Abstract
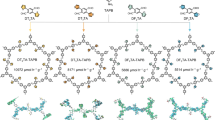
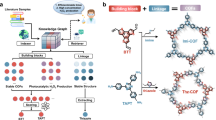
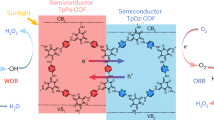
Covalent organic frameworks (COFs) can be used as photocatalysts for the direct photosynthesis of hydrogen peroxide (H2O2) from oxygen, water and sunlight. However, their highly symmetric structure can lead to weak adsorption of O2 and, therefore, unsatisfactory photocatalytic performance. Here we explore the local asymmetric electron distribution induced by Pauli and electron–electron repulsion in COFs to construct localized bonding sites for O2 species, which promotes photocatalytic H2O2 production. Experimental results and theoretical calculations reveal that TAPT–FTPB COFs (where TAPT is 1,3,5-tris-(4-aminophenyl) triazine and FTPB is 5-(5-formylthiophen-2-yl)thiophene-2-carbaldehyde) with an asymmetric electron distribution show strong O2 adsorption interaction and a record-breaking solar-to-chemical conversion efficiency of 1.22% for direct photosynthesis of H2O2 from oxygen and water, which is higher than in the photosynthesis of plants (~0.1%). A flow-type photocatalytic microreactor integrated with TAPT–FTPB COFs exhibits 100% sterilization efficiency for killing bacteria and 97.8% conversion for photocatalytic 2-thiophene methylamine coupling. This work reports a strategy for manipulating the local electron distribution in COFs, opening the door for research on the rational design of high-performance photocatalysis with a local asymmetric electron distribution.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the Article and its Supplementary Information.
References
Kim, D., Sakimoto, K. K., Hong, D. & Yang, P. Artificial photosynthesis for sustainable fuel and chemical production. Angew. Chem. Int. Ed. 54, 3259–3266 (2015).
Xia, C., Xia, Y., Zhu, P., Fan, L. & Wang, H. Direct electrosynthesis of pure aqueous H2O2 solutions up to 20% by weight using a solid electrolyte. Science 366, 226–231 (2019).
Liu, Y. et al. Fluorination of covalent organic framework reinforcing the confinement of Pd nanoclusters enhances hydrogen peroxide photosynthesis. J. Am. Chem. Soc. 145, 19877–19884 (2023).
Zhao, D. et al. Boron-doped nitrogen-deficient carbon nitride-based Z-scheme heterostructures for photocatalytic overall water splitting. Nat. Energy 6, 388–397 (2021).
Hou, H., Zeng, X. & Zhang, X. Production of hydrogen peroxide by photocatalytic processes. Angew. Chem. Int. Ed. 59, 17356–17376 (2020).
Jung, E. et al. Atomic-level tuning of Co–N–C catalyst for high-performance electrochemical H2O2 production. Nat. Mater. 19, 436–442 (2020).
Tan, H. et al. Photocatalysis of water into hydrogen peroxide over an atomic Ga–N5 site. Nat. Synth. 2, 557–563 (2023).
Bi, S., Meng, F., Wu, D. & Zhang, F. Synthesis of vinylene-linked covalent organic frameworks by monomer self-catalyzed activation of knoevenagel condensation. J. Am. Chem. Soc. 144, 3653–3659 (2022).
Jin, E. et al. A nanographene-based two-dimensional covalent organic framework as a stable and efficient photocatalyst. Angew. Chem. Int. Ed. 61, e202114059 (2022).
He, T. & Zhao, Y. Covalent organic frameworks for energy conversion in photocatalysis. Angew. Chem. Int. Edit. 62, e202303086 (2023).
Liu, Y. et al. A universal room-temperature approach to large-area continuous COFs film for photocatalytic coupling of amines. Adv. Funct. Mater. 33, 2302874 (2023).
Zhou, Z.-B. et al. Toward azo-linked covalent organic frameworks by developing linkage chemistry via linker exchange. Nat. Commun. 13, 2180 (2022).
Chen, Z. et al. Tuning excited state electronic structure and charge transport in covalent organic frameworks for enhanced photocatalytic performance. Nat. Commun. 14, 1106 (2023).
Li, W. et al. Thiazolo[5,4-d]thiazole-based donor–acceptor covalent organic framework for sunlight-driven hydrogen evolution. Angew. Chem. Int. Ed. 60, 1869–1874 (2021).
Qian, Y., Li, D., Han, Y. & Jiang, H.-L. Photocatalytic molecular oxygen activation by regulating excitonic effects in covalent organic frameworks. J. Am. Chem. Soc. 142, 20763–20771 (2020).
Liu, W. et al. A scalable general synthetic approach toward ultrathin imine-linked two-dimensional covalent organic framework nanosheets for photocatalytic CO2 reduction. J. Am. Chem. Soc. 141, 17431–17440 (2019).
Banerjee, T., Gottschling, K., Savasci, G., Ochsenfeld, C. & Lotsch, B. V. H2 evolution with covalent organic framework photocatalysts. ACS Energy Lett. 3, 400–409 (2018).
Kou, M. et al. Molecularly engineered covalent organic frameworks for hydrogen peroxide photosynthesis. Angew. Chem. Int. Ed. 61, e202200413 (2022).
Chen, R. et al. Charge separation via asymmetric illumination in photocatalytic Cu2O particles. Nat. Energy 3, 655–663 (2018).
Chen, K. et al. Iron phthalocyanine with coordination induced electronic localization to boost oxygen reduction reaction. Nat. Commun. 11, 4173 (2020).
Gillespie, R. J. The VSEPR model revisited. Chem. Soc. Rev. 21, 59–69 (1992).
Gillespie, R. J. & Robinson, E. A. Electron domains and the VSEPR model of molecular geometry. Angew. Chem. Int. Ed. Engl. 35, 495–514 (1996).
Lei, Z. et al. Boosting lithium storage in covalent organic framework via activation of 14-electron redox chemistry. Nat. Commun. 9, 576 (2018).
Liu, M. et al. Two-dimensional covalent organic framework films prepared on various substrates through vapor induced conversion. Nat. Commun. 13, 1411 (2022).
Liu, Y. et al. Two-dimensional metal–organic framework film for realizing optoelectronic synaptic plasticity. Angew. Chem. Int. Ed. 60, 17440–17445 (2021).
Liu, Y. et al. Cu2O/2D COFs core/shell nanocubes with antiphotocorrosion ability for efficient photocatalytic hydrogen evolution. ACS Nano 17, 5994–6001 (2023).
Ascherl, L. et al. Solvatochromic covalent organic frameworks. Nat. Commun. 9, 3802 (2018).
Li, D. et al. Metal-free thiophene-sulfur covalent organic frameworks: precise and controllable synthesis of catalytic active sites for oxygen reduction. J. Am. Chem. Soc. 142, 8104–8108 (2020).
Chen, L. et al. Acetylene and diacetylene functionalized covalent triazine frameworks as metal-free photocatalysts for hydrogen peroxide production: a new two-electron water oxidation pathway. Adv. Mater. 32, 1904433 (2020).
Teng, Z. et al. Atomically dispersed antimony on carbon nitride for the artificial photosynthesis of hydrogen peroxide. Nat. Catal. 4, 374–384 (2021).
Shiraishi, Y. et al. Resorcinol–formaldehyde resins as metal-free semiconductor photocatalysts for solar-to-hydrogen peroxide energy conversion. Nat. Mater. 18, 985–993 (2019).
Li, Y. et al. Photo-charge regulation of metal-free photocatalyst by carbon dots for efficient and stable hydrogen peroxide production. J. Mater. Chem. A 9, 25453–25462 (2021).
Zhang, Y. et al. H2O2 generation from O2 and H2O on a near-infrared absorbing porphyrin supramolecular photocatalyst. Nat. Energy 8, 361–371 (2023).
Zhang, H. et al. The long-distance charge transfer process in ferrocene-based MOFs with FeO6 clusters boosts photocatalytic CO2 chemical fixation. Appl. Catal. B 337, 122909 (2023).
Li, Y. et al. In situ photodeposition of platinum clusters on a covalent organic framework for photocatalytic hydrogen production. Nat. Commun. 13, 1355 (2022).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865 (1996).
Acknowledgements
This study was financially supported by National Science Fund for Distinguished Young Scholars (no. 52025133), National Key R&D Program of China (no. 2022YFE0128500), National Natural Science Foundation of China (nos. 52025133, 52261135633, 52303363 and 52302207), China National Petroleum Corporation-Peking University Strategic Cooperation Project of Fundamental Research, the Beijing Natural Science Foundation (no. Z220020), New Cornerstone Science Foundation through the XPLORER PRIZE, CNPC Innovation Found (no. 2021DQ02-1002), China National Postdoctoral Program for Innovative Talents (no. BX20220009) and Project funded by China Postdoctoral Science Foundation (nos. 2022M720225 and 2023M730029). We acknowledge the assistance of Rongjuan Feng in data collection and analysis for in situ FT-IR spectra.
Author information
Authors and Affiliations
Contributions
S.G. conceived the project. Y.L. designed and performed the experiments. Y.L. and L.L. performed the DFT calculation. Y.L. conducted data analyses with the help of L.L., Z. Sang, H.T., N.Y., C.S., Z. Sun and M.L. All the authors participated in discussions of the research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Liqun Ye and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–49 and Tables 1–3.
Supplementary Data 1 (download XLSX )
Statistical source data for supplementary figures.
Supplementary Video 1 (download MP4 )
Supplementary Video 1.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data for Fig. 1.
Source Data Fig. 2 (download XLSX )
Statistical source data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Statistical source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Statistical source data for Fig. 4.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Li, L., Sang, Z. et al. Enhanced hydrogen peroxide photosynthesis in covalent organic frameworks through induced asymmetric electron distribution. Nat. Synth 4, 134–141 (2025). https://doi.org/10.1038/s44160-024-00644-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-024-00644-z
This article is cited by
-
Towards single-crystalline two-dimensional poly(arylene vinylene) covalent organic frameworks
Nature Chemistry (2026)
-
Synthesis of covalent organic frameworks for photocatalytic hydrogen peroxide production guided by large language models
Nature Communications (2026)
-
Construction of Modifiable Phthalocyanine-Based Covalent Organic Frameworks with Irreversible Linking for Efficient Photocatalytic CO2 Reduction
Nano-Micro Letters (2026)
-
Photocatalytic H2O2 Production over Ultrathin Layered Double Hydroxide with 3.92% Solar-to-H2O2 Efficiency
Nano-Micro Letters (2026)
-
Thiazolo[5,4-d]thiazole-based CMPs as bifunctional photocatalysts for H2O2 production and organic pollutant degradation
Research on Chemical Intermediates (2026)