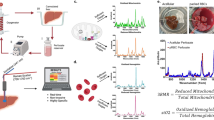
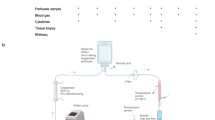
Abstract
Background
Mitochondrial dysfunction is a critical factor in several diseases, but current in situ assessment methods are severely limited. Non-invasive monitoring of mitochondrial redox state using resonance Raman Spectroscopy (RRS) offers a promising solution. This study aims to demonstrate RRS utility with liver models of warm ischemia-reperfusion injury in organ transplantation.
Methods
Lewis rat (female) and Yorkshire pig (both sexes) livers were evaluated during reperfusion by subnormothermic machine perfusion, with 3-6 replicates per study group, and statistical comparisons using unpaired two-tailed Student’s t-tests with Welch’s correction for potentially unequal variance. RRS provides in situ quantification of the overall mitochondrial redox state, and herein further refined to resolve the redox state of individual complex III and IV.
Results
Here we show that RRS can differentiate non-viable rat livers (3 h warm ischemia, WI) from viable 1 h WI and fresh controls as early as 30 mins into reperfusion. RRS also identifies dysfunction at complex III characterized by hyperoxidation during reperfusion. This guides us to test methylene blue, which acts as an alternate electron donor to bypass complex III, as treatment rescuing mitochondria from WI-induced reperfusion injury. When tested on pig marginal livers with extended WI (30-45 mins), our RRS-guided treatment enables recovery of hemodynamics and oxygen/lactate values that approached controls without WI.
Conclusions
RRS assessment and guided treatment with methylene blue provide two lines of evidence indicating that mitochondrial hyperoxidation, specifically at complex III, is a critical mechanism underlying warm ischemia-reperfusion injury. This study demonstrates the potential of RRS for transplantation and broader applications.
Plain language summary
Healthy mitochondria are crucial for organ function, and their functional failure contributes to poor organ transplantation outcomes. In this study, we used a light-based method called resonance Raman spectroscopy (RRS) to assess mitochondrial health directly on the liver surface, without removing tissue. We find that long periods of warm ischemia (i.e., no blood flow at body temperature) caused significant mitochondrial stress, especially at complex III, which is important for energy production. We then tested methylene blue, an FDA-approved drug that helps mitochondria maintain function while bypassing complex III. In pig livers with injury similar to marginal human donor organs, methylene blue improves oxygen use and blood flow profiles. These results highlight the potential of RRS for improving transplant outcomes.
Similar content being viewed by others
Data availability
All data relevant to this study are included in this published article and its Supplementary Material file. Additional information can be provided by the corresponding author upon reasonable request. Source data underlying Figs. 1–5 can be accessed from Supplementary Data 1.
References
Brand, M. D. et al. The role of mitochondrial function and cellular bioenergetics in ageing and disease. Br. J. Dermatol. 169, 1–8 (2013).
Jastroch, M. et al. Mitochondrial proton and electron leaks. Essays Biochem. 47, 53–67 (2010).
Mazat, J. P., Devin, A. & Ransac, S. Modelling mitochondrial ROS production by the respiratory chain. Cell. Mol. Life Sci. 77, 455–465 (2020).
Bardaweel, S. K. et al. Reactive oxygen species: the dual role in physiological and pathological conditions of the human body. Eurasian J. Med. 50, 193–201 (2018).
Fromenty, B. & Roden, M. Mitochondrial alterations in fatty liver diseases. J. Hepatol. 78, 415–429 (2023).
Prag H. A. et al. Chapter 26—Mitochondrial ROS production during ischemia-reperfusion injury. in Oxidative Stress (ed. Sies, H.) 513–538 (Academic Press, 2020).
Schatten, H., Sun, Q. Y. & Prather, R. The impact of mitochondrial function/dysfunction on IVF and new treatment possibilities for infertility. Reprod. Biol. Endocrinol. 12, 111 (2014).
Li, J. L. et al. Mitochondrial function and Parkinson’s disease: from the perspective of the electron transport chain. Front. Mol. Neurosci. 14, 797833 (2021).
Bruinsma, B. G. et al. Peritransplant energy changes and their correlation to outcome after human liver transplantation. Transplantation 101, 1637–1644 (2017).
Eden, J. et al. Assessment of liver graft quality during hypothermic oxygenated perfusion: the first international validation study. J. Hepatol. 82, 523–534 (2025).
Logan, A. et al. Assessing the mitochondrial membrane potential in cells and in vivo using targeted click chemistry and mass spectrometry. Cell Metab. 23, 379–385 (2016).
Sivandzade, F., Bhalerao, A. & Cucullo, L. Analysis of the mitochondrial membrane potential using the cationic JC-1 dye as a sensitive fluorescent probe. Bio. Protoc. 9, e3128 (2019).
Meszaros, A. T. et al. Mitochondrial respiration during normothermic liver machine perfusion predicts clinical outcome. EBioMedicine 85, 104311 (2022).
Kuznetsov, A. V. et al. Evaluation of mitochondrial respiratory function in small biopsies of liver. Anal. Biochem. 305, 186–194 (2002).
Koliaki, C. et al. Adaptation of hepatic mitochondrial function in humans with non-alcoholic fatty liver is lost in steatohepatitis. Cell Metab. 21, 739–746 (2015).
de Vries, R. J. et al. Non-invasive quantification of the mitochondrial redox state in livers during machine perfusion. PLoS ONE 16, e0258833 (2021).
Jain, R. et al. Real-time monitoring of mitochondrial oxygenation during machine perfusion using resonance Raman spectroscopy predicts organ function. Sci. Rep. 14, 7328 (2024).
Perry, D. A. et al. Responsive monitoring of mitochondrial redox states in heart muscle predicts impending cardiac arrest. Sci. Transl. Med. 9, eaan0117 (2017).
Jassem, W. & Heaton, N. D. The role of mitochondria in ischemia/reperfusion injury in organ transplantation. Kidney Int. 66, 514–517 (2004).
Tennankore, K. K. et al. Prolonged warm ischemia time is associated with graft failure and mortality after kidney transplantation. Kidney Int. 89, 648–658 (2016).
Abt, P. L. et al. Survival following liver transplantation from non-heart-beating donors. Ann. Surg. 239, 87–92 (2004).
White, C. W. et al. Transplantation of hearts donated after circulatory death. Front. Cardiovasc. Med. 5, 8 (2018).
Eden, J. et al. Utilization of livers donated after circulatory death for transplantation—an international comparison. J. Hepatol. 78, 1007–1016 (2023).
Beuth, J. et al. New strategies to expand and optimize heart donor pool: ex vivo heart perfusion and donation after circulatory death: a review of current research and future trends. Anesth. Analg. 128, 406–413 (2019).
Jawitz, O. K. et al. Increasing the United States heart transplant donor pool with donation after circulatory death. J. Thorac. Cardiovasc. Surg. 159, e307–e309 (2020).
Goldaracena, N. et al. Expanding the donor pool for liver transplantation with marginal donors. Int. J. Surg. 82S, 30–35 (2020).
Tolboom, H. et al. Recovery of warm ischemic rat liver grafts by normothermic extracorporeal perfusion. Transplantation 87, 170–177 (2009).
Xue, H., Thaivalappil, A. & Cao, K. The Potentials of methylene blue as an anti-aging drug. Cells 10, 3379 (2021).
Tessier, S. N. et al. Partial freezing of rat livers extends preservation time by 5-fold. Nat. Commun. 13, 4008 (2022).
Chen, M. et al. Contrast-enhanced ultrasound to quantify perfusion in a machine-perfused pig liver. In Proc. Annual International Conference of the IEEE Engineering in Medicine and Biology Society 2018, 3128–3131 (IEEE, 2018).
Bruinsma, B. G. et al. Subnormothermic machine perfusion for ex vivo preservation and recovery of the human liver for transplantation. Am. J. Transplant. 14, 1400–1409 (2014).
Olverson, G. 4th et al. Cardiac loading using passive left atrial pressurization and passive afterload for graft assessment. J. Vis. Exp. 210, (2024).
Bruinsma, B. G. et al. Supercooling preservation and transplantation of the rat liver. Nat. Protoc. 10, 484–494 (2015).
Shi, Z. F. et al. Methylene blue ameliorates brain edema in rats with experimental ischemic stroke via inhibiting aquaporin 4 expression. Acta Pharmacol. Sin. 42, 382–392 (2021).
Xin, J. et al. Spatial transcriptomics analysis of zone-dependent hepatic ischemia-reperfusion injury murine model. Commun. Biol. 6, 194 (2023).
Palladini, G. et al. Lobe-specific heterogeneity and matrix metalloproteinase activation after ischemia/reperfusion injury in rat livers. Toxicol. Pathol. 40, 722–730 (2012).
Covian, R. et al. Spectroscopic identification of the catalytic intermediates of cytochrome c oxidase in respiring heart mitochondria. Biochim. Biophys. Acta Bioenerg. 1864, 148934 (2023).
Glancy, B. & Balaban, R. S. Protein composition and function of red and white skeletal muscle mitochondria. Am. J. Physiol. Cell Physiol. 300, C1280–C1290 (2011).
Balaban, R. S., Mootha, V. K. & Arai, A. Spectroscopic determination of cytochrome c oxidase content in tissues containing myoglobin or hemoglobin. Anal. Biochem. 237, 274–278 (1996).
Ljungdahl, P. O., Pennoyer, J. D. & Trumpower, B. L. [18] Purification of cytochrome bc1 complexes from phylogenically diverse species by a single method. in Methods in Enzymology (eds. Fleischer, S. & Fleischer, B.)181–191 (Academic Press, 1986).
Spiro, T. G. Resonance raman spectroscopic studies of heme proteins. Biochim. Biophys. Acta Rev. Bioenerg. 416, 169–189 (1975).
Callahan, P. M. & Babcock, G. T. Insights into heme structure from Soret excitation Raman spectroscopy. Biochemistry 20, 952–958 (1981).
Hinchliffe, P. & Sazanov, L. A. Organization of iron-sulfur clusters in respiratory complex I. Science 309, 771–774 (2005).
Baradaran, R. et al. Crystal structure of the entire respiratory complex I. Nature 494, 443–448 (2013).
Valyushok, D. S. et al. Identification of a new b-type cytochrome from the whitefish Coregonidae eggs. FEBS Lett. 332, 61–63 (1993).
Schägger, H. & Pfeiffer, K. The ratio of oxidative phosphorylation complexes I–V in bovine heart mitochondria and the composition of respiratory chain supercomplexes*. J. Biol. Chem. 276, 37861–37867 (2001).
Siletsky, S. A. & Konstantinov, A. A. Cytochrome c oxidase: charge translocation coupled to single-electron partial steps of the catalytic cycle. Biochim. Biophys. Acta Bioenerg. 1817, 476–488 (2012).
Bruinsma, B. G. et al. Metabolic profiling during ex vivo machine perfusion of the human liver. Sci. Rep. 6, 22415 (2016).
Ogita, M. et al. Antimycin A-induced cell death depends on AIF translocation through NO production and PARP activation and is not involved in ROS generation, cytochrome c release and caspase-3 activation in HL-60 cells. J. Antibiot. 62, 145–152 (2009).
Tolboom, H. et al. Subnormothermic machine perfusion at both 20°C and 30°C recovers ischemic rat livers for successful transplantation. J. Surg. Res. 175, 149–156 (2012).
Aburawi, M. M. et al. Synthetic hemoglobin-based oxygen carriers are an acceptable alternative for packed red blood cells in normothermic kidney perfusion. Am. J. Transplant. 19, 2814–2824 (2019).
Liu, J. & Man, K. Mechanistic insight and clinical implications of ischemia/reperfusion injury post liver transplantation. Cell. Mol. Gastroenterol. Hepatol. 15, 1463–1474 (2023).
Nemeth, N. et al. Hemorheological and microcirculatory factors in liver ischemia-reperfusion injury—an update on pathophysiology, molecular mechanisms and protective strategies. Int. J. Mol. Sci. 22, 1864 (2021).
Mahboub, P. et al. Gradual rewarming with a hemoglobin-based oxygen carrier improves viability of donation after circulatory death in rat livers. Front. Transplant. 3, 1353124 (2024).
Minor, T. et al. Controlled oxygenated rewarming as novel end-ischemic therapy for cold stored liver grafts. A randomized controlled trial. Clin. Transl. Sci. 15, 2918–2927 (2022).
van Rijn, R. et al. Hypothermic machine perfusion in liver transplantation—a randomized trial. N. Engl. J. Med. 384, 1391–1401 (2021).
McGiffin, D. C. et al. Hypothermic oxygenated perfusion (HOPE) safely and effectively extends acceptable donor heart preservation times: results of the Australian and New Zealand trial. J. Heart Lung Transplant. 43, 485–495 (2024).
Yu, B., Bloch, K. D. & Zapol, W. M. Hemoglobin-based red blood cell substitutes and nitric oxide. Trends Cardiovasc. Med. 19, 103–107 (2009).
Cabrales, P. & Friedman, J. M. HBOC vasoactivity: interplay between nitric oxide scavenging and capacity to generate bioactive nitric oxide species. Antioxid. Redox Signal. 18, 2284–2297 (2013).
Song, B. K. et al. Effects of a hemoglobin-based oxygen carrier (HBOC-201) and derivatives with altered oxygen affinity and viscosity on systemic and microcirculatory variables in a top-load rat model. Microvasc. Res. 95, 124–130 (2014).
Acknowledgements
This work was supported by generous funding to S.N.T. from the US National Institutes of Health (R01DK134590). The authors also gratefully acknowledge funding to S.N.T. from the US National Institute of Health (K99/R00 HL1431149; R01HL157803; R24OD034189), National Science Foundation (EEC 1941543), Polsky Family Foundation, and Shriners Children’s Boston (Grant #BOS-85115). In addition, the authors acknowledge funding to K.U. from the US National Institute of Health (R01DK114506, R01DK096075), and support for R.J. by Grant #LIFER23-263034 from the American Association for the Study of Liver Diseases Foundation. This work was also supported by the Office of the Assistant Secretary of Defense for Health Affairs through the Peer Reviewed Medical Research Program under Award Number W81XWH-19-1-0472 to J.N.K. Opinions, interpretations, conclusions, and recommendations are those of the author and are not necessarily endorsed by the Department of Defense. The authors express deep gratitude for expert support from highly skilled technicians of the MGH Knight Surgery for the pig liver procurements, that of Pathology Core from Harvard Medical School for histology studies, and that of Mass Spectroscopy Core from Shriners Children’s Boston for the liver energy status evaluation. The authors would like to extend their gratitude to Dr. Robert Balaban and Dr. Armel Femnou from the NIH National Heart, Lung, and Blood Institute for their expertise in mitochondrial energetics and their important assistance in developing the RRS library of individual mitochondrial complexes. In addition, the authors would like to thank Ms. Jesslyn James from Xavier University of Louisiana for her support with pig liver machine perfusion and RRS measurements at MGH during a research experience for undergraduate (REU) program funded by the NSF Grant No. EEC 1941543. The authors would like to thank Dr. Alissa Cutrone and Dr. Arnaud Lyon for their surgical support with pig liver procurement. The authors also deeply appreciate Dr. Reinier J de Vries and Ms. Casie A. Pendexter for their valuable contributions to the human liver experiments. Finally, the authors express sincere gratitude to Mr. Zafiris Zafirelis and HbO2 Therapeutics for their provision of hemoglobin-based oxygen carrier (HBOC) used in this study.
Author information
Authors and Affiliations
Contributions
K.T.N., O.S.O. and S.N.T. designed the study. K.T.N. and O.S.O. performed the rat liver machine perfusion. C.T. and K.T.N. performed the pig liver machine perfusion with support from O.S.O. and R.J. K.T.N. was responsible for resonance Raman Spectroscopy (RRS) data collection and interpretation. O.S.O. was responsible for rat and pig liver procurements. K.T.N., O.S.O., C.T., and T.P. contributed to organ viability data generation and interpretation. K.T.N., R.J., and P.R contributed to RRS algorithm optimization and data analysis with support from E.A. and D.V. additionally. J.N.K., P.R., and D.V. contributed to developing the RRS library for individual complex III and IV. K.T.N. performed the data curation, and R.J. performed the statistical hypothesis testing with support from K.T.N., O.S.O., and A.M. Overall, S.N.T. is responsible for all aspects of the research project with support for laboratory resources from K.U., surgical techniques and pig liver resources from S.A.R. and A.A.O., as well as engineering support for RRS technology development from D.V. and P.R. All authors reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare competing interests. S.N.T., J.N.K., and P.R. have provisional patent applications relevant to this study. D.V. and P.R. are employees and shareholders of Pendar Technologies. S.N.T.’s are managed by MGH and Partners HealthCare in accordance with their conflict-of-interest policies. J.N.K.’s competing interests are managed by BCH’s conflict-of-interest policies. D.V. and P.R.’s competing interests are subject to the Research Integrity Policy of Pendar Technologies. The following patented technologies have been used in this study: US2020/0281474A1 In-vivo monitoring of cellular energetics with Raman spectroscopy (application). Additional patent applications for use in ophthalmology, tissue viability, and burn injury assessment using Resonance Raman Spectroscopy have been submitted, where R.J. is also an inventor. Finally, a patent disclosure was submitted for the use of methylene blue to overcome ischemia-reperfusion injury, where K.T.N., O.S.O., and S.N.T. are co-inventors. This does not alter our adherence to journal policies on sharing data and materials. All other authors do not have competing interests.
Peer review
Peer review information
Communications Medicine thanks Oner Ulger, Kosuke Dodo, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Nguyen, K.T., Ozgur, O.S., Jain, R. et al. Mitochondrial hyperoxidation contributes to warm ischemia-reperfusion injury in rat and pig livers. Commun Med (2026). https://doi.org/10.1038/s43856-026-01551-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43856-026-01551-4