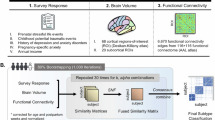
Abstract
Background
Postpartum depression is a common and disabling condition that differs from major depressive disorder and shows marked variation in symptoms and outcomes. Identifying distinct biological subtypes could improve diagnosis and treatment. The present study aims to uncover neurophysiological subtypes of postpartum depression and explore their underlying neural and molecular features.
Methods
We analyzed structural brain images from a cohort of postpartum women recruited at the West China Second Hospital, Sichuan University, including 76 patients with postpartum depression (age range: 24-39 years) and 62 healthy postpartum women (age range: 23-40 years). An unsupervised clustering approach was applied to gray matter volume patterns to identify neurobiological subtypes. Individualized structural covariance networks were then constructed to compare subtype-specific connectivity. Transcriptomic profiles and neurotransmitter density maps were further integrated to examine molecular mechanisms underlying the structural alterations.
Results
Here we show that postpartum depression can be divided into two neurobiological subtypes. Subtype 1 displays reduced gray matter volume in the dorsal attention network, consistent with cognitive impairments. Subtype 2 shows increased gray matter volume in the default mode network, reflecting emotional dysregulation. Subtype 2 also exhibits weaker structural connectivity between the middle temporal gyrus, parahippocampus, and amygdala. Molecular analysis indicates that Subtype 1 is related to energy metabolism and the neurotransmitter receptor mGluR5, whereas Subtype 2 is associated with synaptic regulation, neuroplasticity, and neurotransmitter receptors such as 5-HT1B, dopamine D2, cholinergic M1 and μ-opioid receptor (MOR).
Conclusions
These findings suggest that postpartum depression comprises two biologically distinct forms with different cognitive and emotional characteristics. Recognizing these subtypes may enhance our understanding of its neuropathology and support the development of personalized therapeutic strategies.
Plain language summary
After childbirth, some women experience a serious form of depression called postpartum depression. This condition affects thinking, emotions, and daily life, but not all women experience it in the same way. In this study, we used brain scans to see if there are different types of postpartum depression based on brain structure. We found two groups: one showing changes in brain areas linked to attention and thinking, and another showing changes in regions involved in emotions. These differences were also reflected in brain chemistry and gene activity. Our findings suggest that postpartum depression is not one single disorder, and understanding its biological types may help doctors offer more personalized care and treatment in the future.
Similar content being viewed by others
References
Payne, J. L. & Maguire, J. Pathophysiological mechanisms implicated in postpartum depression. Front. Neuroendocrinol. 52, 165–180 (2019).
Falah-Hassani, K., Shiri, R. & Dennis, C. L. Prevalence and risk factors for comorbid postpartum depressive symptomatology and anxiety. J. Affect. Disord. 198, 142–147 (2016).
Batt, M. M., Duffy, K. A., Novick, A. M., Metcalf, C. A. & Epperson, C. N. Is postpartum depression different from depression occurring outside of the perinatal period? A review of the evidence. Focus (Am. Psychiatr. Publ.) 18, 106–119 (2020).
Pearlstein, T., Howard, M., Salisbury, A. & Zlotnick, C. Postpartum depression. Am. J. Obstet. Gynecol. 200, 357–364 (2009).
Cheng, B. et al. Postpartum depression and major depressive disorder: the same or not? Evidence from resting-state functional MRI. Psychoradiology 2, 121–128 (2022).
Wisner, K. L., Moses-Kolko, E. L. & Sit, D. K. Postpartum depression: a disorder in search of a definition. Arch. Women’s. Ment. Health 13, 37–40 (2010).
Farr, S. L., Dietz, P. M., O’Hara, M. W., Burley, K. & Ko, J. Y. Postpartum anxiety and comorbid depression in a population-based sample of women. J. Women’s Health (2002) 23, 120–128 (2014).
Goodman, J. H. Paternal postpartum depression, its relationship to maternal postpartum depression, and implications for family health. J. Adv. Nurs. 45, 26–35 (2004).
Hendrick, V., Altshuler, L., Strouse, T. & Grosser, S. Postpartum and nonpostpartum depression: differences in presentation and response to pharmacologic treatment. Depress. Anxiety 11, 66–72 (2000).
Cheng, B. et al. Abnormal dynamics of resting-state functional activity and couplings in postpartum depression with and without anxiety. Cereb. Cortex 32, 5597–5608 (2022).
Cheng, B. et al. Social support mediates the influence of cerebellum functional connectivity strength on postpartum depression and postpartum depression with anxiety. Transl. Psychiatry 12, 54 (2022).
Cheng, B. et al. Altered functional connectivity density and couplings in postpartum depression with and without anxiety. Soc. Cogn. Affect. Neurosci. 17, 756–766 (2022).
Brucar, L. R., Feczko, E., Fair, D. A. & Zilverstand, A. Current approaches in computational psychiatry for the data-driven identification of brain-based subtypes. Biol. Psychiatry 93, 704–716 (2023).
Drysdale, A. T. et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat. Med. 23, 28–38 (2017).
Chen, J. et al. Neural–genetic–environmental evidence for a disease factor in mental and physical health multimorbidity. Nat. Commun. 16, 10088 (2025).
Wen, J. V. E. et al. Subtyping brain diseases from imaging data. In Machine Learning for Brain Disorders Ch. 16 (ed., Colliot, O.) (Humana, 2023).
Wang, H., Jin, X., Zhang, Y. & Wang, J. Single-subject morphological brain networks: connectivity mapping, topological characterization and test–retest reliability. Brain Behav. 6, e00448 (2016).
Sun, H. et al. Mapping individual structural covariance network in development brain with dynamic time warping. Cereb. Cortex 34, bhae039 (2024).
Kassem, M. S. et al. Stress-induced grey matter loss determined by mri is primarily due to loss of dendrites and their synapses. Mol. Neurobiol. 47, 645–661 (2013).
Gogtay, N. & Thompson, P. M. Mapping gray matter development: implications for typical development and vulnerability to psychopathology. Brain Cogn. 72, 6–15 (2010).
Cheng, B. et al. Prolactin mediates the relationship between regional gray matter volume and postpartum depression symptoms. J. Affect. Disord. 301, 253–259 (2022).
Alexander-Bloch, A., Giedd, J. N. & Bullmore, E. Imaging structural co-variance between human brain regions. Nat. Rev. Neurosci. 14, 322–336 (2013).
Mechelli, A., Friston, K. J., Frackowiak, R. S. & Price, C. J. Structural covariance in the human cortex. J. Neurosci. 25, 8303–8310 (2005).
Zielinski, B. A., Gennatas, E. D., Zhou, J. & Seeley, W. W. Network-level structural covariance in the developing brain. Proc. Natl. Acad. Sci. USA 107, 18191–18196 (2010).
Montembeault, M. et al. The impact of aging on gray matter structural covariance networks. NeuroImage 63, 754–759 (2012).
Zhang, S. et al. Using individualized structural covariance networks to analyze the heterogeneity of cerebral small vessel disease with cognitive impairment. J. Stroke Cerebrovasc. Dis. 33, 107829 (2024).
Han, S. et al. Resolving heterogeneity in depression using individualized structural covariance network analysis. Psychol. Med. 53, 5312–5321 (2023).
Liu, Z. et al. Resolving heterogeneity in schizophrenia through a novel systems approach to brain structure: individualized structural covariance network analysis. Mol. Psychiatry 26, 7719–7731 (2021).
Zhang, H. et al. Reorganization of cortical individualized differential structural covariance network is associated with regional morphometric changes in chronic subcortical stroke. NeuroImage Clin. 45, 103735 (2025).
Li, W. et al. Disentangling the switching behavior in functional connectivity dynamics in autism spectrum disorder: insights from developmental cohort analysis and molecular-cellular associations. Adv. Sci. (Weinh.) 12, e2403801 (2025).
Cox, J. L., Holden, J. M. & Sagovsky, R. Detection of postnatal depression. Development of the 10-item Edinburgh Postnatal Depression Scale. Br. J. Psychiatry 150, 782–786 (1987).
Beck, A. T., Ward, C. H., Mendelson, M., Mock, J. & Erbaugh, J. An inventory for measuring depression. Arch. Gen. Psychiatry 4, 561–571 (1961).
Beck, A. T., Epstein, N., Brown, G. & Steer, R. A. An inventory for measuring clinical anxiety: psychometric properties. J. Consult Clin. Psychol. 56, 893–897 (1988).
Buysse, D. J., Reynolds, C. F. 3rd, Monk, T. H., Berman, S. R. & Kupfer, D. J. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 28, 193–213 (1989).
Logsdon, M. C. & Usui, W. M. The Postpartum Support Questionnaire: psychometric properties in adolescents. J. Child Adolesc. Psychiatr. Nurs. 19, 145–156 (2006).
Cohen, S. & Williamson, G. M. Perceived stress in a probability sample of the United States. In S. Spacapan & S. Oskamp (Eds.), In CLAR Symposium Soc. Psychol. Health, 31–67 (Sage Publications, Inc. 1988).
Ashburner, J. A fast diffeomorphic image registration algorithm. Neuroimage 38, 95–113 (2007).
Margulies, D. S. et al. Situating the default-mode network along a principal gradient of macroscale cortical organization. Proc. Natl. Acad. Sci. USA 113, 12574–12579 (2016).
Hawrylycz, M. J. et al. An anatomically comprehensive atlas of the adult human brain transcriptome. Nature 489, 391–399 (2012).
Markello, R. D. et al. Standardizing workflows in imaging transcriptomics with the abagen toolbox. Elife 10, https://doi.org/10.7554/eLife.72129 (2021).
Arnatkeviciute, A., Fulcher, B. D. & Fornito, A. A practical guide to linking brain-wide gene expression and neuroimaging data. Neuroimage 189, 353–367 (2019).
Schaefer, A. et al. Local–global parcellation of the human cerebral cortex from intrinsic functional connectivity MRI. Cereb. Cortex 28, 3095–3114 (2018).
Hansen, J. Y. et al. Mapping neurotransmitter systems to the structural and functional organization of the human neocortex. Nat. Neurosci. 25, 1569–1581 (2022).
Bauer, A. E. et al. Identification and validation of postpartum depression subtypes: a population-based cohort study. EClinicalMedicine 89, 103540 (2025).
Chen, K. et al. Shared and disorder-specific large-scale intrinsic and effective functional network connectivities in postpartum depression with and without anxiety. Cereb. Cortex 34, https://doi.org/10.1093/cercor/bhae478 (2024).
Chen, K. X. et al. Molecular basis underlying default mode network functional abnormalities in postpartum depression with and without anxiety. Hum. Brain Mapp. 45, e26657 (2024).
Batty, M. J. et al. Cortical gray matter in attention-deficit/hyperactivity disorder: a structural magnetic resonance imaging study. J. Am. Acad. Child Adolesc. Psychiatry 49, 229–238 (2010).
Cafiero, P. J. & Justich Zabala, P. Postpartum depression: impact on pregnant women and the postnatal physical, emotional, and cognitive development of their children. An ecological perspective. Arch. Argent. Pediatr. 122, e202310217 (2024).
Qiu, T., Wen, H., Liu, Z. X., Pan, X. P. & Zeng, T. Investigation regarding early cognitive function of women in the postpartum period and the analysis of influencing factors. Risk Manag. Health Policy 14, 3747–3754 (2021).
Ives-Deliperi, V. & Butler, J. T. Mechanisms of cognitive impairment in temporal lobe epilepsy: a systematic review of resting-state functional connectivity studies. Epilepsy Behav. 115, 107686 (2021).
Huang, S., Li, M., Huang, C. & Liu, J. Acute limbic system connectivity predicts chronic cognitive function in mild traumatic brain injury: an individualized differential structural covariance network study. Pharm. Res. 206, 107274 (2024).
Liu, Y. et al. Transcriptional patterns of amygdala functional connectivity in first-episode, drug-naive major depressive disorder. Transl. Psychiatry 14, 351 (2024).
Ramasubbu, R. et al. Reduced intrinsic connectivity of amygdala in adults with major depressive disorder. Front. Psychiatry 5, 17 (2014).
Sun, H. et al. Functional connectivity analyses of individual hippocampal subregions in major depressive disorder with electroconvulsive therapy. Psychoradiology 4, kkae030 (2024).
Moreira, L. K. S. et al. Post-partum depression: from clinical understanding to preclinical assessments. Front. Psychiatry 14, 1173635 (2023).
Okun, M. L. Disturbed sleep and postpartum depression. Curr. Psychiatry Rep. 18, 66 (2016).
Tabaeifard, R. et al. Association between oxidative balance score and risk of postpartum depression in Iranian women: a prospective cohort study. Sci. Rep. 15, 8590 (2025).
Gorman-Sandler, E. et al. Mitochondrial might: powering the peripartum for risk and resilience. Front. Behav. Neurosci. 17, 1286811 (2023).
McEwen, A. M. et al. Increased glutamate levels in the medial prefrontal cortex in patients with postpartum depression. Neuropsychopharmacology 37, 2428–2435 (2012).
Ghuman, A. et al. Prospective investigation of glutamate levels and percentage gray matter in the medial prefrontal cortex in females at risk for postpartum depression. Curr. Neuropharmacol. 20, 1988–2000 (2022).
Quoidbach, J., Mikolajczak, M. & Gross, J. J. Positive interventions: an emotion regulation perspective. Psychol. Bull. 141, 655–693 (2015).
Brown, J. V. E. et al. Antidepressant treatment for postnatal depression. Cochrane Database Syst. Rev. 2, CD013560, (2021).
Raichle, M. E. The brain’s default mode network. Annu. Rev. Neurosci. 38, 433–447 (2015).
Raichle, M. E. et al. A default mode of brain function. Proc. Natl. Acad. Sci. USA 98, 676–682 (2001).
Chase, H. W., Moses-Kolko, E. L., Zevallos, C., Wisner, K. L. & Phillips, M. L. Disrupted posterior cingulate–amygdala connectivity in postpartum depressed women as measured with resting BOLD fMRI. Soc. Cogn. Affect. Neurosci. 9, 1069–1075 (2014).
Duan, C., Cosgrove, J. & Deligiannidis, K. M. Understanding peripartum depression through neuroimaging: a review of structural and functional connectivity and molecular imaging research. Curr. Psychiatry Rep. 19, 70 (2017).
Kim, P. et al. The plasticity of human maternal brain: longitudinal changes in brain anatomy during the early postpartum period. Behav. Neurosci. 124, 695–700 (2010).
Wang, J. et al. The critical mediating roles of the middle temporal gyrus and ventrolateral prefrontal cortex in the dynamic processing of interpersonal emotion regulation. Neuroimage 300, 120789 (2024).
Šimić, G. et al. Understanding emotions: origins and roles of the amygdala. Biomolecules 11, https://doi.org/10.3390/biom11060823 (2021).
Zhu, Y. et al. Emotion regulation of hippocampus using real-time fMRI neurofeedback in healthy human. Front. Hum. Neurosci. 13, 242 (2019).
Landi, N. et al. Maternal neural responses to infant cries and faces: relationships with substance use. Front. Psychiatry 2, 32 (2011).
Chen, G. et al. Environmental enrichment attenuates depressive-like behavior in maternal rats by inhibiting neuroinflammation and apoptosis and promoting neuroplasticity. Neurobiol. Stress 30, 100624 (2024).
Post, C. & Leuner, B. The maternal reward system in postpartum depression. Arch. Women’s Ment. Health 22, 417–429 (2019).
Jacob, Y. et al. Neural correlates of rumination in major depressive disorder: a brain network analysis. Neuroimage Clin. 25, 102142 (2020).
Benarroch, E. E. Synaptic effects of cannabinoids: complexity, behavioral effects, and potential clinical implications. Neurology 83, 1958–1967 (2014).
Ribeiro, S. C., Kennedy, S. E., Smith, Y. R., Stohler, C. S. & Zubieta, J. K. Interface of physical and emotional stress regulation through the endogenous opioid system and mu-opioid receptors. Prog. Neuropsychopharmacol. Biol. Psychiatry 29, 1264–1280 (2005).
Delgado, P. L. Depression: the case for a monoamine deficiency. J. Clin. Psychiatry 61(Suppl. 6), 7–11 (2000).
Della Corte, L. et al. Prevalence and associated psychological risk factors of postpartum depression: a cross-sectional study. J. Obstet. Gynaecol. 42, 976–980 (2022).
Hopkins, J., Marcus, M. & Campbell, S. B. Postpartum depression: a critical review. Psychol. Bull. 95, 498–515 (1984).
Costa-Brito, A. R., Goncalves, I. & Santos, C. R. A. The brain as a source and a target of prolactin in mammals. Neural Regen. Res. 17, 1695–1702 (2022).
Xu, Y. et al. The association between prolactin levels and cognitive function in female patients with severe mental disorders. Psychiatry Investig. 21, 832–837 (2024).
Acknowledgements
This study was supported by Yunnan Fundamental Research Projects (202501AV070005), National Natural Science Foundation of China (32560206), Yunnan Provincial International Joint R&D Center for Precision Healthcare in Maternal-Fetal Medicine (202503AP140051), Key Research and Development Plan of Yunnan Province (202403AC100002), Sichuan Science and Technology Program (2024TFFK0361), Natural Science Foundation of Chongqing (CSTB2024NSCQ-MSX1116), and Open Research Fund of the State Key Laboratory of Brain–Machine Intelligence, Zhejiang University (BMI2500002).
Author information
Authors and Affiliations
Contributions
J.W., K.C., B.C., and X.D. contributed to the conception and design of the study. J.C., Y.L., W.L., M.C., and Y.W. contributed to the acquisition and analysis of data; P.X., X.T., and T.Z. contributed to material preparation and data collection; J.C., Y.L., B.C., K.C., and J.W. wrote and edited the manuscript. All the authors discussed the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks Yasmin Harrington, Xiaoyu Tong and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, J., Liang, Y., Li, W. et al. Mapping neurophysiological biotypes of postpartum depression and underlying neural and molecular basis. Commun Med (2026). https://doi.org/10.1038/s43856-026-01477-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43856-026-01477-x