Abstract
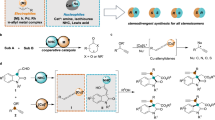
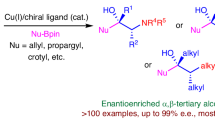
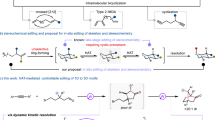
Developing general protocols that control multiple types of selectivity in a single transformation is challenging yet highly desirable for the synthesis of complex molecules. Here we describe a N-heterocyclic carbene–nickel catalyst that enables simultaneous control over chemo-, regio-, E/Z-, diastereo- and enantioselectivity in a dynamic kinetic resolution (DKR) asymmetric aldehyde–alkyne reductive coupling reaction. This method provides a straightforward synthesis of enantioenriched allylic alcohols from easily available substrates, and has broad scope and excellent functional group compatibility. We applied this protocol to the modification of profen-type drugs and the formal synthesis of sphingosine. Control experiments reveal that the stereocontrol is influenced by the rate of racemization. We expect the reported process to further stimulate the development of dynamic kinetic resolution chemistry and simplify the synthesis of valuable complex molecules.

This is a preview of subscription content, access via your institution
Access options






Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the article and its Supplementary Information. Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers 2416723 (6fa) and 2416724 (4ta). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. Source data are provided with this paper.
References
Trost, B. M. Selectivity: the key to synthetic efficiency. Science 219, 245–250 (1983).
Trost, B. M. & Fleming, I. Comprehensive Organic Synthesis: Selectivity, Strategy, and Efficiency in Modern Organic Chemistry (Pergamon, 1991).
Caddick, S. & Jenkins, K. Dynamic resolutions in asymmetric synthesis. Chem. Soc. Rev. 25, 447–456 (1996).
Bhat, V., Welin, R. E., Guo, X. & Stoltz, M. B. Advances in stereoconvergent catalysis from 2005 to 2015: transition-metal-mediated stereoablative reactions, dynamic kinetic resolutions, and dynamic kinetic asymmetric transformations. Chem. Rev. 117, 4528–4561 (2017).
Huerta, F. F., Minidis, E. B. A. & Bäckvall, E. J. Racemization in asymmetric synthesis. Dynamic kinetic resolution and related processes in enzyme and metal catalysis. Chem. Soc. Rev. 30, 321–331 (2001).
You, H., Rideau, E., Sidera, M. & Fletcher, P. S. Non-stabilized nucleophiles in Cu-catalysed dynamic kinetic asymmetric allylic alkylation. Nature 517, 351–355 (2015).
Choi, J. & Fu, G. C. Transition metal-catalyzed alkyl–alkyl bond formation: another dimension in cross-coupling chemistry. Science 356, eaaf7230 (2017).
Dehovitz, S. J. et al. Static to inducibly dynamic stereocontrol: the convergent use of racemic ketones. Science 369, 1113–1118 (2020).
Huang, M., Zhang, L., Pan, T. & Luo, S. Deracemization through photochemical E/Z isomerization of enamines. Science 375, 869–874 (2022).
Shimizu, H. et al. Developments in asymmetric hydrogenation from an industrial perspective. Acc. Chem. Res. 40, 1385–1393 (2007).
Noyori, R. et al. Stereoselective hydrogenation via dynamic kinetic resolution. J. Am. Chem. Soc. 111, 9134–9135 (1989).
Xie, J.-H. & Zhou, Q.-L. Catalytic asymmetric hydrogenation of α-substituted ketones and aldehydes via dynamic kinetic resolution: efficient approach to chiral alcohols. Aldrichimica Acta. 48, 33–40 (2015).
Steward, M. K., Gentry, C. E. & Johnson, S. J. Dynamic kinetic resolution of α-keto esters via asymmetric transfer hydrogenation. J. Am. Chem. Soc. 134, 7329–7332 (2012).
Wang, F. et al. Asymmetric transfer hydrogenation of α‑substituted-β-keto carbonitriles via dynamic kinetic resolution. J. Am. Chem. Soc. 143, 2477–2483 (2021).
Liu, J., Krajangsri, S., Yang, J., Li, J. & Andersson, G. P. Iridium-catalysed asymmetric hydrogenation of allylic alcohols via dynamic kinetic resolution. Nat. Catal. 1, 438–443 (2018).
Bartlett, S. L. & Johnson, J. S. Synthesis of complex glycolates by enantioconvergent addition reactions. Acc. Chem. Res. 50, 2284–2296 (2017).
Zavesky, B. P. & Johnson, J. S. Direct zinc(II)-catalyzed enantioconvergent additions of terminal alkynes to α-keto esters. Angew. Chem. Int. Ed. 56, 8805–8808 (2017).
Bartlett, S. L., Keiter, K. M. & Johnson, J. S. Synthesis of complex tertiary glycolates by enantioconvergent arylation of stereochemically labile α‑keto esters. J. Am. Chem. Soc. 139, 3911–3916 (2017).
Corbett, M. T. & Johnson, J. S. Dynamic kinetic asymmetric transformations of β-stereogenic α-ketoesters by direct aldolization. Angew. Chem. Int. Ed. 53, 255–259 (2014).
Ruan, L.-X., Sun, B., Liu, J. & Shi, S.-L. Dynamic kinetic asymmetric arylation and alkenylation of ketones. Science 379, 662–670 (2023).
Sun, B. et al. Dynamic kinetic asymmetric allylation, propargylation and crotylation of ketones using copper catalysis. Nat. Synth. 3, 1091–1103 (2024).
Cai, Y. & Shi, S.-L. Enantioconvergent arylation of racemic secondary alcohols to chiral tertiary alcohols enabled by nickel/N-heterocyclic carbene catalysis. J. Am. Chem. Soc. 143, 11963–11968 (2021).
Zhang, J.-W. et al. Enantioconvergent Negishi cross-coupling of racemic sec-alkylzinc reagent with aryl halides enabled by bulky N-heterocyclic carbene–Pd catalyst. CCS Chem. https://doi.org/10.31635/ccschem.025.202505591 (2025).
Hoshimoto, Y., Ohashi, M. & Ogoshi, S. Nickel-catalyzed selective conversion of two different aldehydes to cross-coupled esters. J. Am. Chem. Soc. 133, 4668–4671 (2011).
Rodrigo, S. K. et al. Efficient and regioselective nickel-catalyzed [2 + 2 + 2] cyclotrimerization of ynoates and related alkynes. Org. Biomol. Chem. 11, 7653–7657 (2013).
Lage, M. L., Bader, S. J., Sa-ei, K. & Montgomery, J. Chemoselective hydrosilylation of hydroxyketones. Tetrahedron 69, 5609–5613 (2013).
Chaulagain, M., Mahandru, G. M. & Montgomery, J. Alkyne hydrosilylation catalyzed by nickel complexes of N-heterocyclic carbenes. Tetrahedron 62, 7560–7566 (2006).
Janssen-Müller, D., Schlepphorst, C. & Glorius, F. Privileged chiral N-heterocyclic carbene ligands for asymmetric transition-metal catalysis. Chem. Soc. Rev. 46, 4845–4854 (2017).
Wang, Z.-C. & Shi, S.-L. Induced-fit chiral N-heterocyclic carbene ligands for asymmetric catalysis. Acc. Chem. Res. 58, 1376–1380 (2025).
Cai, Y. et al. Copper-catalyzed enantioselective Markovnikov protoboration of α-olefins enabled by a buttressed N-heterocyclic carbene ligand. Angew. Chem. Int. Ed. 57, 1376–1380 (2018).
Shen, D., Xu, Y. & Shi, S.-L. A bulky chiral N-heterocyclic carbene palladium catalyst enables highly enantioselective Suzuki–Miyaura cross-coupling reactions for the synthesis of biaryl atropisomers. J. Am. Chem. Soc. 141, 14938–14945 (2019).
Zhang, W.-B., Chen, G. & Shi, S.-L. Enantioselective Ni/N-heterocyclic carbene-catalyzed redox-economical coupling of aldehydes, alkynes, and enones for rapid construction of acyclic all-carbon quaternary stereocenters. J. Am. Chem. Soc. 144, 130–136 (2022).
Wang, Z.-C. et al. Enantioselective C–C cross-coupling of unactivated alkenes. Nat. Catal. 6, 1087–1097 (2023).
Liu, J.-M. et al. Chemodivergent, enantio- and regioselective couplings of alkynes, aldehydes and silanes enabled by nickel/N-heterocyclic carbene catalysis. Sci. Bull. 70, 674–682 (2025).
Miller, K., Huang, W.-S. & Jamison, T. Catalytic asymmetric reductive coupling of alkynes and aldehydes: enantioselective synthesis of allylic alcohols and α-hydroxy ketones. J. Am. Chem. Soc. 125, 3442–3443 (2003).
Wang, H. et al. NHC ligands tailored for simultaneous regio- and enantiocontrol in nickel-catalyzed reductive couplings. J. Am. Chem. Soc. 139, 9317–9324 (2017).
Han, F.-S. Transition-metal-catalyzed Suzuki–Miyaura cross-coupling reactions: a remarkable advance from palladium to nickel catalysts. Chem. Soc. Rev. 42, 5270–5298 (2013).
Ager, D. J., Prakash, I. & Schaad, D. R. 1,2-Amino alcohols and their heterocyclic derivatives as chiral auxiliaries in asymmetric synthesis. Chem. Rev. 96, 835–876 (1996).
Li, G., Chang, H.-T. & Sharpless, K. B. Catalytic asymmetric aminohydroxylation of olefins. Angew. Chem. Int. Ed. 35, 451–454 (1996).
Xie, J.-H. et al. Highly enantioselective and diastereoselective synthesis of chiral amino alcohols by ruthenium-catalyzed asymmetric hydrogenation of α-amino aliphatic ketones. J. Am. Chem. Soc. 131, 4222–4223 (2009).
Reetz, M. T., Drewes, M. W. & Schmitz, A. Stereoselective synthesis of β-amino alcohols from optically active α-amino acids. Angew. Chem. Int. Ed. 26, 1141–1143 (1987).
Merrill, A. & Sandhoff K. Sphingolipids: Metabolism and Cell Signaling in Biochemistry of Lipids, Lipoprotein, and Membranes (Elsevier, 2002).
Koolath, S. et al. Chiral combinatorial preparation and biological evaluation of unique ceramides for inhibition of sphingomyelin synthase. Chirality. 32, 308–313 (2020).
Acknowledgements
This work is supported by the National Key R&D Program of China (2022YFA1503700), the National Natural Science Foundation of China (22325110, 92256303, 22171280, 22501291), the Strategic Priority Research Program of the CAS (XDB0610000, XDA0540000), the Program of Shanghai Academic Research Leader (22XD1424900), the CAS Youth Interdisciplinary Team (JCTD-2021-11) and the Ningbo Natural Science Foundation (2022J017) (S.-L.S.).
Author information
Authors and Affiliations
Contributions
G.C., J.-M.L. and L.-X.R. developed the catalytic method and conducted the mechanistic studies. S.-L.S. conceived of the projects and directed the investigations. S.-L.S. and G.C. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Experimental details, Supplementary Sections 1–14, Tables 1–9 and Figs. 1–3.
Supplementary Data 1
X-ray crystallographic data for 6fa, CCDC 2416723.
Supplementary Data 2
X-ray crystallographic data for 4ta, CCDC 2416724.
Source data
Source Data Fig. 6 (download XLSX )
Source data for Fig. 6b.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, G., Liu, JM., Ruan, LX. et al. Selective dynamic kinetic asymmetric aldehyde–alkyne reductive coupling. Nat. Synth 4, 1630–1639 (2025). https://doi.org/10.1038/s44160-025-00887-4
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00887-4