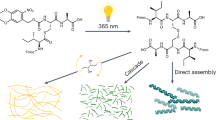
Abstract
A hallmark of life is its capacity to grow and make changes in response to specific conditions, which drives adaptation and variation within populations. Replicating this ability in artificial systems marks a significant milestone in creating lifelike behaviours and probing the origin of life. Here we present an adaptive supramolecular system that exhibited complex growth and phase behaviours driven by the interplay between external environmental factors (light input) and intrinsic chemical activity. This process is powered by light-activated bond scission of strained cyclic disulfides in monomers, followed by the formation of diverse oligomers with linear disulfide linkages. Through self-organization, the information of chemical reactions is translated into protocellular growth and structural transition by coupling supramolecular aggregation and liquid–liquid phase separation. Our findings highlight the emergence of chemical complexity and the formation of cell-like structures from simple molecular systems, offering insights into the mechanisms of self-organization and compartmentalization, which are crucial to understanding the origin of life.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Information. Source data for Figs. 2–5 are provided with this paper.
References
Mann, S. Systems of creation: the emergence of life from nonliving matter. Acc. Chem. Res. 45, 2131–2141 (2012).
Blain, J. C. & Szostak, J. W. Progress toward synthetic cells. Annu. Rev. Biochem. 83, 615–640 (2014).
Guindani, C., da Silva, L. C., Cao, S., Ivanov, T. & Landfester, K. Synthetic cells: from simple bio-inspired modules to sophisticated integrated systems. Angew. Chem. Int. Ed. 61, e202110855 (2022).
Jia, H. & Schwille, P. Bottom-up synthetic biology: reconstitution in space and time. Curr. Opin. Biotechnol. 60, 179–187 (2019).
Gözen, I. et al. Protocells: milestones and recent advances. Small 18, 2106624 (2022).
Elani, Y. Interfacing living and synthetic cells as an emerging frontier in synthetic biology. Angew. Chem. Int. Ed. 60, 5602–5611 (2021).
Liu, Z., Zhou, W., Qi, C. & Kong, T. Interface engineering in multiphase systems toward synthetic cells and organelles: from soft matter fundamentals to biomedical applications. Nat. Commun. 32, 2002932 (2020).
Engelhart, A. E., Adamala, K. P. & Szostak, J. W. A simple physical mechanism enables homeostasis in primitive cells. Nat. Chem. 8, 448–453 (2016).
Podolsky, K. A. & Devaraj, N. K. Synthesis of lipid membranes for artificial cells. Nat. Rev. Chem. 5, 676–694 (2021).
Weiss, M. et al. Sequential bottom-up assembly of mechanically stabilized synthetic cells by microfluidics. Nat. Mater. 17, 89–96 (2018).
Marguet, M., Bonduelle, C. & Lecommandoux, S. Multicompartmentalized polymeric systems: towards biomimetic cellular structure and function. Chem. Soc. Rev. 42, 512–529 (2013).
Peters, R. J. R. W. et al. Cascade reactions in multicompartmentalized polymersomes. Angew. Chem. Int. Ed. 53, 146–150 (2014).
Belluati, A. et al. Artificial cell synthesis using biocatalytic polymerization-induced self-assembly. Nat. Chem. 16, 564–574 (2024).
Wang, X., Liu, X. & Huang, X. Bioinspired protein-based assembling: toward advanced life-like behaviors. Adv. Mater. 32, 2001436 (2020).
Städler, B. et al. Polymer hydrogel capsules: en route toward synthetic cellular systems. Nanoscale 1, 68–73 (2009).
Ghosh, B., Bose, R. & Tang, T. Y. D. Can coacervation unify disparate hypotheses in the origin of cellular life? Curr. Opin. Colloid Interface Sci. 52, 101415 (2021).
Abbas, M., Lipiński, W. P., Wang, J. & Spruijt, E. Peptide-based coacervates as biomimetic protocells. Chem. Soc. Rev. 50, 3690–3705 (2021).
Zhan, P., Jahnke, K., Liu, N. & Göpfrich, K. Functional DNA-based cytoskeletons for synthetic cells. Nat. Chem. 14, 958–963 (2022).
Altenburg, W. J. et al. Programmed spatial organization of biomacromolecules into discrete, coacervate-based protocells. Nat. Commun. 11, 6282 (2020).
Mu, W. et al. Membrane-confined liquid-liquid phase separation toward artificial organelles. Sci. Adv. 7, eabf9000 (2021).
Hanczyc, M. M., Fujikawa, S. M. & Szostak, J. W. Experimental models of primitive cellular compartments: encapsulation, growth, and division. Science 302, 618–622 (2003).
Nakashima, K. K., van Haren, M. H. I., André, A. A. M., Robu, I. & Spruijt, E. Active coacervate droplets are protocells that grow and resist Ostwald ripening. Nat. Commun. 12, 3819 (2021).
Chang, M.-Y., Ariyama, H., Huck, W. T. S. & Deng, N.-N. Division in synthetic cells. Chem. Soc. Rev. 52, 3307–3325 (2023).
Steinkühler, J. et al. Controlled division of cell-sized vesicles by low densities of membrane-bound proteins. Nat. Commun. 11, 905 (2020).
Ianeselli, A. et al. Non-equilibrium conditions inside rock pores drive fission, maintenance and selection of coacervate protocells. Nat. Chem. 14, 32–39 (2022).
Kurihara, K. et al. Self-reproduction of supramolecular giant vesicles combined with the amplification of encapsulated DNA. Nat. Chem. 3, 775–781 (2011).
Adamski, P. et al. From self-replication to replicator systems en route to de novo life. Nat. Rev. Chem. 4, 386–403 (2020).
Lu, H. et al. Small-molecule autocatalysis drives compartment growth, competition and reproduction. Nat. Chem. 16, 70–78 (2024).
Zhang, S. et al. Engineering motile aqueous phase-separated droplets via liposome stabilisation. Nat. Commun. 12, 1673 (2021).
Rifaie-Graham, O. et al. Photoswitchable gating of non-equilibrium enzymatic feedback in chemically communicating polymersome nanoreactors. Nat. Chem. 15, 110–118 (2023).
Buddingh, B. C., Elzinga, J. & van Hest, J. C. M. Intercellular communication between artificial cells by allosteric amplification of a molecular signal. Nat. Commun. 11, 1652 (2020).
Dupin, A. & Simmel, F. C. Signalling and differentiation in emulsion-based multi-compartmentalized in vitro gene circuits. Nat. Chem. 11, 32–39 (2019).
Bolognesi, G. et al. Sculpting and fusing biomimetic vesicle networks using optical tweezers. Nat. Commun. 9, 1882 (2018).
Wang, D., Yang, Y., Chen, F., Lyu, Y. & Tan, W. Network topology–directed design of molecular CPU for cell-like dynamic information processing. Sci. Adv. 8, eabq0917 (2022).
Samanta, A., Hörner, M., Liu, W., Weber, W. & Walther, A. Signal-processing and adaptive prototissue formation in metabolic DNA protocells. Nat. Commun. 13, 3968 (2022).
Li, H. et al. Artificial receptor-mediated phototransduction toward protocellular subcompartmentalization and signaling-encoded logic gates. Sci. Adv. 9, eade5853 (2023).
Chen, H. et al. DNA-based artificial receptors as transmembrane signal transduction systems for protocellular communication. Angew. Chem. Int. Ed. 62, e202301559 (2023).
Monreal Santiago, G., Liu, K., Browne, W. R. & Otto, S. Emergence of light-driven protometabolism on recruitment of a photocatalytic cofactor by a self-replicator. Nat. Chem. 12, 603–607 (2020).
Liu, K. et al. Light-driven eco-evolutionary dynamics in a synthetic replicator system. Nat. Chem. 16, 79–88 (2024).
Robinson, W. E., Daines, E., van Duppen, P., de Jong, T. & Huck, W. T. S. Environmental conditions drive self-organization of reaction pathways in a prebiotic reaction network. Nat. Chem. 14, 623–631 (2022).
Scheutz, G. M. et al. Harnessing strained disulfides for photocurable adaptable hydrogels. Macromolecules 53, 4038–4046 (2020).
Nelson, B. R. et al. Photoinduced dithiolane crosslinking for multiresponsive dynamic hydrogels. Adv. Mater. 36, 2211209 (2023).
Mishra, D. et al. Photochemical transformation of lipoic acid-based ligands: probing the effects of solvent, ligand structure, oxygen and pH. Phys. Chem. Chem. Phys. 20, 3895–3902 (2018).
Narayan, M., Welker, E., Wedemeyer, W. J. & Scheraga, H. A. Oxidative folding of proteins. Acc. Chem. Res. 33, 805–812 (2000).
Gao, Y., Li, X., Li, P. & Lin, Y. A brief guideline for studies of phase-separated biomolecular condensates. Nat. Chem. Biol. 18, 1307–1318 (2022).
Yin, H., Lin, Y., Huang, J. & Ye, J. Temperature-induced vesicle aggregation in catanionic surfactant systems: the effects of the headgroup and counterion. Langmuir 23, 4225–4230 (2007).
Yin, Y. et al. Non-equilibrium behaviour in coacervate-based protocells under electric-field-induced excitation. Nat. Commun. 7, 10658 (2016).
Saleh, O. A., Wilken, S., Squires, T. M. & Liedl, T. Vacuole dynamics and popping-based motility in liquid droplets of DNA. Nat. Commun. 14, 3574 (2023).
Kato, T., Takeuchi, H. & Seimiya, T. Concentration dependence of micellar size and composition in mixed anionic/cationic surfactant solutions studied by light scattering and pulsed-gradient FT-NMR spectroscopy. J. Phys. Chem. 96, 6839–6843 (1992).
Caillet, C., Hebrant, M. & Tondre, C. Sodium octyl sulfate/cetyltrimethylammonium bromide catanionic vesicles: aggregate composition and probe encapsulation. Langmuir 16, 9099–9102 (2000).
Banerjee, P. R., Milin, A. N., Moosa, M. M., Onuchic, P. L. & Deniz, A. A. Reentrant phase transition drives dynamic substructure formation in ribonucleoprotein droplets. Angew. Chem. Int. Ed. 56, 11354–11359 (2017).
Andes-Koback, M. & Keating, C. D. Complete budding and asymmetric division of primitive model cells to produce daughter vesicles with different interior and membrane compositions. J. Am. Chem. Soc. 133, 9545–9555 (2011).
Zong, W. et al. A fissionable artificial eukaryote-like cell model. J. Am. Chem. Soc. 139, 9955–9960 (2017).
Aldeek, F. et al. UV and sunlight driven photoligation of quantum dots: understanding the photochemical transformation of the ligands. J. Am. Chem. Soc. 137, 2704–2714 (2015).
Acknowledgements
We thank the National Natural Science Foundation of China (number 22172007 to Y.L. and number T2425001 to Y.Q.), the Science Fund for Creative Research Groups of the National Natural Science Foundation of China (number 52221006 to Y.L.), the Strategic Priority Research Program of the Chinese Academy of Sciences (number XDB0480000 to Y.Q.) and the Fundamental Research Funds for the Central Universities (number buctrc202015 to Y.L.) for financial support.
Author information
Authors and Affiliations
Contributions
Y.L. led the project. K.S. and P.S. performed the experiments. K.S. and Y.L. conceived the experiments. K.S., P.S., J.L., Y.Q. and Y.L. analysed the data and wrote the manuscript. All authors discussed the results and have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Job Boekhoven, Jianbo Liu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Experimental details, Supplementary sections 1–3, Figs. 1–37.
Supplementary Video 1 (download MP4 )
A macroscopic video shows an A/B solution (molar ratio of 5:5, total concentration of 5 mM, pH 7.5) turns cloudy as irradiated by 365 nm UV light, indicating the light-triggered formation of coacervate droplets (5 seconds off, 10 seconds on).
Supplementary Video 2 (download AVI )
A fluorescence microscopy video captures the coacervation process in an A/B solution (molar ratio of 5:5, total concentration of 5 mM, pH 7.5) under UV light illumination from the microscope. Green fluorescent HPTS was doped into the coacervate droplets for imaging, and the video shows the continuous growth and coalescence of coacervate droplets as they were exposed to the UV-light.
Supplementary Video 3 (download AVI )
An optical microscopy video demonstrates the coacervation process in an A/B solution ([molar ratio of 5:5, total concentration of 5 mM, pH 7.5) under UV light illumination from the microscope. The video reveals the continuous growth and coalescence of coacervate droplets as the solution was exposed to the UV-light.
Supplementary Video 4 (download MP4 )
A macroscopic video shows a progression where the solution changed from transparent to turbid, and then back to transparent. The increase in turbidity was attributed to the formation of coacervate droplets induced by 365 nm UV light (5 seconds off, 10 seconds on) in an A/B solution (molar ratio of 6:4, total concentration of 5 mM, pH 7.5).
Supplementary Video 5 (download AVI )
A fluorescence microscopy video captures the UV light-triggered dynamics of the vacuolation–collapse cycles in an A/B solution (molar ratio of 6:4, total concentration of 5 mM, pH 7.5). The video shows the formation and subsequent collapse of vacuoles within the coacervate structures as the solution was exposed to UV light. This process was repeated upon continuous UV light illumination.
44160_2025_844_MOESM8_ESM.avi (download AVI )
Supplementary Video 6 A fluorescence microscopy video captures the degradation of coacervate droplets upon the addition of glucose (10 mM). The coacervates, loaded with glucose oxidase (0.1 mg·mL⁻¹, 22.3 U·mL⁻¹), catalysed the oxidation of glucose into gluconic acid using oxygen. This reaction ultimately generated hydrogen peroxide, which oxidized and cleaved the disulfide bonds, resulting in the breakdown of the coacervate structures. The coacervate droplets were prepared from A/B solution (molar ratio of 5:5, total concentration of 5 mM, pH 7.5) after UV-light irradiation.
Source data
Source Data Fig. 2 (download XLSX )
Statistical Source Data.
Source Data Fig. 3 (download XLSX )
Statistical Source Data.
Source Data Fig. 4 (download XLSX )
Statistical Source Data.
Source Data Fig. 5 (download XLSX )
Statistical Source Data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shi, K., Song, P., Li, J. et al. Light-fuelled growth dynamics and structural transition of synthetic protocells. Nat. Synth 4, 1359–1368 (2025). https://doi.org/10.1038/s44160-025-00844-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00844-1
This article is cited by
-
Recent advances in coacervate protocells from passive catalysts to chemically programmable systems
Communications Chemistry (2026)
-
From light to life-like protocells
Nature Synthesis (2025)