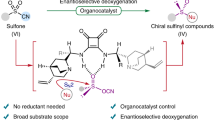
Abstract
The unique properties of stereogenic sulfur centres make sulfoxides highly valuable in medicinal chemistry, organic synthesis and other fields. Axially chiral sulfoxides are a class of complex organosulfur compounds with promising applications, but their synthesis remains challenging. Here we report a streamlined synthetic strategy for axially chiral sulfoxides. By leveraging the combination of rationally designed reagents and tailored catalytic systems, the method simultaneously establishes chiral sulfoxide and axial chirality in a single reaction step. This method was applied to a wide variety of substrates and resulted in products with broad functionality. The sulfinylation reaction was studied through a series of mechanistic experiments. This study provides a route to chiral sulfoxides with underexplored functionalities for potential future applications.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data relating to the characterization of materials and products, general methods, optimization studies, mechanistic studies, HPLC chromatograms and NMR spectra are available in the Supplementary Information. Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2335616 (3a), CCDC 2362413 (3b), CCDC 2362414 (3p) and CCDC 2442967 (10). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. The data that support the findings of this study are available in the article and its Supplementary Information.
References
Devendar, P. & Yang, G. F. Sulfur-containing agrochemicals. Top. Curr. Chem. 375, 82 (2017).
Drabowicz, J. Stereochemistry of organic sulfur compounds: more than 100 years of history, current state and further challenges. Phosphorus Sulfur Silicon Relat. Elem. 192, 145–148 (2017).
Colomer, I., Velado, M., Fernández De La Pradilla, R. & Viso, A. From allylic sulfoxides to allylic sulfenates: fifty years of a never-ending [2,3]-sigmatropic rearrangement. Chem. Rev. 117, 14201–14243 (2017).
Otocka, S., Kwiatkowska, M., Madalińska, L. & Kiełbasiński, P. Chiral organosulfur ligands/catalysts with a stereogenic sulfur atom: applications in asymmetric synthesis. Chem. Rev. 117, 4147–4181 (2017).
Kaiser, D. et al. Bond-forming and -breaking reactions at sulfur(IV): sulfoxides, sulfonium salts, sulfur ylides, and sulfinate salts. Chem. Rev. 119, 8701–8780 (2019).
Wojaczyńska, E. & Wojaczyński, J. Modern stereoselective synthesis of chiral sulfinyl compounds. Chem. Rev. 120, 4578–4611 (2020).
Wang, M. & Jiang, X. Prospects and challenges in organosulfur chemistry. ACS Sustain. Chem. Eng. 10, 671–677 (2022).
Matsui, T., Dekishima, Y. & Ueda, M. Biotechnological production of chiral organic sulfoxides: Current state and perspectives. Appl. Microbiol. Biotechnol. 98, 7699–7706 (2014).
Feng, M., Tang, B., Liang, S. H. & Jiang, X. Sulfur containing scaffolds in drugs: synthesis and application in medicinal chemistry. Curr. Top. Med. Chem. 16, 1200–1216 (2016).
Scott, K. A. & Njardarson, J. T. Analysis of US FDA-approved drugs containing sulfur atoms. Top. Curr. Chem. 376, 5 (2018).
Wojaczyńska, E. & Wojaczyński, J. Sulfoxides in medicine. Curr. Opin. Chem. Biol. 76, 102340–102348 (2023).
Agranat, I. & Caner, H. Intellectual property and chirality of drugs. Drug Discov. Today 4, 313–321 (1999).
Lipinski, C. & Hopkins, A. Navigating chemical space for biology and medicine. Nature 432, 855–861 (2004).
Bentley, R. Role of sulfur chirality in the chemical processes of biology. Chem. Soc. Rev. 34, 609–624 (2005).
Carreão, M. C. Applications of sulfoxides to asymmetric synthesis of biologically active compounds. Chem. Rev. 95, 1717–1760 (1995).
Fernández, I. & Khiar, N. Recent developments in the synthesis and utilization of chiral sulfoxides. Chem. Rev. 103, 3651–3705 (2003).
Sipos, G., Drinkel, E. E. & Dorta, R. The emergence of sulfoxides as efficient ligands in transition metal catalysis. Chem. Soc. Rev. 44, 3834–3860 (2015).
Yang, M. M., Wang, S. & Dong, Z. B. Recent advances for chiral sulfoxides in asymmetric catalysis. Synthesis 54, 5168–5185 (2022).
Trost, B. M. & Rao, M. Development of chiral sulfoxide ligands for asymmetric catalysis. Angew. Chem. Int. Ed. 54, 5026–5043 (2015).
Jia, T., Wang, M. & Liao, J. Chiral sulfoxide ligands in asymmetric catalysis. Top. Curr. Chem. 377, 8 (2019).
Allen, J. V., Bower, J. F. & Williams, J. M. J. Enantioselective palladium catalysed allylic substitution. Electronic and steric effects of the ligand. Tetrahedron: Asymmetry 5, 1895–1898 (1994).
Hiroi, K., Watanabe, K., Abe, I. & Koseki, M. New chiral sulfoxide ligands in catalytic asymmetric Diels–Alder reactions: double acceleration by the chiralities of the sulfoxides and oxazolines. Tetrahedron Lett. 42, 7617–7619 (2001).
Ichikawa, E. et al. New entries in Lewis acid–Lewis base bifunctional asymmetric catalyst: catalytic enantioselective Reissert reaction of pyridine derivatives. J. Am. Chem. Soc. 126, 11808–11809 (2004).
Mariz, R. et al. A chiral bis-sulfoxide ligand in late-transition metal catalysis; rhodium-catalyzed asymmetric addition of arylboronic acids to electron-deficient olefins. J. Am. Chem. Soc. 130, 2172–2173 (2008).
Hoshi, T. et al. A diastereomeric pair of sulfoxide-containing chiral MOP-type ligands: preparation and application to rhodium-catalyzed asymmetric 1,4-addition reactions. Chem. Lett. 47, 800–802 (2018).
Zhao, G. Z. et al. Electronic and steric tuning of an atropisomeric disulfoxide ligand motif and its use in the Rh(I)-catalyzed addition reactions of boronic acids to a wide range of acceptors. J. Org. Chem. 83, 9741–9755 (2018).
Liu, W., Ke, J. & He, C. Sulfur stereogenic centers in transition-metal-catalyzed asymmetric C–H functionalization: generation and utilization. Chem. Sci. 12, 10972–10984 (2021).
Choppin, S. & Wencel-Delord, J. Sulfoxide-directed or 3d-metal catalyzed C–H activation and hypervalent iodines as tools for atroposelective synthesis. Acc. Chem. Res. 56, 189–202 (2023).
Marqués, P. S. & Kammerer, C. Aryl sulfoxides: a traceless directing group for catalytic C–H activation of arenes. Chempluschem 89, e202300728 (2024).
Wang, Y.-B. & Tan, B. Construction of axially chiral compounds via asymmetric organocatalysis. Acc. Chem. Res. 51, 534–547 (2018).
Cheng, J. K., Xiang, S. H. & Tan, B. Organocatalytic enantioselective synthesis of axially chiral molecules: development of strategies and skeletons. Acc. Chem. Res. 55, 2920–2937 (2022).
Bai, X. F., Cui, Y. M., Cao, J. & Xu, L. W. Atropisomers with axial and point chirality: synthesis and applications. Acc. Chem. Res. 55, 2545–2561 (2022).
Kagan, H. B. (ed.) in Organosulfur Chemistry in Asymmetric Synthesis 1–29 (Wiley‐VCH, 2009).
Wojaczyńska, E. & Wojaczyński, J. Enantioselective synthesis of sulfoxides: 2000–2009. Chem. Rev. 110, 4303–4356 (2010).
Han, J., Soloshonok, V. A., Klika, K. D., Drabowicz, J. & Wzorek, A. Chiral sulfoxides: advances in asymmetric synthesis and problems with the accurate determination of the stereochemical outcome. Chem. Soc. Rev. 47, 1307–1350 (2018).
Zhang, X., Wang, F. & Tan, C. H. Asymmetric synthesis of S(IV) and S(VI) stereogenic centers. JACS Au 3, 700–714 (2023).
Broutin, P. E. & Colobert, F. An asymmetric biaryl Suzuki cross-coupling reaction: stereogenic benzylic carbinols as chiral auxiliaries. Eur. J. Org. Chem. 6, 1113–1128 (2005).
Colobert, F. et al. Axial chirality control during Suzuki–Miyaura cross-coupling reactions: the tert-butylsulfinyl group as an efficient chiral auxiliary. Org. Lett. 11, 5130–5133 (2009).
Leermann, T., Broutin, P. E., Leroux, F. R. & Colobert, F. Construction of the biaryl-part of vancomycin aglycon via atropo-diastereoselective Suzuki–Miyaura coupling. Org. Biomol. Chem. 10, 4095–4102 (2012).
Wesch, T., Leroux, F. R. & Colobert, F. Atropodiastereoselective C–H olefination of biphenyl p-tolyl sulfoxides with acrylates. Adv. Synth. Catal. 355, 2139–2144 (2013).
Hazra, C. K., Dherbassy, Q., Wencel-Delord, J. & Colobert, F. Synthesis of axially chiral biaryls through sulfoxide-directed asymmetric mild C–H activation and dynamic kinetic resolution. Angew. Chem. Int. Ed. 53, 13871–13875 (2014).
Dherbassy, Q. et al. Enantiopure sulfoxides: efficient chiral directing group for stereoselective C‒H bond activation: towards the control of axial chirality. Phosphorus Sulfur Silicon Relat. Elem. 190, 1339–1351 (2015).
Dherbassy, Q. et al. 1,1,1,3,3,3-Hexafluoroisopropanol as a remarkable medium for atroposelective sulfoxide-directed Fujiwara–Moritani reaction with acrylates and styrenes. Chem. Eur. J. 22, 1735–1743 (2016).
Clayden, J., Senior, J. & Helliwell, M. Atropisomerism at C–S bonds: asymmetric synthesis of diaryl sulfones by dynamic resolution under thermodynamic control. Angew. Chem. Int. Ed. 48, 6270–6273 (2009).
Jin, L. et al. Atroposelective synthesis of conjugated diene-based axially chiral styrenes via Pd(II)-catalyzed thioether-directed alkenyl C–H olefination. J. Am. Chem. Soc. 143, 12335–12344 (2021).
Vroemans, R. et al. Synthesis of homochiral sulfanyl- and sulfoxide-substituted naphthyltriazoles and study of the conformational stability. Org. Biomol. Chem. 19, 6521–6526 (2021).
Wen, W. et al. Bifunctional squaramide-catalyzed oxidative kinetic resolution: Simultaneous access to axially chiral thioether and sulfoxide. Adv. Sci. 11, 2402429 (2024).
Berkessel, A., Gröger, H. & MacMillan, D. W. C. (eds) Asymmetric Organocatalysis: From Biomimetic Concepts to Applications in Asymmetric Synthesis (Wiley-VCH, 2005).
MacMillan, D. W. C. The advent and development of organocatalysis. Nature 455, 304–308 (2008).
List, B. (ed.) Asymmetric Organocatalysis (Springer, 2009).
Song, C. E. (ed.) Cinchona Alkaloids in Synthesis and Catalysis: Ligands, Immobilization and Organocatalysis (Wiley-VCH, 2009).
Zhang, X. et al. Synthesis of chiral sulfinate esters by asymmetric condensation. Nature 604, 298–303 (2022).
Huang, S. et al. Organocatalytic asymmetric deoxygenation of sulfones to access chiral sulfinyl compounds. Nat. Chem. 15, 185–193 (2023).
Zhang, Y. Q. et al. Nickel-catalysed enantioselective hydrosulfenation of alkynes. Nat. Catal. 6, 487–494 (2023).
Gao, L. et al. Nickel-catalyzed enantioselective synthesis of dienyl sulfoxide. Angew. Chem. Int. Ed. 63, e202317626 (2024).
Liao, M. et al. Enantioselective sulfinylation of alcohols and amines by condensation with sulfinates. Chem 10, 1541–1552 (2024).
Wei, T. et al. Enantioselective construction of stereogenic-at-sulfur(IV) centres via catalytic acyl transfer sulfinylation. Nat. Chem. 16, 1301–1311 (2024).
Mishra, A. & Hanessian, S. 1,2-trans-Diaminocyclohexane (DACH) in asymmetric catalysis: nearing fifty years of faithful service and counting. Synthesis 56, 2747–2885 (2024).
Lee, H. J., Kang, S. H. & Kim, D. Y. Highly enantioselective conjugate addition of 3-substituted oxindoles to vinyl sulfone catalyzed by binaphthyl-modified tertiary amines. Synlett 11, 1559–1562 (2011).
Jang, H. S., Kim, Y. & Kim, D. Y. Enantioselective addition of diphenyl phosphonate to ketimines derived from isatins catalyzed by binaphthyl-modified organocatalysts. Beilstein J. Org. Chem. 12, 1551–1556 (2016).
Kim, K. S. & Kim, D. Y. Organocatalytic enantioselective one-pot synthesis of dihydrocoumarins via C–H oxidation and cyclization cascade. Asian J. Org. Chem. 12, e202300355 (2023).
Rayner, D. R., Gordon, A. J. & Mislow, K. Thermal racemization of diaryl, alkyl aryl, and dialkyl sulfoxides by pyramidal inversion. J. Am. Chem. Soc. 90, 4854–4860 (1968).
Tsurutani, Y., Yamashita, T. & Horie, K. Photoinversion reaction of a pyrenyl sulfoxide in solution and in polymer matrices. Polym. J. 30, 11–16 (1998).
Simmons, E. M. & Hartwig, J. F. On the interpretation of deuterium kinetic isotope effects in C–H bond functionalizations by transition-metal complexes. Angew. Chem. Int. Ed. 51, 3066–3072 (2012).
Satyanarayana, T., Abraham, S. & Kagan, H. B. Nonlinear effects in asymmetric catalysis. Angew. Chem. Int. Ed. 48, 456–494 (2009).
Wu, M. et al. Organocatalytic Si–Caryl bond functionalization-enabled atroposelective synthesis of axially chiral biaryl siloxanes. J. Am. Chem. Soc. 145, 20646–20654 (2023).
Zhu, X., Li, Y. & Bao, H. Mechanistic applications of nonlinear effects in first-row transition-metal catalytic systems. Chinese J. Chem. 41, 3097–3114 (2023).
Thierry, T., Frey, J., Geiger, Y. & Bellemin-Laponnaz, S. Non-linear effects in asymmetric catalysis. Actual. Chim. 15, 40–49 (2024).
Yin, S., Liu, J., Weeks, K. N. & Aponick, A. Catalytic enantioselective synthesis of axially chiral imidazoles by cation-directed desymmetrization. J. Am. Chem. Soc. 145, 28176–28183 (2023).
Balcells, D., Maseras, F. & Khiar, N. Base-catalyzed inversion of chiral sulfur centers. A computational study. Org. Lett. 6, 2197–2200 (2004).
Shibata, N. et al. Cinchona alkaloid/sulfinyl chloride combinations: enantioselective sulfinylating agents of alcohols. J. Am. Chem. Soc. 127, 1374–1375 (2005).
Shibata, N. et al. Cinchona alkaloid–sulfinyl chloride combinations: catalytic enantioselective sulfinylation of alcohols. Synlett 11, 1699–1702 (2005).
Acknowledgements
This study was supported by the Science and Technology Innovation Key R&D Program of Chongqing (grant number CSTB2022TIAD-STX0015 (H.Y.)), the Natural Science Foundation of Chongqing (grant numbers CSTB2023NSCQ-MSX0057 (W.Q.), CSTC2021JCYJ-JQX0019 (H.Y.)), the Graduate Research and Innovation Foundation of Chongqing, China (grant number CYB23075 (H.Y.)), the Fundamental Research Funds for the Central Universities (grant number 2023CDJKYJH009 (H.Y.)) and the National Natural Science Foundation of China (grant numbers 22271032 (H.Y.), 22471026 (W.Q.)). We thank X. Gong for X-ray crystallographic analysis.
Author information
Authors and Affiliations
Contributions
H.Y., W.Q. and P.W. conceived and directed the project. Y.C., G.Z. and D.X. designed and performed experiments. Y.C. and P.W. analysed and interpreted the experimental data. Y.C. prepared the Supplementary Information. H.Y., W.Q., P.W. and Y.C. wrote the paper. All authors discussed the results and commented on the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Hai-Ming Guo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Experimental details, Supplementary Figs. 1–12 and Tables 1–14.
Supplementary Data 1
X-ray crystallographic data for 3a, CCDC 2335616.
Supplementary Data 2
Structure factors for 3a, CCDC 2335616.
Supplementary Data 3
X-ray crystallographic data for 3b, CCDC 2362413.
Supplementary Data 4
Structure factors for 3b, CCDC 2362413.
Supplementary Data 5
X-ray crystallographic data for 3p, CCDC 2362414.
Supplementary Data 6
Structure factors for 3p, CCDC 2362414.
Supplementary Data 7
X-ray crystallographic data for 10, CCDC 2442967.
Supplementary Data 8
Structure factors for 10, CCDC 2442967.
Source data
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3, the data of the racemization experiment of 3a.
Source Data Fig. 5 (download XLSX )
Source data files for Fig. 5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chang, Y., Zhou, G., Xu, D. et al. Enantioselective organocatalytic construction of axially chiral sulfoxides. Nat. Synth 4, 1587–1597 (2025). https://doi.org/10.1038/s44160-025-00877-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00877-6