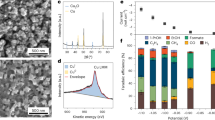
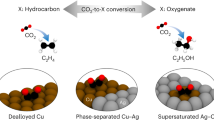
Abstract
Electroreduction of CO2 or CO can produce renewable ethanol—a valuable industrial chemical. However, the limited energy and carbon efficiencies of reported systems present practical challenges. Here we introduce p-block elements into copper catalysts, enabling electrosynthesis of ethanol from CO for 200 h and delivering a full-cell energy efficiency of 22% and a carbon efficiency of 50%, seven-fold better than state-of-the-art. Density functional theory calculations indicate that the lead-doped copper catalyst enhances the cleavage of C–O bonds in *OCHx molecules formed during CO hydrogenation. This enhances the cross-coupling reaction between *CO and *CHx species favouring ethanol production, as opposed to the conventional *CO dimerization pathway that primarily yields ethylene. Using a set of in situ spectroscopic techniques, we show that the addition of lead to copper catalysts expedites the generation of *OCHx species and increases the coverage of *CHx species, thereby enhancing their coupling with *CO and improving ethanol production, in agreement with our theoretical predictions.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets analysed and generated during the current study are included in the paper and its Supplementary Information. Source data are provided with this paper.
References
Ethanol Market Size, Share & COVID-19 Impact Analysis, by Type (Synthetic and Biobased), by Application (Fuel, Industrial Solvent, Food & Beverages, Chemicals, and Others), and Regional Forecast, 2023–2030 (Fortune Business Insights, 2024).
Fry, M. et al. Capturing and Utilizing CO2 from Ethanol: Adding Economic Value and Jobs to Rural Economies and Communities While Reducing Emissions (State CO2-EOR Deployment Work Group, 2017).
Chen, C. et al. Highly efficient electroreduction of CO2 to C2+ alcohols on heterogeneous dual active sites. Angew. Chem. Int. Ed. 132, 16601–16606 (2020).
Li, F. et al. Cooperative CO2-to-ethanol conversion via enriched intermediates at molecule–metal catalyst interfaces. Nat. Catal. 3, 75–82 (2020).
Wang, X. et al. Efficient electrically powered CO2-to-ethanol via suppression of deoxygenation. Nat. Energy 5, 478–486 (2020).
Gu, Z. et al. Efficient electrocatalytic CO2 reduction to C2+ alcohols at defect-site-rich Cu surface. Joule 5, 429–440 (2021).
Huang, J. E. et al. CO2 electrolysis to multicarbon products in strong acid. Science 372, 1074–1078 (2021).
Crandall, B. S. et al. Kilowatt-scale tandem CO2 electrolysis for enhanced acetate and ethylene production. Nat. Chem. Eng. 1, 421–429 (2024).
Jouny, M., Hutchings, G. S. & Jiao, F. Carbon monoxide electroreduction as an emerging platform for carbon utilization. Nat. Catal. 2, 1062–1070 (2019).
Ozden, A. et al. Energy-and carbon-efficient CO2/CO electrolysis to multicarbon products via asymmetric ion migration–adsorption. Nat. Energy 8, 179–190 (2023).
Ozden, A. et al. Cascade CO2 electroreduction enables efficient carbonate-free production of ethylene. Joule 5, 706–719 (2021).
Chen, C. et al. Exploration of the bio-analogous asymmetric C–C coupling mechanism in tandem CO2 electroreduction. Nat. Catal. 5, 878–887 (2022).
da Silva, A. H. M., Karaiskakis, G., Vos, R. E. & Koper, M. T. M. Mechanistic insights into the formation of hydroxyacetone, acetone, and 1, 2-propanediol from electrochemical CO2 reduction on copper. J. Am. Chem. Soc. 145, 15343–15352 (2023).
Li, J. et al. Intercepting elusive intermediates in Cu-mediated CO electrochemical reduction with alkyl species. J. Am. Chem. Soc. 144, 20495–20506 (2022).
Ren, D. et al. Atomic layer deposition of ZnO on CuO enables selective and efficient electroreduction of carbon dioxide to liquid fuels. Angew. Chem. Int. Ed. 58, 15036–15040 (2019).
Iyengar, P., Kolb, M. J., Pankhurst, J. R., Calle-Vallejo, F. & Buonsanti, R. Elucidating the facet-dependent selectivity for CO2 electroreduction to ethanol of Cu–Ag tandem catalysts. ACS Catal. 11, 4456–4463 (2021).
Kong, S. et al. Delocalization state-induced selective bond breaking for efficient methanol electrosynthesis from CO2. Nat. Catal. 6, 6–15 (2023).
Liu, Z. et al. Switching CO2 electroreduction toward ethanol by delocalization state-tuned bond cleavage. J. Am. Chem. Soc. 146, 14260–14266 (2024).
Yan, T. et al. Multiscale CO2 electrocatalysis to C2+ products: reaction mechanisms, catalyst design, and device fabrication. Chem. Rev. 123, 10530–10583 (2023).
Li, J. et al. Constraining CO coverage on copper promotes high-efficiency ethylene electroproduction. Nat. Catal. 2, 1124–1131 (2019).
Zheng, T. et al. Copper-catalysed exclusive CO2 to pure formic acid conversion via single-atom alloying. Nat. Nanotechnol. 16, 1386–1393 (2021).
Vasileff, A., Xu, C., Jiao, Y., Zheng, Y. & Qiao, S.-Z. Surface and interface engineering in copper-based bimetallic materials for selective CO2 electroreduction. Chem 4, 1809–1831 (2018).
Tröger, L. et al. Full correction of the self-absorption in soft-fluorescence extended X-ray-absorption fine structure. Phys. Rev. B 46, 3283 (1992).
Shao, F. et al. In situ spectroelectrochemical probing of CO redox landscape on copper single-crystal surfaces. Proc. Natl Acad. Sci. USA 119, e2118166119 (2022).
Long, C. et al. Regulating reconstruction of oxide-derived Cu for electrochemical CO2 reduction toward n-propanol. Sci. Adv. 9, eadi6119 (2023).
Wen, Y. et al. Introducing Brønsted acid sites to accelerate the bridging-oxygen-assisted deprotonation in acidic water oxidation. Nat. Commun. 13, 4871 (2022).
Zhang, N. et al. Dynamical evolution of CO2 and H2O on garnet electrolyte elucidated by ambient pressure X-ray spectroscopies. Nat. Commun. 15, 2777 (2024).
Palomino, R. M. et al. Interfaces in heterogeneous catalytic reactions: ambient pressure XPS as a tool to unravel surface chemistry. J. Electron. Spectrosc. Relat. Phenom. 221, 28–43 (2017).
Leclerc, M., Etxebarria, A., Ye, Y., Crumlin, E. J. & Brisard, G. M. An APXPS probe of Cu/Pd bimetallic catalyst surface chemistry of CO2 toward co in the presence of H2O and H2. J. Chem. Phys. 124, 17085–17094 (2020).
Ferrah, D. et al. Wet chemical growth and thermocatalytic activity of Cu-based nanoparticles supported on TiO2 nanoparticles/HOPG: in situ ambient pressure XPS study of the CO2 hydrogenation reaction. ACS Catal. 9, 6783–6802 (2019).
Gengenbach, T. R., Major, G. H., Linford, M. R. & Easton, C. D. Practical guides for X-ray photoelectron spectroscopy (XPS): interpreting the carbon 1s spectrum. J. Vac. Sci. Technol. A 39, 013204 (2021).
Ma, X. et al. Stabilizing Cu0–Cu+ sites by Pb-doping for highly efficient CO2 electroreduction to C2 products. Green Chem. 25, 7635–7641 (2023).
Han, S. et al. CO2 electroreduction to C2+ products over Cu–Pb heterojunction catalyst. ChemCatChem 16, e202300918 (2024).
Niu, W. et al. Pb-rich Cu grain boundary sites for selective CO-to-n-propanol electroconversion. Nat. Commun. 14, 4882 (2023).
Gabardo, C. M. et al. Continuous carbon dioxide electroreduction to concentrated multi-carbon products using a membrane electrode assembly. Joule 3, 2777–2791 (2019).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Rad. 12, 537–541 (2005).
Yao, K. et al. In situ copper faceting enables efficient CO2/CO electrolysis. Nat. Commun. 15, 1749 (2024).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558 (1993).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal–amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251 (1994).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953 (1994).
Perdew, J. P., Ernzerhof, M. & Burke, K. Rationale for mixing exact exchange with density functional approximations. J. Chem. Phys. 105, 9982–9985 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu. J. Chem. Phys. 132, 154104 (2010).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Yao, Z., Chan, M. K. Y. & Wolverton, C. Exploring the origin of anionic redox activity in super Li-rich iron oxide-based high-energy-density cathode materials. Chem. Mater. 34, 4536–4547 (2022).
Ma, X. et al. Electrochemical CC coupling between CO2 and formaldehyde into ethanol. Chem Catal. 2, 3207–3224 (2022).
Xu, Q. et al. Identifying and alleviating the durability challenges in membrane-electrode-assembly devices for high-rate CO electrolysis. Nat. Catal. 6, 1042–1051 (2023).
Li, J. et al. Weak CO binding sites induced by Cu–Ag interfaces promote CO electroreduction to multi-carbon liquid products. Nat. Commun. 14, 698 (2023).
Wang, P. et al. Boosting electrocatalytic CO2-to-ethanol production via asymmetric C–C coupling. Nat. Commun. 13, 3754 (2022).
Wang, X. et al. Efficient electrosynthesis of n-propanol from carbon monoxide using a Ag–Ru–Cu catalyst. Nat. Energy 7, 170–176 (2022).
Acknowledgements
This work was financially supported by the National Key Research and Development Program of China (2023YFA1507500 and 2022YFA1505100 to J.L.), the National Natural Science Foundation of China (BE3250011 to J.L. and 52373228 to Z.Y), the Fundamental Research Funds for the Central Universities (23×010301599 and 24×010301678 to J.L.), the Shanghai Pilot Program for Basic Research—Shanghai Jiao Tong University (21TQ1400227 to J.L.), Shanghai Municipal Science and Technology Major Project to J.L., the Ontario Research Foundation: Research Excellence Program to D.S., the Natural Sciences and Engineering Research Council (NSERC) of Canada to D.S., and TOTAL SE to D.S. A.O. gratefully acknowledges the financial support of Khalifa University for this work through the grant FSU-2025-006. Part of the XAS described in this paper was performed in May 2022 at the Canadian Light Source, a national research facility of the University of Saskatchewan, which is supported by NSERC, the Canada Foundation for Innovation (CFI), the National Research Council (NRC), the Canadian Institutes of Health Research (CIHR), the Government of Saskatchewan and the University of Saskatchewan. We acknowledge the Shanghai Synchrotron Radiation Facility, China, for the provision of synchrotron radiation beamtime at beamlines BL13SSW and BL02B01. Support from Canada Research Chairs Program is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
J.L. conceived the idea and supervised the project. P.L., N.S. and Y.W. carried out all the electrochemical experiments. P.P., A.O., R.K.M. and Y.X. contributed to electrode fabrication and assisted with the electrochemical testing. J.S. performed DFT calculations under the supervision of Z.Y., in which H.W., X.W. and Z.W. assisted with calculations and data analysis. P.L., N.S., H. Liu, Z.P. and Y.Z. performed XAS, NAP-XPS and Raman measurements, in which M.S., J.Z. and Y.H. assisted with the XAS testing, and S.S. and H.Z. assisted with the NAP-XPS testing. S.C. assisted with transmission electron microscopy characterizations. J.L. wrote the paper. D.S., Z.Y., H. Liang, A.O. and P.L. contributed to paper editing. All authors discussed the results and assisted during the paper preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Miho Yamauchi, Tao Yao and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–34, Discussion, Tables 1–6 and Note 1.
Supplementary Data (download XLSX )
Statistical source data for Supplementary Figs. 1, 2, 4, 6, 9, 10, 12, 13, 14, 15, 18, 21–29, 31–33.
Source data
Source Data Figs. (download XLSX )
2c,d, 3c–f, 4a–d and 5a–g.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, P., Sun, N., Su, J. et al. Electrosynthesis of ethanol via CO–CHx cross-coupling on copper alloy catalysts with engineered oxygen affinity. Nat. Synth 4, 1462–1472 (2025). https://doi.org/10.1038/s44160-025-00868-7
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00868-7