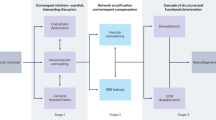
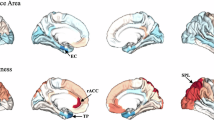
Abstract
Background
Obesity is a major health concern linked to chronic conditions such as diabetes and cardiovascular disease. However, most neurological studies have focused on specific metabolic states, limiting understanding of how brain function changes from fasting to satiety. Furthermore, hypothesis-driven approaches may introduce bias and fail to capture complex neural interactions. This study aimed to identify brain connectivity patterns associated with obesity across different metabolic states using a data-driven approach.
Methods
Electroencephalography data were collected from 30 women with obesity and 30 women without obesity over a four-hour period encompassing fasting and post-meal states. All subjects were aged 20 to 65 years. Functional connectivity was calculated from source-localized signals, and a machine learning framework incorporating a feature selection method was applied to identify the most discriminative connectivity features between groups.
Results
Here we show that six connectivity features classify obesity with 95% accuracy across metabolic states. Reduced connectivity are observed within food-reward processing regions in the obese group, with the dorsal anterior cingulate cortex emerging as a central hub. This pattern reflects a persistent alteration in energy prediction and craving regulation that is independent of metabolic state.
Conclusions
These findings demonstrate that disrupted brain connectivity is a fundamental characteristic of obesity. The results highlight the dorsal anterior cingulate cortex as a key region underlying maladaptive reward processing and suggest that targeting this area through neuromodulation therapies may offer a promising intervention for obesity treatment.
Plain Language Summary
Obesity is a growing health issue that affects both the body and the brain. In this study, we wanted to understand how brain activity differs between people with and without obesity, and whether these differences change from fasting to after eating. We recorded brain signals from women with and without obesity while they moved from hunger to fullness. Using computer-based analysis, we found specific patterns of brain communication that could accurately distinguish the two groups. People with obesity showed weaker connections in brain areas related to food reward and self-control, especially in a region called the anterior cingulate cortex. These results suggest that changes in brain connectivity may underlie overeating and could guide future brain-based treatments for obesity.
Similar content being viewed by others
Data availability
The datasets generated during this study, including anonymized EEG recordings, anthropometric data, and behavioural measures, are available upon reasonable request due to ethical and privacy restrictions. Access is granted for non-commercial, academic purposes only, subject to approval and a signed data use agreement. Requests should be directed to the corresponding author (yuan.yue@otago.ac.nz). Requests will be reviewed and responded to within four weeks. Use of the data is restricted to the approved research purpose, and redistribution to third parties is not permitted. Source data for Figs. 2–4, 6, and 7 are available on Figshare (https://doi.org/10.6084/m9.figshare.30351280.v3)92.
Code availability
The code used in this study, along with mock datasets, is publicly available at https://github.com/yuan410/ObesityMeta/tree/main. Detailed instructions for running the code are provided in the README file within the repository. The code is also archived on Figshare (https://doi.org/10.6084/.m9.figshare.30351418.v1)93.
References
World Health Organization et al. Noncommunicable Diseases Country Profiles 2018 (WHO, 2018).
Scully, T., Ettela, A., LeRoith, D. & Gallagher, E. J. Obesity, type 2 diabetes, and cancer risk. Front. Oncol. 10, 615375 (2021).
Rao, K. R., Lal, N. & Giridharan, N. Genetic & epigenetic approach to human obesity. Indian J. Med. Res. 140, 589–603 (2014).
Castelo-Branco, C. & Soveral, I. The immune system and aging: a review. Gynecol. Endocrinol. 30, 16–22 (2014).
Glaser, R. & Kiecolt-Glaser, J. K. Stress-induced immune dysfunction: implications for health. Nat. Rev. Immunol. 5, 243–251 (2005).
Ochoa-Repáraz, J. & Kasper, L. H. The second brain: is the gut microbiota a link between obesity and central nervous system disorders? Curr. Obes. Rep. 5, 51–64 (2016).
Afzaal, M. et al. Human gut microbiota in health and disease: unveiling the relationship. Front. Microbiol. 13, 999001 (2022).
Carnell, S., Gibson, C., Benson, L., Ochner, C. & Geliebter, A. Neuroimaging and obesity: current knowledge and future directions. Obes. Rev. 13, 43–56 (2012).
Amen, D. G., Wu, J., George, N. & Newberg, A. Patterns of regional cerebral blood flow as a function of obesity in adults. J. Alzheimers. Dis. 77, 1331–1337 (2020).
Syan, S. K. et al. Dysregulated resting state functional connectivity and obesity: a systematic review. Neurosci. Biobehav. Rev. 131, 270–292 (2021).
Price, D. D. Psychological and neural mechanisms of the affective dimension of pain. Science 288, 1769–1772 (2000).
De Ridder, D., Maciaczyk, J. & Vanneste, S. The future of neuromodulation: smart neuromodulation. Expert Rev. Med. Devices 18, 307–317 (2021).
Abreu, A. M. et al. “Action anticipation beyond the action observation network: a functional magnetic resonance imaging study in expert basketball players. Eur. J. Neurosci. 35, 1646–1654 (2012).
Wijngaarden, M. et al. Obesity is marked by distinct functional connectivity in brain networks involved in food reward and salience. Behav. Brain Res. 287, 127–134 (2015).
Milano, W. et al. Depression and obesity: analysis of common biomarkers. Diseases 8, 23 (2020).
Kokka, I., Mourikis, I. & Bacopoulou, F. Psychiatric disorders and obesity in childhood and adolescence—a systematic review of cross-sectional studies. Children 10, 285 (2023).
Saruco, E. & Pleger, B. “A systematic review of obesity and binge eating associated impairment of the cognitive inhibition system. Front. Nutr. 8, 609012 (2021).
Lengton, R., Schoenmakers, M., Penninx, B. W., Boon, M. R. & van Rossum, E. F. Glucocorticoids and hpa axis regulation in the stress–obesity connection: a comprehensive overview of biological, physiological and behavioural dimensions. Clin. Obes. 15, e12725 (2025).
Chen, M.-H. et al. Role of obesity in systemic low-grade inflammation and cognitive function in patients with bipolar i disorder or major depressive disorder. CNS Spectr. 26, 521–527 (2021).
De Ridder, D., Vanneste, S. & Freeman, W. “The bayesian brain: phantom percepts resolve sensory uncertainty. Neurosci. Biobehav. Rev. 44, 4–15 (2014).
Mathar, D., Neumann, J., Villringer, A. & Horstmann, A. Failing to learn from negative prediction errors: Obesity is associated with alterations in a fundamental neural learning mechanism. Cortex 95, 222–237 (2017).
Kullmann, S. et al. The effect of hunger state on hypothalamic functional connectivity in response to food cues. Hum. Brain Mapp. 44, 418–428 (2023).
Pursey, K. M., Contreras-Rodriguez, O., Collins, C. E., Stanwell, P. & Burrows, T. L. Food addiction symptoms and amygdala response in fasted and fed states. Nutrients 11, 1285 (2019).
Wiemerslage, L. et al. A resting-state fmri study of obese females between pre-and postprandial states before and after bariatric surgery. Eur. J. Neurosci. 45, 333–341 (2017).
Zhang, B. et al. Altered baseline brain activity differentiates regional mechanisms subserving biological and psychological alterations in obese men. Sci. Rep. 5, 11563 (2015).
Hogenkamp, P. S. et al. Higher resting-state activity in reward-related brain circuits in obese versus normal-weight females independent of food intake. Int. J. Obes. 40, 1687–1692 (2016).
De Ridder, D. et al. The brain, obesity and addiction: an eeg neuroimaging study. Sci. Rep. 6, 34122 (2016).
Park, B. -y et al. Whole-brain functional connectivity correlates of obesity phenotypes. Hum. Brain Mapp. 41, 4912–4924 (2020).
Wang, J. et al. Multivariate resting-state functional connectomes predict and characterize obesity phenotypes. Cereb. Cortex 33, 8368–8381 (2023).
Baldini, S. et al. Detection of epileptic activity in presumably normal eeg. Brain Commun. 2, fcaa104 (2020).
Griffith, L., Haddad, E. H. & Tonstad, S. Postprandial effects of consuming a staggered meal on gut peptide and glycemic responses in obese women and men. Obes. Res. Clin. Pract. 10, 264–274 (2016).
Haase, L., Green, E. & Murphy, C. Males and females show differential brain activation to taste when hungry and sated in gustatory and reward areas. Appetite 57, 421–434 (2011).
Habas, M. E. & Macdonald, I. Metabolic and cardiovascular responses to liquid and solid test meals. Br. J. Nutr. 79, 241–247 (1998).
Blackman, R. B. & Tukey, J. W. The measurement of power spectra from the point of view of communications engineering—part i. Bell Syst. Tech. J. 37, 185–282 (1958).
Vitacco, D., Brandeis, D., Pascual-Marqui, R. & Martin, E. Correspondence of event-related potential tomography and functional magnetic resonance imaging during language processing. Hum. Brain Mapp. 17, 4–12 (2002).
Worrell, G. A. et al. Localization of the epileptic focus by low-resolution electromagnetic tomography in patients with a lesion demonstrated by mri. Brain Topogr. 12, 273–282 (2000).
Dierks, T. et al. Spatial pattern of cerebral glucose metabolism (pet) correlates with localization of intracerebral eeg-generators in alzheimer’s disease. Clin. Neurophysiol. 111, 1817–1824 (2000).
Zumsteg, D., Friedman, A., Wieser, H. G. & Wennberg, R. A. Propagation of interictal discharges in temporal lobe epilepsy: correlation of spatiotemporal mapping with intracranial foramen ovale electrode recordings. Clin. Neurophysiol. 117, 2615–2626 (2006).
Mazziotta, J. et al. A probabilistic atlas and reference system for the human brain: International consortium for brain mapping (icbm). Philos. Trans. R. Soc. Lond. Ser. B 356, 1293–1322 (2001).
Klimesch, W. Eeg alpha and theta oscillations reflect cognitive and memory performance: a review and analysis. Brain Res. Rev. 29, 169–195 (1999).
Hindriks, R. Relation between the phase-lag index and lagged coherence for assessing interactions in eeg and meg data. Neuroimage Rep. 1, 100007 (2021).
Pascual-Marqui, R. D. et al. Assessing interactions in the brain with exact low-resolution electromagnetictomography. Philos. Trans. R. Soc. A 369, 3768–3784 (2011).
Pascual-Marqui, R. D. Instantaneous and lagged measurements of linear and nonlinear dependence between groups of multivariate time series: frequency decomposition. Preprint at https://arxiv.org/abs/0711.1455 (2007).
De Ridder, D., Manning, P., Leong, S. L., Ross, S. & Vanneste, S. Allostasis in health and food addiction. Sci. Rep. 6, 37126 (2016).
He, H. & Garcia, E. A. Learning from imbalanced data. IEEE Trans. Knowl. Data Eng. 21, 1263–1284 (2009).
Verma, S. A Comparative Study of Oversampling Techniques on Binary Classification for Detecting Duplicate Advertisement. Ph.D. dissertation (National College of Ireland, 2018).
Lee, H. S., Kim, J. H., Son, J., Park, H. & Choi, J. Machine learning models for predicting early hemorrhage progression in traumatic brain injury. Sci. Rep. 14, 11690 (2024).
Nielsen, R. L. et al. Data-driven identification of predictive risk biomarkers for subgroups of osteoarthritis using interpretable machine learning. Nat. Commun. 15, 2817 (2024).
Su, Q. et al. Faecal microbiome-based machine learning for multi-class disease diagnosis. Nat. Commun. 13, 6818 (2022).
Rosenblatt, M., Tejavibulya, L., Jiang, R., Noble, S. & Scheinost, D. Data leakage inflates prediction performance in connectome-based machine learning models. Nat. Commun. 15, 1829 (2024).
Yue, Y. et al. Discovering functional connectivitybased neural signatures of obesity via dual-layer incremental wrapper-based feature selection. In International Conference on Neural Information Processing, 377–391 (Springer, 2024).
Theng, D. & Bhoyar, K. K. Feature selection techniques for machine learning: a survey of more than two decades of research. Knowl. Inf. Syst. 66, 1575–1637 (2024).
Huang, N., Lu, G. & Xu, D. A permutation importance-based feature selection method for short-term electricity load forecasting using random forest. Energies 9, 767 (2016).
Fernández-Delgado, M., Cernadas, E., Barro, S. & Amorim, D. Do we need hundreds of classifiers to solve real world classification problems? J. Mach. Learn. Res. 15, 3133–3181 (2014).
Wu, J. et al. Hyperparameter optimization for machine learning models based on bayesian optimization. J. Electron. Sci. Technol. 17, 26–40 (2019).
Li, G. et al. Brain functional and structural magnetic resonance imaging of obesity and weight loss interventions. Mol. Psychiatry 28, 1466–1479 (2023).
Weis, S. et al. Sex classification by resting state brain connectivity. Cereb. Cortex 30, 824–835 (2020).
Zhao, Z. et al. Classification of schizophrenia by combination of brain effective and functional connectivity. Front. Neurosci. 15, 651439 (2021).
Dinh, S. T. et al. Brain dysfunction in chronic pain patients assessed by resting-state electroencephalography. Pain 160, 2751–2765 (2019).
Rolls, E. T. Taste and smell processing in the brain. Handb. Clin. Neurol. 164, 97–118 (2019).
Byun, M. S. et al. Region-specific association between basal blood insulin and cerebral glucose metabolism in older adults. NeuroImage Clin. 22, 101765 (2019).
Alexander, W. H. & Brown, J. W. The role of the anterior cingulate cortex in prediction error and signaling surprise. Top. Cogn. Sci. 11, 119–135 (2019).
Seabrook, L. T. & Borgland, S. L. The orbitofrontal cortex, food intake and obesity. J. Psychiatry Neurosci. 45, 304–312 (2020).
Donoso, M., Collins, A. G. & Koechlin, E. “Foundations of human reasoning in the prefrontal cortex. Science 344, 1481–1486 (2014).
Wallis, J. D. & Kennerley, S. W. “Contrasting reward signals in the orbitofrontal cortex and anterior cingulate cortex. Ann. N. Y. Acad. Sci. 1239, 33–42 (2011).
De Ridder, D. & Vanneste, S. Visions on the future of medical devices in spinal cord stimulation: what medical device is needed? Expert Rev. Med. devices 13, 233–242 (2016).
Rolls, E. T., Feng, R., Cheng, W. & Feng, J. Orbitofrontal cortex connectivity is associated with food reward and body weight in humans. Soc. Cogn. Affect. Neurosci. 18, nsab083 (2023).
Stillman, C. M. et al. Changes in cerebral perfusion following a 12-month exercise and diet intervention. Psychophysiology 58, e13589 (2021).
Tarantini, S., Csiszar, A. & Ungvari, Z. Midlife obesity impairs neurovascular coupling responses. Obesity 29, 17–17 (2021).
Moulton, E. A. et al. Aversion-related circuitry in the cerebellum: responses to noxious heat and unpleasant images. J. Neurosci. 31, 3795–3804 (2011).
Park, B. -y, Chung, C.-S., Lee, M. J. & Park, H. Accurate neuroimaging biomarkers to predict body mass index in adolescents: a longitudinal study. Brain Imaging Behav. 14, 1682–1695 (2020).
Tuovinen, N. et al. Neurometabolite correlates with personality and stress in healthy emerging adults: a focus on sex differences. Neuroimage 247, 118847 (2022).
Peters, A., McEwen, B. S. & Friston, K. Uncertainty and stress: why it causes diseases and how it is mastered by the brain. Prog. Neurobiol. 156, 164–188 (2017).
Matsumori, K., Koike, Y. & Matsumoto, K. A biased bayesian inference for decision-making and cognitive control. Front. Neurosci. 12, 734 (2018).
Hobson, J. A. & Friston, K. J. Waking and dreaming consciousness: neurobiological and functional considerations. Prog. Neurobiol. 98, 82–98 (2012).
van Pelt, S. et al. Beta-and gamma-band activity reflect predictive coding in the processing of causal events. Soc. Cogn. Affect. Neurosci. 11, 973–980 (2016).
Ng, J., Stice, E., Yokum, S. & Bohon, C. An fmri study of obesity, food reward, and perceived caloric density. does a low-fat label make food less appealing? Appetite 57, 65–72 (2011).
Livneh, Y. et al. Estimation of current and future physiological states in insular cortex. Neuron 105, 1094–1111 (2020).
Arnal, L. H., Doelling, K. B. & Poeppel, D. Delta–beta coupled oscillations underlie temporal prediction accuracy,. Cereb. Cortex 25, 3077–3085 (2015).
Mattavelli, G., Gorrino, I., Tornaghi, D. & Canessa, N. Cognitive and motor impulsivity in the healthy brain, and implications for eating disorders and obesity: a coordinate-based meta-analysis and systematic review. Cortex 171, 90–112 (2024).
Yokum, S. & Stice, E. Relation of overweight/obesity to reward region response to food reward and the moderating effects of parental history of eating pathology in adolescent females. Nutrients 15, 2558 (2023).
Buschman, T. J., Denovellis, E. L., Diogo, C., Bullock, D. & Miller, E. K. Synchronous oscillatory neural ensembles for rules in the prefrontal cortex. Neuron 76, 838–846 (2012).
Tataranni, P. A. et al. Neuroanatomical correlates of hunger and satiation in humans using positron emission tomography. Proc. Natl. Acad. Sci. 96, 4569–4574 (1999).
Mulert, C. et al. Integration of fmri and simultaneous eeg: towards a comprehensive understanding of localization and time-course of brain activity in target detection. Neuroimage 22, 83–94 (2004).
Nguyen-Danse, D. A. et al. Feasibility of reconstructing source functional connectivity with low-density eeg. Brain Topogr. 34, 709–719 (2021).
Volpe, U. et al. The cortical generators of p3a and p3b:a loreta study. Brain Res. Bull. 73, 220–230 (2007).
Pizzagalli, D. et al. Functional but not structural subgenual prefrontal cortex abnormalities in melancholia. Mol. Psychiatry 9, 393–405 (2004).
Zumsteg, D., Lozano, A. M. & Wennberg, R. A. Mesial temporal inhibition in a patient with deep brain stimulation of the anterior thalamus for epilepsy. Epilepsia 47, 1958–1962 (2006).
Clemens, B. et al. Three-dimensional localization of abnormal eeg activity in migraine: a low resolution electromagnetic tomography (loreta) study of migraine patients in the pain-free interval. Brain Topogr. 21, 36–42 (2008).
Clemens, B. et al. Eeg-loreta endophenotypes of the common idiopathic generalized epilepsy syndromes,. Epilepsy Res. 99, 281–292 (2012).
Coveleskie, K. et al. Altered functional connectivity within the central reward network in overweight and obese women. Nutr. Diab. 5, e148–e148 (2015).
Yue, Y. Source data for Machine Learning-Based Identification of Abnormal Functional Connectivity in Obesity Across Different Metabolic States. figshare, https://doi.org/10.6084/m9.figshare.30351280.v3 (2025).
Code script for Machine Learning-Based Identification of Abnormal Functional Connectivity in Obesity Across Different Metabolic States. figshare, https://doi.org/10.6084/m9.figshare.30351418.v1 (2025).
Acknowledgements
We thank all participants who contributed to this study. This research received no external funding.
Author information
Authors and Affiliations
Contributions
Conceptualization: Y.Y., P.M., D.D.R., J.D., D.B.A., M.H.; Methodology: Y.Y., J.D., D.D.R., P.M., D.B.A., M.H., S.R.; Data collection and processing: P.M., S.R., Y.Y.; Data analysis: Y.Y.; Writing—first draft: Y.Y., P.M., D.D.R.; Writing—review and editing: Y.Y., P.M., D.D.R., J.D., D.B.A., M.H. Code implementation: Y.Y., D.A.C. All authors have critically reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Medicine thanks Philipp G. Sämann and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yue, Y., Manning, P., De Ridder, D. et al. Machine learning-based identification of abnormal functional connectivity in obesity across different metabolic states. Commun Med (2026). https://doi.org/10.1038/s43856-026-01518-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43856-026-01518-5