Abstract
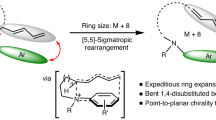
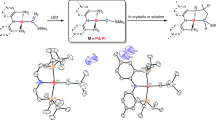
For over a century, scientists have been fascinated by the unique electronic, structural and bonding properties of the three isomers of benzyne, a highly reactive organic intermediate derived from benzene by removing two hydrogen atoms. Although o- and p-benzynes have been extensively studied following the establishment of reliable synthetic methods to prepare them, m-benzyne in the ground state has remained experimentally inaccessible. We report herein the room-temperature and atmospheric-pressure synthesis of m-benzyne in solution. Experimental and theoretical investigations revealed that owing to the inner bond inside the benzene ring between C1 and C3 atoms, m-benzyne behaves as a potent electrophile with a Mayr’s electrophilicity parameter E of around −2 but shows weak free-radical character. The bonding appears similar to the inverted σ-bond, the so-called charge-shift bond, in [1.1.1]propellane. By utilizing the unique bonding character of m-benzyne, we established halogenations and C–N and C–C coupling reactions, as well as a successive m-benzyne generation and trapping sequence that provides access to 1,3,5-trisubstituted benzenes.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available in this article and the Supplementary Information (experimental procedures and characterization data). Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2061582 (8a), CCDC 2268762 (8b), CCDC 2268732 (8c) and CCDC 2061583 (25). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. Source data are provided with this paper.
References
Wentrup, C. The benzyne story. Aust. J. Chem. 63, 979–986 (2010).
Wenk, H. H., Winkler, M. & Sander, W. One century of aryne chemistry. Angew. Chem. Int. Ed. 42, 502–528 (2003).
Sander, W. m-Benzyne and p-benzyne. Acc. Chem. Res. 32, 669–676 (1999).
Kraka, E. & Cremer, D. Ortho-, meta-, and para-benzyne. A comparative CCSD (T) investigation. Chem. Phys. Lett. 216, 333–340 (1993).
Wenthold, P. G. & Squires, R. R. Biradical thermochemistry from collision-induced dissociation threshold energy measurements. Absolute heats of formation of ortho-, meta-, and para-benzyne. J. Am. Chem. Soc. 116, 6401 (1994).
Fisher, I. P. & Lossing, F. P. Ionization potential of benzyne. J. Am. Chem. Soc. 85, 1018–1019 (1963).
Marquardt, R., Sander, W. & Kraka, E. 1,3-Didehydrobenzene (m-benzyne). Angew. Chem. Int. Ed. 35, 746–748 (1996).
Bertorello, H. E., Rossi, R. A. & de Rossi, R. H. Thermal decomposition of carboxybenzenediazonium salts. II. 1,3-Dehydroaromatic compounds from carboxybenzenediazonium salts. J. Org. Chem. 35, 3332–3338 (1970).
Berry, R. S., Clardy, J. & Schafer, M. E. Decomposition of benzenediazonium-3-carboxylate: transient 1,3-dehydrobenzene. Tetrahedron Lett. 6, 1011–1017 (1965).
Sander, W. et al. Vibrational spectrum of m-benzyne: a matrix isolation and computational study. J. Am. Chem. Soc. 124, 13072–13079 (2002).
Winkler, M. & Sander, W. The structure of meta-benzyne revisited – a close look into σ-bond formation. J. Phys. Chem. A 105, 10422–10432 (2001).
Wei, H., Hrovat, D. A., Mo, Y., Hoffmann, R. & Borden, W. T. The contributions of through-bond interactions to the singlet–triplet energy difference in 1,3-dehydrobenzene. J. Phys. Chem. A 113, 10351–10358 (2009).
Dilmaç, A. M., Spuling, E., de Meijere, A. & Bräse, S. Propellanes—from a chemical curiosity to ‘explosive’ materials and natural products. Angew. Chem. Int. Ed. 56, 5684–5718 (2017).
Varvoglis, A. The Organic Chemistry of Polycoordinated Iodine (VCH, 1992).
Yoshimura, A. & Zhdankin, V. V. Advances in synthetic applications of hypervalent iodine compounds. Chem. Rev. 116, 3328–3435 (2016).
Wirth, T. (ed) Hypervalent Iodine Chemistry (Springer, 2016).
Ochiai, M. Hypervalent aryl-, alkynyl-, and alkenyl-λ3-bromanes. Synlett 2009, 159–173 (2009).
Miyamoto, K. Chemistry of Hypervalent Bromine. In Patai’s Chemistry of Functional Groups (ed. Rappoport, Z.) 781–806 (Wiley, 2018).
Okuyama, T., Takino, T., Sueda, T. & Ochiai, M. Solvolysis of cyclohexenyliodonium salt, a new precursor for the vinyl cation: remarkable nucleofugality of the phenyliodonio group and evidence for internal return from an intimate ion-molecule pair. J. Am. Chem. Soc. 117, 3360–3367 (1995).
Nakajima, M., Miyamoto, K., Hirano, K. & Uchiyama, M. Diaryl-λ3-chloranes: versatile synthesis and unique reactivity as aryl cation equivalent. J. Am. Chem. Soc. 141, 6499–6503 (2019).
Thoen, K. K. & Kenttämaa, H. I. Reactivity of a substituted m-benzyne biradical. J. Am. Chem. Soc. 121, 800–805 (1999).
Harada, T., Chiba, M. & Oku, A. Novel homologation reaction of arylzincates bearing a leaving group at the ortho and meta positions. J. Org. Chem. 64, 8210–8213 (1999).
Miyamoto, K. et al. Room-temperature chemical synthesis of C2. Nat. Commun. 11, 2134 (2020).
Kitamura, T. et al. A new and efficient hypervalent iodine–benzyne precursor, (phenyl)[o-(trimethylsilyl)phenyl]iodonium triflate: generation, trapping reaction, and nature of benzyne. J. Am. Chem. Soc. 121, 11674–11679 (1999).
Kitamura, T. Synthetic methods for the generation and preparative application of benzyne. Aust. J. Chem. 63, 987–1001 (2010).
Ochiai, M., Toyonari, M., Nagaoka, T., Chen, D.-W. & Kida, M. Stereospecific synthesis of vinyl(phenyl)iodonium tetrafluoroborates via boron-iodane exchange of vinylboronic acids and esters with hypervalent phenyliodanes. Tetrahedron Lett. 38, 6709–6712 (1997).
Miyamoto, K. et al. Benchtop-stable hypervalent bromine(III) compounds: versatile strategy and platform for air- and moisture-stable λ3-bromanes. J. Am. Chem. Soc. 143, 9327–9331 (2021).
Scherübl, M., Daniliuc, C. G. & Studer, A. Arynes as radical acceptors: TEMPO-mediated cascades comprising addition, cyclization, and trapping. Angew. Chem. Int. Ed. 60, 711–715 (2021).
Iida, T. et al. Practical and facile access to bicyclo[3.1.1]heptanes: potent bioisosteres of meta-substituted benzenes. J. Am. Chem. Soc. 144, 21848–21852 (2022).
Frank, N. et al. Synthesis of meta-substituted arene bioisosteres from [3.1.1]propellane. Nature 611, 721–726 (2022).
Nolte, C., Ammer, J. & Mayr, H. Nucleofugality and nucleophilicity of fluoride in protic solvents. J. Org. Chem. 77, 3325–3335 (2012).
Kolomeitsev, A. A., Vorobyev, M. & Gillandt, H. Versatile application of trifluoromethyl triflate. Tetrahedron Lett. 49, 449–454 (2008).
Grushin, V. V. & Marshall, W. J. Fluorination of nonactivated haloarenes via arynes under mild conditions, resulting from further studies toward Ar-F reductive elimination from palladium(II). Organometallics 27, 4825–4828 (2008).
Graskemper, J. W., Wang, B., Qin, L., Neumann, K. D. & DiMagno, S. G. Unprecedented directing group ability of cyclophanes in arene fluorinations with diaryliodonium salts. Org. Lett. 13, 3158–3161 (2011).
Diemer, V., Garcia, J. S., Leroux, F. R. & Colobert, F. Aryne-mediated fluorination: synthesis of fluorinated biaryls via a sequential desilylation–halide elimination–fluoride addition process. J. Fluorine Chem. 134, 146–155 (2012).
Minegishi, S., Loos, R., Kobayashi, S. & Mayr, H. Kinetics of the reactions of halide anions with carbocations: quantitative energy profiles for SN1 reactions. J. Am. Chem. Soc. 127, 2641–2649 (2005).
Perrin, C. L., Rodgers, B. L. & O’Connor, J. M. Nucleophilic addition to a p-benzyne derived from an enediyne: a new mechanism for halide incorporation into biomolecules. J. Am. Chem. Soc. 129, 4795–4799 (2007).
Liu, S., Li, Y. & Lan, Y. Mechanistic study of the fluoride-induced activation of a kobayashi precursor: pseudo-SN2 pathway via a pentacoordinated silicon ate complex. Eur. J. Org. Chem. 2017, 6349–6353 (2017).
Johnson, W. T. G. & Cramer, C. Substituent effect on benzyne electronic structures. J. Phys. Org. Chem. 14, 597–603 (2001).
Shi, J., Li, L. & Li, Y. o‑Silylaryl triflates: a journey of Kobayashi aryne precursors. Chem. Rev. 121, 3892–4044 (2021).
Medina, J. M., Mackey, J. L., Garg, N. K. & Houk, K. N. The role of aryne distortions, steric effects, and charges in regioselectivities of aryne reactions. J. Am. Chem. Soc. 136, 15798–15805 (2014).
Nathel, N. F. F., Morrill, L. A., Mayr, H. & Garg, N. K. Quantification of the electrophilicity of benzyne and related intermediates. J. Am. Chem. Soc. 138, 10402–10405 (2016).
Kanzian, T., Nigst, T. A., Maier, A., Pichl, S. & Mayr. H. Nucleophilic reactivities of primary and secondary amines in acetonitrile. Eur. J. Org. Chem. 2009, 6379–6385 (2009).
Ammer, J., Baidya, M., Kobayashi, S. & Mayr, H. Nucleophilic reactivities of tertiary alkylamines. J. Phys. Org. Chem. 23, 1029–1035 (2010).
Brotzel, F., Chu, Y. C. & Mayr, H. Nucleophilicities of primary and secondary amines in water. J. Org. Chem. 72, 3679–3688 (2007).
Mizukoshi, Y., Mikamo, K. & Uchiyama, M. Aryne polymerization enabling straightforward synthesis of elusive poly(ortho-arylene)s. J. Am. Chem. Soc. 137, 74–77 (2015).
Terrier, F., Pouet, M.-J., Halle, J.-C., Kizilian, E. & Buncel, E. Electrophilic aromatic substitutions: reactions of hydroxy- and methoxy-substituted benzenes with 4,6-dinitrobenzofuroxan: kinetics and mechanism. J. Phys. Org. Chem. 11, 707–714 (1998).
Shi, J., Qiu, D., Wang, J., Xu, H. & Li, Y. Domino aryne precursor: efficient construction of 2,4-disubstituted benzothiazoles. J. Am. Chem. Soc. 137, 5670–5673 (2015).
Qiu, D., He, J., Yue, X., Shi, J. & Li, Y. Diamination of domino aryne precursor with sulfonamides. Org. Lett. 18, 3130–3133 (2016).
Huang, Y. et al. Direct synthesis of ortho-halogenated arylphosphonates via a three-component reaction involving arynes. J. Org. Chem. 86, 7010–7018 (2021).
Schleyer, P. v. R., Jiao, H., Glukhovtsev, M. N., Chandrasekhar, J. & Krakas, E. Double aromaticity in the 3,5-dehydrophenyl cation and in cyclo[6]carbon. J. Am. Chem. Soc. 116, 10129–10134 (1994).
Toriumi, N., Muranaka, A., Kayahara, E., Yamago, S. & Uchiyama, M. In-plane aromaticity in cycloparaphenylene dications: a magnetic circular dichroism and theoretical study. J. Am. Chem. Soc. 137, 82–85 (2015).
Acknowledgements
This research was supported in part by Grants-in-Aid for Scientific Research (Nos. 17H03017 and 20H02720 to K.M., and Nos. 17H06173, 22H00320 and JP22H05125 to M.U.) from Japan Society for the Promotion of Science (JSPS) (Japan), Research Fellowships for Young Scientists (No. 21J21600 to K.K.) from JSPS (Japan), Core Research for Evolutionary Science and Technology (CREST) (No. JPMJCR19R2 to M.U.) from Japan Science and Technology Agency (Japan), NAGASE Science Technology Foundation, Naito Foundation, Chugai Foundation and Uehara Memorial Foundation (to M.U.).
Author information
Authors and Affiliations
Contributions
K.K. and K.M. carried out the experimental work. K.K. carried out computational studies and performed X-ray crystal structure analyses of diaryl-λ3-iodanes and compound 25. K.M. and M.U. designed, advised and directed the project, and wrote the paper. All authors edited the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Wolfram Sander and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Peter Seavill, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Experimental details, Supplementary Figs. 1–5 and Tables 1–3.
Supplementary Data 1
X-Ray crystallographic data for compound 8a, CCDC 2061582.
Supplementary Data 2
X-Ray crystallographic data for compound 8b, CCDC 2268762.
Supplementary Data 3
X-Ray crystallographic data for compound 8c, CCDC 2268732.
Supplementary Data 4
X-Ray crystallographic data for compound 25, CCDC 2061583.
Source data
Source Data Fig. 4 (download XLSX )
Source data for the graphs in Fig. 4b.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Koyamada, K., Miyamoto, K. & Uchiyama, M. Room-temperature synthesis of m-benzyne. Nat. Synth 3, 1083–1090 (2024). https://doi.org/10.1038/s44160-024-00572-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-024-00572-y