRNA), microRNA (miRNA), and antisense oligonucleotide (ASO), are currently being studied for the treatment of regenerative diseases. These types of RNA contain distinctive features that enable/disable their use during specific therapies. Amongst these, siRNAs are currently the most widely used RNA that function by downregulating gene expression through RNA-induced silencing complex (RISC)-mediated mRNA degradation. Similarly, miRNAs also recruit RISC to the complementary mRNA sequences, and miRNA mimics are designed to upregulate the mRNA expression25. Conversely, mRNAs are used when a dysfunctional protein replacement is required, or antigen encoding is needed for a longer-term immunity to pathogens such as SARS-CoV-2. A distinctive disadvantage of mRNA is that the transcription occurs in vitro and therefore they cannot have site-specific chemical modifications25. On the other hand, ASOs act by either inducing the degradation of the mRNA or by inhibiting the progression of splicing or gene translation19,26. The RNA size is also taken into account when designing delivery systems. siRNAs and ASOs are much smaller than mRNAs and can therefore be delivered through small conjugates or antibodies; however, mRNAs are large and need to be encapsulated before any conjugation25.
Additionally, other emerging non-viral technologies are also being used for RNA delivery in regenerative medicine, such as engineered extracellular vesicles (EVs) and tissue nanotransfection27. Extracellular vesicles are lipid-bound vesicles that function as endogenous intercellular cargo transfer systems for RNA moieties due to their natural cell-targeting abilities, pharmacokinetics, and biodistribution27. Overall, EVs can be further classified as exosomes and microvesicles, whereby exosomes are formed from endosomal compartments while microvesicles are produced through budding and pinching from the cell membranes27,28,29. EVs offer the advantage of natural cell-targeting ability as opposed to lipid nanoparticles that are often engulfed and exocytosized by endosomes. Reports have shown successful loading of mRNA, noncoding RNAs, and miRNAs into EVs for regenerative medicine applications, and studies have indicated that, depending on the source of EVs, they may show lesser immunogenic properties due to fewer transmembrane proteins on their surface as compared to the parental cells in regenerative medicine29. On the other hand, tissue nanotransfection has also paved its way through advancements to become the future of regenerative medicine by permitting direct cytosolic delivery of regenerative factors using a non-invasive array of nanochannels supplied with focused electroporation30. Interestingly, researchers have demonstrated that the combination of RNA-based EVs with tissue nanotransfection can trigger cellular or tissue regeneration more effectively30, however, more in-depth studies are still required to generate a proof-of-concept of these technologies in regenerative medicine. Interestingly, researchers have demonstrated that the combination of RNA-based EVs with tissue nanotransfection can trigger cellular or tissue regeneration more effectively30; however, more in-depth studies are still required to generate a proof-of-concept of these technologies in regenerative medicine. Following these advancements, innovative strategies are being explored to enhance regenerative outcomes.
Emerging technologies in regenerative medicine
Ocular diseases
The FDA approval of Luxturna®, an adeno-associated virus vector-based gene therapy for the treatment of a form of Leber Congenital Amaurosis (LCA) that causes inherited blindness, has opened new avenues for RNA nanomedicine in the treatment of ocular diseases31. Studies have discussed the potential of RNA nanomedicine in eye diseases and disorders. Ocular blindness is caused by the degeneration of PRs, RPEs, and retinal ganglion cells, and he emphasized that over the past decade, retinal regenerative medicine has permitted the generation of specific cells from progenitor cells for cell repair and restoration that can be strategized through RNA nanomedicine32. Also, a surge in RNA nanotherapies for the treatment of ocular diseases has been witnessed over the past few years, owing to their ability to penetrate complex ocular barriers such as the corneal-retinal and blood retinal barriers33,34. This further contributes to minimal to no extra-ocular delivery, thereby limiting off-target toxicities. Recently, Dulla et al. investigated an antisense oligonucleotide (ASO) based treatment of retinitis pigmentosa caused by USH2A exon 13 mutations in a zebrafish model35. The results validated the ASO QR-421a as a candidate for an exon-skipping therapy and are currently being clinically tested in a phase ½ trial (NCT03780257).
Neurodegenerative disorders
Although advances in stem cell therapy have enabled neuron replacement and reconstruction in ischemic stroke patients, most stem cells do not exclusively differentiate into functional neurons, resulting in inefficient clinical translation. In this regard, nanoparticles offer an unprecedented advantage of co-delivering stem cells with genes that favor neuronal differentiation. In a recent investigation, Lin et al. studied the co-delivery of neuronal stem cells with Pnky siRNA that silenced the gene responsible for the inhibition of neuronal differentiation of neuronal stem cells, and surprisingly, the results indicated that this combination of nanomedicine promoted neuronal differentiation and hence enhanced the therapeutic efficacy of regenerative medicine in stroke36. In addition to this, the convergence of nanomedicine and regenerative medicine also holds immense scope for the treatment of devastating neurodegenerative diseases. The past decade has witnessed stem cell transplantation in these diseases, along with nanotechnology to enable these therapies across the blood-brain barrier (BBB) as the most promising strategy for the treatment of neurodegenerative diseases37. Previously, a clinical study (NCT01640067) demonstrated the therapeutic effect in reducing the progression of Amyotrophic Lateral Sclerosis (ALS) post exogenous transplantation of neural stem cells; however, utilizing nanoparticles in conjunction with this therapy could have also activated endogenous neurons along with the exogenous populations for a more potent pharmacological effect38,39.
Pancreatic diseases
Tremendous advancement has been witnessed in pancreatic regenerative medicine to prevent and treat diabetes. With substantial progress in understanding the disease, several researchers are designing pancreatic stem cells for differentiation into pancreatic β-cells and islet-like organoids that efficiently regulate glucose and secrete insulin40,41. Most recently, Melamed et al. have developed mRNA LNPs that can be specifically targeted to the pancreas for the treatment of pancreatic diseases and disorders. In this study, the intraperitoneal route of delivery was utilized as a strategy to specifically deliver the mRNA to the pancreas located in the peritoneal cavity and reduce systemic toxicity associated with the intravenous route of delivery. The results indicated excellent mRNA-induced protein expression in the insulin-producing β cells with LNPs formulated using three different ionizable lipidoids, and approximately 50% increase in the protein expression was observed in the pancreas when the LNPs were administered intraperitoneally as compared to intravenous administration. Additionally, a proof-of-concept was established to show that the mRNA was delivered by peritoneal macrophage extracellular vesicle transfer, and the mRNA efficacy was not associated with any systemic toxicity42.
Liver diseases
Liver, as an organ, presents exceptional ability for regeneration. However, liver diseases are the 11th leading cause of death worldwide, accounting for 4% of all deaths worldwide, with liver transplantation being the mainstay for patients with end-stage liver disease incapable of inherent regeneration43,44. Also, the major anti-fibrotic therapies employed during non-alcoholic steatohepatitis, liver fibrosis, and cirrhosis present low therapeutic efficiency and high side effects. To overcome these challenges, in the past few decades, several researchers have been investigating RNA nanomedicine for the treatment of chronic liver disease and regenerative medicine45,46,47,48,49. Most recently, Mitchell’s group has developed a combinatorial library of anisamide ligand-tethered lipidoids (AA-lipidoids)50. These lipidoids showed a high potency and selectivity to deliver RNA payloads to activated HSCs. HSP47 siRNA (si-HSP47)-loaded AA-T3A-C12 LNP, which are specifically targeted to activated myofibroblasts, performed approximatively 65% knockdown and strongly decreased liver fibrosis, with remarkably reduced collagen fibers and pseudolobules with minimal collagen deposition. Moreover, AA-T3A-C12/si-HSP47 LNP-treated fibrotic mice demonstrated almost normal histological structure and had a lower number of fibrotic septa and apoptotic hepatocytes, indicating exceptional potential for RNA nanomedicine to target undruggable liver diseases with a potential for regenerative medicine. Overall, RNA nanomedicine represents an exciting field of research for the treatment of end-organ diseases through regenerative medicine.
Strategies to deliver RNA nanomedicine directly into target cells/tissues
Tissue/cell-specific delivery
Since most nanoparticles are systematically administered, an urgent challenge lies in the specific delivery of the circulated mRNA molecules to the site of interest. They are also highly prone to nuclease degradation and, therefore, susceptible to degradation prior to reaching the organ of interest, further affecting therapeutic efficacy. Moreover, the mRNA may require cellular penetration for gene editing; hence, strategies for cytosolic delivery also need to be considered51,52. To overcome these obstacles, different surface ligands and careful design of nanoparticles are necessary for a successful targeted and efficacious therapy. Recently, for lung targeting, Qui et al. reported a novel PEGylated KL4 peptide (PEG12KL4) that acts as a pulmonary surfactant protein B (SP-B) mimic and can be conjugated on nanoparticle surfaces for effective delivery to the lung epithelial cells53,54. Additionally, researchers have also discovered that although the majority of LNPs are made of ionizable lipids, utilizing ionizable lipids with low pKa and unsaturated hydrocarbons and/or 0.5% PEG during LNP formulation elicits greater transfection efficiency in the retinal pigment epithelium (RPE) cells of the eye following subretinal injections55.
Similarly, while most lipid nanoparticles accumulate in the liver, it is crucial to identify the liver cells of interest more specifically for therapeutic targeting and to avoid any off-target distribution and toxicity. Considering this, Han et al. have recently developed an anisamide ligand-tethered lipidoid (AA-T3A-C12) as a high-affinity ligand for the overexpressed sigma receptors on activated fibroblasts and HSCs50. These novel LNPs showed greater RNA delivery to activated fibroblasts in the liver compared to the current FDA-approved MC3 ionizable lipid. Also, 2-fold fibroblast uptake and decreased liver fibrosis were evident using the AA-T3A-C12 formulated LNPs with heat shock protein 47 siRNA as the therapeutic target for liver fibrosis50. Alternatively, researchers have discovered that apolipoprotein E (ApoE) and N-acetylgalactosamine (GalNAc) can direct the nanoparticle uptake specifically to hepatocytes through the lipoprotein (LDLR) and asialoglycoprotein (ASGPR) receptors, respectively46,56,57. Hepatocyte targeting is crucial for several diseases, including hereditary amyloidosis that is currently being treated by GalNAc-mediated hepatocyte targeting LNPs – Onpattro.
Subsequently, RNA nanomedicine has also shown promise in cardiac regeneration for myocardial infarction and gene modulation in inherited cardiac diseases; however, insufficient RNA delivery to the cardiac tissue is often an obstacle. Researchers have been studying different approaches to maximize cardiac tissue delivery by optimizing the various excipients used in LNP formulation development. In a recent study, a positively charged ionizable lipid C12-200, along with higher dioleoylphosphatidylethanolamine (DOPE) and lower cholesterol, was used to formulate LNPs for cardiomyocyte targeted gene therapy, and the results indicated that these novel LNPs were able to target the cardiomyocytes safely and effectively58. Collectively, such approaches suggest that specifically targeted nanomedicine holds great potential in cardiac regeneration by targeting the hard-to-transfect cardiomyocytes.
While LNPs mostly tend to accumulate in the liver, which facilitates liver targeting as opposed to targeting other organs, liver localization is not an inherent property of all nano carriers, and chemical modifications in the nanoparticle design can allow minimal liver involvement. This has evidently been demonstrated by the discovery of a selective organ targeting (SORT) strategy to rationally develop nanoparticles for extrahepatic RNA delivery. Dilliard et al. added a SORT molecule as a fifth component to the conventional LNPs that are comprised of PEG, cholesterol, amphipathic phospholipids, and ionizable cationic lipids to alter the biodistribution profile of the nanoparticles59. Most importantly, the SORT molecule is selected based on the cells or organ of interest and which proteins would most abundantly absorb to the LNPs for extrahepatic delivery, for example, increased pulmonary delivery has been observed with LNPs that contain a quaternary ammonium headgroup as a SORT molecule60.
Local delivery
All speakers and discussants agreed to the fact that the route of delivery is extremely crucial for specifically targeting organs and tissues. Studies have indicated that different routes of administration, such as intranasal, intravitreal, and topical, have shown enhanced delivery to the lungs, brain, eye, and skin, respectively. Pulmonary delivery has been considered challenging due to its specialized cell types, mucus barrier, and mucociliary clearance. Researchers are aiming for inhalational and intratracheal delivery formulations for lung diseases and lung tissue regeneration. For the treatment of cystic fibrosis, MRT5005 is reportedly the first-in-human inhaled mRNA candidate currently in Phase I/II clinical trials that delivers a codon-optimized CFTR mRNA via a nebulizer61. The ongoing study results indicate that this aerosol delivery was safe and well tolerated during the 28-day follow-up post-treatment completion, and no severe adverse effects or off-target effects were reported except minor adverse events such as cough and headache62. Although improvements on nanoparticle stability for nebulization are still required, in vivo studies have shown highly efficient pulmonary mRNA delivery and gene editing in the lung with excellent cellular uptake63. Also, minimal to no off-target distribution was evident by selecting this route of administration for lung delivery.
Furthermore, intranasal delivery has been the preferred route of drug administration for neuron regeneration in neurodegenerative diseases. Although the current standard of care for most CNS diseases is oral medications, it has been proven that oral therapy has a limited therapeutic effect due to its poor absorption and inability to cross the BBB to reach the brain and cells of interest. For this reason, researchers are taking advantage of the nerve pathways, blood vessels, lymphatic system, and cerebrospinal fluid located within the nasal system that enable routing nanoparticles to the brain64. Also, nasal instillation allows the drug to immediately enter the systemic circulation, thereby avoiding any drug degradation that occurs in the gastrointestinal tract during oral therapy. Most essentially, nanomedicine enables efficient drug delivery of low molecular lipophilic molecules past the BBB, which is equipped with abundant tight junctions and undergoes limited pinocytosis for the maintenance of CNS homeostasis65.
Similarly, for targeting the degenerative diseases of the eye, researchers have been focusing on subretinal and intravitreal injections of RNA nanoparticles. Studies have shown how RNA nanoparticles can be designed to not only specifically deliver the gene therapy to the eye, but also to specifically deliver target genes in different cells of the eye by designing specific target peptides for specific cells. Studies have indicated that lipid nanoparticles (LNPs) are able to predominantly deliver gene therapy to retinal pigment epithelium RPE cells and Muller glia cells through subretinal or intravitreal routes; however, LNPs have been unable to penetrate the neural retina for photoreceptor (PR) targeting. To overcome this challenge, researchers have successfully developed and evaluated novel peptides for mRNA delivery to the neural retina in mouse models. Moreover, these chemically decorated LNPs showed excellent delivery in a retinal degenerative nonhuman primate model with potent protein expression in RPE, Muller glia, and PRs, opening the scope for eye regenerative medicine66,67,68.
Design considerations for regenerative medicine: mRNA vs small RNA for RNA vaccines
Amongst others, one crucial hurdle is the immune response as a viral defense mechanism experienced during RNA therapy that has previously led to the termination of clinical studies. Although this could favor while designing RNA vaccines, immunogenicity might not be the ideal response during RNA therapy, and therefore it is extremely important to understand this phenomenon and take into consideration while designing RNA vaccinesfor regenerative medicine69. Most importantly, both single and double-stranded RNA moieties are recognized by the immune system via toll-like receptor (TLR) signaling that further activates several pathways resulting in a type I interferon response, production of inflammatory cytokines, and activation of nuclear factor-kB (NF-kB)70. As a proof-of-concept, Kariko et al. demonstrated that mRNAs with naturally modified nucleotides had a higher translational capacity than unmodified mRNAs. Also, only the unmodified mRNAs depicted immunogenic behavior with elevated levels of interferon α as opposed to the modified mRNAs71.
To understand this, researchers have extensively studied self-amplifying RNA (saRNA) versus mRNA for the development of vaccines and how different cargos are sensed intracellularly to produce the immune response. Researchers have been studying whether different RNAs have shown variations in the optimal parameters for cellular activation, and the studies indicated that most parameters remained the same irrespective of the RNA; however, the size of RNA had an impact on the encapsulation efficacy and eventually the mechanism of action. Also, researchers are employing second and third-generation chemical modifications, such as 2′-ribose modifications on siRNAs and phosphoramidate morpholino oligomers (PMOs), to reduce the associated immunogenicity by neutralizing charge and restricting protein interactions that could trigger immune system activation72,73,74,75.
Most importantly, the stability and rapid degradation of RNAs critically determine the fate of the nanoparticle in the body. Many researchers have been studying ways to improve the RNA half-life for optimal efficacy. Reports have indicated the importance of the Cap, untranslated regions (UTRs), and poly(A) tail located at the 5′- and 3′- ends of the mRNA structure for stable and effective mRNA. Some of the ways to improve mRNA stability include (a) increasing G-C fractions in mRNA, and (b) fine-tuning the poly(A) tail, 5′ Cap, 5′-UTR/3′-UTR, and rare codon76. Similarly, chemical modifications on siRNAs and ASOs also enhance RNA stability and protect them from rapid nuclease degradation. The most common ASO and siRNA modification is a phosphorothioate modification that enhances DNase resistance and improves cellular uptake due to an increase in hydrophobicity77,78,79.
Additionally, several other potential solutions are being investigated, such as performing more in-depth immune-related adverse event screening in human studies with TLR interaction methods, using short 7–8 nucleotide RNAs that show successful low immunogenicity in preclinical studies, administering metronomic (regular and frequent) doses of RNA therapy, and combination treatments of RNA and immune-based therapies.
Overall, the lessons learnt from the COVID-19 mRNA vaccine are important to drive RNA nanotherapeutics in autoimmune diseases, inflammatory conditions, and end-organ diseases as regenerative medicine.
Barriers to clinical translation: understanding the factors contributing to the translational gap in chronic and end-organ diseases for regenerative medicine
Most importantly, a better understanding of the underlying disease mechanisms is needed for the successful clinical translation of regenerative medicine. Currently, most of the regenerative medicine research is based on genetically engineered animal models that do not offer a precise model of the complex traits in the human disease80,81. Subsequently, researchers have also been working to utilize human cells; however, the hurdles in specific cell isolation from whole tissues cannot be overlooked. Most recently, technological advances have enabled researchers to generate human models of different diseases by developing and expanding different types of differentiated cells from both human induced pluripotent stem cells (iPSC) and human embryonic stem cells (hESC) from patients with the specific disease82. However, the biggest challenge still lies in the perfect isolation and expansion of certain rare cell populations in order to completely differentiate them, and the high variability in the cell lines.
Furthermore, when particularly studying cardiovascular regenerative medicine, the heterogeneity of the cardiac progenitor cells that convert to cardiomyocytes in the heart, as opposed to their conversion to arterial and venous smooth muscles, endothelial cells, atrial and ventricular cardiomyocytes, and cells of the conduction system in vivo, presents a critical challenge that is extremely difficult to overcome83,84. Therefore, for an optimal model development for studying cardiac regenerative medicine, it is extremely important to understand these progenitor cells and their corresponding pathways that dictate their fate in the heart.
Additionally, overcoming the immunological barriers in regenerative medicine limits its translation from bench to bedside. The immunological responses are usually minimal when the cells are of autologous origin; however, any allogenic or genetically modified cells generate host immune responses, further inducing rejection85. Some possible strategies to overcome this hurdle include: administering immunosuppressant drugs or monoclonal antibodies that prevent or treat allograft rejection, adoptive transfer of APCs, NKT cells, T cells, or mesenchymal stromal cells to induce tolerance, hematopoietic stem cell transplantation to induce tolerance, and developing biomaterials that restrict contact between donor and host immune systems, such as nanomedicine85.
Most importantly, even after careful consideration and design of preclinical studies with RNA nanotherapies, several factors affect their clinical translation. First, species variability highly affects the pharmacokinetics and pharmacodynamics of RNA therapies due to differences in serum lipids, basal metabolic rates, and organ/body ratios25,86,87. These variations have led to difficulties in correlating small animal biodistribution, cellular interactions, and toxicities to those in human studies88. To solve this problem, researchers are studying the use of species-agnostic nanoparticle delivery screening (SANDS) that enables determining a species-dependent correlation factor between the delivery of RNA therapies in murine, human cells in vivo, and nonhuman primates88.
Another crucial factor determining the translational gap between preclinical and clinical studies is the unavailability of well-characterized models that can reproduce preclinical results during clinical research. Although several diseases can be reproduced in small animals, there are likely to be certain differences that result in failure of clinical trials despite promising preclinical data. This is due to several factors, including diverse anatomy, pathophysiology, disease phenotype, and transcriptomic profiles in human versus research animals. Nevertheless, several diseases are still in their preliminary research stages, which contributes to an insufficient understanding of the disease. There has been an emerging advancement in 3D organ reconstitution by re-cellularizing scaffolds from animal or non-transplantable human organs for studying various diseases. To date, the greatest progress has been achieved in developing epithelial models of the lungs and skin89,90. Subsequently, researchers working in the field of dermatology have also experienced a lack of ideal animal models due to the diversity in the skin morphology and disease pathology in humans versus small animals. Studies have indicated that there is 70% genetic variation between the skin of mice and humans. Consequently, researchers have revolutionized preclinical testing by developing microfluidic organ-on-a-chip tissue cultures that act as 3D organ models to evaluate topical RNA nanomedicine in skin regeneration89,91,92,93. These in situ and ex vivo gene editing systems using transfected skin and transgenic skin sheets mimic the human setting of the skin diseases, as opposed to animal skin, further offering an advantage since it is possible to combine different cells and extracellular matrices to study a combined pharmacological response. This discussion implies that there is an urgent need for further studies utilizing interdisciplinary approaches for a successful RNA-based nanotherapy to address the issue of immune activation.
Conclusion
Call to action: short-term and long-term goals
Currently, organ transplantation remains the standard of care for most end-stage organ diseases, and this mostly varies patient to patient depending on various factors, including disease state, presence of other diseases, disease heterogeneity, and the ability to metabolize drugs. Unfortunately, certain patients are also ineligible for organ transplantation, and therefore, a dire need for alternate therapeutic options arises. Over the past decades, regenerative medicine has offered novel insights into end-stage organ disease management and could emerge as an excellent therapeutic option for several diseases that are currently unmanageable. Also, owing to the challenges in the development of safe and efficacious regenerative medicine, RNA nanotechnologies present with exceptional features that could accelerate the development of safe, effective, and targeted regenerative therapies in patients with chronic and end-organ disease. Furthermore, combination therapies of RNA therapeutics with cell therapy or tissue engineering approaches have also shown exceptional progress in the field of cardiac regeneration through pre-treatment of hiPSC-derived cardiomyocytes with modified RNAs encoding for salutary factors that can improve engraftment and/or functional outcomes in preclinical models. This combined approach shows tremendous hope in the advancement of regenerative medicine. Through this paper, we envision the unprecedented promise of RNA nanotechnologies for the development of novel regenerative medicine.
References
Kim, Y. K. RNA therapy: rich history, various applications and unlimited future prospects. Exp. Mol. Med. 54, 455–465 (2022).
Payne, D. RNA therapies. Nature 574, S1 (2019).
Yin, W. & Rogge, M. Targeting RNA: a transformative therapeutic strategy. Clin. Transl. Sci. 12, 98–112 (2019).
Jasinski, D., Haque, F., Binzel, D. W. & Guo, P. Advancement of the emerging field of RNA nanotechnology. ACS Nano 11, 1142–1164 (2017).
Zilberzwige-Tal, S. et al. Engineered riboswitch nanocarriers as a possible disease-modifying treatment for metabolic disorders. ACS Nano 16, 11733–11741 (2022).
Pirollo, K. F. & Chang, E. H. Targeted delivery of small interfering RNA: approaching effective cancer therapies. Cancer Res. 68, 1247–1250 (2008).
Thompson, M. G., Sacco, M. T. & Horner, S. M. How RNA modifications regulate the antiviral response. Immunol. Rev. 304, 169–180 (2021).
Nguyen, D. N. et al. Drug delivery-mediated control of RNA immunostimulation. Mol. Ther. 17, 1555–1562 (2009).
Pattanayak, V. et al. High-throughput profiling of off-target DNA cleavage reveals RNA-programmed Cas9 nuclease specificity. Nat. Biotechnol. 31, 839–843 (2013).
Rodriguez, P. L. et al. Minimal “Self” peptides that inhibit phagocytic clearance and enhance delivery of nanoparticles. Science 339, 971–975 (2013).
Pardi, N. et al. Expression kinetics of nucleoside-modified mRNA delivered in lipid nanoparticles to mice by various routes. J. Control Release 217, 345–351 (2015).
Ball, R. L., Hajj, K. A., Vizelman, J., Bajaj, P. & Whitehead, K. A. Lipid nanoparticle formulations for enhanced co-delivery of siRNA and mRNA. Nano Lett. 18, 3814–3822 (2018).
Baden, L. R. et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 384, 403–416 (2021). Key Finding: The mRNA-1273 vaccine demonstrated 94.1% efficacy against COVID-19 illness at median 64 days follow-up, with efficacy maintained at 93.1% through median 148 days of blinded follow-up. Provided substantial protection against asymptomatic infection (63% efficacy) and proved highly effective across diverse populations including those ≥65 years and with comorbidities.
Polack, F. P. et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 383, 2603–2615 (2020). Key Finding: The BNT162b2 vaccine showed 95% efficacy in preventing COVID-19, with similar efficacy across age, sex, race, and ethnic groups. This landmark trial established the clinical viability of mRNA vaccine technology for infectious disease.
Walsh, E. E. et al. Safety and immunogenicity of two RNA-based Covid-19 vaccine candidates. N. Engl. J. Med. 383, 2439–2450 (2020).
Jackson, L. A. et al. An mRNA vaccine against SARS-CoV-2 - preliminary report. N. Engl. J. Med. 383, 1920–1931 (2020).
Pardi, N., Hogan, M. J., Porter, F. W. & Weissman, D. mRNA vaccines - a new era in vaccinology. Nat. Rev. Drug Discov. 17, 261–279 (2018). Key Finding: Comprehensive review establishing the fundamental mechanisms of mRNA vaccines, including nucleoside modifications (pseudouridine incorporation) that enhance translation, reduce immunogenicity, and increase mRNA stability. Outlined the transformative potential of mRNA vaccines beyond infectious diseases.
GBD 2021 Diseases and Injuries Collaborators. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 403, 2133–2161 (2024).
Michalopoulos, G. K. & Bhushan, B. Liver regeneration: biological and pathological mechanisms and implications. Nat. Rev. Gastroenterol. Hepatol. 18, 40–55 (2021).
Gilgenkrantz, H. & Collin de l’Hortet, A. Understanding liver regeneration: from mechanisms to regenerative medicine. Am. J. Pathol. 188, 1316–1327 (2018).
Michalopoulos, G. K. Hepatostat: liver regeneration and normal liver tissue maintenance. Hepatology 65, 1384–1392 (2017).
Hora, S. & Wuestefeld, T. Liver injury and regeneration: current understanding, new approaches, and future perspectives. Cells 12, 2129 (2023).
Adams, D. et al. Patisiran, an RNAi therapeutic, for hereditary transthyretin amyloidosis. N. Engl. J. Med 379, 11–21 (2018). Key Finding: Patisiran, the first FDA-approved RNAi therapeutic, significantly improved neuropathy scores (difference of −19.8 points in mNIS+7) compared to placebo at 18 months. This demonstrated that lowering transthyretin levels by up to 90% through targeted mRNA degradation can halt or reverse disease progression in hereditary transthyretin amyloidosis with polyneuropathy.
Kulkarni, J. A., Cullis, P. R. & van der Meel, R. Lipid nanoparticles enabling gene therapies: from concepts to clinical utility. Nucleic Acid Ther. 28, 146–157 (2018). Key Finding: Detailed the critical role of ionizable lipids in LNP formulations that enable pH-dependent endosomal escape, allowing efficient cytosolic delivery of nucleic acids. Established design principles for LNP optimization including the importance of pKa values between 6.2-6.5 for optimal delivery.
Paunovska, K., Loughrey, D. & Dahlman, J. E. Drug delivery systems for RNA therapeutics. Nat. Rev. Genet. 23, 265–280 (2022).
Crooke, S. T. Molecular mechanisms of antisense oligonucleotides. Nucleic Acid Ther. 27, 70–77 (2017). Key Finding: Comprehensive analysis of ASO mechanisms of action, including RNase H1-mediated mRNA degradation, splice modulation, and effects on RNA processing. Established structure-activity relationships and chemical modification strategies (2’-methoxyethyl, phosphorothioate linkages) that improve stability, binding affinity, and reduce immunostimulation.
Murphy DE. et al. Extracellular vesicle-based therapeutics: natural versus engineered targeting and trafficking. Exp. Mol. Med. 51, 1–12 (2019).
Andaloussi, S. E. L., Mäger, I., Breakefield, X. O. & Wood, M. J. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat. Rev. Drug Discov. 12, 347–357 (2013).
Lai, R. C. et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 4, 214–222 (2010).
Gallego-Perez, D. et al. Topical tissue nano-transfection mediates non-viral stroma reprogramming and rescue. Nat. Nanotechnol. 12, 974–979 (2017).
Kumar, S. et al. RNA-targeting strategies as a platform for ocular gene therapy. Prog. Retin Eye Res. 92, 101110 (2023).
Van Gelder, R. N. et al. Regenerative and restorative medicine for eye disease. Nat. Med. 28, 1149–1156 (2022).
Tang, Z., Fan, X., Chen, Y. & Gu, P. Ocular nanomedicine. Adv. Sci. 9, e2003699 (2022).
Sahle, F. F. et al. Nanotechnology in regenerative ophthalmology. Adv. Drug Deliv. Rev. 148, 290–307 (2019).
Dulla, K. et al. Antisense oligonucleotide-based treatment of retinitis pigmentosa caused by USH2A exon 13 mutations. Mol. Ther. 29, 2441–2455 (2021).
Lin, B. et al. Nanomedicine directs neuronal differentiation of neural stem cells via silencing long noncoding RNA for stroke therapy. Nano Lett. 21, 806–815 (2021).
Vissers, C., Ming, G. L. & Song, H. Nanoparticle technology and stem cell therapy team up against neurodegenerative disorders. Adv. Drug Deliv. Rev. 148, 239–251 (2019).
Mazzini, L. et al. Human neural stem cell transplantation in ALS: initial results from a phase I trial. J. Transl. Med. 13, 17 (2015).
Santos, T., Boto, C., Saraiva, C. M., Bernardino, L. & Ferreira, L. Nanomedicine approaches to modulate neural stem cells in brain repair. Trends Biotechnol. 34, 437–439 (2016).
Wang, X., Gao, M., Wang, Y. & Zhang, Y. The progress of pluripotent stem cell-derived pancreatic β-cells regeneration for diabetic therapy. Front. Endocrinol. 13, 927324 (2022).
Hu, X. et al. Human hypoimmune primary pancreatic islets avoid rejection and autoimmunity and alleviate diabetes in allogeneic humanized mice. Sci. Transl. Med. 15, eadg5794 (2023).
Melamed, J. R. et al. Ionizable lipid nanoparticles deliver mRNA to pancreatic β cells via macrophage-mediated gene transfer. Sci. Adv. 9, eade1444 (2023). Key Finding: Discovered that intraperitoneal administration of LNPs with cationic helper lipids produces robust and specific protein expression in pancreatic β cells through an unexpected mechanism involving macrophage-mediated horizontal gene transfer via exosomes. This represents a breakthrough for potential diabetes gene therapies.
Griffin, C. et al. Underestimation of cirrhosis-related mortality in the Medicare eligible population, 1999-2018. Clin. Gastroenterol. Hepatol. 21, 223–5.e3 (2023).
Byass, P. The global burden of liver disease: a challenge for methods and for public health. BMC Med. 12, 159 (2014).
Li, Y. et al. The diagnosis of hepatic fibrosis by magnetic resonance and near-infrared imaging using dual-modality nanoparticles. RSC Adv. 8, 6699–6708 (2018).
Bakrania, A., Zheng, G. & Bhat, M. Nanomedicine in hepatocellular carcinoma: a new frontier in targeted cancer treatmen. Pharmaceutics 14, 41 (2021).
Bakrania, A., Mo, Y., Zheng, G. & Bhat, M. RNA nanomedicine in liver diseases. Hepatology 81, 1847–1877 (2025).
Bakrania, A., To, J., Zheng, G. & Bhat, M. Targeting Wnt-β-catenin signaling pathway for hepatocellular carcinoma nanomedicine. Gastro Hep Adv. 2, 948–963 (2023).
Bakrania, A., Joshi, N., Zhao, X., Zheng, G. & Bhat, M. Artificial intelligence in liver cancers: decoding the impact of machine learning models in clinical diagnosis of primary liver cancers and liver cancer metastases. Pharm. Res. 189, 106706 (2023).
Han, X. et al. Ligand-tethered lipid nanoparticles for targeted RNA delivery to treat liver fibrosis. Nat. Commun. 14, 75 (2023). Key Finding: Engineered LNPs with vitamin A-coupled lipids specifically target hepatic stellate cells, the primary drivers of liver fibrosis. Demonstrated effective mRNA delivery for targeted protein expression and therapeutic intervention in liver fibrosis models, representing a significant advance in cell-specific delivery within organs.
Hamilton, A. G., Swingle, K. L. & Mitchell, M. J. Biotechnology: overcoming biological barriers to nucleic acid delivery using lipid nanoparticles. PLoS Biol. 21, e3002105 (2023).
Breda, L. et al. In vivo hematopoietic stem cell modification by mRNA delivery. Science 381, 436–443 (2023). Key Finding: Demonstrated successful in vivo genetic modification of hematopoietic stem cells using CD117-targeted LNPs delivering base editor mRNA and guide RNAs. Achieved durable editing in long-term repopulating stem cells without myeloablation, opening new possibilities for treating blood disorders.
Qiu, Y. et al. Effective mRNA pulmonary delivery by dry powder formulation of PEGylated synthetic KL4 peptide. J. Control Release 314, 102–115 (2019).
Qiu, Y. et al. From pulmonary surfactant, synthetic KL4 peptide as effective siRNA delivery vector for pulmonary delivery. Mol. Pharm. 14, 4606–4617 (2017).
Ryals, R. C. et al. The effects of PEGylation on LNP based mRNA delivery to the eye. PLoS ONE 15, e0241006 (2020).
Kularatne, R. N., Crist, R. M. & Stern, S. T. The future of tissue-targeted lipid nanoparticle-mediated nucleic acid delivery. Pharmaceuticals 15, 897 (2022).
Akinc, A. et al. Targeted delivery of RNAi therapeutics with endogenous and exogenous ligand-based mechanisms. Mol. Ther. 18, 1357–1364 (2010).
Scalzo, S. et al. Ionizable lipid nanoparticle-mediated delivery of plasmid DNA in cardiomyocytes. Int. J. Nanomed. 17, 2865–2881 (2022).
Dilliard, S. A., Cheng, Q. & Siegwart, D. J. On the mechanism of tissue-specific mRNA delivery by selective organ targeting nanoparticles. Proc. Natl. Acad. Sci. USA 118, e2109256118 (2021).
Cheng, Q. et al. Selective organ targeting (SORT) nanoparticles for tissue-specific mRNA delivery and CRISPR-Cas gene editing. Nat. Nanotechnol. 15, 313–320 (2020). Key Finding: Demonstrated that adding permanent cationic lipids to LNP formulations enables tissue-specific mRNA delivery beyond the liver, achieving targeted delivery to lungs, spleen, and other organs. This breakthrough addresses a major limitation in RNA therapeutic delivery and enables extrahepatic applications.
Chow, M. Y. T., Qiu, Y. & Lam, J. K. W. Inhaled RNA therapy: from promise to reality. Trends Pharmacol. Sci. 41, 715–729 (2020).
Rowe, S. M. et al. Inhaled mRNA therapy for treatment of cystic fibrosis: Interim results of a randomized, double-blind, placebo-controlled phase 1/2 clinical study. J. Cyst. Fibros. 22, 656–664 (2023).
Li, B. et al. Combinatorial design of nanoparticles for pulmonary mRNA delivery and genome editing. Nat. Biotechnol. 41, 1410–1415 (2023).
Rabiee, N. et al. Polymeric nanoparticles for nasal drug delivery to the brain: relevance to Alzheimer’s disease. Adv. Ther. 4, 2000076 (2021).
van Woensel, M. et al. Formulations for intranasal delivery of pharmacological agents to combat brain disease: a new opportunity to tackle GBM? Cancers 5, 1020–1048 (2013).
Herrera-Barrera, M. et al. Peptide-guided lipid nanoparticles deliver mRNA to the neural retina of rodents and nonhuman primates. Sci. Adv. 9, eadd4623 (2023).
Gautam, M., Ryals, R. C. & Sahay, G. Novel lipid nanoparticle variants for in-vivo mRNA delivery to photoreceptors. Invest. Ophthalmol. Vis. Sci. 63, 69 – A0042–69 – A (2022).
Ryals, R. C. et al. Lipid nanoparticles transfect multiple retinal cell types in the non-human primate. Invest. Ophthalmol. Vis. Sci. 63, 3475 (2022).
Winkle, M., El-Daly, S. M., Fabbri, M. & Calin, G. A. Noncoding RNA therapeutics - challenges and potential solutions. Nat. Rev. Drug Discov. 20, 629–651 (2021).
Barton, G. M. & Medzhitov, R. Toll-like receptor signaling pathways. Science 300, 1524–1525 (2003).
Karikó, K. et al. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol. Ther. 16, 1833–1840 (2008).
Sioud, M. Single-stranded small interfering RNA are more immunostimulatory than their double-stranded counterparts: a central role for 2’-hydroxyl uridines in immune responses. Eur. J. Immunol. 36, 1222–1230 (2006).
Morrissey, D. V. et al. Potent and persistent in vivo anti-HBV activity of chemically modified siRNAs. Nat. Biotechnol. 23, 1002–1007 (2005).
Judge, A. D., Bola, G., Lee, A. C. & MacLachlan, I. Design of noninflammatory synthetic siRNA mediating potent gene silencing in vivo. Mol. Ther. 13, 494–505 (2006).
Cirak, S. et al. Exon skipping and dystrophin restoration in patients with Duchenne muscular dystrophy after systemic phosphorodiamidate morpholino oligomer treatment: an open-label, phase 2, dose-escalation study. Lancet 378, 595–605 (2011).
Kim, S. C. et al. Modifications of mRNA vaccine structural elements for improving mRNA stability and translation efficiency. Mol. Cell Toxicol. 18, 1–8 (2022).
Irie, A., Sato, K., Hara, R. I., Wada, T. & Shibasaki, F. An artificial cationic oligosaccharide combined with phosphorothioate linkages strongly improves siRNA stability. Sci. Rep. 10, 14845 (2020).
Hassler, M. R. et al. Comparison of partially and fully chemically-modified siRNA in conjugate-mediated delivery in vivo. Nucleic Acids Res. 46, 2185–2196 (2018).
Selvam, C., Mutisya, D., Prakash, S., Ranganna, K. & Thilagavathi, R. Therapeutic potential of chemically modified siRNA: recent trends. Chem. Biol. Drug Des. 90, 665–678 (2017).
Chien, K. R. Regenerative medicine and human models of human disease. Nature 453, 302–305 (2008).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Osafune, K. et al. Marked differences in differentiation propensity among human embryonic stem cell lines. Nat. Biotechnol. 26, 313–315 (2008).
Laugwitz, K. L. et al. Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature 433, 647–653 (2005).
Moretti, A. et al. Multipotent embryonic isl1+ progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 127, 1151–1165 (2006).
Zakrzewski, J. L., van den Brink, M. R. & Hubbell, J. A. Overcoming immunological barriers in regenerative medicine. Nat. Biotechnol. 32, 786–794 (2014).
Havel, P. J., Kievit, P., Comuzzie, A. G. & Bremer, A. A. Use and importance of nonhuman primates in metabolic disease research: current state of the field. Ilar J. 58, 251–268 (2017).
Patel, S. et al. Boosting intracellular delivery of lipid nanoparticle-encapsulated mRNA. Nano Lett. 17, 5711–5718 (2017).
Hatit, M. Z. C. et al. Species-dependent in vivo mRNA delivery and cellular responses to nanoparticles. Nat. Nanotechnol. 17, 310–318 (2022).
Weinhart, M., Hocke, A., Hippenstiel, S., Kurreck, J. & Hedtrich, S. 3D organ models-revolution in pharmacological research? Pharm. Res. 139, 446–451 (2019).
Zscheppang, K. et al. Human pulmonary 3D models for translational research. Biotechnol. J. 13, 1700341 (2018).
Jaymand, M. Chemically modified natural polymer-based theranostic nanomedicines: are they the golden gate toward a de novo clinical approach against cancer? ACS Biomater. Sci. Eng. 6, 134–166 (2020).
Feng, J. J. & Hedtrich, S. A similarity scaling approach for organ-on-chip devices. Lab Chip 22, 3663–3667 (2022).
Witting, M., Obst, K., Friess, W. & Hedtrich, S. Recent advances in topical delivery of proteins and peptides mediated by soft matter nanocarriers. Biotechnol. Adv. 33, 1355–1369 (2015).
Acknowledgements
We would like to thank MbD CWG for funding us to organize this workshop that brought together renowned scientists in the field of regenerative medicine.
Author information
Authors and Affiliations
Contributions
A.B. and A.L. participated in the literature search, writing and revising the manuscript; R.V., M.L., E.J., H.H., L.R., S.H., G.S., A.B., M.S., B.L., and G.Z. participated in the workshop as speakers and discussants. They also participated in reviewing, providing critical feedback, and editing the manuscript. M.B. and G.Z. led the workshop and reviewed the manuscript at each stage. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bakrania, A., Nguyen Lefebvre, AT., de Vries, R. et al. Advancing regenerative medicine with RNA nanotechnology for chronic and end organ diseases. Commun Med 6, 214 (2026). https://doi.org/10.1038/s43856-026-01476-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43856-026-01476-y