Abstract
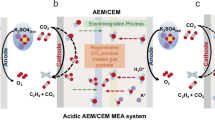
CO2 electrolysis offers an attractive route for the sustainable production of ethylene. However, electrolysis in membrane electrode assembly (MEA) systems using conventional copper electrocatalysts is limited by low current densities and high operating voltages. Here we report a design strategy involving cobalt-based subsurface dopants to construct and stabilize catalytically active sites. In-depth experimental and theoretical investigations revealed that the dopants induce a shift in the rate-determining step for ethylene from CO* coupling to the chemical step: OCCO* + H* → OCCHO* + *. A Tafel slope of 54 mV per decade is observed, which is considerably lower than the value of 124 mV per decade seen for a reference copper catalyst. This enables MEA operation with a low full-cell voltage of 1.89 V at 0.5 A and stable operation for 145 h at 1 A. We showcase record MEA CO2-to-ethylene conversion at a current of 4 A, with a Faradaic efficiency of 70.6% and a full-cell energy efficiency of 25.2%. Techno-economic assessment indicates potential for profitability with a production cost close to the ethylene market price under optimistic conditions.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information files. Should any raw data files be needed in another format, they are available from the corresponding author upon reasonable request. Source data are provided with this paper.
References
Arquer, F. P. G. D. et al. CO2 electrolysis to multicarbon products at activities greater than 1 A cm−2. Science 367, 661–666 (2020).
Qiao, J., Liu, Y., Hong, F. & Zhang, J. A review of catalysts for the electroreduction of carbon dioxide to produce low-carbon fuels. Chem. Soc. Rev. 43, 631–675 (2014).
Stephens, I. E. L. et al. 2022 roadmap on low temperature electrochemical CO2 reduction. J. Phys. Energy 4, 042003 (2022).
Chu, S., Cui, Y. & Liu, N. The path towards sustainable energy. Nat. Mater. 16, 16–22 (2016).
Hepburn, C. et al. The technological and economic prospects for CO2 utilization and removal. Nature 575, 87–97 (2019).
Ross, M. B. et al. Designing materials for electrochemical carbon dioxide recycling. Nat. Catal. 2, 648–658 (2019).
Clark, E. L. et al. Standards and protocols for data acquisition and reporting for studies of the electrochemical reduction of carbon dioxide. ACS Catal. 8, 6560–6570 (2018).
Wang, Y., Liu, J. & Zheng, G. Designing copper-based catalysts for efficient carbon dioxide electroreduction. Adv. Mater. 33, e2005798 (2021).
De Luna, P. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science 364, eaav3506 (2019).
Bushuyev, O. S. et al. What should we make with CO2 and how can we make it? Joule 2, 825–832 (2018).
Jouny, M., Luc, W. & Jiao, F. General techno-economic analysis of CO2 electrolysis systems. Ind. Eng. Chem. Res. 57, 2165–2177 (2018).
Shin, H., Hansen, K. U. & Jiao, F. Techno-economic assessment of low-temperature carbon dioxide electrolysis. Nat. Sustain. 4, 911–919 (2021).
Masel, R. I. et al. An industrial perspective on catalysts for low-temperature CO2 electrolysis. Nat. Nanotechnol. 16, 118–128 (2021).
Weng, L.-C., Bell, A. T. & Weber, A. Z. Towards membrane-electrode assembly systems for CO2 reduction: a modeling study. Energy Environ. Sci. 12, 1950–1968 (2019).
Ge, L. et al. Electrochemical CO2 reduction in membrane-electrode assemblies. Chem 8, 663–692 (2022).
Zhang, Z. et al. Membrane electrode assembly for electrocatalytic CO2 reduction: principle and application. Angew. Chem. Int. Ed. 62, e202302789 (2023).
Hori, Y. in Modern Aspects of Electrochemistry Vol. 42 (eds Vayenas, C. G. et al.) 89–189 (Springer, 2008).
Fan, L. et al. Strategies in catalysts and electrolyzer design for electrochemical CO2 reduction toward C2+ products. Sci. Adv. 6, eaay3111 (2020).
Dinh, C.-T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Gao, D., Arán-Ais, R. M., Jeon, H. S. & Roldan Cuenya, B. Rational catalyst and electrolyte design for CO2 electroreduction towards multicarbon products. Nat. Catal. 2, 198–210 (2019).
Zhao, Z., Zhang, J., Lei, M. & Lum, Y. Reviewing the impact of halides on electrochemical CO2 reduction. Nano Res. Energy 2, e9120044 (2023).
Nitopi, S. et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem. Rev. 119, 7610–7672 (2019).
Kuhl, K. P., Cave, E. R., Abram, D. N. & Jaramillo, T. F. New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces. Energy Environ. Sci. 5, 7050–7059 (2012).
Li, J. et al. Electrokinetic and in situ spectroscopic investigations of CO electrochemical reduction on copper. Nat. Commun. 12, 3264 (2021).
Kim, D., Kley, C. S., Li, Y. & Yang, P. Copper nanoparticle ensembles for selective electroreduction of CO2 to C2–C3 products. Proc. Natl Acad. Sci. USA 114, 10560–10565 (2017).
Li, Z., Zhang, T., Raj, J., Roy, S. & Wu, J. Revisiting reaction kinetics of CO electroreduction to C2+ products in a flow electrolyzer. Energy Fuels 37, 7904–7910 (2023).
Lu, X., Shinagawa, T. & Takanabe, K. Product distribution control guided by a microkinetic analysis for CO reduction at high-flux electrocatalysis using gas-diffusion Cu electrodes. ACS Catal. 13, 1791–1803 (2023).
Zhang, H., Gao, J., Raciti, D. & Hall, A. S. Promoting Cu-catalysed CO2 electroreduction to multicarbon products by tuning the activity of H2O. Nat. Catal. 6, 807–817 (2023).
Fletcher, S. Tafel slopes from first principles. J. Solid State Electrochem. 13, 537–549 (2008).
Wuttig, A. & Surendranath, Y. Impurity ion complexation enhances carbon dioxide reduction catalysis. ACS Catal. 5, 4479–4484 (2015).
Cheng, T., Xiao, H. & Goddard, W. A. 3rd Full atomistic reaction mechanism with kinetics for CO reduction on Cu(100) from ab initio molecular dynamics free-energy calculations at 298 K. Proc. Natl Acad. Sci. USA 114, 1795–1800 (2017).
Garza, A. J., Bell, A. T. & Head-Gordon, M. Mechanism of CO2 reduction at copper surfaces: pathways to C2 products. ACS Catal. 8, 1490–1499 (2018).
Sandberg, R. B., Montoya, J. H., Chan, K. & Nørskov, J. K. CO–CO coupling on Cu facets: coverage, strain and field effects. Surf. Sci. 654, 56–62 (2016).
Yi, H. et al. Structure and properties of Prussian blue analogues in energy storage and conversion applications. Adv. Funct. Mater. https://doi.org/10.1002/adfm.202006970 (2020).
Peterson, A. A. Global optimization of adsorbate–surface structures while preserving molecular identity. Top. Catal. 57, 40–53 (2013).
Birdja, Y. Y. et al. Advances and challenges in understanding the electrocatalytic conversion of carbon dioxide to fuels. Nat. Energy 4, 732–745 (2019).
Peng, H. J., Tang, M. T., Halldin Stenlid, J., Liu, X. & Abild-Pedersen, F. Trends in oxygenate/hydrocarbon selectivity for electrochemical CO2 reduction to C2 products. Nat. Commun. 13, 1399 (2022).
Montoya, J. H., Shi, C., Chan, K. & Norskov, J. K. Theoretical insights into a CO dimerization mechanism in CO2 electroreduction. J. Phys. Chem. Lett. 6, 2032–2037 (2015).
Wang, Y. et al. Catalyst synthesis under CO2 electroreduction favours faceting and promotes renewable fuels electrosynthesis. Nat. Catal. 3, 98–106 (2019).
Sundararaman, R. et al. JDFTx: software for joint density-functional theory. SoftwareX 6, 278–284 (2017).
Sundararaman, R., Goddard, W. A. III & Arias, T. A. Grand canonical electronic density-functional theory: algorithms and applications to electrochemistry. J. Chem. Phys. 146, 114104 (2017).
Aguila, D., Prado, Y., Koumousi, E. S., Mathoniere, C. & Clerac, R. Switchable Fe/Co Prussian blue networks and molecular analogues. Chem. Soc. Rev. 45, 203–224 (2016).
Zakaria, M. B. & Chikyow, T. Recent advances in Prussian blue and Prussian blue analogues: synthesis and thermal treatments. Coord. Chem. Rev. 352, 328–345 (2017).
Zhang, H. et al. On the activity and selectivity of 5-hydroxymethylfurfural electrocatalytic oxidation over cation-defective nickel hydroxides. ACS Catal. 14, 9565–9574 (2024).
Liu, K., Smith, W. A. & Burdyny, T. Introductory guide to assembling and operating gas diffusion electrodes for electrochemical CO2 reduction. ACS Energy Lett. 4, 639–643 (2019).
Schouten, K. J. P., Pérez Gallent, E. & Koper, M. T. M. Structure sensitivity of the electrochemical reduction of carbon monoxide on copper single crystals. ACS Catal. 3, 1292–1295 (2013).
De Gregorio, G. L. et al. Facet-dependent selectivity of Cu catalysts in electrochemical CO2 reduction at commercially viable current densities. ACS Catal. 10, 4854–4862 (2020).
Lum, Y. & Ager, J. W. Stability of residual oxides in oxide-derived copper catalysts for electrochemical CO2 reduction investigated with 18O labeling. Angew. Chem. Int. Ed. 57, 551–554 (2018).
Mandal, L. et al. Investigating the role of copper oxide in electrochemical CO2 reduction in real time. ACS Appl. Mater. Interfaces 10, 8574–8584 (2018).
An, H. et al. Sub-second time-resolved surface-enhanced raman spectroscopy reveals dynamic CO intermediates during electrochemical CO2 reduction on copper. Angew. Chem. Int. Ed. 60, 16576–16584 (2021).
Wei, P. et al. Coverage-driven selectivity switch from ethylene to acetate in high-rate CO2/CO electrolysis. Nat. Nanotechnol. 18, 299–306 (2023).
Gameel, K. M., Sharafeldin, I. M., Abourayya, A. U., Biby, A. H. & Allam, N. K. Unveiling CO adsorption on Cu surfaces: new insights from molecular orbital principles. Phys. Chem. Chem. Phys. 20, 25892–25900 (2018).
Limaye, A. M., Zeng, J. S., Willard, A. P. & Manthiram, K. Bayesian data analysis reveals no preference for cardinal Tafel slopes in CO2 reduction electrocatalysis. Nat. Commun. 12, 703 (2021).
Xiao, H., Cheng, T. & Goddard, W. A. 3rd Atomistic mechanisms underlying selectivities in C1 and C2 products from electrochemical reduction of CO on Cu(111). J. Am. Chem. Soc. 139, 130–136 (2017).
Gao, W., Xu, Y., Fu, L., Chang, X. & Xu, B. Experimental evidence of distinct sites for CO2-to-CO and CO conversion on Cu in the electrochemical CO2 reduction reaction. Nat. Catal. 6, 885–894 (2023).
Lum, Y. & Ager, J. W. Evidence for product-specific active sites on oxide-derived Cu catalysts for electrochemical CO2 reduction. Nat. Catal. 2, 86–93 (2018).
Fang, W. et al. Durable CO2 conversion in the proton-exchange membrane system. Nature 626, 86–91 (2024).
Hao, S. et al. Improving the operational stability of electrochemical CO2 reduction reaction via salt precipitation understanding and management. Nat. Energy 10, 266–277 (2025).
Overa, S. et al. Enhancing acetate selectivity by coupling anodic oxidation to carbon monoxide electroreduction. Nat. Catal. 5, 738–745 (2022).
Ling, N. et al. Acidic media impedes tandem catalysis reaction pathways in electrochemical CO2 reduction. Angew. Chem. Int. Ed. 62, e202308782 (2023).
Chen, J., Jia, M., Hu, P. & Wang, H. CATKINAS: a large-scale catalytic microkinetic analysis software for mechanism auto-analysis and catalyst screening. J. Comput. Chem. 42, 379–391 (2021).
Chen, J.-F., Mao, Y., Wang, H.-F. & Hu, P. Reversibility iteration method for understanding reaction networks and for solving microkinetics in heterogeneous catalysis. ACS Catal. 6, 7078–7087 (2016).
Chen, J., Jia, M., Lai, Z., Hu, P. & Wang, H. SSIA: a sensitivity-supervised interlock algorithm for high-performance microkinetic solving. J. Chem. Phys. 154, 024108 (2021).
Yue, K. et al. Stabilized Cu0–Cu1+ dual sites in a cyanamide framework for selective CO2 electroreduction to ethylene. Nat. Commun. 15, 7820 (2024).
Fang, M. et al. Anionic ionomer: released surface-immobilized cations and an established hydrophobic microenvironment for efficient and durable CO2-to-ethylene electrosynthesis at high current over one month. J. Am. Chem. Soc. 146, 27060–27069 (2024).
Li, F. et al. Molecular tuning of CO2-to-ethylene conversion. Nature 577, 509–513 (2020).
Li, W. et al. Bifunctional ionomers for efficient co-electrolysis of CO2 and pure water towards ethylene production at industrial-scale current densities. Nat. Energy 7, 835–843 (2022).
Liu, W. et al. Electrochemical CO2 reduction to ethylene by ultrathin CuO nanoplate arrays. Nat. Commun. 13, 1877 (2022).
Acknowledgements
We thank B. Johannessen and acknowledge the use of the Australian Synchrotron Facility at the Australian Nuclear Science and Technology Organisation (ANSTO) for collection of the in situ X-ray absorption spectroscopy (XAS) data used in this work. We also acknowledge the use of the X-ray Absorption Fine structure for catalysis (XAFCA) beamline of the Singapore Synchrotron Light Source (SSLS) for collection of the ex situ and in situ XAS data used in this work. In situ XAS, including X-ray absorption near edge structure (XANES) and extended X-ray absorption fine structure (EXAFS), was also performed in fluorescence mode in an in situ photocatalytic cell using a silicon drift detector at the BL32A beamline of TPS, National Synchrotron Radiation Research Center (NSRRC), Taiwan. We thank W. Nie and C. Hu for useful advice on Raman spectra analysis. Y.L. acknowledges support and funding from the A*STAR (Agency for Science, Technology and Research) under its LCER FI programme (award number U2102d2002) and an NRF Fellowship (award number NRF-NRFF14-2022-0003). This research is supported by the National Research Foundation, Prime Minister’s Office, Singapore, under its Campus for Research Excellence and Technological Enterprise (CREATE) programme (Development of Advanced Catalysts for Electrochemical Carbon Abatement, project code 370184872). The computational study is supported by the Marsden Fund Council from government funding (21-UOA-237) and Catalyst: Seeding General Grant (24-UOA-048-CSG), managed by Royal Society Te Apārangi. All DFT calculations were carried out on the New Zealand eScience Infrastructure (NeSI) high-performance computing facilities.
Author information
Authors and Affiliations
Contributions
Y.L. and Z.W. supervised the project. Y.L. and Q.Y. conceived the idea and designed the experiments. Q.Y. carried out the experimental work. X.W. and Y.M. performed the computational work. J.Z. carried out the isotope experiments and analysis. Y.Z., S.X., Y.-C.L., Y.-H.H. and S.-F.H. carried out the XAS experiments. L.Z. performed the high-resolution STEM. S.B.D. carried out the X-ray diffraction measurements. M.W. prepared the Cu/PTFE and Cu/CP catalysts. B.W. prepared the IrOx-coated titanium mesh electrodes. M.Z. carried out the XPS measurements. W.R.L. contributed to data analysis and manuscript editing. Q.Y., X.W., Z.W. and Y.L. co-wrote the manuscript. All authors discussed the results and assisted with the manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
Q.Y. and Y.L. are co-inventors on a patent application ‘Method of converting carbon dioxide to a multi-carbon compound’, PCT/SG2024/050727. The other authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks William Goddard III, Jingshan Luo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Peter Seavill, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–86, Tables 1–38 and Notes 1–3.
Supplementary Data 1 (download PDF )
Atomic coordinates of the optimized computational models.
Source data
Source Data Fig. 2 (download XLSX )
Figure2_SourceData (excel).
Source Data Fig. 3 (download XLSX )
Figure3_SourceData (excel).
Source Data Fig. 4 (download XLSX )
Figure4_SourceData (excel).
Source Data Fig. 5 (download XLSX )
Figure5_SourceData (excel).
Source Data Fig. 6 (download XLSX )
Figure6_SourceData (excel).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, Q., Wang, X., Zhang, J. et al. Ethylene electrosynthesis at low voltages enabled by dopant-induced modulation of the rate-determining step. Nat. Synth 4, 1396–1407 (2025). https://doi.org/10.1038/s44160-025-00850-3
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00850-3