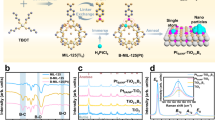
Abstract
Photocatalytic oxidation represents an effective means of hydrocarbon valorization, but achieving high-selectivity intermediate products at high substrate conversion remains challenging. Here single-atom Pd–O catalytic sites with different coordination environments are synthesized. The Pd–O3 coordinated single sites show the best catalytic performance for selective toluene oxidation, exhibiting a high benzaldehyde selectivity of 95% at 95% toluene conversion, and a benzaldehyde yield rate up to 12,000 μmol g−1 h−1, surpassing previously reported results. The Pd–O3 catalytic sites serve as the location of photogenerated charge separation and C(sp3)–H bond activation. These sites can trap the photogenerated holes effectively to enhance the charge-separation efficiency, improving the yield rate by a factor of 2.9 relative to the Bi2WO6 support. Additionally, the coordination structure weakens benzaldehyde adsorption, reducing overoxidation and improving reaction selectivity. This work highlights the importance of single-atom site coordination in optimizing the activity and selectivity of photocatalytic oxidation reactions.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information. Source data are provided with this paper.
Change history
12 May 2025
A Correction to this paper has been published: https://doi.org/10.1038/s44160-025-00820-9
References
Xiong, L. & Tang, J. Strategies and challenges on selectivity of photocatalytic oxidation of organic substances. Adv. Energy Mater. 11, 2003216 (2021).
Maldotti, A., Molinari, A. & Amadelli, R. Photocatalysis with organized systems for the oxofunctionalization of hydrocarbons by O2. Chem. Rev. 102, 3811–3836 (2002).
Cao, X., Han, T., Peng, Q., Chen, C. & Li, Y. Modifications of heterogeneous photocatalysts for hydrocarbon C–H bond activation and selective conversion. Chem. Commun. 56, 13918–13932 (2020).
Shiraishi, Y. & Hirai, T. Titanium oxide-based photocatalysts for selective organic transformations. J. Jpn. Pet. Inst. 55, 287–298 (2012).
Luo, H., Chen, W., Hu, J., Zhang, F. & Hu, X. Highly selective oxidation of toluene to benzaldehyde in alkaline systems. Ind. Eng. Chem. Res. 62, 10051–10056 (2023).
Sterckx, H., Morel, B. & Maes, B. U. W. Catalytic aerobic oxidation of C(sp3)–H bonds. Angew. Chem. Int. Ed. 58, 7946–7970 (2019).
Fujihira, M., Satoh, Y. & Osa, T. Heterogeneous photocatalytic oxidation of aromatic compounds on TiO2. Nature 293, 206–208 (1981).
Song, S. et al. A selective Au–ZnO/TiO2 hybrid photocatalyst for oxidative coupling of methane to ethane with dioxygen. Nat. Catal. 4, 1032–1042 (2021).
Zhou, Y. et al. Monolayered Bi2WO6 nanosheets mimicking heterojunction interface with open surfaces for photocatalysis. Nat. Commun. 6, 8340 (2015).
Cao, X. et al. Engineering lattice disorder on a photocatalyst: photochromic BiOBr nanosheets enhance activation of aromatic C–H bonds via water oxidation. J. Am. Chem. Soc. 144, 3386–3397 (2022).
Zhou, G., Lei, B. & Dong, F. Lewis acid sites in (110) facet-exposed BiOBr promote C–H activation and selective photocatalytic toluene oxidation. ACS Catal. 14, 4791–4798 (2024).
Sun, X. et al. Insight into the role of TiO2 facets in photocatalytic selective oxidation of p-xylene. ACS Catal. 14, 5356–5365 (2024).
Cao, X. et al. a photochromic composite with enhanced carrier separation for the photocatalytic activation of benzylic C–H Bonds in toluene. Nat. Catal. 1, 704–710 (2018).
Shi, Y. et al. Photocatalytic toluene oxidation with nickel-mediated cascaded active units over Ni/Bi2WO6 monolayers. Nat. Commun. 15, 4641 (2024).
Zheng, K. et al. breaking the activity–selectivity trade-off for CH4-to-C2H6 photoconversion. J. Am. Chem. Soc. 146, 12233–12242 (2024).
Zhang, Q. et al. Cl2·− mediates direct and selective conversion of inert C(sp3)–H bonds into aldehydes/ketones. Angew. Chem. Int. Ed. 62, e202304699 (2023).
Boochakiat, S., Inceesungvorn, B., Nattestad, A. & Chen, J. Bismuth‐based oxide photocatalysts for selective oxidation transformations of organic compounds. ChemNanoMat 9, e202300140 (2023).
Singha, A., Kaishyop, J., Khan, T. S. & Chowdhury, B. Visible-light-driven toluene oxidation to benzaldehyde over WO3 nanostructures. ACS Appl. Nano Mater. 6, 21818–21828 (2023).
Wang, H. et al. Achieving high selectivity in photocatalytic oxidation of toluene on amorphous BiOCl nanosheets coupled with TiO2. J. Am. Chem. Soc. 145, 16852–16861 (2023).
Shen, L., Kang, H., Deng, L., Chen, Z. & Yang, M.-Q. Solvent-free oxidation of benzylic C(sp3)–H bond over MA3Bi2Br9 perovskite photocatalyst by efficient singlet oxygen generation. J. Catal. 434, 115499 (2024).
Zhang, W. et al. High-performance photocatalytic nonoxidative conversion of methane to ethane and hydrogen by heteroatoms-engineered TiO2. Nat. Commun. 13, 2806 (2022).
Luo, L. et al. Synergy of Pd atoms and oxygen vacancies on In2O3 for methane conversion under visible light. Nat. Commun. 13, 2930 (2022).
Xue, Z. et al. Efficient benzylic C–H bond activation over single-atom yttrium supported on TiO2 via facilitated molecular oxygen and surface lattice oxygen activation. ACS Catal. 14, 249–261 (2024).
Wang, B., Cai, H. & Shen, S. Single metal atom photocatalysis. Small Methods 3, 1800447 (2019).
Martín, N. & Cirujano, F. G. Supported single atom catalysts for C–H Activation: selective c–h oxidations, dehydrogenations and oxidative C–H/C–H couplings. ChemCatChem 13, 2751–2765 (2021).
Gao, C. et al. Heterogeneous single-atom photocatalysts: fundamentals and applications. Chem. Rev. 120, 12175–12216 (2020).
Jang, E. J., Lee, J., Oh, D. G. & Kwak, J. H. CH4 oxidation activity in Pd and Pt–Pd bimetallic catalysts: correlation with surface PdOx quantified from the DRIFTS study. ACS Catal. 11, 5894–5905 (2021).
Liu, P. et al. Photochemical route for synthesizing atomically dispersed palladium catalysts. Science 352, 797–800 (2016).
Zhang, W. et al. Light-driven flow synthesis of acetic acid from methane with chemical looping. Nat. Commun. 14, 3047 (2023).
Guo, Y. et al. Photo-thermo semi-hydrogenation of acetylene on Pd1/TiO2 single-atom catalyst. Nat. Commun. 13, 2648 (2022).
Keating, J., Sankar, G., Hyde, T. I., Kohara, S. & Ohara, K. Elucidation of structure and nature of the PdO–Pd transformation using in situ PDF and XAS techniques. Phys. Chem. Chem. Phys. 15, 8555–8565 (2013).
Wang, H. et al. PdBi single-atom alloy aerogels for efficient ethanol oxidation. Adv. Funct. Mater. 31, 2103465 (2021).
Kesavan, L. et al. Solvent-free oxidation of primary carbon-hydrogen bonds in toluene using Au–Pd alloy nanoparticles. Science 331, 195–199 (2011).
Dai, Y., Poidevin, C., Ochoa-Hernandez, C., Auer, A. A. & Tueysuez, H. A supported bismuth halide perovskite photocatalyst for selective aliphatic and aromatic C–H bond activation. Angew. Chem. Int. Ed. 59, 5788–5796 (2020).
Bai, Z.-J. et al. Cs3Bi2Br9 nanodots stabilized on defective BiOBr nanosheets by interfacial chemical bonding: modulated charge transfer for photocatalytic C(sp3)–H bond activation. ACS Catal. 12, 15157–15167 (2022).
Mai, H. et al. Synthesis of layered lead-free perovskite nanocrystals with precise size and shape control and their photocatalytic activity. J. Am. Chem. Soc. 145, 17337–17350 (2023).
Li, S. et al. Chromium-doped nickel oxide and nickel nitride mediate selective electrocatalytic oxidation of sterol intermediates coupled with H2 evolution. Angew. Chem. Int. Ed. 62, e202306553 (2023).
Rozen, A. M. & Kostanyan, A. E. Scaling-up effect in chemical engineering. Theor. Found. Chem. Eng. 36, 307–313 (2002).
Yuan, R. et al. Chlorine-radical-mediated photocatalytic activation of C–H bonds with visible light. Angew. Chem. Int. Ed. 52, 1035–1039 (2013).
Ghalta, R. & Srivastava, R. Remarkably improved photocatalytic selective oxidation of toluene to benzaldehyde with O2 over metal-free delaminated g-C3N4 nanosheets: synergistic effect of enhanced textural properties and charge carrier separation. Catal. Sci. Technol. 13, 1541–1557 (2023).
Zou, J. et al. Localized surface plasmon resonance assisted photothermal catalysis of CO and toluene oxidation over Pd–CeO2 catalyst under visible light irradiation. J. Phys. Chem. C 120, 29116–29125 (2016).
Li, F. et al. Transient photovoltage study of the kinetics and synergy of electron/hole co-extraction in MoS2/Ag–In–Zn–S/carbon dot photocatalysts for promoted hydrogen production. Chem. Eng. J. 439, 135759 (2022).
Ou, H. et al. Carbon nitride photocatalysts with integrated oxidation and reduction atomic active centers for improved CO2 conversion. Angew. Chem. Int. Ed. 61, e202206579 (2022).
Liu, D. et al. In situ constructing atomic interface in ruthenium-based amorphous hybrid-structure towards solar hydrogen evolution. Nat. Commun. 14, 1720 (2023).
Cheng, C. et al. Verifying the charge-transfer mechanism in S-scheme heterojunctions using femtosecond transient absorption spectroscopy. Angew. Chem. Int. Ed. 62, e202218688 (2023).
Jiao, X. et al. Defect-mediated electron–hole separation in one-unit-cell ZnIn2S4 layers for boosted solar-driven CO2 reduction. J. Am. Chem. Soc. 139, 7586–7594 (2017).
Fan, J. et al. Transient in-situ DRIFTS investigation of catalytic oxidation of toluene over α‐, γ‐ and β‐MnO2. ChemCatChem 12, 1046–1054 (2020).
Chen, Z. et al. Performance and mechanism of photocatalytic toluene degradation and catalyst regeneration by thermal/UV treatment. Environ. Sci. Technol. 54, 14465–14473 (2020).
Li, J., Na, H., Zeng, X., Zhu, T. & Liu, Z. In situ DRIFTS investigation for the oxidation of toluene by ozone over Mn/HZSM-5, Ag/HZSM-5 and Mn–Ag/HZSM-5 catalysts. Appl. Surf. Sci. 311, 690–696 (2014).
Yu, K. et al. Efficient catalytic combustion of toluene at low temperature by tailoring surficial Pt0 and interfacial Pt–Al(OH)x species. iScience 24, 102689 (2021).
Soichi, H., Hiroshi, H. & Noriyuki, K. Infrared spectra and molecular configuration of dimeric carboxylic acids. Bull. Inst. Chem. Res. Kyoto Univ. 46, 213–218 (1969).
Chai, Y. et al. Dual-atom catalyst with N-colligated Zn1Co1 species as dominant active sites for propane dehydrogenation. J. Am. Chem. Soc. 146, 263–273 (2024).
Tang, S. & Liang, B. Scale formation and its mechanism in the liquid-phase oxidation of toluene by air. Ind. Eng. Chem. Res. 46, 7826–7829 (2007).
Hermans, I., Peeters, J., Vereecken, L. & Jacobs, P. A. Mechanism of thermal toluene autoxidation. ChemPhysChem 8, 2678–2688 (2007).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant number 22275109) and the Fundamental Research Center of Single-Atom Catalysis (grant number 22388102). X.C. thanks the China Postdoctoral Science Foundation (2022M722834, 2023T160587). We greatly appreciate L. Sun for his support on this work. The DRIFTS measurements were performed at the Research Center for Eco-Environmental Sciences with the help of C. Chen. C. He, Y. Zeng and L. Chen offered valuable suggestions about the measurements. We thank the SPring-8 (Japan) 12B2 Taiwan beamline of the National Synchrotron Radiation Research Center (NSRRC) and the BL11B station at the Shanghai Synchrotron Radiation Facility for XAFS measurement. We acknowledge the support of the Analysis Center of Tsinghua University and S. Du from the Dalian Institute of Chemical Physics for XPS experiments.
Author information
Authors and Affiliations
Contributions
R.S., X.C. and J.M. performed the experimental work and analysed the results. H.-C.C. helped with the XAFS experiments. C.C. provided fruitful advice on the work. R.S., X.C., J.M., Q.P. and Y.L. conceived and designed the experiments. All authors contributed to the overall scientific discussions and co-wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Liyong Gan, Shuang-Feng Yin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Details of experiments and calculations, Supplementary Figs. 1–43 and Tables 1–7.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, R., Cao, X., Ma, J. et al. Regulation of Pd single-atom coordination for enhanced photocatalytic oxidation of toluene to benzaldehyde. Nat. Synth 4, 965–975 (2025). https://doi.org/10.1038/s44160-025-00782-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00782-y
This article is cited by
-
What solar fuel technologies can learn from each other
Nature Reviews Clean Technology (2026)